Materials and Reagents
- EDTA blood (extracted blood kept in tubes with EDTA for anticoagulant purposes)
- PBS
- Ficoll-Hypaque solution (density 1.077 g/L, Ficoll-Paque PLUS) (General Electric Company, catalog number: 17-1440-03 )
- FBS
- Complete RPMI-10 (RPMI, 10% FBS)
Equipment
- 15- or 50 ml conical centrifuge tube
- Beckman GPR centrifuge with GH-3.7 horizontal rotor (or equivalent temperature controlled centrifuge)
Procedure
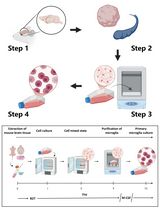
- Place fresh EDTA blood into 15- or 50 ml conical centrifuge tube. Using a sterile pipet, add an equal volume of room temperature PBS. Mix well.
- Set a layer of the Ficoll-Hypaque solution at the bottom of the centrifuge tube. Use 3 ml Ficoll-Hypaque per 10 ml blood/PBS mixture.
- Slowly layer the blood/PBS over the Ficoll at room temperature. Pipette very slowly the mixture against the wall of the tube so that it does not mix with the Ficoll layer.
- Centrifuge 30 min in a GH-3.7 rotor at 900 x g at room temperature, with NO brake.
- With a pipette remove the upper layer that contains the plasma and most of the platelets. Transfer the aggregates of mononuclear cells to another centrifuge tube (the intermediate layer contains aggregates of cells presenting white colour and floating over the Ficoll).
- Wash the mononuclear cells three times with PBS. Add three times the volume of mononuclear cell layer of PBS. Centrifuge 10 min at 400 x g at room temperature.
- Resuspend cells in complete RPMI-10 (RPMI, 10% FBS) (10 ml for plating or 1 ml 10% DMSO for freezing). Freeze or plate the cells.
Acknowledgments
This study was funded by grants SAF2010-17555, RD06/0014/0038 (RECAVA) and, RD06/0010/1013 (TERCEL) from the Spanish Ministry of Economy and Competition (MINECO) and EU FP7-ITN 215761 (NotchIT) to J.L.d.l.P. G.L. had a PhD fellowship from the MINECO (FPI Program, BES-2008-002904).
References
- John E. Coligan, Ada M. Kruisbeek, David H. Margulies, Ethan M. Shevach, and Warren Strober (eds.) (2004). Curr Protocols Immunol. John Wiley & Sons, Inc.
- Luxan, G., Casanova, J. C., Martinez-Poveda, B., Prados, B., D'Amato, G., MacGrogan, D., Gonzalez-Rajal, A., Dobarro, D., Torroja, C., Martinez, F., Izquierdo-Garcia, J. L., Fernandez-Friera, L., Sabater-Molina, M., Kong, Y. Y., Pizarro, G., Ibanez, B., Medrano, C., Garcia-Pavia, P., Gimeno, J. R., Monserrat, L., Jimenez-Borreguero, L. J. and de la Pompa, J. L. (2013). Mutations in the NOTCH pathway regulator MIB1 cause left ventricular noncompaction cardiomyopathy. Nat Med 19(2): 193-201.
Article Information
Copyright
© 2013 The Authors; exclusive licensee Bio-protocol LLC.
Category
Cell Biology > Cell isolation and culture > Cell isolation