- Protocols
- Articles and Issues
- About
- Become a Reviewer
Articles In Press
Articles In Press are peer reviewed and have been accepted for publication. Please note that these versions may be subject to further edits before their final online publication. Nevertheless, Articles In Press are citable using the DOI. Upon the formal online publication, the article will no longer be listed here, but existing links will automatically redirect to the final version in the corresponding issue.
Versatile Dual Mounting Enables Larval Zebrafish Imaging Across Microscope Configurations
Larval zebrafish are often mounted laterally to ensure consistent anatomical positioning and to standardize imaging of body axes across early development. However, this conventional approach often tethers sample orientation to a single microscope configuration and limits optical accessibility. We present a mounting protocol for larval zebrafish that enables optical access from both dorsal and ventral orientations while preserving lateral sample position. This approach uses common laboratory consumables to establish a mounting platform that eliminates any need to remount samples between the use of upright and inverted microscopes. By establishing a hydrophobic seal, mounted embryos can be inverted with ease to access the sample from either orientation. A seamless transition here facilitates reliable identification and longitudinal tracking of the same biological region of interest across microscope configurations. This protocol is broadly applicable to live imaging experiments requiring flexibility in imaging geometry, minimal sample handling, and high reproducibility.
Measuring Electrophysiological Activity in Acute Brain Slices, Spheroids, and Organoids Using 3D High-Density Multielectrode Arrays
Animal and human stem cell–derived three-dimensional models to study physio-pathological brain functioning are becoming a gold standard for in vitro electrophysiology, as they enable the recapitulation of complex network properties by accounting for spatial architectural features that better reflect in vivo conditions than simpler 2D models. Standard planar multielectrode arrays (MEAs), typically providing tens of recording electrodes, are commonly used to record activity from 2D neuronal cultures. However, when adapted for use with 3D models, planar 2D MEAs showed limited effectiveness. The main issues are limited specimen adhesion to the chip, a low number of sensing elements, inability to retrieve signals from within the tissue, and reduced perfusion and vitality of the tissue in contact with sensors. To overcome these limitations, a new generation of microchip-based 3D high-density MEAs (3D HD-MEA) has been developed and validated in recent years. This technological advancement has improved the sensing capabilities and the vitality of 3D models, providing a tool tailored to maximize their potential. Here, we present an optimized protocol for neural network activity recordings in 3D models (including acute slices, brain spheroids, and organoids) from various brain regions using 3D HD-MEAs. First, we summarize the critical steps for 1) obtaining viable acute slices from the mouse cerebellum, cortico-hippocampal circuit, and prefrontal cortex, 2) establishing efficient coupling of the slices with the chip, and 3) performing recordings and analyses. We then describe the main procedures required to obtain human and animal brain spheroids and neural organoids, as well as standardized routines to perform effective recordings and analyses. For each section, we highlight the crucial steps, identify tips for specific applications, and propose troubleshooting procedures. For example, the same type of preparation (e.g., acute slices) requires different adjustments when working with different brain areas. The specific information provided here is intended to assist researchers in their daily efforts to obtain efficient and reproducible functional recordings from 3D models by using the cutting-edge technique of 3D HD-MEA.
ROOT-ExM: Super-Resolution Imaging of Proteins in Arabidopsis Roots by Expansion Microscopy
Conventional light microscopy is limited in resolution by the diffraction limit of light, restricting the visualization of the nanoscale organization of biomolecules. Expansion microscopy (ExM) has emerged as a powerful technique to overcome this barrier by physically expanding the specimen embedded in a swellable hydrogel without requiring specialized or high-cost imaging hardware. ExM is widely used in animal models, whereas its application to plant tissues has been challenging due to their multicellularity, in which each cell is encompassed by the rigid cell wall, which resists the expansion forces and prevents isotropic swelling. Here, we describe a robust and optimized ExM protocol specifically designed for Arabidopsis thaliana root tissues. This protocol details critical steps, including immunostaining, anchoring, gelation, denaturation, cell wall digestion, and expansion. Our method achieves an expansion factor of approximately 4.3×, enabling an effective lateral resolution of ~60 nm using a standard confocal microscope. We demonstrate the visualization of microtubules with preserved ultrastructure. This accessible protocol allows plant researchers to perform super-resolution imaging without specialized optical equipment, facilitating detailed structural analysis of plant cells.
