- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
aGPCR-HEK: A Stable High-Expression Inducible Mammalian Cell Expression System for Adhesion GPCR Structural Biology Applications
Published: Vol 16, Iss 5, Mar 5, 2026 DOI: 10.21769/BioProtoc.5621 Views: 95
Reviewed by: David PaulAshish KabraBeatrice Li

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Electrophysiological Evaluation of a Sciatic Nerve Degree III Injury Model in Rats
Linyu Chen [...] Na Dou
May 20, 2025 1822 Views

ZnCl2 Precipitation-Assisted Sample Preparation for Proteomic Analysis
Qiqing He [...] Fuchu He
Jul 20, 2025 2747 Views
Fluorescence Polarization-Based High-Throughput Screening Assay for Inhibitors Targeting Cathepsin L
Keyu Guo [...] Shuyi Si
Jul 20, 2025 2294 Views
Abstract
ADGRL4 is an adhesion G protein–coupled receptor (aGPCR) implicated in multiple tumours. In our experience, conventional insect cell-based baculovirus expression systems have not yielded sufficient correctly folded ADGRL4 protein for purification and cryo-electron microscopy (cryo-EM) analysis. Here, we describe aGPCR-HEK, a six-week protocol that establishes stable tetracycline-inducible mammalian HEK293S GnTI- TetR cell lines expressing N-terminally HA- and GFP-tagged aGPCRs. The method comprises lentiviral production in Lenti-X 293T cells, transduction of target adherent HEK293S GnTI- TetR cells, flow cytometry enrichment of uninduced GFP-positive cells displaying leaky expression, adaptation to suspension culture, and large-scale tetracycline induction and harvesting of cells for downstream purification and cryo-EM. The system yields reproducible, milligram-scale quantities of folded aGPCR suitable for structural and biochemical studies.
Key features
• Establishes a stable, tetracycline-inducible HEK293S GnTI- TetR expression system in adherent or suspension cells for high-level expression of N-terminally HA- and GFP-tagged aGPCRs.
• Uses lentiviral transduction for efficient aGPCR genomic integration and employs a tetracycline-repressor enabling high-level expression following tetracycline induction.
• Uses flow cytometry enrichment of leaky GFP-positive cells to enhance inducibility and yield.
• Provides suspension culture adaptation and large-scale tetracycline induction for structural biology-grade protein production suitable for purification and cryo-EM analysis.
Keywords: Adhesion GPCR (aGPCR)Graphical overview
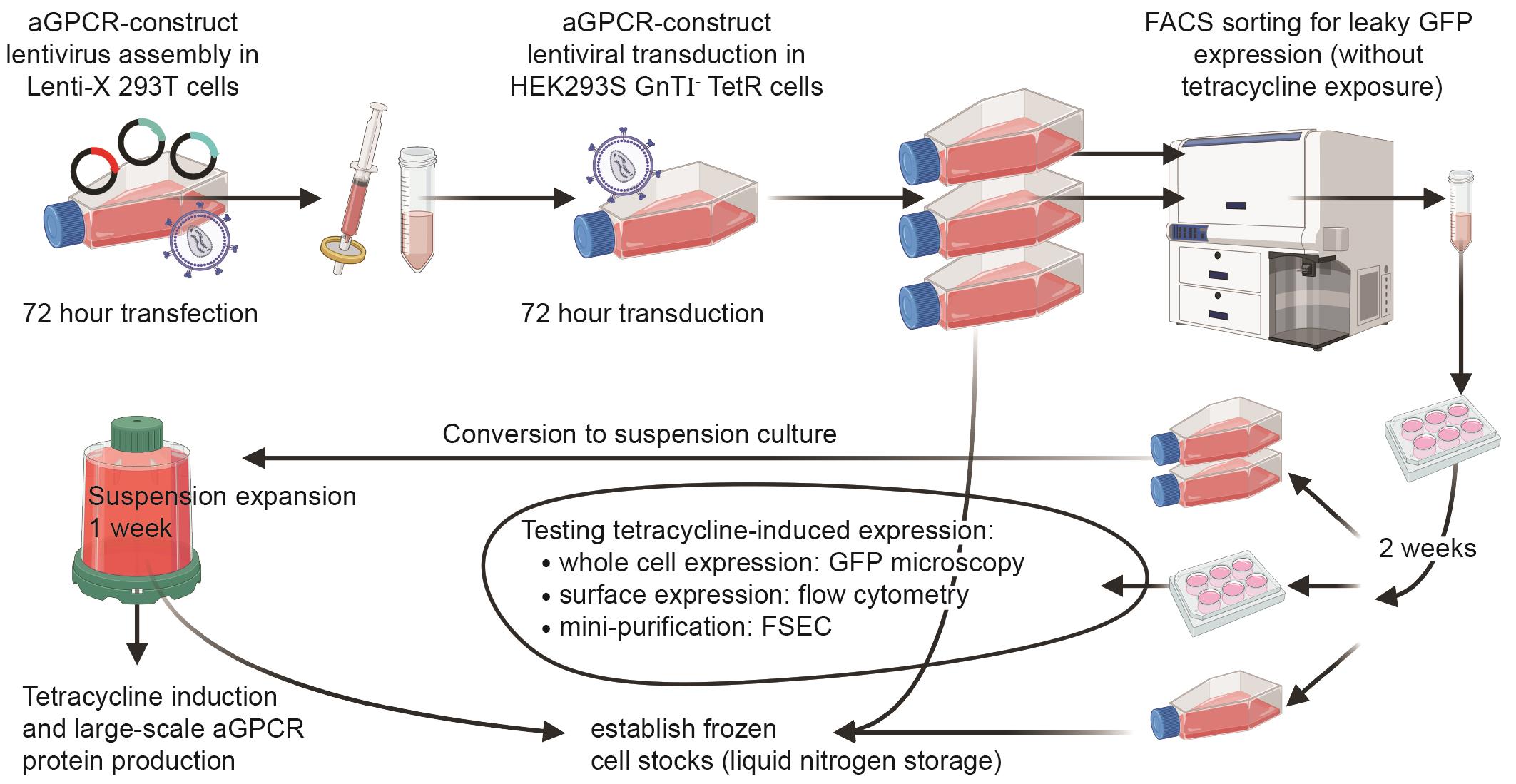
Graphical overview of the adhesion G protein–coupled receptor (aGPCR)-HEK protocol
Background
The adhesion G protein–coupled receptor (aGPCR) family comprises 32 receptors in humans that are involved in a range of important physiological and pathological processes [1]. ADGRL4 is an aGPCR that plays an important role in endothelial biology, is implicated in multiple tumour types, and is a promising therapeutic target [2–22]. Recently, we determined its high-resolution active-state structure using cryo-electron microscopy (cryo-EM) and determined that it is activated by a tethered agonist binding its orthosteric binding pocket [23]. To date, the majority of aGPCR structures determined have been expressed for large-scale protein production in insect cell-based baculovirus expression systems [24–33]. Initially, we attempted to express ADGRL4 in Sf9 (Spodoptera frugiperda) and Tni (Trichoplusia ni) insect cells using the Bac-to-Bac Baculovirus Expression System (Gibco, catalog number: 10359016). Although we were able to express ADGRL4 using this system, it did not provide sufficient high-quality folded protein for purification and cryo-EM microscopy. We thus switched to mammalian expression and developed the aGPCR-HEK protocol. This was successfully employed to express correctly folded ADGRL4 protein at a large scale for structural studies, allowing us to determine the first high-resolution structure of ADGRL4 [23].
Here, we present aGPCR-HEK, a six-week protocol that establishes a stable tetracycline-inducible mammalian HEK293S GnT1- TetR cell line expressing an N-terminally HA- and GFP-tagged aGPCR. This method consists of (i) lentiviral production in Lenti-X 293T cells, (ii) transduction of target adherent HEK293S GnT1- TetR cells, (iii) flow cytometry–positive selection of uninduced GFP-positive HEK293S GnTI- TetR cells (selecting for cells exhibiting leaky low expression of the insert in the absence of tetracycline), (iv) adaptation from adherent to suspension cell culture, and (v) large-scale tetracycline or doxycycline induction for downstream protein purification and cryo-EM. A stable inducible cell line offers superior expression and scalability over transient transfection and minimises the formation of intracellular, misfolded, inactive receptor [34]. Using the aGPCR-HEK protocol, we purified approximately 250 μg of high-quality CTF2B (the unstable active-state version of ADGRL4) from 2 L of HEK293S GnTI- TetR cells [23]. CTF2B denotes the constitutively active C-terminal fragment version of ADGRL4 (residues T407-R690) fused to a C-terminally tethered mini-Gq (βγ-binding “version B”), which stabilises the active-state receptor–G protein complex for downstream purification and cryo-EM. This yield was sufficient for determining the active-state high-resolution structure. The yield is significantly higher when expressing full-length inactive aGPCR receptors, as they are more stable. The ADGRL4 purification protocol for cryo-EM is described in a companion Bio-protocol manuscript [35].
The second-generation lentiviral system used in this method is derived from HIV-1 and has been engineered to be replication-incompetent [36,37]. It comprises three plasmids: (i) a packaging plasmid (psPAX2), (ii) an envelope plasmid (pMD2.G), and (iii) a transfer plasmid. For the transfer plasmid, we modified the pHR-CMV-TetO2_3C-Twin-Strep transfer plasmid [38] by deleting its 3C and Twin Strep sequences.
