- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Recovery and Expansion of Patient-Derived Glioblastoma Cells After Long-term Cryopreservation
Published: Vol 16, Iss 8, Apr 20, 2026 DOI: 10.21769/BioProtoc.5661 Views: 12
Reviewed by: Sébastien GillotinSam PatelAnu Thomas

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Generation of Agarose-Based FFPE Cancer Organoids for Morphology Preservation
Mi Rim Lee [...] Yun-Hee Kim
Oct 5, 2025 1775 Views
Improved Immunohistochemistry of Mouse Eye Sections Using Davidson's Fixative and Melanin Bleaching
Anne Nathalie Longakit [...] Catherine D. Van Raamsdonk
Nov 20, 2025 1627 Views
Abstract
Patient-derived glioblastoma (GBM) cells are valuable models for GBM research due to their rarity and the highly lethal nature of this cancer. Preserving these cells through long-term cryopreservation is therefore essential for advancing future investigations. However, recent studies have reported that standard cell recovery protocols are inefficient, resulting in poor cell survival and limited regrowth. Here, we established an optimized culture protocol that enhances the recovery and expansion of patient-derived GBM cells by combining Matrigel with an increased concentration of fetal bovine serum (FBS). This approach significantly improves cell attachment and recovery after thawing cells that have been cryopreserved for more than a decade. Importantly, the recovered cells retain key phenotypic characteristics and remain suitable for downstream applications, including drug testing and spheroid formation. Together, this optimized protocol provides a novel strategy to increase the availability of patient-derived GBM cells by improving their efficient recovery from long-term cryopreservation, thereby maximizing their utility in GBM research.
Key features
• Optimized for recovery of low-viability adherent cells, including long-term cryopreserved patient-derived GBM.
• Combined use of Matrigel coating and elevated FBS to enhance post-thaw attachment and recovery.
• Recovered cells maintain their morphology, marker expression, and functionality.
• Simple, effective protocol applicable to GBM and potentially to other adherent cell types.
Keywords: Adherent cell recoveryGraphical overview
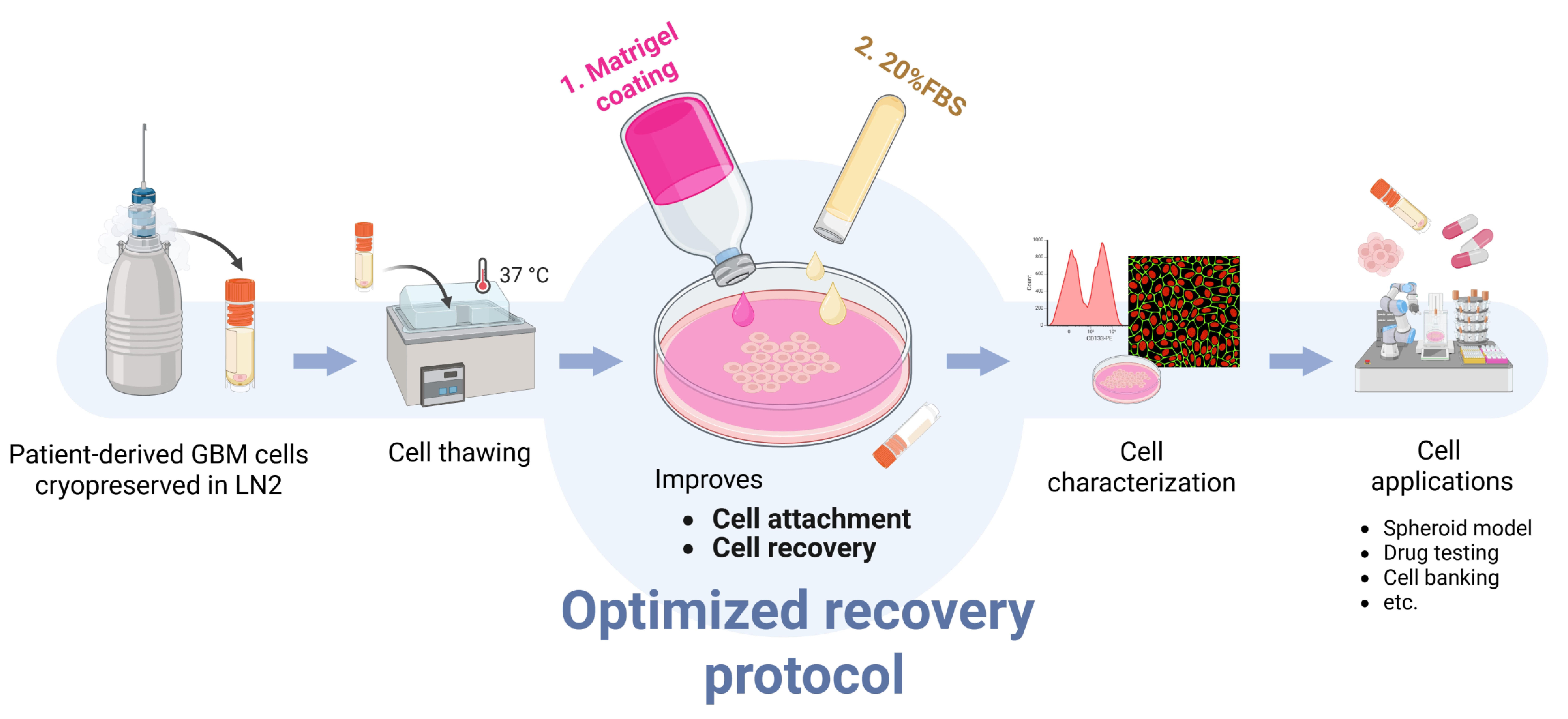
Overview of the optimized recovery protocol for patient-derived glioblastoma (GBM) cells after long-term cryopreservation
Background