Oligo(dT) Fluorescence In Situ Hybridization to Visualize the Poly(A) mRNAs in the Internal Tissues of Drosophila
Fluorescence in situ hybridization (FISH) is a cytological method used to visualize specific oligonucleotide sequences within the cell. This method relies on the specific binding of a fluorescence-tagged probe, a short stretch of single-stranded polynucleotide, to its complementary sequence in the DNA or RNA, forming stable double-stranded hybrids. Fluorochromes, such as fluorescein, Alexa Fluor, cyanine dyes, or rhodamine, are attached to these probes to help in detecting their presence within the cell. Based on sequence complementarity, FISH allows for the visualization of the DNA or RNA with which they have hybridized. The distribution of these fluorochrome-tagged probes can be observed under a fluorescence or confocal microscope. The oligo(dT) FISH technique specifically utilizes a fluorochrome-tagged stretch of 40–50 thymidine (T) oligonucleotides that binds to the poly(A) tails of mature mRNAs within the cell. Newly transcribing pre-mRNAs and certain non-coding RNAs may not have poly(A) tails and therefore cannot be detected by this method. This step-by-step protocol outlines the oligo(dT) FISH technique for visualizing the cellular distribution of polyadenylated mRNAs in the tissues of Drosophila and other related model organisms.
Comprehensive Protocol for Handling Human Small Airway Epithelial Cells (HSAECs) to Establish Air–Liquid Interface (ALI) Cultures With TEER-Based Barrier Integrity Assessment
Understanding epithelial barrier function is essential for studying both its normal physiology and its role in disease, yet choosing an appropriate experimental model remains challenging. Animal models are commonly used but often suffer from interspecies differences that limit translational relevance. Human-derived cell lines offer a more suitable alternative, although establishing them often requires immortalisation strategies that involve overexpression of oncogenes, which can introduce phenotypic and functional changes. In contrast, primary cells, such as human small airway epithelial cells (HSAECs), provide a more physiologically accurate model. A critical aspect of replicating the native respiratory environment is maintaining continuous air exposure, which can be achieved through air–liquid interface (ALI) culture. This protocol provides a unified, step-by-step workflow for cultivating primary HSAECs under ALI conditions, covering the entire process from initial recovery after cryopreservation to the formation of a barrier-like layer. The protocol incorporates non-invasive methods such as transepithelial electrical resistance (TEER) measurements to monitor its integrity. While individual elements of this workflow have been described separately in different studies, a consolidated version encompassing the full workflow has not been widely available. This resource is intended for researchers with limited experience in airway epithelial culture and offers practical, clear guidance through each step of the process.
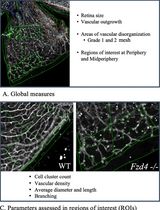
Computational Quantification of Mouse Retinal Vasculature Using ImageJ
Postnatal mouse retinal vascular development is a widely used model for studying retinal vascular diseases and evaluating candidate therapies. This is particularly relevant for inherited disorders such as familial exudative vitreoretinopathy (FEVR), in which impaired vascular growth and organization are central to disease pathogenesis. Numerous approaches have been used to assess retinal vasculature in mouse flat mounts, ranging from qualitative descriptions to limited quantitative measurements of vascular growth. However, phenotypic variability across genetic models, including different models of FEVR, complicates comparisons and underscores the need for standardized, comprehensive multi-parameter analyses that are suitable for rapid and cost-effective screening studies. We describe a standardized morphometric protocol using ImageJ software to quantitatively analyze mouse retinal vasculature in a reproducible manner. The protocol begins with measurement of areas of vascular disorganization (meshes) as well as total vascular and retinal area. Two defined regions in the peripheral and midperipheral retina are then selected to quantify cell clusters, followed by image processing, binarization, and skeletonization. From these processed images, vascular density, branch number, branch length and thickness, junction number, triple points, and box-counting fractal dimension and lacunarity are quantified. Overall, this protocol provides a rapid, cost-effective, and standardized framework for quantifying retinal vascular phenotypes across diverse mouse models. By capturing multiple structural features and accommodating phenotypic variability, it is well-suited for comparative studies and therapeutic screening in retinal vascular disease.
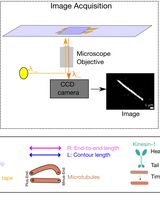
Quantification of Spatial Patterns of Microtubule Transport by Kinesin-1 Head and Tail
The conventional kinesin-1 is a plus-end-directed microtubule-dependent motor protein with distinct motor head, stalk, and tail domains. Along with the motor head, which binds and walks along microtubules in an adenosine 5’-triphosphate (ATP) dependent manner, kinesin also contains a C-terminal microtubule binding tail. Motor-driven collective motility is well characterized using in vitro gliding assays, which show uninterrupted, smooth trajectories of transport. However, gliding assays driven by the full-length Drosophila kinesin-1 with both head and tail resulted in the emergence of spontaneous spatial microtubule patterns and stop-and-go motion. This was reproduced by an equimolar ratio of the active head and passive tail. Here, we describe the detailed protocol to reconstitute these microtubule gliding assays using multiple motor types: the full-length kinesin-1, the motor head or tail, mixtures of both head and tail, and a rigor mutant of the kinesin. We provide details of the approach taken to acquire the image time-series, to then quantify the spatial patterns that result from these motor combinations. Our approach provides a framework to systematically characterize the spatiotemporal effects of molecular motor-driven collective microtubule transport.