Lenti-X 293T cells were chosen for lentivirus production due to their high-level expression of lentivirus. Viral transduction was then performed on HEK293S GnTI- TetR cells. HEK293S GnTI- TetR cells are HEK293S cells lacking the N-acetylglucosaminyltransferase I (GnTI) enzyme encoded by MGAT1 [39,40]. GnTI performs the first step in complex N-glycan processing within the Golgi and, without its glycans, cannot be converted into complex, heterogeneous sugar chains. This ensures that GnTI- cells produce glycoproteins with minimal glycan heterogeneity. The HEK293S GnTI- cell line and its later TetR inducible derivative cell line were originally developed for X-ray crystallography because the presence of complex heterogeneous N-glycans hinders crystal packing and X-ray diffraction [39,40]. Improved homogeneity of N-glycosylated proteins expressed in GnTI- cells makes interpretation of western blots easier because discrete bands are observed rather than a smear produced in wild-type HEK293S cells.
During its original development, the HEK293S GnTI- TetR cell line was stably transfected under blasticidin selection with a pcDNA6/TR plasmid [39,40], integrating a tetracycline repressor (TetR) [41] under constitutive CMV promoter expression as well as a blasticidin resistance gene. HEK293S GnTI - TetR cells thus require culture in medium supplemented with 5 μg/mL blasticidin. This protocol is also applicable to using the T-REx system (Invitrogen, catalog number: K102001) with full-length N-glycans.
In the absence of tetracycline or doxycycline, TetR binds to tetracycline operator (TetO) sequences present within the tetracycline response element (TRE) downstream of the transgene’s CMV promoter. This represses transcription of the aGPCR transgene. When tetracycline or doxycycline is present, they bind to TetR, causing a conformational change that prevents TetR from binding TetO sites. This releases the promoter from repression and permits robust transgene expression. A small subset of transduced HEK293S GnTI- TetR cells displays low-level leaky transgene expression in the absence of tetracycline or doxycycline [42]. Because these cells typically produce higher transgene levels upon induction than non-leaky cells, they are preferentially selected in the aGPCR-HEK protocol by GFP-based fluorescence-activated cell sorting (FACS) [43,44].
Finally, all procedures involving lentivirus must be conducted within a Class II microbiological safety cabinet according to institutional and national biosafety guidelines. Personnel must wear appropriate protective equipment (laboratory coat, gloves, eye protection), and work surfaces and equipment must be decontaminated with a virucidal disinfectant (e.g., Virkon) after handling lentiviral material. All liquids and materials potentially contaminated with lentivirus should be inactivated and disposed of according to institutional and national biosafety rules.
Materials and reagents
Biological materials
1. Lenti-X 293T cell line (Takara, catalog number: 632180); growth medium: DMEM/F-12 with 10% tetracycline-free FBS
2. HEK293S GnTI- TetR cell line (Dr Philip J Reeves [39,40], University of Essex, UK); growth medium: DMEM with 10% tetracycline-free FBS and 5 μg/mL blasticidin.
Note: The aGPCR-HEK protocol is also applicable when using the T-REx expression system with full-length N-glycans (Invitrogen, catalog number: K102001); growth medium: DMEM with 10% tetracycline-free FBS and 5 μg/mL blasticidin.
3. Lentivirus packaging plasmids:
a. psPAX2 packaging plasmid (Addgene, catalog number: 12260)
b. pMD2.G envelope plasmid (Addgene, catalog number: 12259)
c. pHR-CMV-TetO2 transfer plasmid; generated by deleting the 3C-Twin-Strep sequences (bases 8294-8419) from the pHR-CMV-TetO2_3C-Twin-Strep plasmid [38] (Addgene, catalog number: 113883)
4. Adhesion GPCR construct:
a. ADGRL4 CTF2B (C-terminal fragment version 2B [23]: ADGRL4 with N-terminal HA-, GFP-, and purification tags) in pcDNA3.1 plasmid; the CTF2B ADGRL4 sequence was derived from the ADGRL4 Uniprot reference sequence (Q9HBW9) and comprises amino acids T407-R690
Reagents
1. Dulbecco's modified Eagle medium (DMEM) with GlutaMAX supplement, 500 mL (Gibco, catalog number: 31966-021); store at 4 °C
2. DMEM/F-12 medium, 500 mL (Gibco, catalog number: 11330-032); store at 4 °C
3. FreeStyle 293 expression medium, 1,000 mL (Gibco, catalog number: 12338-018); store at 4 °C
4. Fetal bovine serum (FBS) tetracycline-free, 500 mL (Biosera, catalog number: FB-1001T/500); store at -20 °C
5. Phosphate-buffered saline (PBS), pH 7.4, 500 mL (Gibco, catalog number: 10010015)
6. TrypLE Express enzyme, 100 mL (Gibco, catalog number: 12604013); store at 4 °C
7. Lipofectamine LTX reagent with Plus reagent, 0.3 mL (Invitrogen, catalog number: 15338030); store at 4 °C
8. Opti-MEM reduced-serum medium, 500 mL (Gibco, catalog number: 11058021); store at 4 °C
9. Blasticidin S HCl (10 mg/mL stock), 10 mL (Thermo Scientific, catalog number: A1113903); store at -20 °C
10. Polybrene (10 mg/mL stock), 1 mL (Sigma-Aldrich, catalog number: TR-1003-G); store at -20 °C
11. Lenti-X GoStix Plus, 20 tests (Takara, catalog number: 631280)
12. Dimethyl sulfoxide (DMSO), 100 mL (Sigma-Aldrich, catalog number: 34869)
13. Virkon disinfectant virucidal tablets (Day Impex Ltd, catalog number: 330013)
14. Tetracycline 98%–102% (HPLC) 25 g (Sigma-Aldrich, catalog number: T3258-25G); store at -20 °C
15. Absolute ethanol, 2.5 L (Sigma-Aldrich, catalog number: 32221-2.5L-M)
16. Trypan blue solution 0.4%, 100 mL, 2.5 L (Gibo, catalog number: 15250061)
17. Anti-HA monoclonal antibody conjugated to allophycocyanin (APC), 200 μL (Miltenyi Biotec, catalog number: 130-123-553); store at 4 °C
Solutions
1. DMEM supplemented with 10% tetracycline-free FBS (see Recipes)
2. DMEM supplemented with 10% tetracycline-free FBS and 5 μg/mL blasticidin (see Recipes)
3. DMEM/F-12 supplemented with 10% tetracycline-free FBS (see Recipes)
4. FreeStyle 293 supplemented with 1% tetracycline-free FBS and 5 μg/mL blasticidin (see Recipes)
5. Flow cytometry buffer (see Recipes)
6. FACS catch buffer (see Recipes)
7. Cryopreservation medium (see Recipes)
8. 1% Virkon disinfectant solution (see Recipes)
9. Tetracycline solution (see Recipes)
Recipes
Note: All FBS used should be tetracycline-free. All recipes should be prepared under sterile conditions except for Virkon disinfectant solution.
1. DMEM supplemented with 10% tetracycline-free FBS
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| DMEM medium | 90% | 450 mL |
| Tetracycline-free FBS | 10% | 50 mL |
| Total | n/a | 500 mL |
Store at 4 °C.
2. DMEM supplemented with 10% tetracycline-free FBS and blasticidin
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| DMEM medium | 90% | 449.75 mL |
| Tetracycline-free FBS | 10% | 50 mL |
| Blasticidin (10 mg/mL stock) | 5 μg/mL | 250 μL |
| Total | n/a | 500 mL |
Store at 4 °C.
3. DMEM/F-12 supplemented with 10% tetracycline-free FBS
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| DMEM/F-12 medium | 90% | 450 mL |
| Tetracycline-free FBS | 10% | 50 mL |
| Total | n/a | 500 mL |
Store at 4 °C.
4. FreeStyle 293 supplemented with 1% tetracycline-free FBS and blasticidin
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| FreeStyle 293 medium | 99% | 989.5 mL |
| Tetracycline-free FBS | 1% | 10 mL |
| Blasticidin (10 mg/mL stock) | 5 μg/mL | 500 μL |
| Total | n/a | 1,000 mL |
Store at 4 °C.
5. Flow cytometry buffer
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| PBS | 98% | 19.6 mL |
| Tetracycline-free FBS | 2% | 0.4 mL |
| Total | n/a | 20 mL |
Store at 4 °C. Keep on ice when in use.
6. FACS catch buffer
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| DMEM | 90% | 17.99 mL |
| Tetracycline-free FBS | 10% | 2 mL |
| Blasticidin (10 mg/mL stock) | 5 μg/mL | 10 μL |
| Total | n/a | 20 mL |
Store at 4 °C. Keep on ice when in use.
7. Cryopreservation buffer
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| DMEM | 70% | 6.995 mL |
| Tetracycline-free FBS | 20% | 2 mL |
| Blasticidin (10 mg/mL stock) | 5 μg/mL | 5 μL |
| DMSO | 10% | 1 mL |
| Total | n/a | 10 mL |
Make up fresh before use.
8. 1% Virkon disinfectant solution
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| Tap water | 99% | to a total volume of 500 mL |
| Volume of lentivirus-rich discarded cultured medium | × mL | |
| Virkon disinfectant virucidal tablet | 1% | Single 5 g tablet |
| Total | n/a | 500 mL |
9. Tetracycline stock solution
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| Tetracycline powder | 1 mg/mL | 20 mg |
| Absolute ethanol | ~99.9% | 20 mL |
| Total | n/a | 20 mL |
a. Weigh 20 mg of tetracycline powder (~100% HPLC purity) on an analytical balance using a weighing boat or paper.
b. Add the measured tetracycline to ~15 mL of absolute ethanol (≥95%) in a foil-wrapped 50 mL centrifuge tube. Gently mix until dissolved and then bring to 20 mL with further absolute ethanol.
c. To ensure sterility, filter through a 0.22 μm PES filter and decant into a new foil-wrapped sterile 50 mL centrifuge tube.
d. Under sterile conditions, aliquot as appropriate into foil-covered tubes and protect from light (tetracycline is light sensitive). Store at -20 °C for up to a year.
e. When inducing HEK293S GnTI- TetR cells, the final concentration of tetracycline should be 1 μg/mL.