Glioblastoma (GBM) is the most common and lethal primary brain tumor, characterized by aggressive growth and profound therapeutic resistance driven by intrinsic factors such as cellular heterogeneity and extrinsic barriers, including the blood–brain barrier [1]. Despite extensive research efforts, patient outcomes remain poor, highlighting the continued need for robust experimental models that accurately reflect patient tumor biology. Patient-derived GBM cells are therefore invaluable research tools, as they preserve key genetic, phenotypic, and functional characteristics of the original tumors and offer high translational relevance [2]. Consequently, the successful isolation, preservation, and recovery of these samples are critical for advancing GBM research.
Cryopreservation in liquid nitrogen (LN2) is the standard approach for long-term storage of biological materials, including patient-derived cells [3]. Conventional post-thaw recovery protocols typically involve rapid thawing in a 37 °C water bath, followed by seeding cells into standard culture medium supplemented with approximately 10% fetal bovine serum (FBS) and incubation until cell attachment and proliferation are observed [4,5]. While effective for many established cell lines, this approach often proves inadequate for fragile primary cells, particularly when initial cell numbers are low, cellular integrity at freezing is compromised, storage conditions fluctuate, or samples have been cryopreserved for extended periods [6]. In the case of patient-derived GBM cells stored for long durations, recovery failure can lead to irreversible loss of rare and irreplaceable patient material, as well as wasted resources associated with long-term biobanking. In our experience, GBM cells cryopreserved for more than a decade could not be successfully recovered or expanded using standard post-thaw protocols. These limitations underscore the need for an optimized recovery strategy specifically tailored to low-viability, adherent primary tumor cells.
Here, we describe an optimized post-thaw recovery protocol for patient-derived GBM cells that combines extracellular matrix support via Matrigel-coated culture surfaces with increased serum supplementation during the initial recovery phase. Matrigel has been shown to enhance cell adhesion and survival in fragile or low-adherent cell types, such as induced pluripotent stem cells (iPSCs), and may similarly support stressed GBM cells following long-term cryopreservation [7]. In parallel, elevating FBS concentration provides additional growth factors and survival cues to help cells overcome post-thaw stress and resume proliferation [8]. This protocol enables reliable recovery, expansion, and downstream use of patient-derived GBM cells following long-term cryopreservation and may be broadly applicable to other challenging primary adherent cell types.