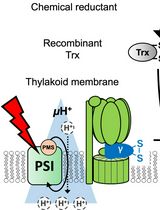
Evaluating Thioredoxin-Mediated CFoCF1 Reduction Using an In Vitro Thylakoid Assay
The activity of chloroplast ATP synthase (CFoCF1) is precisely regulated through a thioredoxin (Trx)-mediated dithiol/disulfide reaction in response to varying light conditions. This regulatory mechanism is further controlled by ΔpH formation across the thylakoid membrane. To better understand this complicating regulatory function of CFoCF1, a method is required to evaluate the extent of CFoCF1 reduction by Trx under controlled ΔpH conditions and to directly evaluate the redox state of CFoCF1. In this study, we present a simple in vitro procedure to assess the CFoCF1 reduction system using spinach thylakoids. The method consists of three key steps: (A) simple preparation of intact thylakoids from spinach leaves; (B) reduction of CFoCF1 on the thylakoid membrane using recombinant Trx under light irradiation; and (C) in situ determination of the redox state of CFoCF1 by labeling thiol groups with a maleimide reagent followed by protein detection using western blotting. The redox state of CFoCF1 was determined by mobility shifts on non-reducing SDS-PAGE. This protocol provides a refined strategy for elucidating the regulatory mechanism controlling energy conversion by CFoCF1 under fluctuating photosynthetic conditions.
One-Step Affinity Purification of MarathonRT Reverse Transcriptase for RNA Sequencing Applications
Transfer RNAs (tRNAs) are important regulators of translation and cellular function. Several high-throughput sequencing methods have been developed to quantitatively analyze tRNA isoacceptors in cells. However, the strong secondary structures and extensive post-transcriptional modification of most tRNA molecules present significant challenges for many reverse transcriptases, negatively impacting sequencing library preparation and causing quantification biases. Currently, the field utilizes processive next-generation reverse transcriptases (ngRTs), such as Induro (New England Biolabs) and UltraMarathonRT (RNAConnect), to address these issues. Despite being used in multiple protocols, these commercial products face little competition and remain costly. However, non-commercial alternatives, such as the original MarathonRT (MRT), are available from gene repositories. MRT is a next-generation reverse transcriptase derived from the Eubacterium rectale group II intron maturase, which can read through RNA secondary structures and chemical modifications. Here, we present a simplified expression and purification protocol for producing highly active MRT that is stable over 1 year. This cost-effective protocol yields a heterogeneous protein preparation with no discernible competing enzymatic activities; it mitigates previously reported precipitation issues, saving one day of laboratory work and eliminating two chromatography-based purification steps. Moreover, the use of the resulting protein preparation has been verified in the mim-tRNAseq pipeline, where it was shown to perform equally to the commercial alternatives Induro and UltraMarathonRT. In addition, we have developed a simple and cost-effective assay for measuring the enzymatic activity of MRT, allowing for batch comparison.
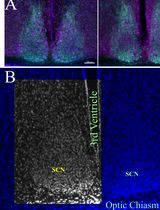
Histomorphometrical Analyses of the Mouse Suprachiasmatic Nucleus
The mammalian central circadian clock resides in the suprachiasmatic nucleus (SCN) of the hypothalamus in the brain and is responsible for coordinating daily rhythms of biological processes spanning from gene expression to behavior. Light, the primary environmental zeitgeber, entrains the SCN via melanopsin-expressing intrinsically photosensitive retinal ganglion cells that project through the retino-hypothalamic tract. Altered circadian rhythms are common in individuals diagnosed with neurodevelopmental and neurodegenerative disorders, and often associated with structural alterations of the SCN and impaired retinal input; importantly, these anomalies can be recapitulated in animal models. Here, we describe step-by-step protocols for quantitative histomorphometrical analysis of the SCN and the assessment of retinal–SCN connectivity, previously used in mouse models of neurodevelopmental and neurodegenerative disorders. These include measurement of the SCN area, perimeter, height, and width using Nissl- or DAPI-stained coronal sections, as well as densitometric and plot profile analyses of cholera toxin β-subunit–labeled retinal projections using Axiovision or Fiji/ImageJ. The protocols incorporate standardized region-of-interest measurements by masked observers and consistent scaling procedures to enhance reproducibility. These methods provide a rigorous framework for detecting structural anomalies and connectivity defects in the circadian system and can be broadly applied to other experimental models of circadian dysfunction.