Laboratory supplies
1. Fisherbrand sterile PES syringe filters, 0.45 μm pore size (Fisher Scientific, catalog number: 15216869)
2. Fisherbrand sterile PES syringe filters, 0.2 μm pore size (Fisher Scientific, catalog number: 15206869)
3. CellTrics 50 μm filters (CellTrics, catalog number: 04-004-2327)
4. Sarstedt sterile 5 mL polypropylene tube (L × Ø): 75 × 12 mm (Sarstedt, catalog number: 55.526.006)
5. Falcon 5 mL sterile round-bottom polypropylene test tube with snap cap (Corning, catalog number: 352063)
6. 50 mL sterile Luer Lock syringes, pack of 25 (Terumo, catalog number: 8SS50L1)
7. 96-well round (U) bottom plates (Thermo Scientific, catalog number: 163320)
8. 50 mL centrifuge tubes (Corning, catalog number: CLS430828-100EA)
9. 6-well CytoOne plates, TC-treated (Starlab, catalog number: CC7682-7506)
10. 24-well CytoOne plates, TC-treated (Starlab, catalog number: CC7682-7524)
11. Falcon T75 cm2 rectangular canted neck cell culture flasks with vented caps (Corning, catalog number: 353136)
12. Falcon T175 cm2 rectangular straight neck cell culture flasks with vented caps (Corning, catalog number: 353112)
13. Corning 500 mL polycarbonate Erlenmeyer flasks with flat cap (Corning, catalog number: 430422)
14. Corning 125 mL polycarbonate Erlenmeyer flask with flat cap (Corning, catalog number: 430421)
15. Corning 850 cm2 polystyrene roller bottle with easy grip cap (Corning, catalog number: 430849)
16. Corning 1.2 mL cryogenic vial (Corning, catalog number: 430487)
17. Mr. Frosty freezing container (Thermo Scientific, catalog number: 51000001)
Equipment
1. Class II biological safety cabinet: LaboGene Mars 1200 Class II Biological Safety Cabinet (LaboGene, catalog number: Mars 1200)
2. Stationary incubator: Sanyo MCO-18AIC CO2 incubator (Sanyo, catalog number: MCO-18AIC)
3. Shaking incubator: Infors HT Multitron Pro Triple Stacked Incubator Shaker (Infors, catalog number: Multitron Pro); shaking incubator (37 °C; 8% CO2; >95% humidity, shaking speed of 125 rpm)
4. Temperature-controlled 1.5 mL tube centrifuge: Eppendorf 5418 R Centrifuge (Eppendorf, catalog number: 5418 R)
5. Temperature-controlled 15–50 mL tube swinging-bucket centrifuge: Beckman Coulter Allegra X-12R Refrigerated Centrifuge (Beckman Coulter, catalog number: X-12R)
6. Temperature-controlled floor-standing centrifuge for 1 L bottles: Beckman Coulter Avanti JXN-26 Refrigerated Centrifuge (Beckman Coulter, catalog number: JXN-26)
7. Microscope (brightfield and fluorescence): Life Technologies EVOS FL microscope (Life Technologies, catalog number: AMF4300)
8. FACs cell sorter: Invitrogen Bigfoot Cell Sorter (Invitrogen, catalog number: PL00304)
9. Flow cytometer (Sony, model: ID7000 Spectral Cell Analyzer)
10. FSEC system: Shimadzu HPLC system comprising the following components: Shimadzu Degasser Module (DGU-20A), Shimadzu Solvent Delivery Module (LC-20AD), Shimadzu Autosampler Module (SIL-20AC), Shimadzu Column Oven Module (CTO-20AC), Shimadzu UV-detector Module (SPD-20A), Shimadzu Fluorescence Detector Module (RF-20A), Shimadzu Fraction Collector Module (FRC-40); HPLC guard column: Agilent Bio SEC-5 guard column (300 Å, 7.8 × 50 mm, 5 μm) (Agilent, catalog number: 5190-2530), HPLC column: Agilent Bio SEC-5 column (300 Å 4.6 × 300 mm 5 μm) (Agilent, catalog number: 5190-2528)
Software and datasets
1. Microsoft Excel (Microsoft, version 16.101.3 (25100321)) for calculations
2. FlowJo (Becton Dickinson & Company, version 10.10.0) for flow cytometry data analysis
Procedure
The key steps in the aGPCR-HEK protocol are summarised in Table 1. This protocol can be completed within six weeks.
Table 1. aGPCR-HEK protocol timeline, reporting key steps
| Timeline | Lenti-X 293T cells | HEK293S GnTI- TetR cells |
|---|---|---|
| Day 1 | Seed cells for transfection | |
| Day 2 | Transfect with lentiviral plasmids | |
| Day 3 | 24 h after transfection: change medium | |
| Day 4 | 48 h after transfection: check cell viability | Seed cells for transduction; seed control flask |
| Day 5 | 72 h after transfection: collect supernatant, filter, and test medium for lentiviral particles | Add polybrene to the collected lentiviral supernatant and transduce HEK293S GnTI- TetR cells |
| Day 6 | 24 h after transduction: change medium | |
| Day 7 | 48 h after transduction: check cell viability | |
| Day 8 | 72 h after transduction: split transduced and control cells (1:3) | |
| Day 8-11 | Expansion | |
| Day 12 | FACS sorting using leaky GFP expression | |
| Day 13–27 | Post-FACS expansion; induction trial | |
| Day 28–35 | Conversion to suspension culture and expansion | |
| Day 36 | Large-scale tetracycline induction | |
| Day 37 | Harvesting for aGPCR structural studies |
Biosafety considerations:
Perform all lentiviral steps in a Class II microbiological safety cabinet according to institutional and national biosafety guidelines. Dispose of lentivirus-contaminated waste in accordance with institutional biosafety rules and decontaminate with Virkon at a final working concentration of 1% (or alternative viricidal disinfectant) before disposal.
A. Plasmid preparation
1. The HIV-1-derived, replication-incompetent, second-generation lentiviral system used in this method comprises three plasmids: (i) a packaging plasmid (psPAX2), (ii) an envelope plasmid (pMD2.G), and (iii) a transfer plasmid (pHR-CMV-TetO2).
2. Ensure that you have correctly cloned the aGPCR gene insert into the pHR-CMV-TetO2 transfer plasmid. For this protocol, we subcloned the active-state C-terminal fragment (CTF) version of ADGRL4 [derived from ADGRL4’s Uniprot reference sequence (Q9HBW9) and corresponding to amino acids T407-R690] from its pcDNA3.1 plasmid to the pHR-CMV-TetO2 transfer plasmid using Gibson Assembly [45], generating the pHR-CMV-TetO2-ADGRL4 transfer plasmid.
Critical: The subcloned aGPCR gene should contain N-terminal HA and GFP tagging; these two tags are critical for the establishment of the aGPCR-HEK cell line. Tags should be placed C-terminal to the signal peptide and can be followed as required by purification tags and a protease cleavage site for downstream protein purification.
3. Verify the sequence integrity of all plasmids by Sanger sequencing. To obtain high-purity DNA stocks, perform maxi-preparations of each plasmid. For ease of use, we recommend keeping all plasmid stocks at the same concentration, for example, 500 ng/μL.
B. Lentiviral production in Lenti-X 293T cells (days 1–5)
The aGPCR-HEK protocol provides instructions for generating one lentivirus and one inducible aGPCR-expressing stable cell line. To generate multiple lentiviruses and stable cell lines simultaneously, scale parameters as required. Lentiviruses are generated over 72 h in Lenti-X 293T cells, after which viral supernatant is harvested, filtered, mixed with polybrene, and then applied to target HEK293S GnTI- TetR cells.
Critical: All lentiviral work must be performed in a Class II microbiological safety cabinet.
Day 1: Seed Lenti-X 293T cells
1. Maintain healthy, low-passage, mycoplasma-free adherent Lenti-X 293T cells in DMEM/F-12 medium (supplemented with 10% tetracycline-free FBS) under standard tissue culture conditions (temperature 37 °C; humidified atmosphere of 5% CO2 and 95% filtered atmospheric air). Passage cells at least twice after thawing before use. For dissociating adherent cells, we use TrypLE Express Enzyme.
2. Twenty-four hours before transfection, seed a T175 flask with 15 × 106 Lenti-X 293T cells in 30 mL of DMEM/F-12 (supplemented with 10% tetracycline-free FBS). This should reach ~70% confluence after 24 h.
Day 2: Transfecting Lenti-X 293T cells
3. Keep Lipofectamine LTX reagent and Plus reagent on ice. Prewarm the following to 37 °C before use: Opti-MEM and DMEM/F-12 (supplemented with 10% tetracycline-free FBS).
Critical: Before use, ensure that all materials designated for prewarming in this protocol have fully reached 37 °C. Warming times will vary depending on the volume of material to be warmed and its initial temperature.
4. Verify that Lenti-X 293T cells are ~70% confluent with healthy morphology before proceeding with transfection.
Critical: Do not proceed with transfection if cells are under- or over-confluent or if there are signs of contamination or significant cell death.
5. Transfect the T175 flask of Lenti-X 293T cells with 45.6 μg of total plasmid DNA in a 4:3:1 mass ratio between the transfer plasmid (pHR-CMV-TetO2-ADGRL4), packaging plasmid (psPAX2), and envelope plasmid (pMD2.G). Table 2 below lists the amounts of all reagents required.