Materials and reagents
Biological materials
1. Patient-derived glioblastoma cells (originally collected, isolated, and cryopreserved over a decade ago by a previous researcher [9]; subsequently thawed and utilized in our recently published study [10] and this protocol development); see Supplementary Materials of [10] for patient data details
Reagents
1. Dulbecco’s modified Eagle medium (DMEM), high glucose (Thermo Fisher Scientific, Gibco, catalog number: 12100046)
2. Fetal bovine serum (FBS) (Sigma-Aldrich, catalog number: F7524)
3. Penicillin-streptomycin mixture (double antibiotics, 100×) (Servicebio, catalog number: G4003)
4. Trypsin-EDTA (0.25%) (Thermo Fisher Scientific, Gibco, catalog number: 25200072)
5. Matrigel® basement membrane matrix, LDEV-free, 10 mL (Corning, catalog number: 354234)
6. Sodium chloride (NaCl) (Sigma-Aldrich, catalog number: S5886)
7. Sodium phosphate dibasic (Na2HPO4) (Sigma-Aldrich, catalog number: 106586)
8. Potassium phosphate monobasic (KH2PO4) (Sigma-Aldrich, catalog number: P0662)
9. APC anti-human CD44 antibody, dilution 1:100 (BioLegend, catalog number: 38806)
10. PE/Cyanine7 anti-human CD133 antibody, dilution 1:100 (BioLegend, catalog number: 393909)
11. Anti-nestin antibody, dilution 1:250 (Sigma-Aldrich, catalog number: MAB5326)
12. Anti-SOX2 antibody, dilution 1:250 (Sigma-Aldrich, catalog number: AB5603)
Solutions
1. Matrigel coating solution (see Recipes)
2. Optimized recovery medium (see Recipes)
3. Standard culture medium (see Recipes)
4. 1× phosphate-buffered saline (PBS) (see Recipes)
5. Trypsin solution 0.05% (see Recipes)
Recipes
1. Matrigel coating solution
| Reagent | Final concentration | Volume (for 6 mL)* |
| Matrigel | 0.3 mg/mL | 0.2 mL |
| Basal DMEM (high glucose) | n/a | 5.8 mL |
*This recipe provides an example for a Matrigel stock concentration of 9.0 mg/mL. The volume of basal DMEM (high glucose) added to the Matrigel aliquot was adjusted according to the lot-specific concentration reported in the manufacturer’s certificate of analysis for each Matrigel lot. To ensure a standardized final concentration of 0.3 mg/mL, the required dilution volume was calculated for each lot using the following formula:
Vmedium = [(Cstock × Valiquot)/0.3 mg/mL] - Valiquot
Vmedium is the required dilution volume of basal DMEM (high glucose) medium;
Cstock is the lot-specific concentration of the Matrigel stock (such as 9.0 mg/mL);
Valiquot is the volume of the frozen Matrigel aliquot (such as 0.2 mL);
Example calculation in this recipe:
Vmedium = [(9 × 0.2)/0.3] - 0.2 = 5.8 mL
Matrigel basement membrane matrix stock was stored at -80 °C until use or aliquoting. Aliquot volumes were prepared according to the manufacturer’s recommended working concentration range for coating (0.2–0.3 mg/mL); in our laboratory, a concentration of 0.3 mg/mL in 6 mL was routinely used as the working solution. For aliquoting, the Matrigel stock was thawed on ice and dispensed into pre-chilled 15 mL centrifuge tubes, which were then stored at -20 °C until use. To prepare the Matrigel coating solution, an aliquot was diluted with ice-cold DMEM (high glucose) to prevent gel formation upon warming. To ensure homogeneity, the diluted solution was incubated overnight at 4 °C. Although fresh preparation was preferred, working solutions were stored at 4 °C for no longer than one week.
2. Optimized recovery medium
| Reagent | Final concentration | Volume (for 50 mL) |
| Basal DMEM (high glucose) | 80% | 40 mL |
| FBS | 20% | 10 mL |
Fresh preparation is preferred, but if stored, the medium should be kept at 4 °C for no longer than one week to minimize the risk of contamination and degradation of nutrients. Warm the medium at 37 °C before use.
3. Standard culture medium
| Reagent | Final concentration | Volume (for 50 mL) |
| Basal DMEM (high glucose) | 79% | 44.5 mL |
| FBS | 10% | 5 mL |
| Penicillin-streptomycin mixture | 1× | 0.5 mL |
The medium should be kept at 4 °C for no longer than 2–4 weeks to minimize the risk of contamination and degradation of nutrients. Warm the medium at 37 °C before use.
4. 1× PBS
| Reagent | Final concentration | Amount (for 1,000 mL) |
| NaCl | 131.0 mM | 7.65 g |
| Na2HPO4 | 7.0 mM | 0.994 g |
| KH2PO4 | 3.0 mM | 0.408 g |
| Milli-Q water | n/a | 1,000 mL |
In our laboratory, 1,000 mL of 1× PBS was prepared by dissolving the chemical powders listed in the recipe in Milli-Q water. The solution was stirred until fully dissolved, and the pH was adjusted to approximately 7.4. The buffer was then brought to a final volume of 1,000 mL and sterilized by autoclaving. After sterilization, the solution was allowed to cool overnight before use. Sterile PBS was stored at room temperature for up to 6 months. Sterile 1× PBS from commercial sources can be used when preparation is not preferred.
5. Trypsin solution (0.05%)
| Reagent | Final concentration | Volume (for 50 mL) |
| 0.25% trypsin | 0.05% | 10 mL |
| 1× PBS | n/a | 40 mL |
Store 0.05% trypsin solution at 4 °C for no longer than 1–2 weeks for reliable enzyme activity. Warm at room temperature before use.