Using Single-Particle Fluorescence Microscopy to Quantify Substrate Binding of Peptidoglycan-Modification Enzymes
Peptidoglycan (PG), a network of glycan strands crosslinked by short peptides, is an essential and bacterial-specific structure that determines cell shape and protects cells from lysis. Understanding how bacteria assemble, maintain, and modify their PG not only addresses fundamental questions in cell biology but also provides a basis for developing strategies to treat bacterial infections. Although several in vitro methods, such as zymography, Remazol Brilliant Blue (RBB) assay, and LC-MS analyses, are available to quantify the activities of PG-modification enzymes, these approaches are not readily applicable in vivo. Here, we describe a single-particle tracking photo-activated localization microscopy (sptPALM)-based method to quantify the binding of enzymes to PG in vivo, which serves as a proxy for their enzymatic activities. Because the PG meshwork is relatively immobile, fluorescently tagged enzymes that transiently or stably bind it exhibit reduced mobility, reflected by lower diffusion coefficients. This approach provides sensitive, quantitative, and real-time insights into enzyme behavior in vivo under diverse physiological conditions or genetic backgrounds. The protocol is particularly valuable for investigating PG-modification enzymes that are essential or functionally redundant, which are often difficult to analyze using traditional genetic methods.
PEPTERGENT: A Peptide-Based Reagent for Detergent-Free Extraction of Membrane Proteins and Purification of Membrane Proteomes
Peptergent is a novel class of amphipathic peptides that enables detergent-free extraction of membrane proteins (MPs) from lipid bilayers. This reagent self-assembles around hydrophobic transmembrane regions, forming stable, water-soluble complexes that can be isolated directly from biological membranes. Peptergent therefore bypasses the limitations imposed by traditional detergents, which often destabilize protein assemblies. Since detergents are completely avoided, MPs are directly amenable to structural and mass spectrometry (MS) analysis, thereby addressing their persistent underrepresentation in proteomic datasets and improving their accessibility in drug-screening strategies. We present here a streamlined protocol for MPs extraction with the Peptergent PDET-1, followed by exchange into His-tagged Peptidiscs for Ni-NTA-based affinity purification. The method encompasses membrane isolation, peptide preparation, protein extraction, clarification, and MPs exchange from Peptergents to Peptidiscs. This workflow yields an enriched membrane proteome compatible with downstream LC-MS/MS analysis for improved identification of multi-pass MPs.
Isolation and Biophysical Characterization of Extracellular Vesicles Released by Myocytes
Extracellular vesicles (EVs) are lipid bilayer–enclosed vesicles released by diverse cell types and found in various body fluids. Because their composition and cargo dynamically respond to physiological and environmental cues, EVs hold promise both as biomarkers and as carriers for therapeutic delivery. Skeletal muscle functions as an endocrine organ, secreting myokines and EVs that modulate a wide range of cellular processes. The murine C2C12 cell line is a widely used in vitro model for investigating muscle biology. Here, we describe a protocol for isolating EVs from differentiated C2C12 myocytes. The isolated EVs are characterized and validated using western blotting, transmission electron microscopy (TEM), and dynamic light scattering (DLS) analysis. This workflow provides a robust platform for studying the molecular composition and functional roles of muscle-derived EVs.
3D Reconstruction of Mature Arabidopsis Ovules Using FIB-SEM to Study Filiform Apparatus Morphology
Volume electron microscopy based on serial sectioning allows for three-dimensional (3D) visualization and analysis of the internal structures of tissues, cells, and organelles. One such technique, focused ion beam (FIB) scanning electron microscopy (SEM), has the advantages of nanoscale sectioning and high z-resolution, but the disadvantage of limited volume processing. Because of this limitation, targeting localized objects by FIB-SEM is difficult. Here, we developed a FIB-SEM observation workflow that enables the analysis of the filiform apparatus of synergid cells enclosed in the Arabidopsis ovule. In this protocol, plant samples are stained, embedded, trimmed, and carbon-coated while maintaining their orientation within the tissue. Then, sequential observations are performed using Cut & See function of FIB-SEM, followed by image processing for 3D reconstruction. Utilization of multi-scanning and image cropping from high-resolution data helps to identify localized targets within plant tissue. The filiform apparatus, which is an invaginated cell wall structure of the synergid cells, shows distinct contrast in each image, allowing for segmentation using brightness-based binarization. Such segmentation avoids the need to manually trace complex structures and facilitates 3D reconstruction by volume electron microscopy.