Table 2. Transfection reagent amounts for a single T175 flask
| Flask size | T175 (single flask) |
|---|---|
| Replacement medium before transfection | 36.46 mL |
| Total DNA required | 45.6 μg of total DNA Breakdown (4:3:1 ratio): • 22.8 μg of pHR-CMV-TetO2-ADGRL4 transfer plasmid (containing ADGRL4) • 17.1 μg of psPAX2 packaging plasmid • 5.7 μg of pMD2.G envelope plasmid |
| DNA dilution | 45.6 μg of DNA adjusted to a final volume of 9.12 mL with Opti-MEM |
| Volume Lipofectamine LTX required | 91.2 μL |
| Plus reagent | 45.6 μL [corresponding to an equal volume (μL) of Plus reagent to the mass (μg) of plasmid added] |
a. Dilute 45.6 μg of total plasmid DNA in a 50 mL sterile centrifuge tube to a final volume of 9.12 mL in Opti-MEM. Label the tube as “transfection mixture.” Table 3 below lists the DNA and Opti-MEM amounts required for plasmids at a stock concentration of 500 ng/μL.
Table 3. Plasmid dilution volumes
| Plasmid concentration | Plasmid DNA needed | Volume required (μL) | |
| pHR-CMV-TetO2-ADGRL4 | 500 ng/μL | 22.8 μg | 45.6 μL |
| psPAX2 | 500 ng/μL | 17.1 μg | 34.2 μL |
| pMD2.G | 500 ng/μL | 5.7 μg | 11.4 μL |
| Total DNA: 45.6 μg | Total DNA volume: 91.2 μL | ||
| Opti-MEM volume: 9,028.8 μL | |||
| Total volume: 9,120 μL | |||
b. Add 45.6 μL of Plus reagent, which corresponds to a 1:1 ratio of μL of Plus reagent per μg of total plasmid DNA used (45.6 μg). Gently mix the Plus reagent vial before use, then add it and incubate at room temperature for 15 min. The total volume in the transfection mixture tube will now be 9120 μL + 45.6 μL = 9165.6 μL.
c. After the 15-min incubation, add 91.2 μL of Lipofectamine LTX to the transfection mixture tube and mix very gently by slow side-to-side rocking of the 50 mL tube (no vortexing or pipette mixing). Incubate for 30 min at room temperature to allow for complex formation. The total volume is 9,165.6 μL + 91.2 μL = 9,256.8 μL.
Caution: Do not shake or mix vigorously.
d. During the 30-min Lipofectamine incubation, remove the T175 Lenti-X 293T flask from the incubator. Under sterile conditions, replace the 30 mL of old medium with 36.46 mL of fresh DMEM/F-12 (supplemented with 10% tetracycline-free FBS) warmed to 37 °C using a pipette. Return the flask to the incubator until incubation is complete.
Caution: When changing medium, avoid detaching adherent cells from the base of the flask.
e. After the 30-min incubation, remove the T175 flask from the incubator. Under sterile conditions, add the 9,256.8 μL transfection mixture dropwise to the medium in the flask. After adding the transfection mixture, the total media volume in the flask will be 36.46 mL + 9.2568 mL = 45.7168 mL.
f. Gently rock the flask to evenly distribute the complexes. Incubate for 24 h at 37 °C with 5% CO2 and 95% filtered atmospheric air.
Day 3: Medium change (24 h after transfection)
6. Prewarm DMEM (supplemented with 10% tetracycline-free FBS) to 37 °C before use. Prepare 450 mL of Virkon disinfectant solution by dissolving one 5 g Virkon virucidal tablet in 450 mL of water in a large beaker. The addition of ~45 mL of discarded cultured medium results in a final Virkon concentration of 1%.
7. Replace the medium within the transfected Lenti-X 293T T175 flask with 30 mL of DMEM (supplemented with 10% tetracycline-free FBS) to remove toxic transfection reagents. Treat discarded medium and lentivirus-exposed pipettes with Virkon at a final concentration of 1% before disposal
Caution: Do not replace medium with DMEM/F-12.
Day 5: Collect lentiviral supernatant (72 h after transfection)
8. Prepare 500 mL of 1% Virkon disinfectant solution in a large beaker.
Critical: All labware that contacts lentiviral supernatant must be disinfected before disposal.
9. Under sterile conditions, collect the lentiviral supernatant from the transfected Lenti-X 293T cells (72-h time point) and transfer to a sterile 50 mL centrifuge tube. The transfected Lenti-X 293T cells can be discarded after supernatant collection.
10. Filter the lentiviral supernatant through a 0.45 μm sterile polyethersulfone (PES) filter using a sterile 50 mL Luer Lock syringe. Avoid smaller pore sizes (which risk shearing viral particles) and do not use nitrocellulose filters. Because of cell debris from lentivirus production, filters may clog. Expect to use 2–3 filters to process the supernatant collected from a T175 flask.
Critical: Filter gently to prevent filter rupture. If resistance increases, switch to a fresh 0.45 μm PES filter.
Pause point: The filtered lentiviral supernatant may be stored at 4 °C for up to a week, though viral titre decreases after 48 h. If storing, do not add polybrene to the supernatant; add it only immediately before transduction. For longer periods (weeks to months), aliquot and freeze at -80 °C (do not snap-freeze in liquid nitrogen or use DMSO). Thaw at 37 °C for 10 min, then keep on ice and use immediately. Avoid repeated freeze-thaw cycles to prevent titre loss.
11. Determine the viral titre using the Takara Lenti-X GoStix Plus kit.
Note: The Lenti-X GoStix assay estimates viral particle levels rather than functional transducing units. Alternative functional titration approaches, such as limiting dilution–based transduction assays, may be used to determine transducing units per volume if required.
a. Pipette 20 μL of filtered lentiviral supernatant into the sample of the GoStix cassette.
b. Add 80 μL of GoStix chase buffer, then incubate at room temperature for 10 min.
c. After incubation, scan the cassette with a smartphone using the GoStix Plus app, which will automatically calculate the viral titre based on test band intensity.
Critical: Change into fresh gloves before handling the smartphone to prevent viral contamination.
d. If no virus is detected, repeat the test with a new cassette. If still negative, concentrate the collected supernatant using the Takara Lenti-X concentrator (Takara, catalog number: 631231) or another validated method, then repeat the GoStix assay. Persistent low detection indicates low viral yield, requiring a repeat transfection with adjusted plasmid DNA or Lipofectamine LTX ratios.
C. Lentiviral transduction of HEK293S GnTI- TetR cells (days 4–8) and initial expansion (days 9–11)
Day 4: Seed HEK293S GnTI- TetR cells
1. Maintain low-passage, mycoplasma-free adherent HEK293S GnTI- TetR cells in DMEM (supplemented with 10% tetracycline-free FBS and 5 μg/mL blasticidin) under standard tissue culture conditions. After thawing, ensure that cells have been grown to 100% confluence and have been passaged twice before seeding for lentiviral transduction.
Caution: Maintain cells for a maximum of 20 passages (approximately 8–10 weeks of continuous culture) to preserve TetR expression. The TetR gene, expressed constitutively under a CMV promoter, is prone to transcriptional silencing during extended culture. Always supplement the medium with blasticidin to prevent downregulation or epigenetic silencing of the integrated TetR and blasticidin-resistance gene cassette.
2. Seed two T175 flasks each with 15 × 106 HEK293S GnTI- TetR cells in 30 mL of DMEM (supplemented with 10% tetracycline-free FBS and 5 μg/mL blasticidin):
a. Flask 1: Cells to be transduced
b. Flask 2: Control cells (essential negative control for downstream flow cytometry)
Incubate for 24 h under standard tissue culture conditions to reach 70% confluence after 24 h.
Day 5: Lentiviral transduction of HEK293S GnTI- TetR cells
3. Prewarm DMEM (supplemented with 10% tetracycline-free FBS and 5 μg/mL blasticidin) to 37 °C before use. Prepare 500 mL of 1% Virkon disinfectant solution in a large beaker. Thaw 10 mg/mL polybrene (hexadimethrine bromide) and keep on ice.
4. Before lentiviral transduction, inspect seeded HEK293S GnTI- TetR cells under a microscope. Cells should be ~70% confluent and healthy in appearance.
Critical: Do not proceed if confluence is <50% or if cells show signs of contamination or significant cell death.
5. Prepare the transduction mixture:
a. In a sterile 50 mL centrifuge tube, combine the filtered lentiviral supernatant (typically ~40 mL after filtration) with 20 mL of fresh DMEM (supplemented with 10% tetracycline-free FBS and 5 μg/mL blasticidin).
b. Add 6 μL of polybrene (10 mg/mL stock) to the supplemented lentiviral supernatant (~60 mL) to achieve a final polybrene concentration of 8 μg/mL.
Note: Polybrene is a positively charged polymer that increases viral entry into cells by neutralising the electrostatic repulsion between virus particles and the negatively charged cell membrane.
c. Incubate the mixture at room temperature for 10 min. Table 4 below lists reagent volumes.
Table 4. Viral supernatant volumes
| Reagent | Volume |
|---|---|
| Filtered lentiviral supernatant | ~40 mL |
| Additional 1/3 volume fresh DMEM (supplemented with 10% tetracycline-free FBS and 5 μg/mL blasticidin) | 20 mL |
| Polybrene (10 mg/mL stock; 8 μg/mL final concentration) | 6 μL |
| Total volume | ~60 mL |
6. Remove the old medium from the T175 flask of HEK293S GnTI- TetR cells for transduction, then replace it with ~60 mL of viral supernatant supplemented with polybrene. Do not add viral supernatant to the control cell flask. Gently rock the flask to ensure that the medium is distributed evenly and incubate for 72 h under standard tissue culture conditions.
Caution: Avoid detaching adherent cells from the flask when adding the viral supernatant.
Critical: Disinfect all labware that contacts lentiviral material in Virkon at a final concentration of 1% before disposal.
Day 6: Assess cells under microscope for viability (24 h after transduction)
7. Examine cells daily for viability and GFP fluorescence. A small subset of cells may already exhibit faint leaky GFP expression (low-level fluorescence observed without tetracycline or doxycycline induction). This population is typically more responsive to induction and will later be enriched by FACS sorting.
Day 7: Assess cells under microscope for viability (48 h after transduction)
Day 8: Stable HEK293S GnTI- TetR cell line established (72 h after transduction)
8. Prepare 440 mL of Virkon disinfectant solution by dissolving one 5 g Virkon virucidal tablet in 440 mL of water in a large beaker. The addition of ~60 mL of discarded cultured medium results in a final Virkon concentration of 1%. Prewarm the following to 37 °C before use: DMEM (supplemented with 10% tetracycline-free FBS and 5 μg/mL blasticidin), PBS, and TrypLE Express enzyme.
9. Examine both transduced and control flasks under a microscope for viability and leaky GFP expression.
10. Split the transduced T175 flask into three T175 flasks:
a. One flask for cryopreservation after reaching confluence (cryopreservation protocol below).
b. Two flasks for FACS sorting of leaky GFP-expressing cells once confluent. FACS will be performed once they reach confluence in ~4 days.
11. Split the non-transduced control HEK293S GnTI- TetR cells into three T175 flasks:
a. Two flasks as a negative control for future flow cytometry assays.
b. One flask for cryopreservation after reaching confluence.
Notes:
1. When splitting for the first time after transduction, wash adherent cells gently three times with 10 mL of prewarmed PBS before adding 4 mL of TrypLE Express enzyme to dissociate cells.
2. Both transduced and non-transduced control cells should be maintained under blasticidin selection.
Critical: Treat all waste (medium, PBS, labware) with Virkon at a final concentration of 1% before disposal.
12. Cryopreservation protocol for a single confluent T175 flask:
a. Make up fresh cryopreservation buffer (see Recipe 7 for details).
b. Under sterile conditions, remove medium, wash cells with 10 mL of prewarmed PBS, remove PBS, and add 4 mL of TrypLE Express enzyme. Incubate for 5 min in a 37 °C incubator under standard conditions.
c. After incubation, add 6 mL of DMEM (supplemented with 10% tetracycline-free FBS and 5 μg/mL blasticidin) to the T175 flask, resuspend cells, and transfer to a 50 mL sterile centrifuge tube.
d. Count cells and determine the total number.
e. Centrifuge cells at 500× g for 5 min at 4 °C, then discard the supernatant carefully without disturbing the pellet.
f. Resuspend the cell pellet in cryopreservation buffer to achieve ~4 × 106 cells/mL. Dispense 1 mL per cryovial, label clearly, and place vials in a controlled-rate freezing container (Mr. Frosty freezing container) equilibrated to room temperature.
g. Transfer the container to a -80 °C freezer for overnight controlled-rate freezing.
h. Transfer vials to liquid nitrogen storage for long-term preservation.
D. Post-transduction FACS sorting and expansion (days 12–27)
Day 12: FACS sorting of stable HEK293S GnTI- TetR cell line
1. FACS scheduling: Book two FACS machines for a 6-h session to allow parallel sorting and minimise sample exposure time in flow cytometry buffer.
2. Prewarm the following to 37 °C before use: DMEM (supplemented with 10% tetracycline-free FBS and 5 μg/mL blasticidin), PBS, and TrypLE Express enzyme.
3. Prepare FACS reagents on ice (see Recipes 5 and 6 for details): flow cytometry buffer and FACS catch buffer. Under sterile conditions, pre-fill sterile 1.5 mL microcentrifuge tubes (catch tubes) with 1.5 mL of FACS catch buffer and keep on ice.
4. FACS sorting for leaky GFP expressors: FACS sorting is used to isolate transduced cells showing low, leaky GFP expression in the absence of tetracycline or doxycycline induction. In the aGPCR-HEK system, only ~0.5%–1% of transduced cells display leaky expression. These cells are selected and expanded because they respond more strongly to induction than non-leaky expressors and produce more receptor.
a. Flasks for sorting:
i. Two transduced confluent T175 flasks for harvesting and sorting.
ii. One non-transduced control T175 flask to set the GFP-negative control threshold during sorting (not sorted or saved).
b. Cell harvesting for each confluent T175 flask:
i. Remove medium, wash with 10 mL of prewarmed PBS, remove PBS, apply 5 mL of TrypLE Express enzyme, and incubate for 5 min in a 37 °C incubator under standard conditions.
ii. After incubation, add 10 mL of flow cytometry buffer to the flask, ensuring that all cells have detached.
iii. Transfer cells to a sterile 50 mL labelled centrifuge tube, count cells, and then centrifuge at 500× g for 5 min at 4 °C.
iv. After centrifugation, carefully remove supernatant without disturbing the cell pellet. Discard the supernatant. Resuspend cells in flow cytometry buffer to a concentration of 10 × 106 cells/mL. This can be diluted later if required.
v. Filter suspensions through sterile CellTrics 50 μm filters into sterile 5 mL polypropylene tubes (maximum 3 mL per tube).
vi. Keep all tubes (cell suspensions and catch tubes) on ice until sorting.
c. FACS sorting:
i. Process cells immediately after preparation.
ii. Operate two sorters in parallel to minimise exposure time in FACS buffer.
iii. Use non-transduced cells to set the GFP-negative threshold, then define the gate for leaky GFP-positive cells. Expect ~0.5%–1% of transduced cells to fall within this leaky population.
d. Post-sorting handling of cells:
i. FACS-sorted GFP-positive transduced cells are collected directly into the pre-filled catch tubes. Record the number of sorted cells collected per catch tube.
ii. Under sterile conditions, pool all FACS-sorted transduced cells together and then centrifuge at 500× g for 5 min at 4 °C.
iii. Carefully remove the supernatant without disturbing the cell pellet. Resuspend the cell pellet in DMEM (supplemented with 10% tetracycline-free FBS and 5 μg/mL blasticidin) using a volume appropriate for the number of sorted cells (see Table 5 below). Seed the cells accordingly and expand at 37 °C under standard tissue culture conditions.
Table 5. Post-FACS seeding details
| Number of sorted GFP-positive cells | Labware for seeding | Medium volume |
|---|---|---|
| 10,000 | 96-well plate: single well | 200 μL medium/well |
| >35,000 | 24-well plate: single well | 1 mL medium/well |
| 100,000–500,000 | 6-well plate: single well | 3 mL medium/well |
| >1,000,000 | T75 flask | 20 mL medium/flask |
Days 13–27: Expansion of FACS-sorted transduced HEK293S GnTI- TetR cells
5. Expand FACS-sorted transduced HEK293S GnTI- TetR cells to three T175 flasks:
a. One flask for cryopreservation after reaching confluence (steps C12a–h for the cryopreservation protocol).
b. Two flasks for conversion to suspension culture.
6. Expand non-transduced control HEK293S GnTI- TetR cells to two T175 flasks for conversion to suspension culture.
7. For workflow efficiency, plan to convert both control and FACS-sorted transduced cells to suspension culture on the same day.
8. Assess cell viability daily by microscopy. FACS-sorted transduced cells should display low-level GFP fluorescence (leaky expression) in the absence of tetracycline or doxycycline. Replace DMEM medium (supplemented with 10% tetracycline-free FBS and 5 μg/mL blasticidin) every 2–3 days and avoid over-confluence to maintain optimal growth.
9. Induction trial in a subset of sorted stable cells:
a. Before large-scale suspension expansion, perform a small-scale induction trial to verify that the transgene is inducible and to determine the optimal induction duration. This protocol uses tetracycline, but doxycycline can also be used for induction.
b. Seed a 6-well plate with 2 mL of 0.3 × 106 cells/mL per well in DMEM (supplemented with 10% tetracycline-free FBS and 5 μg/mL blasticidin). Incubate for 24 h at 37 °C under standard tissue culture conditions. Seed one well with non-transduced cells and five wells with FACS-sorted transduced cells, as shown in Table 6.
Table 6. 6-well plate map for induction trial in adherent cells
| Well | Sample detail | Tetracycline exposure | Harvest |
|---|---|---|---|
| 1 | Control cells | No tetracycline exposure | At 24 h |
| 2 | Sorted transduced cells | No tetracycline exposure | At 24 h |
| 3 | Sorted transduced cells | 1 μg/mL tetracycline induction for 24 h | At 24 h |
| 4 | Sorted transduced cells | 1 μg/mL tetracycline induction for 48 h | At 48 h |
| 5 | Sorted transduced cells | 1 μg/mL tetracycline induction for 72 h | At 72 h |
| 6 | Sorted transduced cells | 1 μg/mL tetracycline induction for 96 h | At 96 h |
c. After 24 h, replace the medium as below and incubate for 24 h under standard tissue culture conditions:
i. Add tetracycline-free medium (DMEM supplemented with 10% tetracycline-free FBS and 5 μg/mL blasticidin) to the control well and one well of sorted transduced cells.
ii. Add medium with 1 μg/mL tetracycline to the remaining four wells. Lower tetracycline concentrations can be sufficient for maximal induction, as doses as low as 0.1 μg/mL have been reported to induce comparable expression levels in a related tetracycline-inducible GPCR expression system [42]. As the optimal induction concentration may vary depending on receptor stability, TetR expression levels, and cellular toxicity, we recommend performing a tetracycline dose–response induction curve to identify the minimal concentration that achieves maximal expression for each GPCR of interest. Incubate for 24 h at 37 °C under standard tissue culture conditions.
d. After 24 h, image all wells by brightfield and fluorescence microscopy. Harvest the control cell well, the non-induced transduced well, and the 24-h tetracycline-induced well. Count cells and assess viability using trypan blue as per standardised cell counting protocols. Stain a subset of harvested cells with anti-HA-APC antibody (see Reagents) and analyse by flow cytometry for i) aGPCR surface expression, and ii) whole-cell GFP fluorescence (using the control cells to set the negative fluorescence threshold). Snap-freeze the remaining induced cells for western blotting and small-scale purification to assess the quality of the induced aGPCR using fluorescence-detection size-exclusion chromatography (FSEC).
e. Repeat microscopy, harvesting, counting, flow cytometry, and small-scale purifications at 48, 72, and 96 h intervals, harvesting the corresponding tetracycline-induced wells at each time point.
f. Compare cell counts, viability, whole-cell GFP and surface HA expression levels, and quality of purified aGPCR across all time points to determine the optimal tetracycline induction duration for your aGPCR stable cell line (defined as the time point yielding maximal cell surface expression and highest quality purified protein, with best cell viability).
E. Conversion to suspension cell culture and expansion (days 28–35)
Days 28–35: Convert to suspension culture and expand
1. Prewarm the following to 37 °C before use: FreeStyle 293 medium supplemented with 1% tetracycline-free FBS and 5 μg/mL blasticidin, PBS, and TrypLE Express enzyme.
2. Convert adherent HEK293S GnTI- TetR cells to suspension culture by switching to FreeStyle 293 medium with reduced FBS.
Critical: Seed suspension HEK293S GnTI- TetR cultures at a minimum 0.5 × 106 cells/mL to maintain optimal growth. Doubling time is approximately 24 h. For optimal gas exchange, use a maximum medium volume of 25% of the flask’s total capacity (e.g., 125 mL medium in 500 mL Erlenmeyer flasks, or 500 mL medium in 2 L roller bottles).
3. Harvest adherent cells and transition to suspension culture:
a. Harvest two confluent T175 flasks per cell line (both FACS-sorted transduced cells and non-transduced control cells) when converting to suspension culture.
b. Remove medium and wash each T175 flask with 10 mL of prewarmed PBS.
c. Discard PBS and add 5 mL of TrypLE Express enzyme to each flask, followed by 5 min of incubation under standard tissue culture conditions.
d. After 5 min of incubation, add 25 mL of prewarmed FreeStyle 293 medium (supplemented with 1% tetracycline-free FBS and 5 μg/mL blasticidin) to each flask and ensure that all cells have detached.
e. Pool transduced cells (~30 mL + ~30 mL) into a labelled 500 mL Erlenmeyer flask and do the same in a separate flask with control cells (~30 mL + ~30 mL).
f. Measure cell density (~60 mL per flask) and dilute each to 0.5 × 106 cells/mL using prewarmed FreeStyle 293 medium supplemented with 1% tetracycline-free FBS and 5 μg/mL blasticidin.
g. Incubate flasks in a shaking incubator at 37 °C, 8% CO2, >95% humidity, and a shaking speed of 125 rpm.
4. Suspension culture maintenance and expansion:
a. Count cells daily.
b. When FACS-sorted transduced cell cultures reach 2 × 106 cells/mL, split into larger flasks and reseed at 0.5 × 106 cells/mL.
c. Maintain non-transduced control cells between 0.5 and 2 × 106 cells/mL. Control cells do not require expansion; they will be used as negative controls for flow cytometry when harvesting large-scale induction.
d. Expand FACS-sorted transduced cells to the desired culture volume for downstream applications, for example, 3–6 L.
5. Small-scale induction validation in suspension: To confirm tetracycline induction performance in suspension culture, perform a separate small-scale induction.
a. Seed 30 mL of 2 × 106 cells/mL FACS-sorted transduced cells into a 125 mL Erlenmeyer flask.
b. Induce cells by adding 30 μL of tetracycline (1 mg/mL stock) for a final concentration of 1 μL/mL and incubate in a shaking incubator under standard conditions (37 °C, 8% CO2, >95% humidity, 125 rpm).
c. Collect samples at 24, 48, 72, and 96 h, and collect non-transduced control cells at each time point. Count cells and assess viability using trypan blue. Stain a subset of harvested cells with anti-HA-APC antibody (see Reagents) and analyse by flow cytometry for aGPCR surface HA expression and whole-cell GFP fluorescence (using control cells to set the negative fluorescence threshold). Snap-freeze the remaining induced cells for western blotting and small-scale purification and assess the quality of the induced aGPCR using FSEC.
d. Determine the optimal induction time point as that with the highest surface expression, best purified protein quality, and best cell viability.
Critical: To protect against downstream failure, cryopreserve a portion of both FACS-sorted transduced and control cells before induction.
6. Cryopreservation protocol for suspension cells:
a. Prepare fresh cryopreservation buffer (see Recipe 7).
b. Collect 50 mL of FACS-sorted transduced cells and deposit in a labelled sterile 50 mL centrifuge tube. Do the same for control cells in a separate labelled 50 mL tube.
c. Count cells and determine total numbers.
d. Centrifuge cells at 500× g for 5 min at 4 °C; then, discard supernatant carefully without disturbing the pellet.
e. Resuspend cells in cryopreservation buffer to a concentration of ~4 × 106 cells/mL.
f. Dispense 1 mL per cryovial, label clearly, and place vials in a controlled-rate freezing container (Mr. Frosty freezing container) equilibrated to room temperature.
g. Transfer the container to a -80 °C freezer for overnight controlled-rate freezing.
h. Transfer vials to liquid nitrogen storage for long-term preservation.
F. Tetracycline induction and harvesting (days 36–37)
Day 36: Tetracycline induction
1. The optimal tetracycline duration and concentration may vary depending on the specific aGPCR construct and expression characteristics. For our aGPCR stable cell line expressing the CTF2B variant of active-state ADGRL4, we induced for 24 h with 1 μg/mL tetracycline (48 h or longer caused excessive cell death and misfolded protein). In contrast, for the inactive state version of ADGRL4, we were able to induce for 48 h.
2. Prewarm FreeStyle 293 (supplemented with 1% tetracycline-free FBS and 5 μg/mL blasticidin) to 37 °C before use.
3. Induction:
a. Induce FACS-sorted transduced cultures once both the target volume (e.g., 4 L) and cell density (2 × 106 cells/mL) have been reached.
b. For a 4 L culture, distribute cells into eight 2 L (850 cm2) roller bottles, each containing 500 mL of cell suspension.
c. Add 500 μL of 1 mg/mL tetracycline stock to each 500 mL of culture, making a final concentration of 1 μg/mL tetracycline per 2 L roller bottle.
d. Incubate cultures for 24 h in a shaking incubator (37 °C, 8% CO2, >95% humidity, 125 rpm shaking speed).
Day 37: Large-scale harvesting
4. Prepare 100 mL of fresh isotonic harvesting buffer supplemented with protease inhibitors. Chill to 4 °C and keep on ice.
Note: The exact composition of the harvesting buffer should be adjusted according to the downstream purification method to be used.
5. Pre-harvest quality control:
a. Collect separate 2 mL samples from both induced transduced and control cell cultures.
b. Count cells and assess viability with trypan blue.
c. Stain 200 μL of each sample with anti-HA-APC antibody and analyse by flow cytometry for whole-cell GFP fluorescence and surface HA expression. Expression patterns should be consistent with those observed in earlier small-scale induction trials.
6. Large-scale harvesting:
a. Proceed with large-scale harvesting and downstream processing of the induced aGPCR cell line. Sterility is no longer required from this stage onwards.
b. Discard all control cultures.
c. Centrifuge cells at 500× g for 15 min at 4 °C in 1 L centrifuge bottles.
d. After centrifugation, carefully remove the supernatant and resuspend the cell pellets with 4 °C isotonic harvesting buffer. Perform a second centrifugation at 4 °C using 50 mL centrifuge tubes. Remove most of the supernatant, leaving approximately 5 mL above each pellet. Proceed with snap-freezing in liquid nitrogen or immediate downstream processing.
Validation of protocol
The aGPCR-HEK protocol was experimentally validated in the following open-access research publication [23]. The method enabled the generation of four independent tetracycline-inducible HEK293S GnTI- TetR stable cell lines expressing distinct aGPCR constructs for structural biology applications. The method was originally developed to express ADGRL4, a receptor that was poorly expressed in insect cell systems. Using protein generated by the aGPCR-HEK protocol, we determined the first high-resolution structure of active-state ADGRL4 using cryo-EM [23].
The figures and tables listed below from the associated publication provide direct experimental validation aGPCR-HEK protocol:
1. Purification of ADGRL4 constructs for structural studies: Supplementary Figure 3a–j.
2. Characterisation of the aGPCR-HEK protocol protein product: Figures 2a–b, 3a–b, 4a–c, 5a–c, and 6a–c; Supplementary Figure 4 and Supplementary Table 1.
General notes and troubleshooting
Troubleshooting
Problem 1: Poor transfection efficiency in Lenti-X 293T cells.
Potential causes: Low-quality plasmid DNA, incorrect DNA ratios, or unhealthy cells (high passage, over- or under-confluent, or contaminated).
Solutions: Use freshly prepared, high-quality plasmid DNA; confirm correct DNA ratios during transfection; transfect only low-passage, healthy Lenti-X 293T cells at ~70% confluence.
Problem 2: Very low or no lentiviral detection when using the Lenti-X GoStix assay.
Potential causes: Inefficient transfection, incorrect filter type or pore size (e.g., 0.2 μm or nitrocellulose), or viral titre loss during storage.
Solutions: Use a 0.45 μm sterile PES filter to avoid viral shearing; test freshly harvested supernatant; if storage is required, aliquot and freeze at -80 °C to prevent titre loss and avoid repeat freeze-thaw cycles.
Problem 3: Inefficient transduction of HEK293S GnTI- TetR cells.
Potential causes: Low viral titre or poor cell health at transduction (over- or under-confluent, contaminated, or high passage); polybrene omitted or added before storage of viral supernatant.
Solutions: Transduce only healthy ~70% confluent, low-passage HEK293S GnTI- TetR cells; add polybrene (8 μg/mL final concentration) immediately before transduction; verify viral titre using Lenti-X GoStix assay and concentrate the virus if necessary.
Problem 4: Failed induction with tetracycline.
Potential causes: Problems 1–3 above, or degraded tetracycline/doxycycline stocks due to light exposure or repeated freeze-thaw cycles.
Solutions: Prepare fresh 1 mg/mL tetracycline in ethanol; protect from light degradation by wrapping in foil; store in aliquots at -20 °C; avoid repeated freeze-thaw cycles.
Problem 5: Gradual loss of inducibility in stable HEK293S GnTI- TetR lines.
Potential causes: Transcriptional silencing of the TetR cassette after extended passaging or loss of blasticidin selection, leading to constitutive aGPCR transgene expression.
Solutions: Maintain continuous blasticidin selection at 5 μg/mL; limit cultures to less than 20 passages (corresponding to 8–10 weeks of continuous culture); re-establish cell line by repeating the AGPCR-HEK protocol.
Acknowledgments
D.M.F. designed and conducted the original study [23] and wrote this protocol. C.G.T. provided guidance and contributed to writing.
The graphical overview figure was created using BioRender and Adobe Illustrator.
This work was supported by funding from Cancer Research UK (CRUK) (grant holder D.M.F.; grant number RCCPOB-May22\100008), from the Medical Research Council, and from the Department of Oncology, University of Cambridge (fellowship holder D.M.F.). C.G.T. was supported by core funding from the Medical Research Council (MRC U105197215).
This protocol is based on the original research manuscript in which the methodology was first described and validated [23]. For the purpose of open access, the MRC Laboratory of Molecular Biology has applied a CC BY public copyright licence to any Author Accepted Manuscript version arising. pMD2.G (Addgene plasmid: 12259; http://n2t.net/addgene:12259) and psPAX2 (Addgene plasmid: 12260; http://n2t.net/addgene:12260) were a gift from Dr Didier Trono. pHR-CMV-TetO2_3C-Twin-Strep was a gift from Dr A. Radu Aricescu (Addgene plasmid: 113883; http://n2t.net/addgene:113883).
Competing interests
The authors declare no conflicts of interest.
References
- Hamann, J., Aust, G., Araç, D., Engel, F. B., Formstone, C., Fredriksson, R., Hall, R. A., Harty, B. L., Kirchhoff, C., Knapp, B., et al. (2015). International Union of Basic and Clinical Pharmacology. XCIV. Adhesion G Protein–Coupled Receptors. Pharmacol Res. 67(2): 338–367. https://doi.org/10.1124/pr.114.009647
- Dieterich, L. C., Mellberg, S., Langenkamp, E., Zhang, L., Zieba, A., Salomäki, H., Teichert, M., Huang, H., Edqvist, P. H., Kraus, T., et al. (2012). Transcriptional profiling of human glioblastoma vessels indicates a key role of VEGF-A and TGFβ2 in vascular abnormalization. J Pathol. 228(3): 378–390. https://doi.org/10.1002/path.4072
- Towner, R. A., Jensen, R. L., Colman, H., Vaillant, B., Smith, N., Casteel, R., Saunders, D., Gillespie, D. L., Silasi-Mansat, R., Lupu, F., et al. (2013). ELTD1, a Potential New Biomarker for Gliomas. Neurosurgery. 72(1): 77–91. https://doi.org/10.1227/neu.0b013e318276b29d
- Masiero, M., Simões, F. C., Han, H. D., Snell, C., Peterkin, T., Bridges, E., Mangala, L. S., Wu, S. Y., Pradeep, S., Li, D., et al. (2013). A Core Human Primary Tumor Angiogenesis Signature Identifies the Endothelial Orphan Receptor ELTD1 as a Key Regulator of Angiogenesis. Cancer Cell. 24(2): 229–241. https://doi.org/10.1016/j.ccr.2013.06.004
- Favara, D. M., Banham, A. H. and Harris, A. L. (2014). A review of ELTD1, a pro-angiogenic adhesion GPCR. Biochem Soc Trans. 42(6): 1658–1664. https://doi.org/10.1042/bst20140216
- Favara, D. M., Banham, A. H. and Harris, A. L. (2019). ADGRL4/ELTD1 is a highly conserved angiogenesis-associated orphan adhesion GPCR that emerged with the first vertebrates and comprises 3 evolutionary variants. BMC Evol Biol. 19(1): 143. https://doi.org/10.1186/s12862-019-1445-9
- Favara, D. M., Zois, C. E., Haider, S., Pires, E., Sheldon, H., McCullagh, J., Banham, A. H. and Harris, A. L. (2019). ADGRL4/ELTD1 Silencing in Endothelial Cells Induces ACLY and SLC25A1 and Alters the Cellular Metabolic Profile. Metabolites. 9(12): 287. https://doi.org/10.3390/metabo9120287
- Favara, D. M., Liebscher, I., Jazayeri, A., Nambiar, M., Sheldon, H., Banham, A. H. and Harris, A. L. (2021). Elevated expression of the adhesion GPCR ADGRL4/ELTD1 promotes endothelial sprouting angiogenesis without activating canonical GPCR signalling. Sci Rep. 11(1): 8870. https://doi.org/10.1038/s41598-021-85408-x
- Li, J., Shen, J., Wang, Z., Xu, H., Wang, Q., Chai, S., Fu, P., Huang, T., Anas, O., Zhao, H., et al. (2019). ELTD1 facilitates glioma proliferation, migration and invasion by activating JAK/STAT3/HIF-1α signaling axis. Sci Rep. 9(1): 13904. https://doi.org/10.1038/s41598-019-50375-x
- Wang, Q., Zhang, C., Pang, Y., Cheng, M., Wang, R., Chen, X., Ji, T., Yang, Y., Zhang, J., Zhong, C., et al. (2024). Comprehensive analysis of bulk, single-cell RNA sequencing, and spatial transcriptomics revealed IER3 for predicting malignant progression and immunotherapy efficacy in glioma. Cancer Cell Int. 24(1): 332. https://doi.org/10.1186/s12935-024-03511-1
- Sun, B. and Zhong, F. J. (2021). ELTD1 Promotes Gastric Cancer Cell Proliferation, Invasion and Epithelial–Mesenchymal Transition Through MAPK/ERK Signaling by Regulating CSK. Int J Gen Med. 14: 4897–4911. https://doi.org/10.2147/ijgm.s325495
- Wang, L., Xu, Y., Jiang, L. and Liu, J. (2025). The elevated expression of ADGRL4 indicates poor prognosis in gastric cancer. Asian J Surg. 48(4): 2503–2505. https://doi.org/10.1016/j.asjsur.2024.07.355
- Sun, J., Zhang, Z., Chen, J., Xue, M. and Pan, X. (2021). ELTD1 promotes invasion and metastasis by activating MMP2 in colorectal cancer. Int J Biol Sci. 17(12): 3048–3058. https://doi.org/10.7150/ijbs.62293
- Abdul Aziz, N. A., Mokhtar, N. M., Harun, R., Mollah, M. M. H., Mohamed Rose, I., Sagap, I., Mohd Tamil, A., Wan Ngah, W. Z. and Jamal, R. (2016). A 19-Gene expression signature as a predictor of survival in colorectal cancer. BMC Med Genomics. 9(1): 58. https://doi.org/10.1186/s12920-016-0218-1
- Sheldon, H., Bridges, E., Silva, I., Masiero, M., Favara, D. M., Wang, D., Leek, R., Snell, C., Roxanis, I., Kreuzer, M., et al. (2021). ADGRL4/ELTD1 Expression in Breast Cancer Cells Induces Vascular Normalization and Immune Suppression. Mol Cancer Res. 19(11): 1957–1969. https://doi.org/10.1158/1541-7786.mcr-21-0171
- Wang, Z. and Zhang, Z. (2024). Single-cell analysis reveals ADGRL4+ renal tubule cells as a highly aggressive cell type in clear cell renal cell carcinoma. Sci Rep. 14(1): 2407. https://doi.org/10.1038/s41598-024-52928-1
- Kan, A., Le, Y., Zhang, Y. f., Duan, F. t., Zhong, X. p., Lu, L. h., Ling, Y. h. and Guo, R. P. (2018). ELTD1 Function in Hepatocellular Carcinoma is Carcinoma-Associated Fibroblast-Dependent. J Cancer. 9(14): 2415–2427. https://doi.org/10.7150/jca.24406
- Mao, D., Wang, H., Guo, H., Che, X., Chen, M., Li, X., Liu, Y., Huo, J. and Chen, Y. (2024). Tanshinone IIA normalized hepatocellular carcinoma vessels and enhanced PD-1 inhibitor efficacy by inhibiting ELTD1. Phytomedicine. 123: 155191. https://doi.org/10.1016/j.phymed.2023.155191
- Li, Z., Nguyen Canh, H., Takahashi, K., Le Thanh, D., Nguyen Thi, Q., Yang, R., Yoshimura, K., Sato, Y., Nguyen Thi, K., Nakata, H., et al. (2024). Histopathological growth pattern and vessel co-option in intrahepatic cholangiocarcinoma. Med Mol Morphol. 57(3): 200–217. https://doi.org/10.1007/s00795-024-00392-1
- Guihurt Santiago, J., Burgos-Tirado, N., Lafontaine, D. D., Mendoza Sierra, J. C., Camacho, R. H., Vecchini Rodríguez, C. M., Morales-Tirado, V. and Flores-Otero, J. (2021). Adhesion G protein-coupled receptor, ELTD1, is a potential therapeutic target for retinoblastoma migration and invasion. BMC Cancer. 21(1): 53. https://doi.org/10.1186/s12885-020-07768-3
- Geng, G., Zhang, L., Yu, Y., Guo, X., Li, Q. and Ming, M. (2025). ADGRL4 Promotes Cell Growth, Aggressiveness, EMT, and Angiogenesis in Neuroblastoma via Activation of ERK/STAT3 Pathway. Curr Mol Med. 25(1): 45–55. https://doi.org/10.2174/0115665240254765231117122210
- Tian, Y., Bi, Z., Ge, S., Ye, B. and Han, W. (2022). STAT5A modulated EndMT via upregulation of ELTD1 expression in diabetic nephropathy. Clin Exp Pharmacol Physiol. 49(6): 686–695. https://doi.org/10.1111/1440-1681.13644
- Chen, Q., Gusach, A., Diamante, A., Patel, J. C., Edwards, P. C., Tate, C. G. and Favara, D. M. (2025). Structure of the Gq-coupled adhesion receptor ADGRL4. Nat Commun. 17(1): e1038/s41467–025–67629–0. https://doi.org/10.1038/s41467-025-67629-0
- Ping, Y. Q., Mao, C., Xiao, P., Zhao, R. J., Jiang, Y., Yang, Z., An, W. T., Shen, D. D., Yang, F., Zhang, H., et al. (2021). Structures of the glucocorticoid-bound adhesion receptor GPR97–Go complex. Nature. 589(7843): 620–626. https://doi.org/10.1038/s41586-020-03083-w
- Barros-Álvarez, X., Nwokonko, R. M., Vizurraga, A., Matzov, D., He, F., Papasergi-Scott, M. M., Robertson, M. J., Panova, O., Yardeni, E. H., Seven, A. B., et al. (2022). The tethered peptide activation mechanism of adhesion GPCRs. Nature. 604(7907): 757–762. https://doi.org/10.1038/s41586-022-04575-7
- Ping, Y. Q., Xiao, P., Yang, F., Zhao, R. J., Guo, S. C., Yan, X., Wu, X., Zhang, C., Lu, Y., Zhao, F., et al. (2022). Structural basis for the tethered peptide activation of adhesion GPCRs. Nature. 604(7907): 763–770. https://doi.org/10.1038/s41586-022-04619-y
- Xiao, P., Guo, S., Wen, X., He, Q.-T., Lin, H., Huang, S.-M., Gou, L., Zhang, C., Yang, Z., Zhong, Y.-N., et al. (2022). Tethered peptide activation mechanism of the adhesion GPCRs ADGRG2 and ADGRG4. Nature. 604: 771–778. https://doi.org/10.1038/s41586-022-04590-8
- Qu, X., Qiu, N., Wang, M., Zhang, B., Du, J., Zhong, Z., Xu, W., Chu, X., Ma, L., Yi, C., et al. (2022). Structural basis of tethered agonism of the adhesion GPCRs ADGRD1 and ADGRF1. Nature. 604(7907): 779–785. https://doi.org/10.1038/s41586-022-04580-w
- Zhu, X., Qian, Y., Li, X., Xu, Z., Xia, R., Wang, N., Liang, J., Yin, H., Zhang, A., Guo, C., et al. (2022). Structural basis of adhesion GPCR GPR110 activation by stalk peptide and G-proteins coupling. Nat Commun. 13(1): 5513. https://doi.org/10.1038/s41467-022-33173-4
- Lin, H., Xiao, P., Bu, R. Q., Guo, S., Yang, Z., Yuan, D., Zhu, Z. L., Zhang, C. X., He, Q. T., Zhang, C., et al. (2022). Structures of the ADGRG2–Gs complex in apo and ligand-bound forms. Nat Chem Biol. 18(11): 1196–1203. https://doi.org/10.1038/s41589-022-01084-6
- Qian, Y., Ma, Z., Liu, C., Li, X., Zhu, X., Wang, N., Xu, Z., Xia, R., Liang, J., Duan, Y., et al. (2022). Structural insights into adhesion GPCR ADGRL3 activation and Gq, Gs, Gi, and G12 coupling. Mol Cell. 82(22): 4340–4352.e6. https://doi.org/10.1016/j.molcel.2022.10.009
- Wang, N., Qian, Y., Xia, R., Zhu, X., Xiong, Y., Zhang, A., Guo, C. and He, Y. (2023). Structural basis of CD97 activation and G-protein coupling. Cell Chem Biol. 30(11): 1343–1353.e1345. https://doi.org/10.1016/j.chembiol.2023.08.003
- Yang, Z., Ping, Y. Q., Wang, M. W., Zhang, C., Zhou, S. H., Xi, Y. T., Zhu, K. K., Ding, W., Zhang, Q. Y., Song, Z. C., et al. (2025). Identification, structure, and agonist design of an androgen membrane receptor. Cell. 188(6): p1589–1604.e24. https://doi.org/10.1016/j.cell.2025.01.006
- Thomas, J. A. and Tate, C. G. (2014). Quality Control in Eukaryotic Membrane Protein Overproduction. J Mol Biol. 426(24): 4139–4154. https://doi.org/10.1016/j.jmb.2014.10.012
- Favara, D. M. and Tate, C. G. (2026). Purification of the Active-State G Protein-Coupled Receptor ADGRL4 for Cryo-Electron Microscopy Using a Modular Tag System and a Tethered mini-Gq. Bio Protoc. 16(5): e5617. https://doi.org/10.21769/BioProtoc.5617
- Naldini, L., Blömer, U., Gallay, P., Ory, D., Mulligan, R., Gage, F. H., Verma, I. M. and Trono, D. (1996). In Vivo Gene Delivery and Stable Transduction of Nondividing Cells by a Lentiviral Vector. Science. 272(5259): 263–267. https://doi.org/10.1126/science.272.5259.263
- Zufferey, R., Dull, T., Mandel, R. J., Bukovsky, A., Quiroz, D., Naldini, L. and Trono, D. (1998). Self-Inactivating Lentivirus Vector for Safe and Efficient In Vivo Gene Delivery. J Virol. 72(12): 9873–9880. https://doi.org/10.1128/jvi.72.12.9873-9880.1998
- Elegheert, J., Behiels, E., Bishop, B., Scott, S., Woolley, R. E., Griffiths, S. C., Byrne, E. F. X., Chang, V. T., Stuart, D. I., Jones, E. Y., et al. (2018). Lentiviral transduction of mammalian cells for fast, scalable and high-level production of soluble and membrane proteins. Nat Protoc. 13(12): 2991–3017. https://doi.org/10.1038/s41596-018-0075-9
- Reeves, P. J., Callewaert, N., Contreras, R. and Khorana, H. G. (2002). Structure and function in rhodopsin: High-level expression of rhodopsin with restricted and homogeneous N-glycosylation by a tetracycline-inducible N-acetylglucosaminyltransferase I-negative HEK293S stable mammalian cell line. Proc Natl Acad Sci USA. 99(21): 13419–13424. https://doi.org/10.1073/pnas.212519299
- Reeves, P. J., Kim, J. M. and Khorana, H. G. (2002). Structure and function in rhodopsin: A tetracycline-inducible system in stable mammalian cell lines for high-level expression of opsin mutants. Proc Natl Acad Sci USA. 99(21): 13413–13418. https://doi.org/10.1073/pnas.212519199
- Yao, F., Svensjö, T., Winkler, T., Lu, M., Eriksson, C. and Eriksson, E. (1998). Tetracycline repressor, tetR, rather than the tetR-mammalian cell transcription factor fusion derivatives, regulates inducible gene expression in mammalian cells. Hum Gene Ther. 9(13): 1939–1950. https://doi.org/10.1089/hum.1998.9.13-1939
- Tate, C. G., Haase, J., Baker, C., Boorsma, M., Magnani, F., Vallis, Y. and Williams, D. (2003). Comparison of seven different heterologous protein expression systems for the production of the serotonin transporter. Biochimica et Biophysica Acta (BBA) – Biomembranes. 1610(1): 141–153. https://doi.org/10.1016/s0005-2736(02)00719-8
- Andréll, J., Edwards, P. C., Zhang, F., Daly, M. and Tate, C. G. (2016). Generation of Tetracycline-Inducible Mammalian Cell Lines by Flow Cytometry for Improved Overproduction of Membrane Proteins. Methods Mol Biol. 1432: 63–78. https://doi.org/10.1007/978-1-4939-3637-3_5
- Helfinger, L. and Tate, C. G. (2022). Expression and Purification of the Human Thyroid-Stimulating Hormone Receptor. Methods Mol Biol. 2507: 313–325. https://doi.org/10.1007/978-1-0716-2368-8_16
- Gibson, D. G., Young, L., Chuang, R. Y., Venter, J. C., Hutchison, C. A. and Smith, H. O. (2009). Enzymatic assembly of DNA molecules up to several hundred kilobases. Nat Methods. 6(5): 343–345. https://doi.org/10.1038/nmeth.1318
Article Information
Publication history
Received: Dec 9, 2025
Accepted: Jan 28, 2026
Available online: Feb 10, 2026
Published: Mar 5, 2026
Copyright
© 2026 The Author(s); This is an open access article under the CC BY-NC license (https://creativecommons.org/licenses/by-nc/4.0/).
How to cite
Favara, D. M. and Tate, C. G. (2026). aGPCR-HEK: A Stable High-Expression Inducible Mammalian Cell Expression System for Adhesion GPCR Structural Biology Applications. Bio-protocol 16(5): e5621. DOI: 10.21769/BioProtoc.5621.
Category
Biological Engineering
Molecular Biology > Protein > Expression
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link









