- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Preparation of Outer Membrane Vesicle from Escherichia coli
Published: Vol 3, Iss 23, Dec 5, 2013 DOI: 10.21769/BioProtoc.995 Views: 23201
Reviewed by: Anonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Extraction of Bacterial Membrane Vesicle and Phage Complex by Density Gradient Ultracentrifugation
Shangru Li [...] Tianyuan Jia
Aug 20, 2024 2041 Views
Mycobacterium smegmatis Ribosome Purification, Co-sedimentation, and Subunit Association Assay
Aneek Banerjee [...] Jayati Sengupta
May 20, 2025 1564 Views
Preparation and Negative Staining for Visualization of Cyanoglobule Lipid Droplets Using Transmission Electron Microscopy
Febri A. Susanto [...] Peter K. Lundquist
Dec 5, 2025 1304 Views
Abstract
Outer membrane vesicles (OMVs) are spherical bilayered phospholipids of 20-200 nm in size produced from all Gram-negative bacteria and Gram-positive bacteria investigated to date. Previous biochemical and proteomic studies have revealed that the Gram-negative bacteria-derived OMVs are composed of various components like outer membrane proteins, lipopolysaccharides, outer membrane lipids, periplasmic proteins, DNA, and RNA. Here, in this protocol, we describe the method to isolate the OMVs from the culture supernatant of Escherichia coli (E. coli).
Keywords: Outer membrane vesiclesMaterials and Reagents
- Phosphate buffered saline (PBS) (Gibco®, catalog number: 70013-032 )
- E. coli (Isolated from the peritoneal lavage fluid of cecal ligation and puncture-operated mice)
- Luria-Bertani broth (LB) medium (Merck KGaA, catalog number: 1.10285.0500 ) (see Recipes)
Equipment
- 2 L Flasks
- Shaking incubator
- Centrifuge
- 500 ml Bottle top filter 43 mm neck (0.45 μm and 0.22 μm) (Corning, catalog number: 430514 , 430513 )
- QuixStand Benchtop System (Amersham Biosciences, catalog number: 56-4107-44 )
- 100-kDa hollow-fiber membrane (Amersham Biosciences, catalog number: 56-4101-33 )
- Vacuum pump
Note: All centrifuge tubes and flasks should be autoclaved before use to avoid contamination
Procedure
- A single colony of E. coli is transferred to 5 ml of LB broth.
- The bacteria are incubated in an orbital bacteria shaking incubator at 200 rpm at 37 °C overnight (8 h).
- LB broth of 500 ml is inoculated with 1/100 volume of the overnight cultured cells.
Notes:- Use 2 L flask when culturing 500 ml. Also, since 1/100 volume of 5 ml is 50 μl, before inoculation, increase the volume by adding about 900 μl of fresh LB medium to reduce cell loss.
- The yield of OMVs in terms of protein amount is 100 μg per 1 liter of E. coli culture.
- The cells are grown for 12 h at 200 rpm at 37 °C.
- The cells are pelleted at 5,000 x g for 15 min.
- The supernatant fraction is collected and pelleted again at 5,000 x g for 15 min.
- The supernatant is collected and filtered through a bottle top filter of pore size 0.45 μm using a vacuum pump.
- The filtered supernatant is concentrated to 50-fold by ultra-filtration with a Quixstand Benchtop System using a 100 kDa hollow-fiber membrane.
Note: Because the yield of OMVs is very low, in order to obtain a visible pellet after ultracentrifugation, the total volume of bacteria culture should be more than 5-7 liters. However, depending on the amount of OMV needed, the volume of bacteria culture could be reduced as well as the degree of concentration. Ex. 7 L of bacteria culture supernatant is concentrated to give about 280 ml of the concentrated supernatant to be filled in the total of four ultra-centrifuge tube (70 ml each). - The concentrated supernatant is filtered once again through a 0.22 μm vacuum filter to remove any remaining debris or bacteria.
- The resulting filtrate is subjected to ultra-centrifugation at 150,000 x g for 3 h at 4 °C.
- The supernatant is removed and the pellet (purified OMV) is resuspended in PBS and stored at – 80 °C until use.
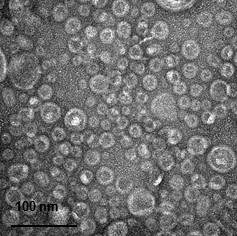
Figure 1. TEM image of E. coli OMV
Recipes
- LB medium
1% Tryptone, 0.5% yeast extract, 200 mM NaCl
Acknowledgments
This protocol was adapted from previously published work (Kim et al., 2013). This work was supported by a grant from the Korean Ministry of Education, Science and Technology, FPR08B1-240 of the 21C Frontier Functional Proteomics Program and Mid-career Researcher Program of National Research Foundation of Korea (NRF) grant funded by the Korea government MEST (No. 20110000215 and No. 20120005634).
References
- Kim, O. Y., Hong, B. S., Park, K. S., Yoon, Y. J., Choi, S. J., Lee, W. H., Roh, T. Y., Lotvall, J., Kim, Y. K. and Gho, Y. S. (2013). Immunization with Escherichia coli outer membrane vesicles protects bacteria-induced lethality via Th1 and Th17 cell responses. J Immunol 190(8): 4092-4102.
- Lee, E. Y., Choi, D. S., Kim, K. P. and Gho, Y. S. (2008). Proteomics in gram-negative bacterial outer membrane vesicles. Mass Spectrom Rev 27(6): 535-555.
- Park, K. S., Choi, K. H., Kim, Y. S., Hong, B. S., Kim, O. Y., Kim, J. H., Yoon, C. M., Koh, G. Y., Kim, Y. K. and Gho, Y. S. (2010). Outer membrane vesicles derived from Escherichia coli induce systemic inflammatory response syndrome. PLoS One 5(6): e11334.
Article Information
Copyright
© 2013 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Kim, O. Y., Hong, B. S., Park, K., Yoon, Y. J., Choi, S. J., Lee, W. H., Roh, T., Kim, Y. and Gho, Y. S. (2013). Preparation of Outer Membrane Vesicle from Escherichia coli. Bio-protocol 3(23): e995. DOI: 10.21769/BioProtoc.995.
Category
Microbiology > Microbial cell biology > Organelle isolation
Cell Biology > Organelle isolation > Outer membrane vesicles
Biochemistry > Lipid > Extracellular lipids
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link