An In Vitro A-431 Epithelial Cell Infection Model for Studying Fungal Pathogenicity and Immune Responses Associated With Vulvovaginal Candidiasis
Vulvovaginal candidiasis (VVC), also known as vaginal thrush, is an infection of the vulvovaginal mucosa caused by fungi of the Candida genus. Particularly for patients suffering from recurrent infection, the disease has a significant impact on their quality of life. The still unknown aspects of disease pathogenesis, as well as factors driving the development of infections and recurrence, represent a challenge for both clinical practitioners and patients. Mouse models and patient studies have suggested important roles of the microbiome, deployment of fungal pathogenicity mechanisms in the vagina, and dysregulated immune responses for VVC pathology. Dissecting their individual contributions can reveal specific processes associated with infection and may inspire novel therapeutic strategies. Epithelial in vitro infection models have been playing a key role in dissecting a crucial interaction during VVC, the invasion and infection of the vaginal mucosa. They have been instrumental in characterizing candidalysin as a fungal toxin that damages epithelial cells and elicits initial inflammatory responses to catalyze downstream inflammation. Moreover, they have also revealed potential protective immune pathways. Such a standardized epithelial cell infection model offers high versatility and compatibility with different downstream assays to link epithelial responses with other processes during VVC. This protocol describes a general A-431 vulvovaginal epithelial cell–Candida infection model in detail and provides several adaptations, such as live-cell imaging and mRNA silencing, as well as possible follow-up readouts, like the quantification of cytokine release, cytotoxicity, and neutrophil recruitment to study diverse processes relevant to VVC research.
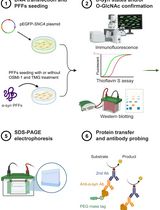
Chemoenzymatic Labeling Method for Detection of O-GlcNAcylated α-Synuclein Proteins by Western Blot
α-Synuclein (α-syn) aggregation has emerged as a key pathogenetic feature in several neurodegenerative disorders. The α-syn protein has various conformational strains, each with unique structural features that influence their cytotoxicity, propagation, and neuroinflammation. A post-translational modification known as O-GlcNAcylation has been found to influence the toxicity of α-syn and its propensity to aggregate. Difficulties in detecting and quantifying this modification are a major challenge to understanding its roles among the conformational forms of α-syn. We now describe a protocol for detecting O-GlcNAcylated α-syn that combines a click chemistry labeling approach and western blotting. This chemoenzymatic method involves the transfer of azido-modified galactose (GalNAz) from UDP-GalNAz to O-GlcNAcylated proteins, enabling their further functionalization with alkyne-containing polyethylene glycol of defined molecular weight. This protocol facilitates the determination of the glycosylation status of varying conformations of α-syn and their stoichiometric ratios.
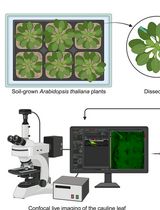
Analysis of Cauline Leaf Development in Arabidopsis thaliana Using Time-Lapse Confocal Microscopy
Understanding cellular growth dynamics in plants requires precise, long-term imaging of developing tissues. Cauline leaves are produced during the transition from vegetative to reproductive development and provide a useful system for studying how laminar organs diversify in form and function. While other laminar organs, such as rosette leaves and sepals, have been extensively studied, early cauline leaf development remains technically challenging to capture due to their concealed position, curved morphology, and the presence of dense trichomes. Here, we provide a complete pipeline for the dissection, confocal imaging, 2.5D segmentation, and image analysis of initiating cauline leaves in Arabidopsis thaliana. This method enables reproducible, high-resolution imaging of cauline leaves, supporting robust quantitative analysis of growth across developmental stages at cellular scale resolution.
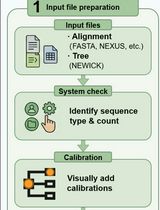
A Step-by-Step GUI-Based Protocol for Molecular Dating Analysis Using PhyloSuite v2
In current genomic research, molecular dating is challenged by both imperfect substitution modeling and analysis efficiency, as genome-scale datasets often exhibit substantial rate heterogeneity and complex patterns of sequence evolution, which can make divergence-time estimation sensitive to modeling assumptions and computational settings. Meanwhile, commonly used molecular dating workflows remain operationally demanding; preparing correctly formatted inputs, implementing model settings, configuring fossil calibrations, and performing basic diagnostics and visualization frequently require multiple tools and extensive manual steps, resulting in high hands-on time and avoidable operational errors. To facilitate the practical implementation of molecular dating analyses and lower the operational barrier for users, this protocol describes a GUI-based workflow in PhyloSuite v2 for molecular dating analysis. Using a dataset of fish nuclear genomes as an example, the tutorial covers multi-format data import, visual configuration of fossil calibrations, automatic selection and implementation of substitution models, automation of complex analytical procedures, and assessment of Markov chain Monte Carlo (MCMC) convergence, along with data visualization. Through this protocol, users can quickly master the full workflow—from input preparation and molecular dating to MCMC sample statistical assessment and timetree visualization—thus significantly enhancing the efficiency of molecular dating analysis and result verification.