Laboratory supplies
1. 6-well clear TC-treated multiple well plates, individually wrapped, sterile (Corning, catalog number: 3516)
2. 25 cm2 rectangular canted neck cell culture flask with vent cap (Corning, catalog number: 430639)
3. 75 cm2 U-shaped canted neck cell culture flask with vent cap (Corning, catalog number: 431464U)
4. Centrifuge tubes with flat cap, 15 mL, sterilized (JET Biofil, catalog number: CFT011150)
5. Centrifuge tubes with flat cap, 50 mL, sterilized (JET Biofil, catalog number: CFT011500)
6. 2 mL internal threaded polypropylene cryogenic vial, self-standing with round bottom (Corning, catalog number: 430488)
7. 10 μL pipette tips, non-filtered, clear (Corning, Axygen®, catalog number: T-300)
8. 1–200 μL pipette tips, graduated, yellow (Corning, catalog number: 4112)
9. 1,000 μL pipette tips (maximum working volume 1,250 μL), graduated, natural (Corning, catalog number: 4114)
Equipment
1. CO2 incubator (Thermo Scientific, FormaTM Steri-CycleTM Incubator, 184 L, Polished Stainless Steel, model: 371)
2. Class II biological safety cabinet (Thermo Fisher Scientific, model: MSC-Advantage BSC 1.8)
3. Inverted microscope (Nikon, model: Eclipse TS100)
4. Water bath at 37 °C (Julabo, model: SW23)
5. Pipetman P1000, 100–1,000 μL (Gilson)
6. Motorized pipette controller (Corning, model: StripettorTM Ultra Pipet Controller 4099)
7. Disposable serological pipette 5, 10, and 25 mL (Wuxi NEST Biotechnology)
8. Laboratory/medical refrigerator at 37 °C (Panasonic, model: MPR-1411)
9. Liquid nitrogen tank (Thermo Fisher Scientific, model: CY509109)
Procedure
A. Preparation of optimized recovery medium and Matrigel-coated culture containers
1. Prepare 6 mL of Matrigel coating solution (see Recipe 1). Store at 4 °C overnight prior to use for container coating.
2. Select the recovery culture container for Matrigel coating.
Caution: When cell numbers in a freezing vial are unknown due to long-term storage, or if previous thawing attempts have been unsuccessful, initiating recovery in smaller containers (such as a single well of a 12- or 6-well plate) is recommended. This approach maximizes cell–cell interactions, improves survival, and enhances recovery efficiency while minimizing resource consumption.
3. Coat the culture container with cold Matrigel coating solution. The coating volume should be adjusted according to the container size (see Table 1) following the manufacturer’s instructions. Distribute the solution to ensure complete coverage of the container surface. Incubate coated containers at 37 °C in a cell culture incubator for at least 1 h to allow the Matrigel matrix to polymerize and set prior to use.
Caution: Always keep the Matrigel coating solution on ice during handling. Coating must be performed while the solution is cold, because Matrigel stays liquid at 4 °C but quickly gels when warmed.
4. Prepare 50 mL of optimized recovery medium supplemented with 20% FBS (see Recipe 2). Incubate the medium at 37 °C for at least 30 min prior to use for cell thawing.
5. Transfer 10 mL of prewarmed optimized recovery medium into a new 15 mL centrifuge tube for subsequent cell thawing.
Table 1. Coating volume according to container size
| Container size | Matrigel coating solution volume |
| Single well of a 12-well plate | 0.5 mL |
| Single well of a 6-well plate | 1 mL |
| 25 cm2 cell culture flask (T25) | 2 mL |
B. Patient-derived GBM cell thawing
1. Remove a cryogenic vial of patient-derived GBM cells stored in freezing medium from the liquid nitrogen tank.
2. Incubate the vial in a 37 °C water bath until the frozen cell suspension is completely thawed.
Critical: Do not fully submerge the vial in the water bath and ensure the water level remains below the cap line to prevent water seepage and microbial contamination. The thawing step should be performed rapidly, as dimethyl sulfoxide (DMSO) in the freezing medium is toxic to cells if exposure is prolonged.
Note: Gentle swirling of the vial in the water bath can speed thawing but ensure to avoid water contact with the vial cap.
3. Gently transfer the whole thawed cell suspension from the vial into a 15 mL centrifuge tube containing 10 mL of prewarmed recovery medium (as described in step A4) using a pipette.
4. Centrifuge cells at 523× g for 5 min to separate the cell pellet from the freezing medium.
5. Carefully remove the supernatant and then gently resuspend the cell pellet in prewarmed optimized recovery medium. Adjust the resuspension volume according to the size of the culture container used for initial recovery (see Table 2).
Table 2. Medium volume according to container size
| Container size | Medium volume |
| Single well of a 12-well plate | 1 mL |
| Single well of a 6-well plate | 2–3 mL |
| 25 cm2 cell culture flask (T25) | 5–7 mL |
| 75 cm2 cell culture flask (T75) | 12–15 mL |
6. Gently mix the cell pellet with the medium using a pipette until the suspension is homogeneous.
7. Transfer the cell suspension into Matrigel-coated culture containers (as described in step A3).
Caution: The suspension volume should not be divided for cell counting at this stage; the priority is to utilize the entire volume for recovery.
Critical: The cell suspension should be evenly distributed across the surface of the culture container to ensure complete coverage and to minimize clumping, which can reduce cell recovery.
8. Incubate cells under standard culture conditions in a humidified incubator under normoxic conditions (21% O2 with 5% CO2 at 37 °C).
Note: To minimize disturbance during recovery, incubate cells in an isolated incubator with minimal door openings.
C. Patient-derived GBM cell recovery
1. Examine the cell culture medium visually and under a microscope on the following day. Confirm that the medium is visually clear.
a. If the medium is cloudy and shows evidence of microbial contamination, discard the culture and thaw a new vial.
b. If the medium is cloudy without signs of contamination but contains floating cells, consider these cells non-viable or too weak to attach to the Matrigel support and continue to the next step.
2. Observe the attached cell morphology and density under a microscope.
a. If the medium is clear, with no floating cells and many attached cells displaying normal morphology, this indicates successful thawing, with most cells overcoming the initial stress and attaching to the Matrigel-coated surface. Proceed to the next step.
b. If the medium is cloudy with floating cells and very few or no attached cells, even when thawing in a smaller container such as a 12- or 6-well plate with Matrigel and 20% FBS support, this indicates poor recovery or poor cell integrity. Discard the culture and thaw a new vial of the same case.
c. If the medium is slightly turbid or cloudy with floating cells but also contains some attached cells, regardless of morphology, this indicates partial recovery. Viable cells are able to adhere to the Matrigel-coated surface, while weaker or non-viable cells remain floating. Proceed to the next step.
Note: See Troubleshooting if thawing of the same case fails repeatedly.
3. Remove the old medium carefully to eliminate floating cells without disturbing the attached cells.
4. Add the same volume of fresh, prewarmed (37 °C) optimized recovery medium to the culture container.
5. Return cultures to the incubator under the same conditions.
6. Replace the medium every other day until confluence is reached (Figure 1).
Notes:
1. Cells typically regain normal morphology within one week, though some cases may require 1–4 weeks to reach confluence.
2. See Troubleshooting if cells remain non-proliferative or fail to reach confluence in a small container (such as 12- or 6-well plates) after one month.
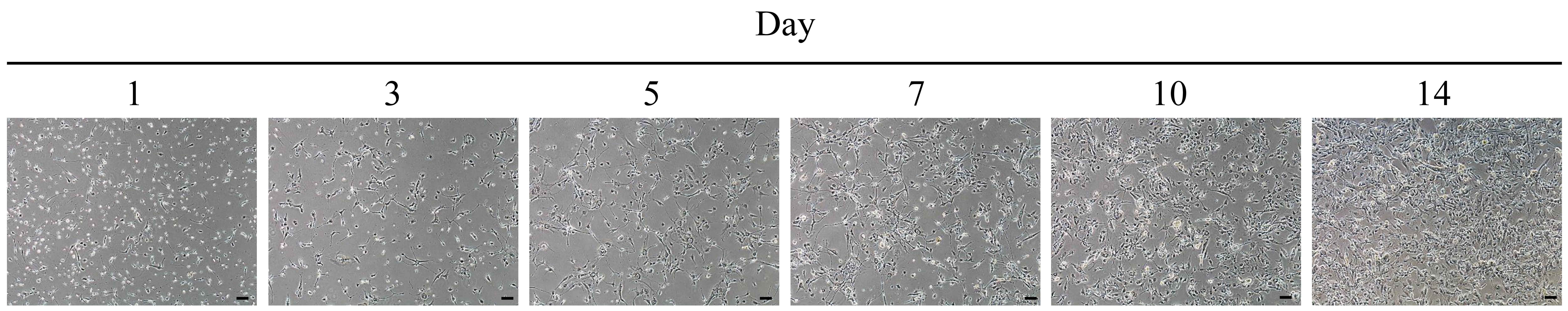
Figure 1. Morphology of long-term cryopreserved patient-derived glioblastoma cells during post-thaw recovery using an optimized recovery protocol. Representative brightfield images show cells regaining adherent morphology and proliferative growth until confluence. Images were acquired at 4× magnification; scale bar, 100 μm.
D. Cell expansion, characterization, and application
1. Sub-culture cells for expansion into larger culture containers (e.g., from 6-well plates into T25 flasks) once cultures reach confluence.
2. Remove the medium and wash cells twice with 1× PBS, following the standard trypsinization protocol.
3. Add 0.05% trypsin solution after washing and completely removing the 1× PBS. Adjust the volume according to container size (see Table 3). Incubate the culture container at 37 °C for 5 min.
Table 3. 0.05% trypsin solution and medium volume according to container size (1:5 ratio, trypsin:medium)
| Container size | 0.05% trypsin solution volume | Medium volume |
| Single well of a 12-well plate | 0.2 mL | 1 mL |
| Single well of a 6-well plate | 0.5 mL | 2.5 mL |
| 25 cm2 cell culture flask (T25) | 1 mL | 5 mL |
| 75 cm2 cell culture flask (T75) | 2 mL | 10 mL |
4. Add prewarmed recovery medium to the trypsinized cells at a ratio of 1:5 (trypsin:medium). Gently flush cells from the surface to ensure complete detachment.
Notes:
1. Cells normally detach after 5 min of incubation. If cells remain adherent, apply gentle tapping.
2. After flushing, ensure minimal or no cells remain attached to the container to avoid loss of yield during transfer.
5. Transfer the cell suspension into a new 15 mL centrifuge tube and centrifuge at 523× g for 5 min to pellet the cells and separate them from the trypsin solution.
6. Remove the supernatant and resuspend the pellet in prewarmed recovery medium (adjust volume to the size of the new container, see Table 2).
7. Transfer the suspension into a new, larger culture container.
Note: Matrigel coating is not required at this stage, as the cells have demonstrated recovery through proliferation and confluence and can be sub-cultured into larger vessels without additional attachment support.
8. Replace the medium every other day until confluence is reached and cells are ready for further subculture.
Note: Once cells expand to T75 flasks, change the medium from optimized recovery medium (20% FBS) to standard culture medium (10% FBS). At this stage, cells are considered fully recovered from cryopreservation and no longer require enhanced nutrient supplementation.
9. Perform characterization after full recovery and expansion in T75 flasks to confirm that recovered GBM cells retain their specific characteristics and marker expression.
a. Flow cytometry: Detect cluster of differentiation (CD) markers on the cell surface. Use CD133 and CD44 as representative GBM markers (Figure 2A).
b. Immunofluorescent staining: Detect GBM-specific markers. Use Nestin and SOX2 as representative GBM markers (Figure 2B).
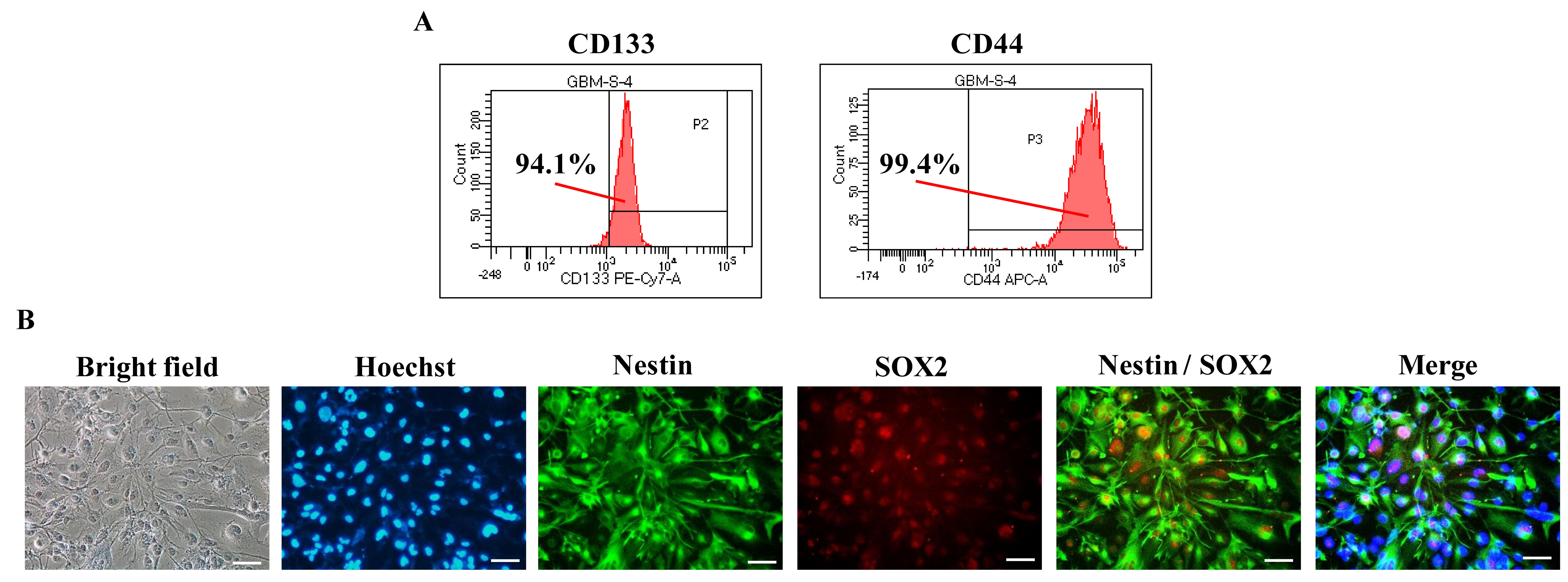
Figure 2. Characterization of recovered long-term cryopreserved patient-derived glioblastoma (GBM) cells following thawing with an optimized recovery protocol. (A) Representative flow cytometry analysis of GBM cell surface markers CD133 and CD44 of recovered GBM cells following the optimized recovery protocol. Numbers indicate the percentage of positive cells for each marker. (B) Representative immunofluorescence images of recovered GBM cells following the optimized recovery protocol. Cells were stained with GBM-specific markers Nestin (green) and SOX2 (red) and with Hoechst (blue) for nuclear staining. Images were acquired at 20× magnification; scale bar, 100 μm.
10. Utilize cells for downstream applications, including drug testing, spheroid formation, and cryopreservation, following confirmation of GBM characterization, as shown in previous studies [10].
Caution: Cells should be used at the earliest possible passages. Based on our laboratory practice, we recommend using cells below 20 passages. Previous studies have reported that patient-derived GBM cultures can typically be maintained for 20–30 passages; however, genomic and transcriptional stability may shift significantly over time [11].
Validation of protocol
This protocol has been used and validated in the following research article:
Khotchawan et al. [10] Recovery of glioblastoma cells derived from patients after a decade of cryopreservation for drug response studies. Science Reports (Figures 1–3, 5–7, and 9).
General notes and troubleshooting
General notes
1. If recovery of the same case fails repeatedly, a new case should be selected to avoid unnecessary resource consumption. Alternatively, consider other recovery assays if continued work with the same case is required.
2. Always keep all tools and materials sterile throughout the process to prevent microbial contamination. This ensures that if cells cannot be recovered using the optimized recovery protocol, the cause can be due to intrinsic cellular factors rather than human error.
3. Always replace the culture medium the day after thawing and passaging cells. This helps remove dead or weak cells and supports the growth of healthy adherent cells.
4. Always keep Matrigel on ice to prevent undesired gel formation during handling.
5. Always record and increase passage numbers after each thawing and passaging step to maintain accurate cell culture history.
Troubleshooting
Problem 1: Repeated failure during thawing of the same case.
Possible cause: The case may have had a low number or initial integrity at the time of cryopreservation.
Solution: Thaw a new vial from the same case or consider using a different case.
Problem 2: After one month, cells remain non-proliferative or fail to reach confluence in small containers (12- or 6-well plates).
Possible cause: The case may have low integrity or be non-responsive to this optimized protocol.
Solution: Thaw a new vial from the same case or switch to a different case. Extending culture beyond one month is not recommended, as this exceeds the limits of prior experience with this optimized protocol.
Acknowledgments
This work was supported by Mahidol University [grant number R016633028 (fund 3)]. The graphical overview was created with Biorender.com. The following figures were created using BioRender: Graphical overview, BioRender.com/nja86mu. This protocol is detailed and expanded from our previous publication [10].
Competing interests
The authors declare no conflicts of interest.
Ethical considerations
The patient-derived glioblastoma cells used in this study were originally collected more than 10 years ago by a previous research group, with informed consent obtained at the time of collection. As all patients have since passed away and the original investigators are now retired, re-contact with legal guardians was not possible. In recognition of the scientific value of these long-term cryopreserved samples, we sought approval from the institutional ethics committee, which reviewed the circumstances and granted authorization for their use in the present study. In addition, this manuscript does not contain any information or images that could lead to identification of the participant. The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Institutional Review Board of Faculty of Medicine Siriraj Hospital, Mahidol University, Bangkok, Thailand (COA no. S0 531/2024, date of approval July 8, 2024).
References
- Pouyan, A., Ghorbanlo, M., Eslami, M., Jahanshahi, M., Ziaei, E., Salami, A., Mokhtari, K., Shahpasand, K., Farahani, N., Meybodi, T. E., et al. (2025). Glioblastoma multiforme: insights into pathogenesis, key signaling pathways, and therapeutic strategies. Mol Cancer. 24(1): e1186/s12943–025–02267–0. https://doi.org/10.1186/s12943-025-02267-0
- Pasupuleti, V., Vora, L., Prasad, R., Nandakumar, D. and Khatri, D. K. (2024). Glioblastoma preclinical models: Strengths and weaknesses. Biochim Biophys Acta Rev Cancer. 1879(1): 189059. https://doi.org/10.1016/j.bbcan.2023.189059
- Khaydukova, I. V., Ivannikova, V. M., Zhidkov, D. A., Belikov, N. V., Peshkova, M. A., Timashev, P. S., Tsiganov, D. I. and Pushkarev, A. V. (2024). Current State and Challenges of Tissue and Organ Cryopreservation in Biobanking. Int J Mol Sci. 25(20): 11124. https://doi.org/10.3390/ijms252011124
- Baust, J. M., Campbell, L. H. and Harbell, J. W. (2017). Best practices for cryopreserving, thawing, recovering, and assessing cells. In Vitro Cell Dev Biol Anim. 53(10): 855–871. https://doi.org/10.1007/s11626-017-0201-y
- Stacey, G. N. and Masters, J. R. (2008). Cryopreservation and banking of mammalian cell lines. Nat Protoc. 3(12): 1981–1989. https://doi.org/10.1038/nprot.2008.190
- Meneghel, J., Kilbride, P. and Morris, G. J. (2020). Cryopreservation as a Key Element in the Successful Delivery of Cell-Based Therapies—A Review. Front Med. 7: e592242. https://doi.org/10.3389/fmed.2020.592242
- Passaniti, A., Kleinman, H. K. and Martin, G. R. (2021). Matrigel: history/background, uses, and future applications. J Cell Commun Signaling. 16(4): 621–626. https://doi.org/10.1007/s12079-021-00643-1
- Weber, T., Malakpour-Permlid, A., Chary, A., D’Alessandro, V., Haut, L., Seufert, S., Wenzel, E. V., Hickman, J., Bieback, K., Wiest, J., et al. (2025). Fetal bovine serum: how to leave it behind in the pursuit of more reliable science. Front Toxicol. 7: e1612903. https://doi.org/10.3389/ftox.2025.1612903
- Sanvoranart, T., Supokawej, A., Kheolamai, P., U-pratya, Y., Poungvarin, N., Sathornsumetee, S. and Issaragrisil, S. (2016). Targeting Netrin-1 in glioblastoma stem-like cells inhibits growth, invasion, and angiogenesis. Tumor Biol. 37(11): 14949–14960. https://doi.org/10.1007/s13277-016-5314-5
- Khotchawan, W., Lorthongpanich, C., Kheolamai, P., Sathornsumetee, S. and Issaragrisil, S. (2025). Recovery of glioblastoma cells derived from patients after a decade of cryopreservation for drug response studies. Sci Rep. 15(1): e1038/s41598–025–22908–0. https://doi.org/10.1038/s41598-025-22908-0
- Baskaran, S., Mayrhofer, M., Kultima, H. G., Bergström, T., Elfineh, L., Cavelier, L., Isaksson, A. and Nelander, S. (2018). Primary glioblastoma cells for precision medicine: a quantitative portrait of genomic (in)stability during the first 30 passages. Neuro Oncol. 20(8): 1080–1091. https://doi.org/10.1093/neuonc/noy024
Article Information
Publication history
Received: Jan 8, 2026
Accepted: Mar 13, 2026
Available online: Mar 23, 2026
Published: Apr 20, 2026
Copyright
© 2026 The Author(s); This is an open access article under the CC BY-NC license (https://creativecommons.org/licenses/by-nc/4.0/).
How to cite
Khotchawan, W., Lorthongpanich, C., Kheolamai, P., Sathornsumetee, S. and Issaragrisil, S. (2026). Recovery and Expansion of Patient-Derived Glioblastoma Cells After Long-term Cryopreservation. Bio-protocol 16(8): e5661. DOI: 10.21769/BioProtoc.5661.
Category
Cancer Biology > General technique > Cell biology assays > Cell viability
Cell Biology > Cell isolation and culture > Cell growth
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link