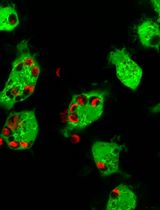
A Versatile In Vitro Quantitative Assay for Macrophage Efferocytosis in Diverse Research Applications
Macrophage efferocytosis is a previously unrecognized key pathogenic event, engulfing apoptotic targets, preventing inflammation and necrosis, and maintaining immune homeostasis. The phagocytic function can be disrupted by harmful factors and toxic substances. This protocol describes a versatile visualized in vitro method that can be used for the detection of general efferocytosis. This method is applicable to a wide range of research scenarios. As a representative application, it can be used to evaluate macrophage efferocytosis dysfunction in diseases linked to harmful exposures, including atherosclerosis, chronic inflammation, and malignant tumors. Among them, the detection of the effects of oxidized low-density lipoprotein (ox-LDL) and arsenite on macrophage efferocytosis capacity is an exemplary application of this protocol. Primary macrophages collected from mice were labeled with a cell-tracking dye and exposed to ox-LDL or arsenite, then co-cultured with apoptotic thymocytes or hepatocytes (labeled with another cell-tracking dye) for 2 h at a ratio of 5:1. Macrophage efferocytosis was visualized using a laser confocal microscope. The results indicate that arsenite impaired macrophage efferocytosis, leading to insufficient clearance of apoptotic thymocytes or hepatocytes. This method can be extended to subsequent studies, including those involving different types of phagocytes, apoptotic cell models, and research related to exposure to various factors.
Biolayer Interferometry (BLI) to Quantify RALF1–Pectin Interactions
Cellular function relies on a network of precisely regulated interactions among macromolecules such as proteins, peptides, carbohydrates, and nucleic acids. These molecular interactions regulate vital processes, including signaling, structural organization, and developmental patterning. Biolayer interferometry (BLI) is a label-free optical biosensing technique that enables real-time quantification of such interactions. This protocol describes how to use BLI to assess the binding affinity between a biotinylated plant peptide hormone (RALF1) and cell wall–derived oligogalacturonides (OG25–50) on the Octet RED96 platform. Streptavidin-coated biosensors are employed to immobilize the ligand, while analyte binding is monitored through wavelength shifts in the reflected light. The protocol includes detailed steps for sensor preparation, assay setup, software configuration, and kinetic data analysis. While optimized for plant peptide–matrix interactions, the method is broadly adaptable to other macromolecular systems across biological disciplines.
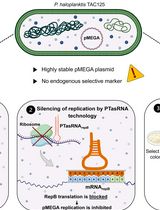
Plasmid Curing of Pseudoalteromonas haloplanktis TAC125 Using Homologous Recombination and PTasRNA Gene Silencing
Pseudoalteromonas haloplanktis TAC125 is a psychrophilic marine bacterium widely used to study cold adaptation and increasingly exploited as a non-conventional platform for biotechnological applications. The strain harbors the endogenous megaplasmid pMEGA (64.7 kb), whose presence may limit its exploitation as a cell factory, making its elimination advantageous to strain engineering. Traditional plasmid-curing approaches based on chemical and physical agents are often inefficient and unsuitable for stable endogenous replicons, such as pMEGA. Here, we describe a targeted protocol for pMEGA curing in P. haloplanktis TAC125 that combines homologous recombination with paired-termini antisense RNA (PTasRNA) gene silencing. First, a selectable marker cassette is inserted into pMEGA by homologous recombination using a suicide vector, enabling selective discrimination between plasmid-positive and plasmid-cured bacteria. Next, PTasRNA gene silencing technology is applied to target a gene essential for the replication of pMEGA, thereby transiently interfering with its replication and promoting its loss. This approach provides a specific method to cure a highly stable endogenous megaplasmid in a psychrophilic non-conventional bacterium, enabling improved functional studies and strain optimization, establishing a broadly applicable framework for targeted curing across diverse bacterial systems.
Simultaneous Immunofluorescence-Based In Situ mRNA Expression and Protein Detection in Bone Marrow Biopsy Samples
Fluorescence in situ hybridization (FISH) can be employed to study the expression and subcellular localization of nucleic acids by using labeled antisense strands that hybridize with the target RNA or DNA molecules. Likewise, immunofluorescence antibody staining (IF) takes advantage of the specific interaction between a fluorophore-labeled antibody and its corresponding antigen. This protocol reports the combination of RNA-FISH and IF antibody staining for simultaneous detection of both RNA transcripts and proteins of interest in routine formalin-fixed paraffin-embedded (FFPE) bone marrow biopsy samples. Herein, we provide a detailed description of the methodology that we have developed and optimized to study the spatial expression of two transcripts—TGFB1 and PDGFA1—in human hematopoietic (CD45+) and non-hematopoietic (CD271+) cells in the bone marrow of patients with acute lymphoblastic leukemia (ALL).
Using combined fluorescent in situ hybridization with Immunohistochemistry to co-localize mRNA in diverse neuronal cell types
Understanding gene expression within defined neuronal populations is essential for dissecting the cellular and molecular diversity of the brain. mRNA assays provide a direct readout of gene expression, capturing transcriptional changes that may precede or occur independently of protein abundance, whereas protein assays reflect the cumulative effects of translation, modification, and degradation. Moreover, in histological analysis, immunohistochemical protein detection results in visually diffuse labeling, which makes it difficult to quantitatively assess levels and locations of expression at high resolution. Here, we present a protocol that allows for mRNA detection in single neuronal cell types with a high degree of sensitivity and anatomical resolution. This protocol combines fluorescent in situ hybridization (FISH) with immunohistochemistry (IHC) on the same tissue section. Briefly, FISH is carried out by ACDBio RNAscope® fluorescent in situ hybridization technology, which involves processing the tissue sections, followed by signal amplification. This involves target retrieval, probe hybridization, and signal enhancement. Then, the tissue section is processed for IHC, which involves blocking nonspecific sites and incubation with primary antibodies, followed by development of a fluorescent signal with secondary antibodies. Typically, visual mRNA detection with FISH can be seen as individual puncta, whereas targeting the protein with an antibody results in filled cells or processes. The variation in staining pattern allows for the quantification of distinct mRNA transcripts within different neuronal populations, which renders co-localization analyses easy and efficient.
Electrophoretic Mobility Shift Assay (EMSA) for Assessing RNA–Protein Binding and Complex Formation Using Recombinant RNA-Binding Proteins and In Vitro–Transcribed RNA
Evaluating RNA–protein interactions is key to understanding post-transcriptional gene regulation. Electrophoretic mobility shift assays (EMSAs) remain a widely used technique to study these interactions, revealing information about binding affinities and binding modalities, including cooperativity and complex formation. Here, we detail, in a step-by-step protocol, how to perform EMSAs. We describe how to generate, purify, and quantitate 32P-radiolabeled RNA by in vitro transcription, as well as the expression and purification of recombinant RNA-binding proteins in E. coli using ELAV as an example. We then describe how to set up binding reactions using serial dilutions in a microtiter plate format of recombinant ELAV and in vitro–transcribed RNA and how to perform EMSAs using native low-crosslinked acrylamide gels, with detailed graphically supported instructions and troubleshooting guides.
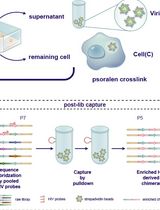
High-resolution mapping of RNA-RNA interactions across the HIV-1 genome with HicapR
The genomes of RNA viruses can fold into dynamic structures that regulate their own infection and immune evasion processes. Proximity ligation methods (e.g., SPLASH) enable genome-wide interaction mapping but lack specificity when dealing with low-abundance targets in complex samples. Here, we describe HiCapR, a protocol integrating in vivo psoralen crosslinking, RNA fragmentation, proximity ligation, and hybridization capture to specifically enrich viral RNA–RNA interactions. Captured libraries are sequenced, and chimeric reads are analyzed via a customized computational pipeline to generate constrained secondary structures. HiCapR generates high-resolution RNA interaction maps for viral genomes. We applied it to resolve the in vivo structure of the complete HIV-1 RNA genome, identifying functional domains, homodimers, and long-range interactions. The protocol's robustness has been previously validated on the SARS-CoV-2 genome. HiCapR combines proximity ligation with targeted enrichment, providing an efficient and specific tool for studying RNA architecture in viruses, with broad applications in virology and antiviral development.
Enhanced RNA-Seq Expression Profiling and Functional Enrichment in Non-model Organisms Using Custom Annotations
Functional enrichment analysis is essential for understanding the biological significance of differentially expressed genes. Commonly used tools such as g:Profiler, DAVID, and GOrilla are effective when applied to well-annotated model organisms. However, for non-model organisms, particularly for bacteria and other microorganisms, curated functional annotations are often scarce. In such cases, researchers often rely on homology-based approaches, using tools like BLAST to transfer annotations from closely related species. Although this strategy can yield some insights, it often introduces annotation errors and overlooks unique species-specific functions. To address this limitation, we present a user-friendly and adaptable method for creating custom annotation R packages using genomic data retrieved from NCBI. These packages can be directly imported as libraries into the R environment and are compatible with the clusterProfiler package, enabling effective gene ontology and pathway enrichment analysis. We demonstrate this approach by constructing an R annotation package for Mycobacterium tuberculosis H37Rv, as an example. The annotation package is then utilized to analyze differentially expressed genes from a subset of RNA-seq dataset (GSE292409), which investigates the transcriptional response of M. tuberculosis H37Rv to rifampicin treatment. The chosen dataset includes six samples, with three serving as untreated controls and three exposed to rifampicin for 1 h. Further, enrichment analysis was performed on genes to demonstrate changes in response to the treatment. This workflow provides a reliable and scalable solution for functional enrichment analysis in organisms with limited annotation resources. It also enhances the accuracy and biological relevance of gene expression interpretation in microbial genomics research.
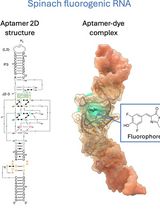
Visualizing diverse RNA functions in living cells with Spinach™ family of fluorogenic aptamers
RNA is now recognized as a highly diverse and dynamic class of molecules whose localization, processing, and turnover are central to cell function and disease. Live-cell RNA imaging is therefore essential for linking RNA behavior to mechanism. Existing approaches include quenched hybridization probes that directly target endogenous transcripts but face delivery and sequestration issues, protein-recruitment tags such as MS2/PP7 that add large payloads and can perturb localization or decay, and CRISPR–dCas13 imaging that requires substantial protein cargo and careful control of background and off-target effects. Here, we present a protocol for live-cell RNA imaging using the SpinachTM family of fluorogenic RNA aptamers. The method details the design and cloning of SpinachTM-tagged RNA constructs, selection and handling of cognate small-molecule fluorophores, expression in mammalian cell lines, dye loading, and image acquisition on standard fluorescence microscopes, followed by quantitative analysis of localization and dynamics. We include controls to verify aptamer expression and signal specificity, guidance for multiplexing with related variants (e.g., Broccoli, Corn, Squash, Beetroot), and troubleshooting for dye permeability and signal optimization. Application examples illustrate use in tracking cellular delivery of mRNA therapeutics, monitoring transcription and decay in response to perturbations, and the forming of toxic RNA aggregates. Compared with prior methods, SpinachTM tags are compact, genetically encodable, and fluorogenic, providing high-contrast imaging in both the nucleus and cytoplasm with single-vector simplicity and multiplexing capability. The protocol standardizes key steps to improve robustness and reproducibility across cell types and laboratories.
Enhancement of RNA Imaging Platforms by the Use of Peptide Nucleic Acid-Based Linkers
RNA imaging techniques enable researchers to monitor RNA localization, dynamics, and regulation in live or fixed cells. While the MS2-MCP system—comprising the MS2 RNA hairpin and its binding partner, the MS2 coat protein (MCP)—remains the most widely used approach, it relies on a tag containing multiple fluorescent proteins and has several limitations, including the potential to perturb RNA function due to the tag’s large mass. Alternative methods using small-molecule binding aptamers have been developed to address these challenges. This protocol describes the synthesis and characterization of RNA-targeting probes incorporating a peptide nucleic acid (PNA)-based linker within the cobalamin (Cbl)-based probe of the Riboglow platform. Characterization in vitro involves a fluorescence turn-on assay to determine binding affinity (KD) and selective 2′-hydroxyl acylation analyzed by primer extension (SHAPE) footprinting analysis to assess RNA-probe interactions at a single nucleotide resolution. To show the advancement of PNA probes in live cells, we present a detailed approach to perform both stress granule (SG) and U-body assays. By combining sequence-specific hybridization with structure-based recognition, our approach enhances probe affinity and specificity while minimizing disruption to native RNA behavior, offering a robust alternative to protein-based RNA imaging systems.
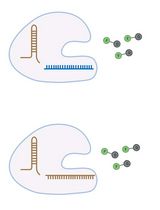
Amplification-Free Detection of Highly Structured RNA Molecules Using SCas12aV2
The CRISPR/Cas12a system has revolutionized molecular diagnostics; however, conventional Cas12a-based methods for RNA detection typically require transcription and pre-amplification steps. Our group has recently developed a diagnostic technique known as the SCas12a assay, which combines Cas12a with a split crRNA, achieving amplification-free detection of miRNA. However, this method still encounters challenges in accurately quantifying long RNA molecules with complex secondary structures. Here, we report an enhanced version termed SCas12aV2 (split-crRNA Cas12a version 2 system), which enables direct detection of RNA molecules without sequence limitation while demonstrating high specificity in single-nucleotide polymorphism (SNP) applications. We describe the general procedure for preparing the SCas12a system and its application in detecting RNA targets from clinical samples.
Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics