- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
A Male Mouse Model of WIN 55,212–2 Self-Administration to Study Cannabinoid Addiction
Published: Vol 16, Iss 7, Apr 5, 2026 DOI: 10.21769/BioProtoc.5652 Views: 26
Reviewed by: Shubham GargFereshteh AzediAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Binging from Food to Alcohol: A Sequential Interaction Between Binging Behaviors in Male Wistar Rats
Sergio Cuesta-Martínez [...] Cruz Miguel Cendán
Aug 5, 2023 1360 Views
Abstract
Despite substantial progress in preclinical cannabinoid research, translational studies on cannabis use disorders (CUD) are still insufficient due to the absence of robust, validated animal models that fully recapitulate the multifactorial clinical phenotype of human CUD. The complex nature of CUD and the incomplete understanding of its underlying neurobiological mechanisms contribute to the limited availability of effective treatments. To address this gap, we developed an operant conditioning–based mouse model that enables the identification of individual vulnerability or resilience to CUD development. This highly translational model is based on the Diagnostic and Statistical Manual of Mental Disorders, 5th Edition (DSM-5) criteria for substance use disorders. The model allows the assessment of addiction-like behaviors by evaluating three behavioral domains: 1) persistence of responding during periods of cannabinoid unavailability, 2) motivation for cannabinoid seeking measured using a progressive ratio schedule, and 3) compulsivity, assessed when cannabinoid reward is paired with an aversive consequence such as a mild electric foot shock. A major strength of this paradigm is its ability to quantify two phenotypic traits proposed as predisposing factors for addiction vulnerability and two parameters related to craving. In addition, the model is specifically designed to evaluate genetic and circuit-level manipulations using chemogenetic approaches, with minor modifications required by surgical viral-vector delivery. Using this protocol, we can determine whether altering the excitability of specific neural networks promotes resilience or vulnerability to developing cannabinoid addiction. Elucidating these mechanisms is expected to facilitate the identification of novel and more effective therapeutic interventions for CUD.
Key features
• Operant conditioning–based mouse model to study cannabis use disorders (CUD) based on DSM-5 substance use disorder criteria.
• Enables assessment of addiction-like behaviors across persistence, motivation (progressive ratio), and compulsivity under punishment, allowing stratification of vulnerable versus resilient individuals.
• Quantifies phenotypic traits linked to cannabinoid addiction vulnerability and behavioral signatures associated with craving for cannabinoids.
• Compatible with genetic and circuit-level manipulations to test how specific neural networks modulate CUD-related behaviors.
Keywords: Cannabis use disorderGraphical overview
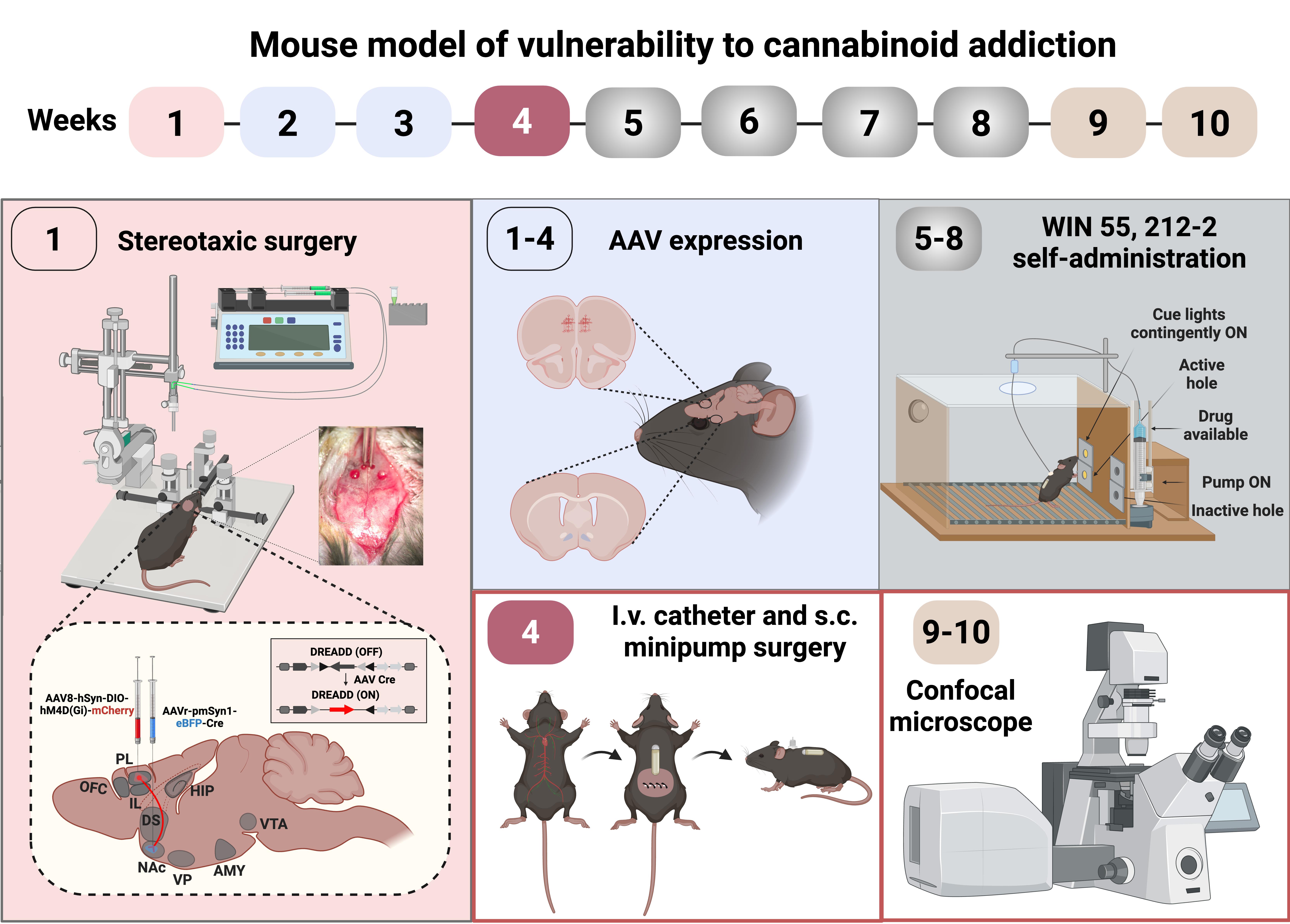
Graphical abstract of the cannabinoid addiction mouse model
Background
Cannabinoids remain the most commonly used illicit drugs worldwide, with consumption increasing in recent years. In Europe, an estimated 8.0%–8.3% of adults aged 15–64 used cannabis in the last year [1]. An increasing perception that cannabinoids are relatively safe has contributed to more permissive attitudes toward use, paralleling rising rates of cannabis use disorder (CUD) [1,2].
CUD is defined as a chronically relapsing neuropsychiatric disorder, diagnosed using the Diagnostic and Statistical Manual of Mental Disorders, 5th edition (DSM-5) criteria, where addiction is equivalent to severe substance use disorder (SUD), requiring fulfillment of at least six of eleven criteria [3]. Cannabis addiction is a complex, multifactorial disorder that emerges from interactions between genetic susceptibility and environmental influences, including epigenetic regulation [4,5]. Not all individuals with repeated exposure transition to addiction, highlighting interindividual variability in vulnerability and resilience [6]. Although numerous cannabinoid-induced neuroadaptations have been described, the mechanisms that specifically confer vulnerability remain incompletely understood, and valid animal models of cannabinoid addiction are still lacking.
Rodent models of operant self-administration have been widely used to study cannabinoid-related behaviors [7–13]. These paradigms have advanced understanding of reward and reinforcement but typically rely on a single behavioral signature and therefore do not capture the multidimensional diagnostic construct of CUD. Moreover, robust self-administration and escalation are more consistently observed with synthetic cannabinoids than with Δ9-tetrahydrocannabinol (THC), whose lower reinforcing efficacy and aversive effects at high doses complicate modeling of cannabis addiction.
The operant conditioning–based protocol described here addresses these limitations by providing a flexible, translational framework to assess addiction-like behaviors induced by synthetic cannabinoids [7–12]. The procedure is aligned with DSM-5 substance use disorder criteria and interrogates three key behavioral domains within the same subjects: (i) persistence of responding during drug unavailability, (ii) motivation to obtain the drug using a progressive ratio schedule, and (iii) compulsive drug seeking when access is paired with an aversive consequence. This design enables longitudinal tracking of addiction-like trajectories at the individual level [7,8].
A major strength of the protocol is its sensitivity to individual differences, allowing classification of animals as vulnerable or resilient based on composite behavioral scores rather than group averages. The task is compatible with viral vector–based approaches and is particularly suited to circuit-specific manipulations, facilitating rapid evaluation of how defined neural networks modulate cannabinoid seeking. Limitations include the need for operant training, specialized equipment, and the potential confounding influence of stress or pain sensitivity on punishment-based measures. Beyond CUD, the framework can be adapted to other drugs of abuse, polysubstance paradigms, developmental exposures, sex differences, and gene–environment interactions, providing a robust platform to dissect the behavioral and circuit-level mechanisms underlying cannabinoid addiction.
Materials and reagents
Biological materials
1. 8–10-week-old C57BL/6J male mice (Charles River, France, or any company that provides mice for laboratories)
Reagents
1. Distilled water
2. Ethanol 70%
3. Iodine, 500 mL (Betadine, MEDA Pharma S.A.U., catalog number: 716720)
4. Physiological saline (0.9%, 250 mL) (Laboratorios ERN, catalog number: 999790.8)
5. Glucose serum (GlucosaVet 5 g/100 mL) (B. Braun Vet Care, catalog number: 1248 ESP)
6. Sodium heparin (Heparina Hospira 5%) (Pfizer, catalog number: 654753.3)
7. Ophthalmic ointment (Xilin night, 5 g) (Visufarma, catalog number: 2919-PS-CM)
8. Blastoestimulina (1%, 30 g) (Almirall, catalog number: 719385)
9. VirkonTM S (Laboratorios Zotal, catalog number: 0065-P)
10. Vetflurane (Isoflurane, 250 mL) (Virbac, catalog number: 2199-ESP)
11. Clozapine N-oxide (CNO, 25 mg) (Enzo Life Sciences, catalog number: BML-NS105-0025); diluted in 0.9% sterile saline (5 mg/mL).
Note: CNO powder is stored at room temperature, protected from light. Stock solutions are prepared in saline, aliquoted, and stored at 4 °C. Working solutions are freshly prepared on the day of injection. Shelf-life: typically stable for 2 years after opening.
12. Anesthesia reagents
a. Ketamine hydrochloride (75 mg/kg of body weight, 10 mL) (Ketamidor) (Richter Pharma AG, catalog number: 580393); dissolved in sterile 0.9% physiological saline
b. Medetomidine hydrochloride (1 mg/kg of body weight) (Domtor) (Esteve, catalog number: 570686); dissolved in sterile 0.9% physiological saline
c. Atipamezole hydrochloride (2.5 mg/kg of body weight) (Revertor) (Virbac, catalog number: 570559); dissolved in sterile 0.9% physiological saline
d. Gentamicine (1 mg/kg of body weight) (Genta-Gobens) (Laboratorios Normon, catalog number: 999037); dissolved in sterile 0.9% physiological saline
e. Meloxicam (2 mg/kg of body weight) (Metacam) (Boehringer Ingelheim, catalog number: 059/02/08CVFPT); dissolved in sterile 0.9% physiological saline
13. Thiopental (Tiobarbital, 0.5 g) (B. Braun)
14. Antibiotic ointment (Bactroban, GlaxoSmithKline)
15. Viral vectors
a. AAV8-hSyn-DIO-hM4D(Gi)-mCherry (1.21 × 1013 gc/mL) (Viral Vector Production Unit of Universitat Autònoma de Barcelona)
b. AAV8-hSyn-DIO-mCherry (1.19 × 1013 gc/mL) (Viral Vector Production Unit of Universitat Autònoma de Barcelona)
c. AAVrg pmSyn1-EBFP-Cre (6 × 1012 vg/mL) (Addgene, catalog number: 51507-AAVrg)
Note: Viral vectors are stored at –80 °C upon arrival and aliquoted to avoid repeated freeze–thaw cycles. Vials are kept on ice during surgical procedures and used within the manufacturer’s recommended stability period. Shelf-life: typically stable for 2 years at -80 °C.
16. WIN 55,212-2 [(R)-(+)-WIN 55,212-2 mesylate salt] (Sigma-Aldrich, catalog number: W102
17. Tween 80 (Sigma-Aldrich, catalog number: P8074)
18. 4% paraformaldehyde (PFA) in 0.1 M Na2HPO4/0.1 M NaH2PO4 buffer (PB) (Sigma-Aldrich, catalog number: P6148)
19. Sucrose (Sigma-Aldrich, catalog number: S7903)
Solutions
1. Heparin 0.0003 mg/mL (see Recipes)
2. Ketamine (7.5 mg/mL) + Medetomidine (0.2 mg/mL) (see Recipes)
3. Atipamezole 1 mg/mL (see Recipes)
4. Gentamicin 0.3 mg/mL (see Recipes)
5. Meloxicam 0.5 mg/mL (see Recipes)
6. WIN 55,212-2 (see Recipes)
Recipes
1. Heparin 0.0003 mg/mL
0.12 mL of sodium heparin + 20 mL of sterile 0.9% physiological saline.
Note: Store heparin at 4 °C in sterile stock vials and prepare fresh working solutions on the day of use to ensure stability and sterility.
2. Ketamine (7.5 mg/mL) + Medetomidine (0.2 mg/mL)
0.75 mL of ketamine hydrochloride (100 mg/mL Ketamidor) + 1 mL of medetomidine hydrochloride (1 mg/mL, Dormitor) + 8.25 mL of sterile 0.9% physiological saline.
3. Atipamezole 1 mg/mL
2 mL of atipamezole hydrochloride (2.5 mg/kg of body weight, Revertor) + 8 mL of sterile 0.9% physiological saline.
4. Gentamicin 0.3 mg/mL
0.06 mL of gentamicin (40 mg/mL, Genta-Gobens) + 10 mL of 0.9% physiological saline.
5. Meloxicam 0.5 mg/mL
0.5 mL of meloxicam (40 mg/mL, Metacam) + 40 mL of glucose serum (GlucosaVet 5 g/100 mL).
6. WIN 55,212-2
Dissolve WIN 55,212-2 powder in one drop of Tween 80 and store at room temperature, protected from light. Prepare stock solutions in saline, aliquot, and store at 4 °C. Prepare working solutions freshly on the day of injection and self-administration session. Shelf-life: typically stable for 2 years.
Laboratory Supplies
1. Scalpel (Agnthos, catalog number: 02-036-04)
2. Manual drill (Plastics One, catalog number: DH-1)
3. Blunt-tipped surgical scissors (Agnthos, catalog number: 03-022-105)
4. Curved iris forceps (Agnthos, catalog number: 08-513-005)
5. Suture clips (Agnthos, catalog number: 08-922-125)
6. Surgical clips (Agnthos, catalog number: 22-620-007)
7. Fine scissors 105 mm-4¼ (Allgaier Instrumente, catalog number: 03-320-105)
8. Curved forceps (Allgaier Instrumente, catalog number: 08-421-100)
9. Bishop-Harman forceps serrated 0.5 mm (Agnthos, catalog number: 11069-08)
10. Graefe forceps straight serrated 0.8 mm (Agnthos, catalog number: 11050-10)
11. 2× Dumont 5 forceps (Fine Science Tools, catalog number: 11251-10)
12. Rolled cotton 100% (Acofarma, Acofar®, catalog number: 4957051)
13. Weighing scale
14. Tygon tubing (0.508 × 1.524 mm) (Tecny fluor, catalog number: S-54-HL)
15. Catheter for mouse (mouse jugular catheter) (Sai Infusions, model: MJC-01)
16. Microsyringe (10 μL, model 1701 N S.Y.R., cemented N.D.L., 26 ga, 2 in, point style 3) (Hamilton company, catalog number: 80039)
17. Polyethylene tubing (PE-20, 0.381 × 1.092 mm) (Plastics One, catalog number: PT-C315CT)
18. Bilateral guide cannula (26-gauge cannula cut 12 mm below pedestal) (Plastics One, catalog number: C235GS-5/Spc)
19. Bilateral internal cannula (33-gauge internal cannula fits 12 mm with 3 mm projection) (Plastics One, catalog number: C235GS-5/Spc)
20. Osmotic minipumps (flow rate of 0.25 μL/h for 28 days) (ALZET, model: 2004, catalog number: 0000298)
21. Suture thread (black braided silk, TB10, 3/8 TRIANG 15 mm 4/0 90 cm) (LorcaMarín, catalog number: 55327-50U)
22. 20 gauge 1½ inch needles (BD Microlance 3, catalog number: 301300)
Equipment
1. Mouse operant self-administration chambers (Med Associates, model: ENV-307A-CT)
Operant chambers are equipped with two nose-poke holes (holes of 1.2 cm diameter), one randomly selected as the active nose poke and the other as the inactive. Nose-poking on the active nose poke results in an infusion of the synthetic cannabinoid agonist WIN 55,212-2 paired with a stimulus-light (2 s, associated-cue) (Med Associates, catalog number: ENV-321M), located above the active nose poke. Nose-poking on the inactive hole has no consequences. A house light is located on the ceiling of each chamber (Med Associates, catalog number: ENV-315M), and two cue lights are positioned at the active nose poke, one inside the hole and one above it (Figure 1). Nose pokes at the active hole trigger a WIN 55,212–2 infusion according to the programmed schedule, coincident with illumination of the cue light above the hole, whereas responses at the inactive hole have no programmed consequences. Chambers consist of aluminum and acrylic walls, enclosed within sound- and light-attenuating cubicles, equipped with ventilating fans that also provide constant white noise at 55–65 dBA. The floor consists of parallel metal bars that deliver foot shocks (0.18 mA) during the punishment test for compulsive-like behavior (Med Associates, catalog number: ENV-307A-GF). The synthetic full agonist cannabinoid WIN 55,212–2 (12.5 μg/kg/infusion) is infused in a volume of 23.5 μL over 2 s via a syringe mounted on a microinfusion pump (Med-Associates, model: PHM-100A), connected through flexible polymer Tygon tubing (0.96 mm outer diameter) (Tecny fluor) to a single-channel liquid swivel (Polysulfone 22 GA single-channel disposable swivel 610014; Harvard apparatus/Panlab) and the mouse intravenous catheter (Sai Infusions, U.S.A). Although Med Associates equipment was used in the present study, functionally equivalent operant conditioning systems and programmable infusion pumps from other manufacturers may be used if they allow precise control of stimulus presentation, response detection, and infusion parameters.
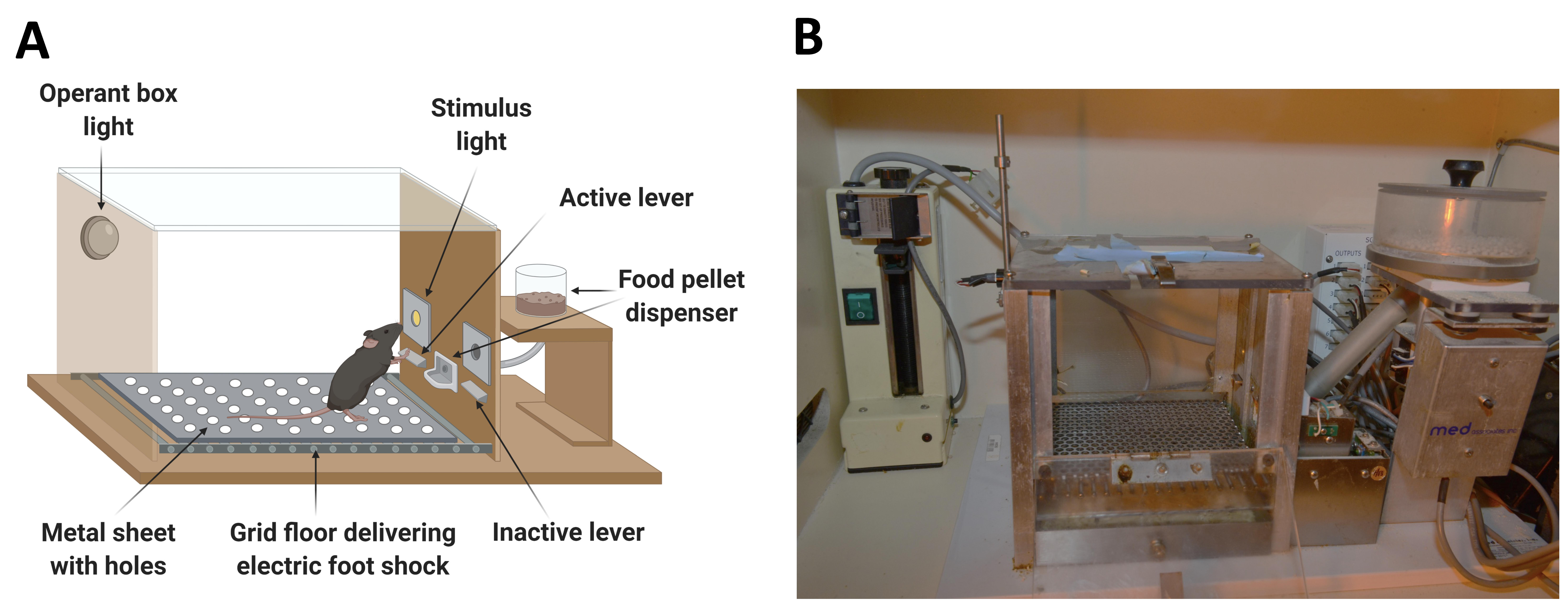
Figure 1. Operant self-administration chamber. (A) Diagram of the operant chamber. The operant chamber is equipped with two nose pokes (active and inactive), a stimulus light, and an infusion pump. The chamber floor is a grid that delivers electric foot shocks during the shock test session. (B) General view of the operant box with the box light, the active nose poke, the cue-light, the grid floor that delivers electric foot shocks, and the infusion pump for drug delivery.
2. Stereotaxic apparatus (100 µm resolution) (Koft instruments, model: 900)
3. Standing magnifier (Carl Zeiss, model: OPMI 1 FR)
4. Microinfusion pump (P.H.D. 2000, Harvard Apparatus, catalog number: MA1 70-20xx)
5. Animal trimmer (Artero, catalog number: M630)
6. Cold light (Leica C.L.S. 150×, Leica Microsystems)
7. Heating pad (60 W, 50 Hz) (Daga, catalog number: N2P 220-230)
8. Hot bead sterilizer (FST 250) (Agnthos, catalog number: 18000-45)
9. Activity boxes (10.8 × 20.3 × 18.6 cm) (Imetronic, Pessac, France)
Software and datasets
1. Med-PC 5 software (Med Associates Inc., U.S.A.) to record all operant behavior in the self-administration chambers
Procedure
The protocol overview begins with stereotaxic surgery to administer the combinatorial viral vector approach, using retro-Cre and inhibitory DREADDs, to reduce the excitability of prelimbic (PL) medial prefrontal cortex (mPFC) neurons projecting to the nucleus accumbens (NAc). After 4 weeks of viral expression, surgeries of intrajugular catheter implantation and minipump subcutaneous insertion are performed. Following surgical recovery, operant conditioning maintained by the cannabinoid full agonist WIN 55,212-2 is established, finalizing with the animals’ categorization into addicted and non-addicted mice and the characterization of phenotypic vulnerability traits. Subsequently, extinction and cue-induced reinstatement are tested to obtain parameters related to craving (Figure 2).
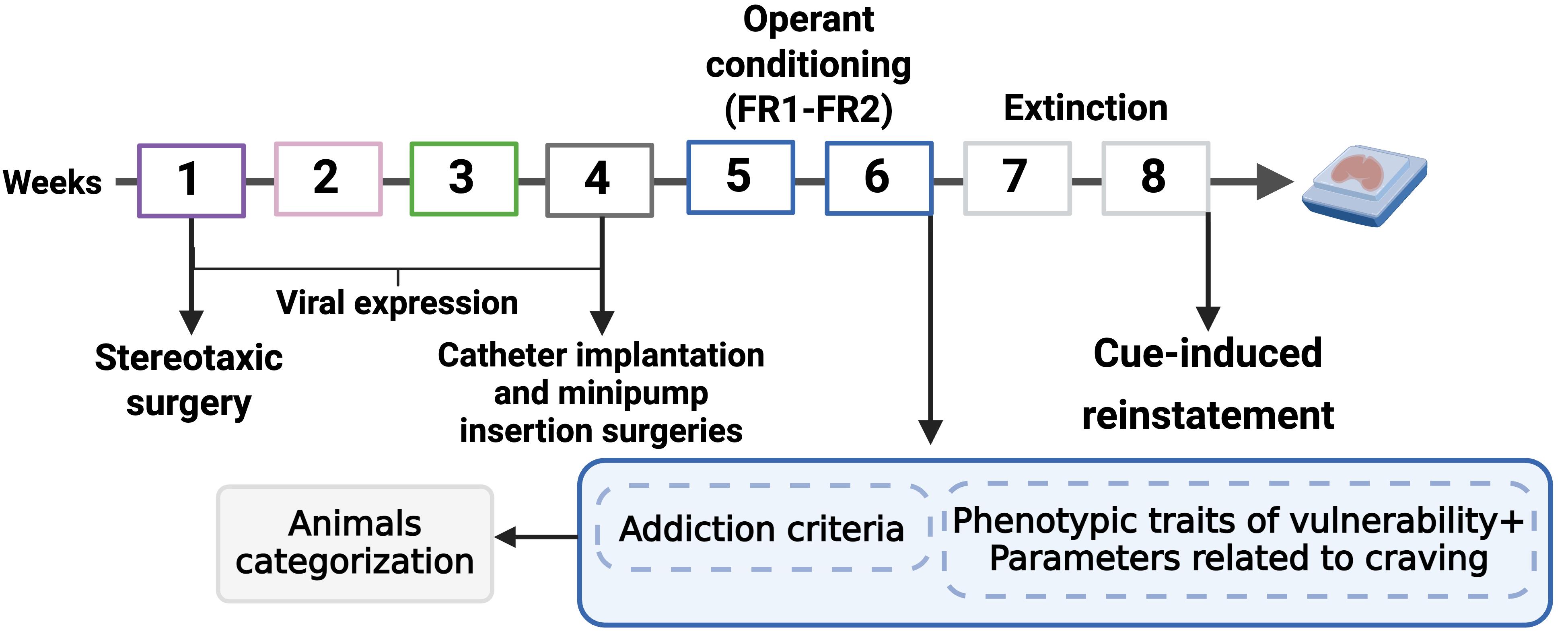
Figure 2. Complete experimental protocol of our mouse model of operant WIN 55,212–2 intravenous self-administration
A. Stereotaxic surgery: DREADD approach with viral vector microinjection
1. Prepare adeno-associated viral vectors: AAV-hM4Di-DREADD [AAV8-hSyn-DIO-hM4D(Gi)-mCherry, 1.21 × 1013 gc/mL] and AAV-retrograde-Cre-EBFP (AAV pmSyn1-EBFP-Cre, 8.2 × 1012 gc/mL).
2. Anesthetize mice by intraperitoneal injection (0.2 mL/10 g) of ketamine hydrochloride (75 mg/kg) and medetomidine hydrochloride (1 mg/kg) in 0.9% sterile saline, and place them in a stereotaxic frame for intracranial adeno-associated virus (AAV) injections.
3. Perform bilateral microinjections using a 26-gauge guide cannula and a 33-gauge internal injection cannula connected via PE-20 tubing to a 10-μL microsyringe; monitor infusion by displacement of an air bubble in the tubing.
4. For PL–NAc pathway inhibition, inject 0.2 μL/site of AAV-hM4Di-DREADD into PL at 0.05 μL/min and 0.4 μL/site of AAV-retrograde-Cre-EBFP into NAc core at 0.10 μL/min. Leave the cannula in place for 10 min post-infusion to allow diffusion, then withdraw slowly while maintaining body temperature at 35 °C on a heating pad; target coordinates (Paxinos and Franklin, 1997): PL, AP +2.10 mm, L ±0.3 mm, DV -2.3 mm; NAc core, AP +1.94 mm, L ±1.0 mm, DV -4.6 mm.
A1. Stereotaxic surgery (Figure 3)
Note: Sterilize the surgical material with a hot bead sterilizer before each animal surgery.
1. Injection cannulas circuit preparation.
a. Remove the microsyringe plunger (10 μL).
b. Attach the polyethylene tubing (50 cm) to the microsyringe.
c. Inject distilled water through the tubing, using a syringe, filling the tubing and the microsyringe. Verify that distilled water flows out through the back of the microsyringe.
d. Insert half of the plunger into the microsyringe.
e. Connect one of the sides of the bilateral internal cannula (33 gauge) to the tubing. Verify that the circuit is assembled correctly by pressing the microsyringe plunger and observing that water flows from the cannula without leakage.
f. Repeat these steps with a second microsyringe, which must be attached to the other side of the same internal cannula as the first microsyringe.
g. Place the microsyringes in the microinfusion pump.
h. Aspirate 1 μL of air into the tubing to form a bubble and monitor the microinjections.
i. Aspirate 4 μL of the viral vector (previously defrosted with ice).
2. Stereotaxic apparatus preparation.
a. Place and fix the bilateral guide cannula in the metal grip of the stereotaxic holder.
b. Insert the bilateral internal cannulae, attached to the tubing, into the bilateral guide cannulae. The bilateral guide cannula is used to keep the internal cannula straight and well-fitted into the holder, and to determine Bregma coordinates before the AAV is loaded. Note that only the internal cannula, filled with the viral vector, penetrates the brain.
3. Mouse preparation.
a. Anesthetize the mouse with a mixture of ketamine hydrochloride and medetomidine hydrochloride dissolved in sterile 0.9% physiological saline. Administer intraperitoneally (75 mg/kg and 1 mg/kg of body weight, respectively).
b. Shave the mouse’s head.
c. Place the mouse in the stereotaxic apparatus. Check that the head is fixed using the ears bars and the nose clamp.
d. Place a heating pad below the mouse to maintain proper body temperature during the surgery.
4. Stereotaxic surgery.
a. Perform a vertical cut in the middle of the head with a scalpel.
b. Scratch the skull carefully to visualize the bregma accurately.
c. Place the cannulas above the bregma and annotate the bregma coordinates.
d. Calculate medial-lateral, dorsal-ventral, and anteroposterior coordinates of your target area where the injection has to be performed.
e. Move the cannulas to the specific injection point by adjusting the stereotaxic apparatus using the medial-lateral, dorsal-ventral, and anterior-posterior readings.
f. Mark the injection point on the skull using a pencil.
g. Following the pencil mark, drill holes into the skull using a manual drill.
h. Clean the injection area with physiological saline.
i. Insert the bilateral cannula into the holes and readjust the dorsal-ventral coordinate.
j. Place the bilateral cannulas precisely at the calculated coordinates.
k. Mark the two sides of the air bubble on the tubing to monitor the injection.
l. Perform viral vector injection.
i) Microinfusion pump parameters [example for prelimbic (PL) and nucleus accumbens core (NAc core) areas]: Target volume: PL 0.2 μL; NAc core 0.4 μL. Infusion rate: PL 0.05 μL/min; NAc core 0.1 μL/min. Infusion time: 4 min.
ii) Leave the cannula in place for 10 min after infusion to prevent reflux.
iii) Withdraw the cannula slowly for 10 min.
m. Clean the injection area with physiological saline.
n. Sew the incision with suture thread.
o. Apply blastoestimulina on the sutured incision to promote proper healing.
p. Inject subcutaneously atipamezole hydrochloride (2.5 mg/kg of body weight) dissolved in sterile 0.9% physiological saline to reverse the anesthesia.
q. Inject intraperitoneally gentamicin (1 mg/kg of body weight) dissolved in sterile 0.9% physiological saline.
r. Inject meloxicam subcutaneously (2 mg/kg of body weight) dissolved in sterile 0.9% physiological saline.
s. Inject glucose serum (0.8 mL) subcutaneously.
t. Place the animal on a heating pad until the animal is awake.
u. During the following 3 post-surgery days, check the mouse and inject 0.8 mL of glucose serum every day.
5. Cleaning.
a. Clean the tubing and cannulas with ethanol, then rinse with distilled water and air-dry.
b. Clean all the surgical material with soap and water.
Note: After bilateral intracranial injection, 4 weeks are required for proper expression of the viral vector.
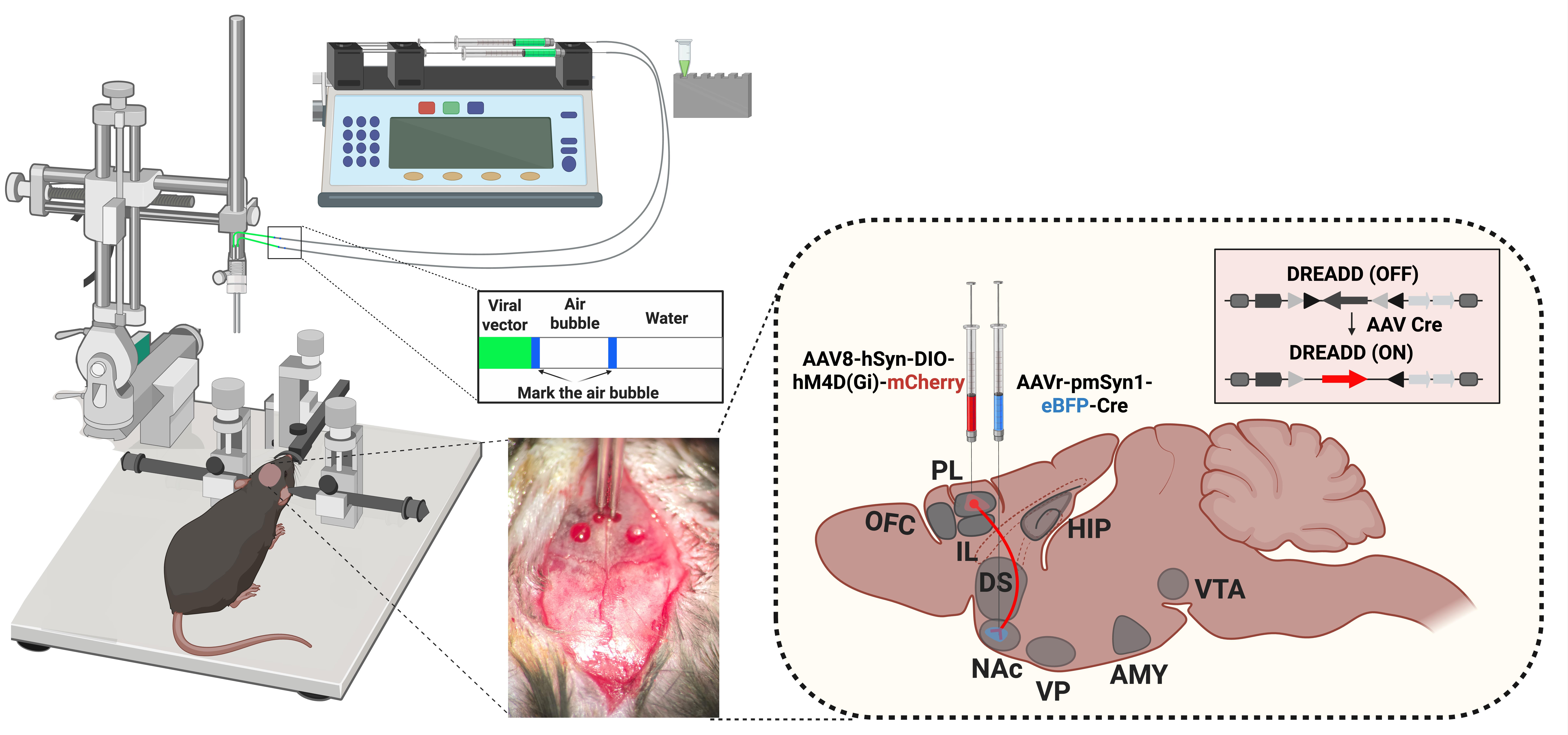
Figure 3. Stereotaxic surgery. Mice are anesthetized and placed in a stereotaxic apparatus for intracranial injection of the viral vector. All the injections are made through a bilateral injection cannula connected to a polyethylene tube. A displacement air bubble within the length of the polyethylene tubing connecting the syringe to the injection needle is used to monitor microinjections. The viral vector is injected at a constant rate by using a microinfusion pump. After infusion, the injection cannula is left in place for an additional 10 min to allow diffusion and prevent reflux, after which it is slowly withdrawn over 10 additional min. In the drawing on the right, the scheme of the mouse brain is shown, with the AAV8-hSyn-DIO-hM4(Gi)-mCherry injection site indicated in the prelimbic (PL) medial prefrontal cortex (mPFC) and the AAVr-pmSyn1-eBFP-Cre in the nucleus accumbens (NAc).
Note: Frequently verify the correct displacement of the air bubble within the polyethylene tube to prevent injection errors caused by cannula obturation or tube fugues.
B. Jugular vein catheterization and osmotic minipump implantation
Note: Jugular vein catheterization and minipump implantation procedures are performed after 4 weeks of viral expression, as shown in the experimental schedule (Figure 2).
1. Anesthetize mice by intraperitoneal injection (0.2 mL/10 g body weight) of ketamine hydrochloride (75 mg/kg) and medetomidine hydrochloride (1 mg/kg) dissolved in 0.9% sterile physiological saline.
2. Implant indwelling intravenous silicone catheters in the right jugular vein as described previously [14]: adapt 6-cm silicone tubing (inner diameter 0.3 mm, outer diameter 0.6 mm) to a 22-gauge steel cannula bent at a right angle and embedded in a dental cement disk with nylon mesh underneath. The step-by-step procedure for jugular vein catheterization (Figure 4) is as follows:
a. Anesthetize the mouse with a ketamine/medetomidine solution (0.2 mL/10 g, intraperitoneal).
b. Apply ophthalmic gel (e.g., xylin) to both eyes to prevent corneal drying during surgery.
c. Shave the chest and back, then disinfect the skin by wiping with 70% ethanol followed by iodine solution over the same area.
d. Identify the interscapular region and make a 1–2 cm skin incision parallel to the spine. Using scissors, gently separate the skin from the underlying musculature to create a subcutaneous pocket for the catheter.
e. With curved forceps, further blunt-dissect the subcutaneous space toward the right shoulder, where the external part of the cannula of the silicon tube will be positioned.
f. Turn the animal onto its back and locate the heartbeat. On the upper right thoracic region, grasp the skin overlying the expected course of the right jugular vein with forceps, make a small skin incision, and bluntly dissect toward the subcutaneous tunnel previously created from the back.
g. Pass the forceps from the thoracic incision through the subcutaneous tunnel to the dorsal incision and pull the catheter through so that it lies over the right shoulder. Confirm catheter integrity by gently flushing with heparinized saline (0.1% heparin in saline).
h. Expose the right jugular vein by carefully removing overlying fat and muscle fibers.
i. Once the vein is visible, insert fine pointed forceps (e.g., Dumont) beneath the vein from the right side, and a second pair from the left side; rotate the forceps to free the vein from surrounding tissue.
j. Pass a bent silk suture under the vein from left to right, then cut the loop to obtain two separate threads. Tie the caudal (lower) thread loosely around the limb to keep it in place. With the cranial (upper) thread, apply a firm single knot to transiently occlude blood flow.
k. Choose a straight segment of the vein for catheterization and puncture it with a 20 G 1½ needle. Remove the needle and insert the catheter guide fully through the needle tract into the lumen (Figure 4A).
l. Advance the silastic catheter tubing into the jugular vein until the silicone bead reaches the vessel entry. Verify correct placement by observing blood backflow into the catheter.
m. Tighten the lower ligature to secure the catheter and slowly inject 0.2 mL of heparinized saline (0.1% heparin) to fill the catheter with fluid (Figure 4B).
n. Tie additional knots with both upper and lower silk threads to firmly anchor the catheter, then add two extra knots on the left and two on the right to stabilize the assembly.
o. Ensure that approximately 1.5 cm of catheter tubing lies within the right jugular vein and that the remaining tubing runs subcutaneously to the dorsal cannula exiting at the midscapular region.
p. Close the thoracic skin incision with sutures and apply a thin layer of antibiotic ointment (e.g., Bactroban).
q. Infuse 0.025 mL heparinized solution (0.3 mg/kg) through the catheter and cap the external port to prevent air entry. Position the tulle of the catheter under the dorsal skin, suture the skin over it to secure the tubing, and close firmly (Figure 4C).
r. Apply topical healing ointment (e.g., Blastoestimulina) to all sutured areas. Administer atipamezole (2 mg/kg, i.p.) to reverse anesthesia, followed by gentamicin (1 mg/kg, s.c.), meloxicam (2 mg/kg, s.c.), and 0.9 mL of glucose-containing saline subcutaneously for hydration (Figure 4B).
s. Allow animals to recover for 3 days before initiating intravenous self-administration sessions.
t. Evaluate catheter patency after the final self-administration session and whenever behavior deviates markedly from previous performance. Assess patency by injecting thiopental (Tiobarbital, 0.5 g) through the catheter at 1.25 mg/kg; if clear anesthetic signs are not evident within 3 s, exclude the mouse from further analysis. For a 12-day protocol, catheter patency should be maintained in >80% of animals. Loss of catheter patency is minimized through careful handling and delicate manipulation of the catheter assembly throughout the experiment. Indicators of catheter blockage or failure include a sudden loss of infusion-contingent responding, resistance during catheter flushing, backflow or leakage at the access port, and subcutaneous swelling observed during infusion. Animals showing evidence of catheter malfunction should be excluded from further testing and subsequent analyses. Final confirmation of catheter patency is performed at the end of the experiment using a thiopental test; however, routine monitoring throughout the study should be conducted to detect potential issues early and prevent data loss.
3. Insert the catheter tubing 1.1 cm into the right jugular vein and secure with a suture; tunnel the remaining tubing subcutaneously to the cannula, exiting at the midscapular region. Suture all incisions and coat with local analgesic (Blastoestimulina).
4. Provide post-surgical care consisting of intraperitoneal antibiotic (gentamicin, 1 mg/kg), subcutaneous analgesic (mixture of glucose serum and meloxicam, 2 mg/kg), and subcutaneous anesthesia reverser atipamezole hydrochloride (2.5 mg/kg), all dissolved in 0.9% sterile physiological saline. Allow mice to recover for 3 days with follow-up analgesia before starting self-administration sessions.
5. At the end of the self-administration sequence, assess intravenous catheter patency by thiopental sodium test; remove any mouse from the study if clear signs of anesthesia are not observed immediately after injection.
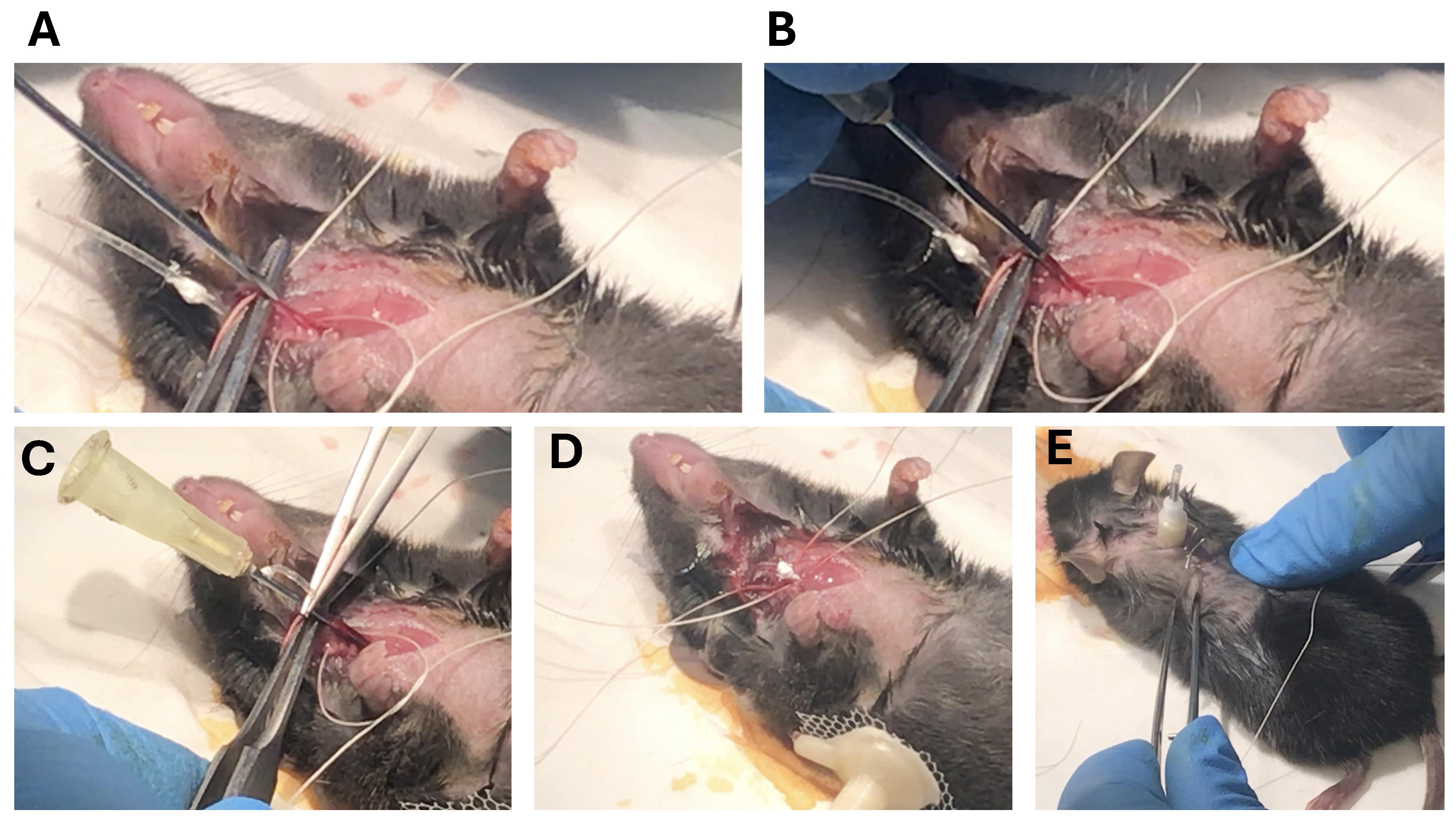
Figure 4. Surgical procedure for intravenous jugular catheterization. Once the jugular vein is located (A), two threads are tied around the vein, using the upper thread to cut the blood supply. Using a 20 G 1½ needle, the vein is punctured (B), the cannula is introduced into the jugular vein (B), and the silicone tube is advanced into the vein (C) until it reaches the catheter’s silicone ball. Then, after checking the catheter entry site, the lower knot is tightened to secure the catheter, and heparin is administered to maintain catheter patency (D). After securing the catheter with a net of sutures, the catheter tulle is placed under the skin, and the skin is sutured over the catheter to fix the tube (E).
6. Prepare Alzet osmotic minipumps filled with clozapine-N-oxide (CNO; 5 mg/mL in 0.9% saline) or vehicle, and implant them subcutaneously in the lower back during jugular vein catheterization surgery to deliver CNO at 0.25 μL/h for 15 days [15]. The step-by-step osmotic minipump implantation protocol (Figure 5) is as follows:
a. Anesthetize the mouse with isoflurane and fix its front and hind legs with tape.
b. Shave the mouse’s lower back.
c. Clean the shaved lower back with ethanol.
d. Make a horizontal cut (1.5 cm), taking as a reference the beginning of the hind legs.
e. Separate the skin from the back muscle, introducing blunt-tipped scissors through the cut.
f. Introduce the osmotic minipump previously filled with CNO (diluted in 0.9% sterile saline; 5 mg/mL) or saline subcutaneously in the middle of the back. The pump cap is in the direction of the animal’s head.
g. Close the cut with surgical clips (approximately 4).
h. Apply iodine to the area.
Note: Correct subcutaneous implantation of the osmotic minipump and meticulous wound closure were performed to minimize the risk of pump displacement, extrusion, or postoperative infection. Under these conditions, incorrect implantation was rarely observed. Animals with suspected pump displacement were excluded from subsequent behavioral testing, and correct pump positioning was verified post-mortem when necessary.
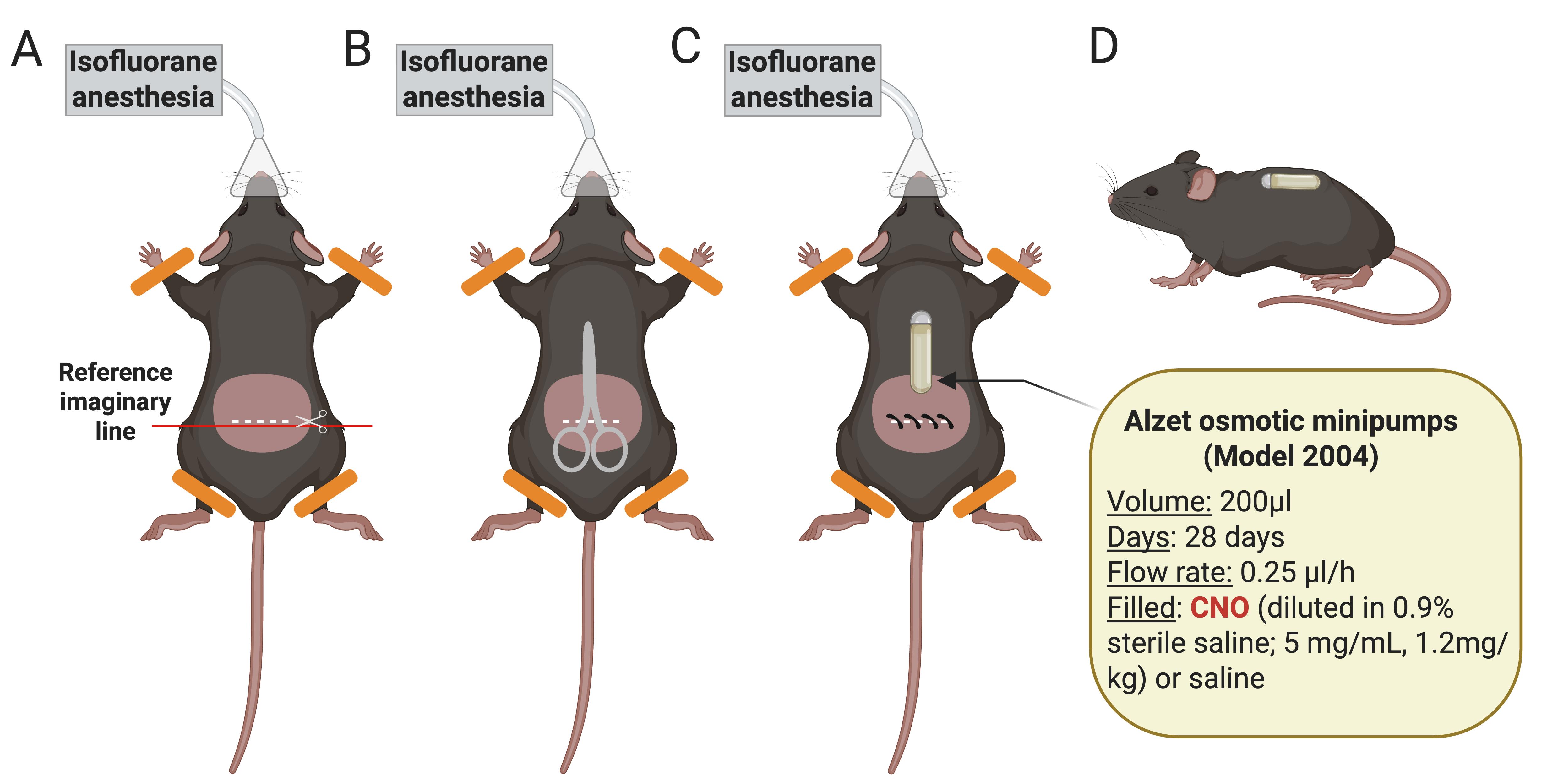
Figure 5. Schematic representation of the osmotic minipump implantation process. (A) Anesthetize the mouse with isoflurane and shave its lower back. Clean the shaved area with ethanol. Make a horizontal cut (1.5 cm), taking as a reference the beginning of the hind legs. (B) Separate the skin from the back muscle, introducing blunt-tipped scissors through the cut. (C) Introduce the osmotic minipump previously filled with CNO or saline subcutaneously in the middle of the back. The pump cap is oriented toward the animal’s head. Close the cut with surgical clips and apply iodine to the area. (D) Side view showing the animal awake with the subcutaneously implanted minipump [15].
Note: Check the correct placement of the minipump on the following days after surgery. If necessary, perform a smooth massage at the minipump placement site to facilitate relocation and prevent skin wounds.
C. WIN 55,212-2 self-administration training
1. Apply an operant drug self-administration procedure based on previously established paradigms [7–10,12,16,17].
2. To minimize acute behavioral suppression and potential aversive or dysphoric-like effects associated with the first exposure to WIN 55,212-2 (e.g., hypolocomotion and anxiety-like responses), mice receive an intraperitoneal injection of WIN 55,212-2 (0.1 mg/kg) 24 h before the first self-administration session [12,18].
3. Train mice to self-administer WIN 55,212-2 intravenously under a fixed-ratio (FR) 1 schedule for 5 consecutive daily sessions, then switch to an FR2 schedule for an additional 5 sessions; conduct all sessions at the same time each day.
4. Start each session with an automatic priming infusion of WIN 55,212-2 via the catheter, followed by two 55-min active periods separated by a 15-min drug-free interval (total session length, 125 min) [11,12,16]. Before connecting the mouse, we press the infusion pump button to produce the sound as a conditioned stimulus.
5. Signal session onset by turning on the house light for 3 s; present cue lights and infusion pump noise as conditioned stimuli during each drug delivery. Implement a 10-s time-out after each infusion, during which the cue light is off, and responses at the active nose poke do not yield drug. Record all responses at active and inactive holes, including those during time-outs.
6. During the drug-free period, deliver neither drug nor cues, and indicate this phase by activating the house light. Terminate the session when either 50 reinforcers have been delivered or 125 min have elapsed, whichever occurs first.
7. Define acquisition of self-administration when all three criteria are met: (i) response rates show at least 80% stability across three consecutive sessions (variance ≤ 20%), (ii) ≥75% of total responses occur at the active hole, and (iii) mice obtain a minimum of five reinforcers per session. Return mice to their home cages after each session. Animals are assigned to home cages in the same room as the operant chambers.
C1. WIN 55,212-2 self-administration training (Figure 6)
1. Twenty-four hours before operant training, an intraperitoneal injection of WIN 55,212-2 (0.1 mg/kg) is performed. Operant conditioning is performed during viral vector expression, which is chronically activated by CNO.
2. Sessions 1–5 (FR1): During these sessions, operant conditioning under the FR1 schedule of reinforcement is applied.
3. Sessions 5–7 (FR2): During these sessions, operant conditioning under the FR2 schedule of reinforcement is performed.
4. Sessions 8–10 (FR2): FR2 sessions to evaluate the criterion of persistence of response and the phenotypic traits of impulsivity and sensitivity to reward.
5. Session 11: Progressive ratio test to evaluate the motivation criteria.
6. Session 12 (FR2): FR2 session before shock test.
7. Session 13: Shock test to evaluate the compulsivity criterion.
8. Sessions 14–23: The extinction phase is applied to evaluate the first parameter related to craving by removing the cue-light, the infusion of WIN 55, 212-2, and the pump noise.
9. Session 24: Cue-induced cannabinoid-seeking test to evaluate the second parameter related to craving.
Note: In cases of failure to acquire self-administration, verify that the operant chamber is functioning properly, ensure that the infusion system is unobstructed, confirm that the animal is correctly connected to the infusion line, and check that the tubing length permits free and unrestricted movement within the chamber. It is also important to assess whether excessive anxiety or stress may be interfering with task engagement; if so, additional habituation and gentle handling may help reduce potential confounding factors. In cases of excessive inactive responding, confirm correct nose-poke functioning and accurate response registration. These measures facilitate distinguishing technical malfunction from behavioral factors and improve protocol reproducibility.
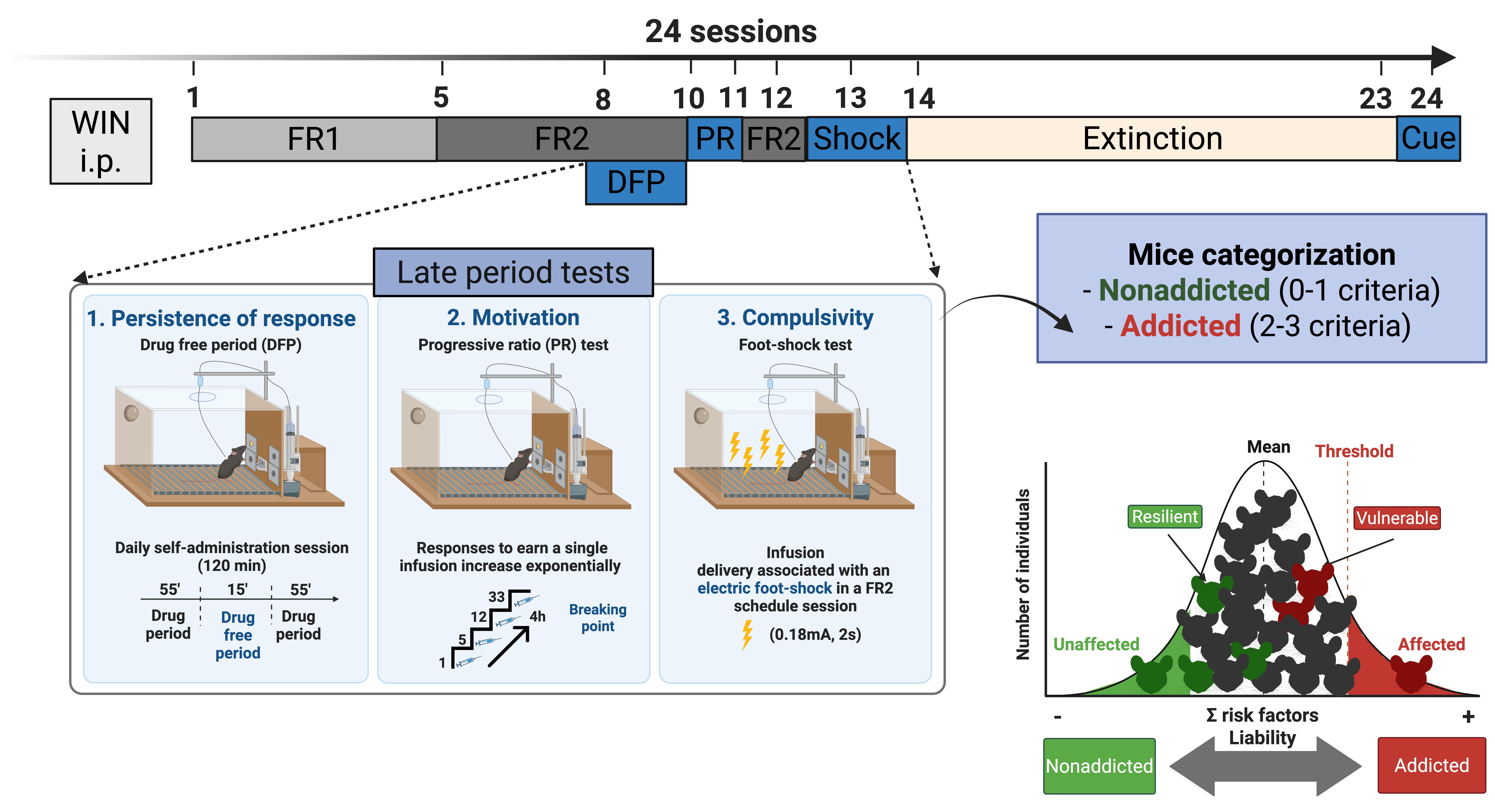
Figure 6. Timeline of the experimental sequence in the cannabinoid addiction mouse model. An intraperitoneal injection of WIN 55,212-2 (0.1 mg/kg) is administered 24 h before the first self-administration session to minimize the aversive effects of the initial cannabinoid exposure. During the first week of operant training, mice acquire WIN 55,212-2 self-administration under fixed-ratio (FR) schedules, with 5 sessions at FR1 followed by 5 sessions at FR2. Sessions 1–10 and 12 include a 15-min drug-free period (DFP), but only the latest 8–10 sessions are used to quantify persistence of responding, the first addiction-like criterion. In session 11, motivation for WIN 55,212-2 is assessed using a progressive-ratio (PR) schedule, providing the second criterion. FR2 session is then re-established in session 12 to stabilize responding before punishment testing. In session 13, compulsivity-like behavior is evaluated in a foot-shock punishment test, yielding the third diagnostic criterion and allowing classification of mice as addicted or non-addicted. Extinction training is conducted in sessions 14–23, and in session 24, a cue-induced reinstatement test is performed to probe relapse-like drug seeking and obtain the parameters related to craving.
D. Three addiction criteria
1. At the end of training, evaluate addictive-like behavior using three DSM-5–based criteria (persistence, motivation, compulsivity) (Figure 7) to compute an addiction score [6,17,19].
2. Persistence of responding (drug seeking): Quantify the number of non-reinforced active nose pokes emitted during the 15-min drug-free period on each of the 3 days preceding the progressive ratio (PR) session.
3. Motivation: Assess motivation for the drug using a PR schedule in which the response requirement for each successive infusion follows the series 1, 2, 3, 5, 12, 21, 33, 51, 75, 90, 120, 155, 180, 225, 260, 300, 350, 410, 465, 540, 630, 730, 850, 1000, 1200, 1500, 1800, 2100, 2400, 2700, 3000, 3400, 3800, 4200, 4600, 5000, and 5500; define the breakpoint (motivation value) as the last ratio completed, with the PR session lasting up to 4 h or ending earlier if mice cease to respond at all nose pokes for 1 h.
4. Compulsivity (resistance to punishment): After a stabilizing FR2 self-administration session following the PR test, place mice in a different operant box and run a 50-min FR2 session in which each infusion is paired with foot-shock punishment; after the first active response, deliver a foot shock (0.18 mA, 2 s), and after the second response, deliver both foot shock and drug plus cue light. If the second response does not occur within 1 min of the first, restart the sequence and use the total number of shocks obtained as the compulsive-like behavior measure. Prior to punishment testing, footshock intensity was calibrated and maintained constant across sessions to ensure consistent aversive contingencies. Excessive shock intensity may produce nonspecific suppression of responding, whereas insufficient intensity may fail to elicit measurable punishment sensitivity. Shock calibration and grid cleanliness were verified before the shock session to rule out technical malfunctions; particular attention was paid to thoroughly cleaning the grid floor to ensure optimal electrical conductivity.
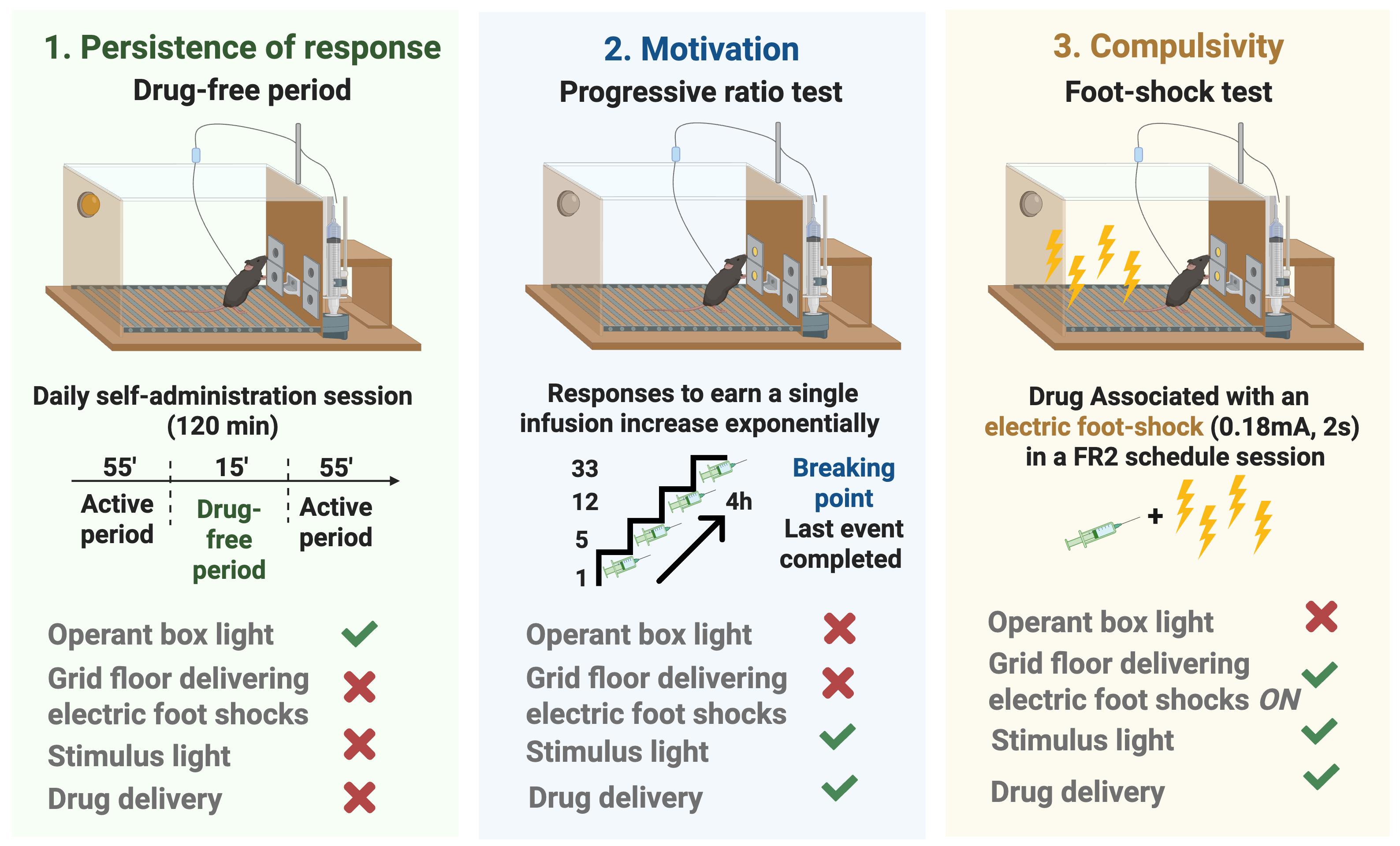
Figure 7. Addictive-like behavior was quantified at the end of training using three DSM-5–based criteria: 1) persistence, 2) motivation, and 3) compulsivity. Persistence was defined as non-reinforced active responses during the 15-min drug-free period across three days before the progressive-ratio (PR) session. Only the operant box house light is on during this period. Motivation was assessed with a PR schedule, with the breakpoint (last ratio completed before cessation of responding) taken as the motivation value. Only the conditioned-stimulus cue light and the drug delivery were active. Compulsivity was measured in a 50-min punished self-administration session under FR2, during which each infusion was paired with a 0.18 mA, 2 s foot shock, and the total number of shocks indexed resistance to punishment. The conditioned-stimulus cue light, the drug delivery, and the unconditioned stimulus of the grid floor delivering foot shocks were active. An overall addiction score was derived from these three behavioral measures.
E. Establishment of mouse subpopulations
1. After completing the three behavioral tests, classify mice as addicted or non-addicted according to the number of addiction-like criteria fulfilled.
2. Define a mouse as positive for a given addiction-like criterion when its behavioral score is at or above the 75th percentile of the saline control distribution.
3. Consider mice meeting 2 or 3 criteria as addicted and categorize them as vulnerable.
4. Consider mice meeting 0 or 1 criterion as non-addicted and categorize them as resilient.
F. Behavioral tests to evaluate addiction-like phenotypic traits
1. Evaluate two additional phenotypic traits as markers of vulnerability to addiction: impulsivity and sensitivity to reward (Figure 8).
2. Impulsivity: Quantify impulsivity-like behavior as the number of non-reinforced active responses emitted during the 10-s time-out period following each WIN 55,212-2 infusion, using the 3 days preceding the PR test for this measure (higher counts indicate greater difficulty in inhibiting responding once initiated).
3. Sensitivity to reward: Assess sensitivity to reward as the total number of reinforcers earned during 2-h sessions across the last three consecutive FR2 self-administration sessions maintained by WIN 55,212-2, where higher reinforcer counts reflect greater reward sensitivity.
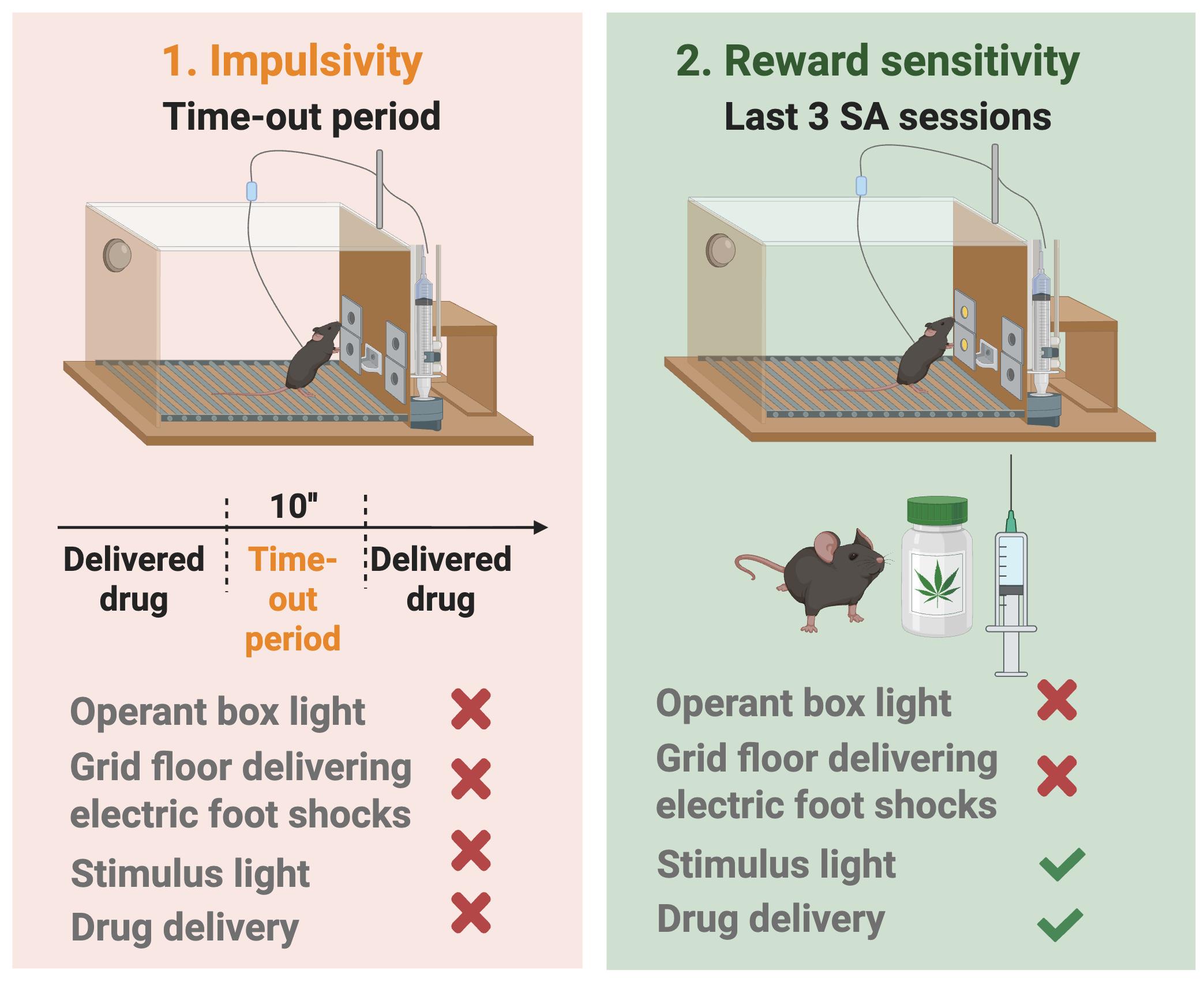
Figure 8. Phenotypic traits as markers of vulnerability to addiction: impulsivity and sensitivity to reward . Impulsivity-like behavior was quantified as the number of non-reinforced active responses emitted during the 10-s time-out period following each WIN 55,212-2 infusion, averaged across the 3 days preceding the progressive-ratio (PR) test (higher counts indicate greater difficulty inhibiting responding once initiated). Sensitivity to reward was assessed as the total number of reinforcers earned during 2-h sessions across the last three consecutive FR2 self-administration sessions maintained by WIN 55,212-2, with higher reinforcer counts reflecting greater reward sensitivity.
G. Extinction and parameters related to craving
1. Include in the extinction phase only mice with patent catheters that satisfied all acquisition criteria. After thiopental testing, allow mice to rest for 1 day and assess locomotor activity in a 2-h session using individual activity boxes (10.8 × 20.3 × 18.6 cm) equipped with infrared sensors for horizontal activity and an infrared plane for rearings.
2. For extinction, run 2-h daily sessions for 10 consecutive days in the same operant chambers used for self-administration, during which neither WIN 55,212-2 priming infusions nor response-contingent WIN 55,212-2 infusions or associated cues are delivered after active nose pokes. Define extinction as performing active responding <35% of the mean active responses previously done during the last 3 days of WIN 55,212-2 self-administration, maintained for 3 consecutive extinction sessions; only mice meeting this criterion proceed to craving-related tests (Figure 9).
3. Resistance to extinction (craving-related measure 1): During the first 2-h extinction session, record the total number of active nose pokes; higher values indicate greater resistance to extinction and increased drug seeking when access to WIN 55,212-2 is prevented.
4. Drug-seeking behavior (cue-induced reinstatement, craving-related measure 2): The day after extinction criterion is reached, perform a single reinstatement session in the same chamber under conditions identical to acquisition, except that active responses are never reinforced by drug; conduct a 120-min FR2 session in which each active nose poke produces all conditioned cues (cue light, pump noise, priming-injection light) without WIN 55,212-2 delivery [16].
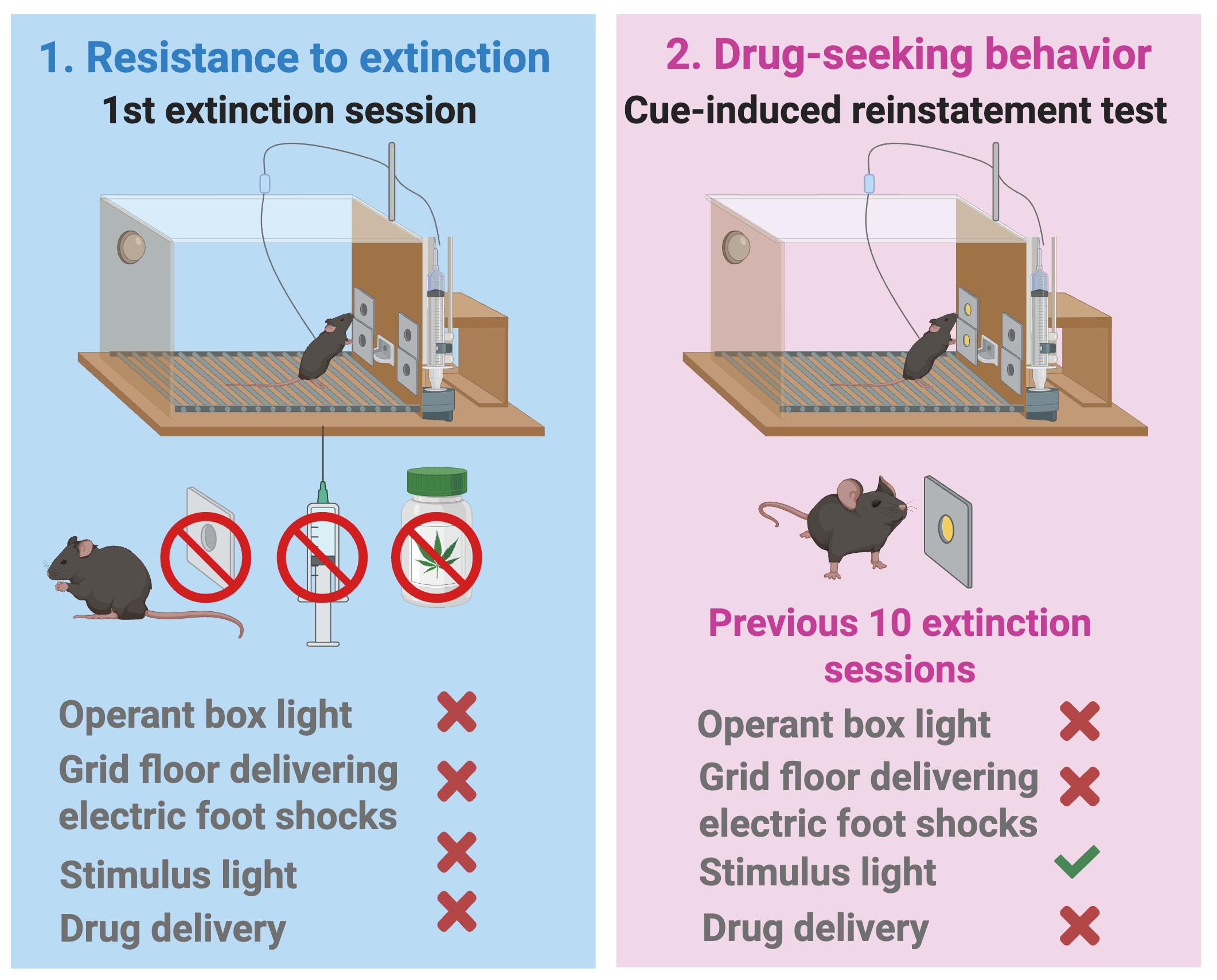
Figure 9. Resistance to extinction and drug-seeking behavior as parameters related to craving. Only mice with patent catheters that met acquisition criteria entered extinction. Resistance to extinction consisted of the first 2-h session in the same operant chambers without WIN 55,212-2 priming, response-contingent infusions, or associated cues after active nose pokes. This parameter (craving-related measure 1) was defined as the total number of active nose pokes during the first 2-h extinction session, with higher values indicating greater resistance to extinction. Drug-seeking behavior (cue-induced reinstatement, craving-related measure 2) was tested the day after 10 extinction sessions in a single 120-min FR2 session identical to acquisition but without drug delivery, where each active nose poke produced the conditioned cues (cue light, pump noise, priming-injection light) instead of WIN 55,212-2 [16].
H. Histology verification
1. Verify viral expression.
a. Perfuse animals using intracardiac perfusion with 4% paraformaldehyde (PFA) in 0.1 M Na2HPO4/0.1 M NaH2PO4 buffer (PB), pH 7.5, delivered with a peristaltic pump at 30 mL per min for 2 min. Animals are previously profoundly anesthetized by intraperitoneal injection (0.2 mL/10 g of body weight) of a mixture of ketamine/medetomidine.
b. Extract the brains and post-fix with 4% PFA for 24 h. Transfer them to a solution of 30% sucrose at 4 °C.
c. Make coronal sections (30 μm) using a freezing microtome and store them in a 5% sucrose solution at 4 °C until the immunofluorescence study.
d. Perform an immunofluorescence study using specific antibodies for your viral vector fluorescent reporter used.
e. Visualize the stained sections of the brain with a confocal microscope to evaluate histological verification of correct injection placement.
f. Make representative immunofluorescence pictures (Figure 10).
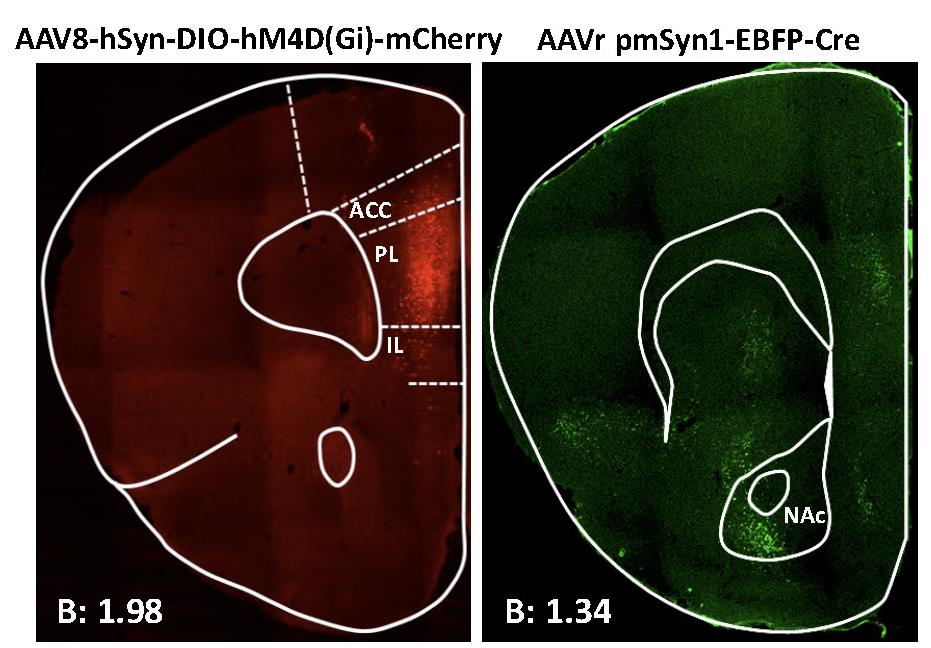
Figure 10. Representative immunofluorescence image showing Cre-dependent hM4Di-mCherry detected at prelimbic (PL) injection site (left) and Cre recombinase at the nucleus accumbens (NAc) core (right). Data derived from Domingo-Rodriguez et al., 2020 [17].
Data analysis
A. Statistical analyses
Comparisons between two experimental groups rely on parametric or nonparametric tests, depending on data distribution. Specifically, Student’s t-test is used when the Kolmogorov–Smirnov test confirms normality, whereas the Mann–Whitney U test is applied for non-normally distributed variables. When repeated measurements over time are available, repeated-measures ANOVA is used to evaluate temporal evolution, followed by Fisher’s least significant difference post hoc tests for pairwise group comparisons.
Correlational and categorical analyses are used to relate behavioral readouts to addiction status. Pearson’s correlation coefficient quantifies the association between continuous scores in each addiction-like criterion and the total number of criteria fulfilled by each mouse, whereas chi-square tests compare the proportion of animals classified as addicted versus non-addicted between groups.
Sample size is determined a priori by power analysis to ensure sufficient sensitivity to detect group differences. Using a two-sample t-test with an alpha level of 0.05, group sizes of 13–16 mice provide a statistical power greater than 80% for the expected effect sizes.
To capture shared variance across multiple behavioral dimensions, a principal component analysis (PCA) is conducted on the addiction-relevant measures. PCA with varimax rotation is applied to the three addiction-like criteria, the two craving-related parameters, and the two vulnerability-related phenotypic traits, reducing them to the smallest set of orthogonal components that explain most of the variance in the dataset. Components with eigenvalues greater than 1 are retained following standard criteria, facilitating visualization of trends, clusters, and outliers in the multidimensional behavioral space.
B. Exclusion criteria
Animals that did not respond in <25% of all FR5 sessions and did not achieve the acquisition criteria are excluded from the remaining experimental sequence.
Animals with a viral vector expression outside the brain regions of interest are excluded from the experiment.
Animals with a negative thiopental test are also excluded from data analysis.
C. Representative results
1. Number of reinforcers: The mean number of active and inactive nose pokes obtained in each session during the entire cannabinoid addiction protocol is shown in Figure 11.
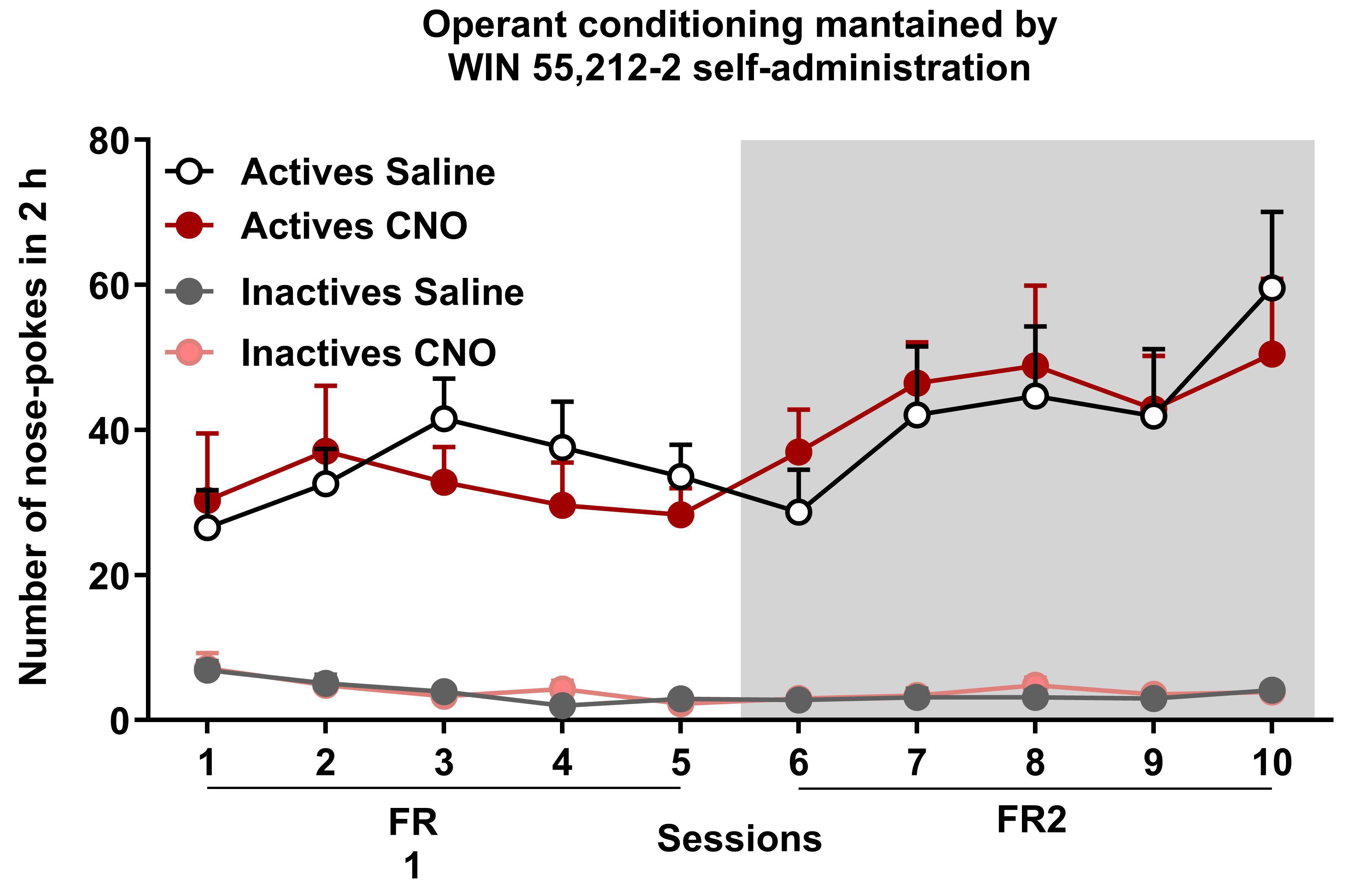
Figure 11. WIN 55,212-2 operant self-administration maintained by WIN 55,212-2 in mice with an inhibition of the prelimbic to nucleus accumbens (PL-NAc) core pathway activated by clozapine-N-oxide (CNO) or corresponding saline controls. Number of total active and inactive nose pokes performed by CNO- and saline-treated groups obtained during 2 h of operant self-administration maintained by intravenous infusions of WIN 55,212-2 in both FR1 and FR2 schedules of reinforcement (mean ± S.E.M., repeated measures ANOVA). Data derived from Cajiao et al., 2023 [7].
2. Behavioral tests of the three addiction-like criteria (persistence to response, motivation, and compulsivity) are shown in Figure 12.
a. Persistence to response: Mean of the total number of non-reinforced active responses during 3 consecutive daily 10-min drug-free periods.
b. Motivation: Breaking point achieved in 4 h of the progressive ratio test.
c. Compulsivity: Number of shocks that mice received in 50 min in the shock test, in which each drug delivery is associated with a foot-shock.
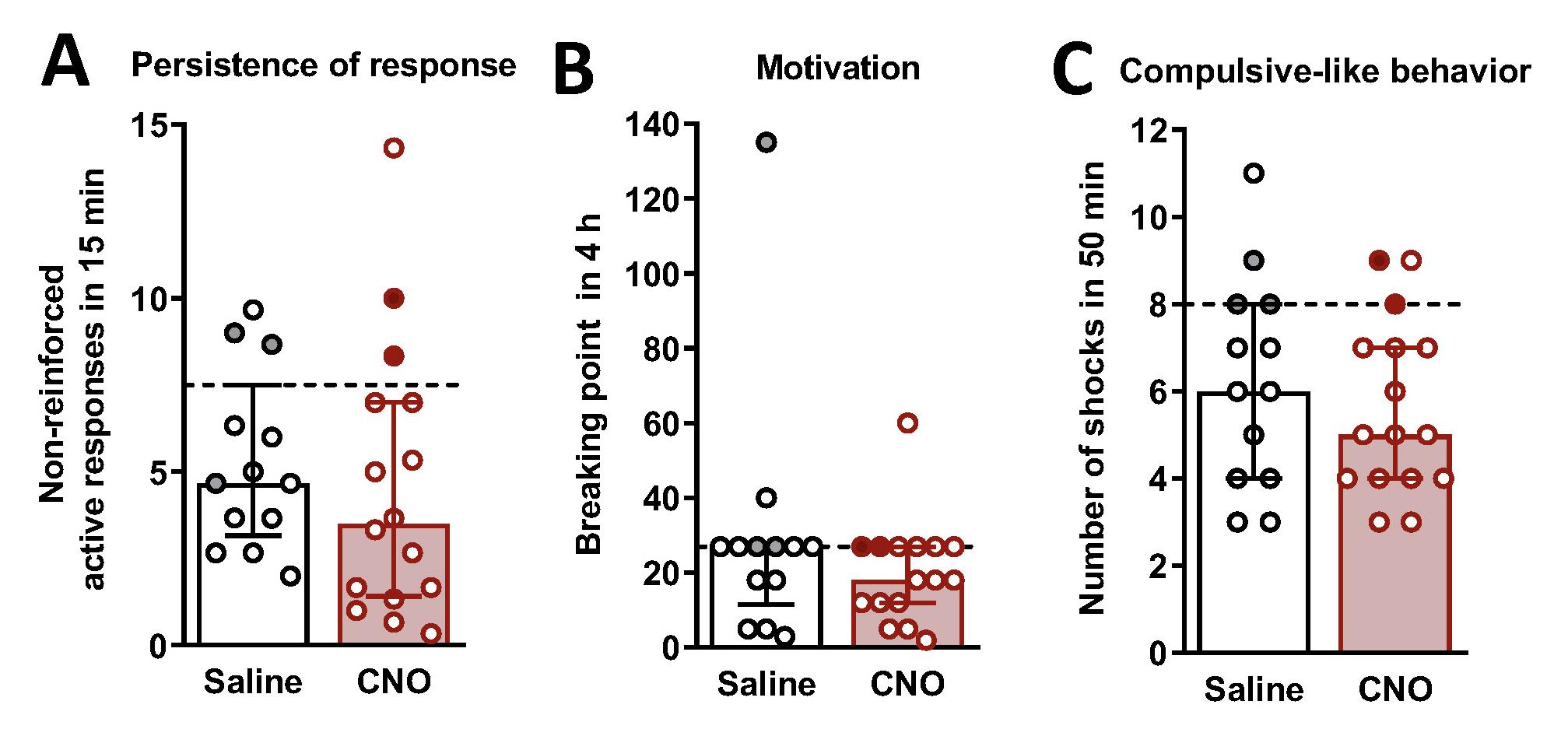
Figure 12. Behavioral tests of the three addiction-like criteria. (A) Persistence of response, (B) motivation, and (C) compulsivity in clozapine-N-oxide (CNO)-treated mice with an inhibition of the prelimbic to nucleus accumbens (PL-NAc) core pathway (individual values with the median and the interquartile range, U Mann–Whitney, **P < 0.01). The dashed horizontal line indicates the 75th percentile of the distribution of mice treated with saline. Addicted mice in gray-filled circles for saline-treated mice (n = 13) and red for CNO-treated mice (n = 16). Data derived from Cajiao et al., 2023 [7].
3. Categorization of mice in addicted and non-addicted animals: The percentage of addicted and non-addicted animals in each group is shown in Figure 13.
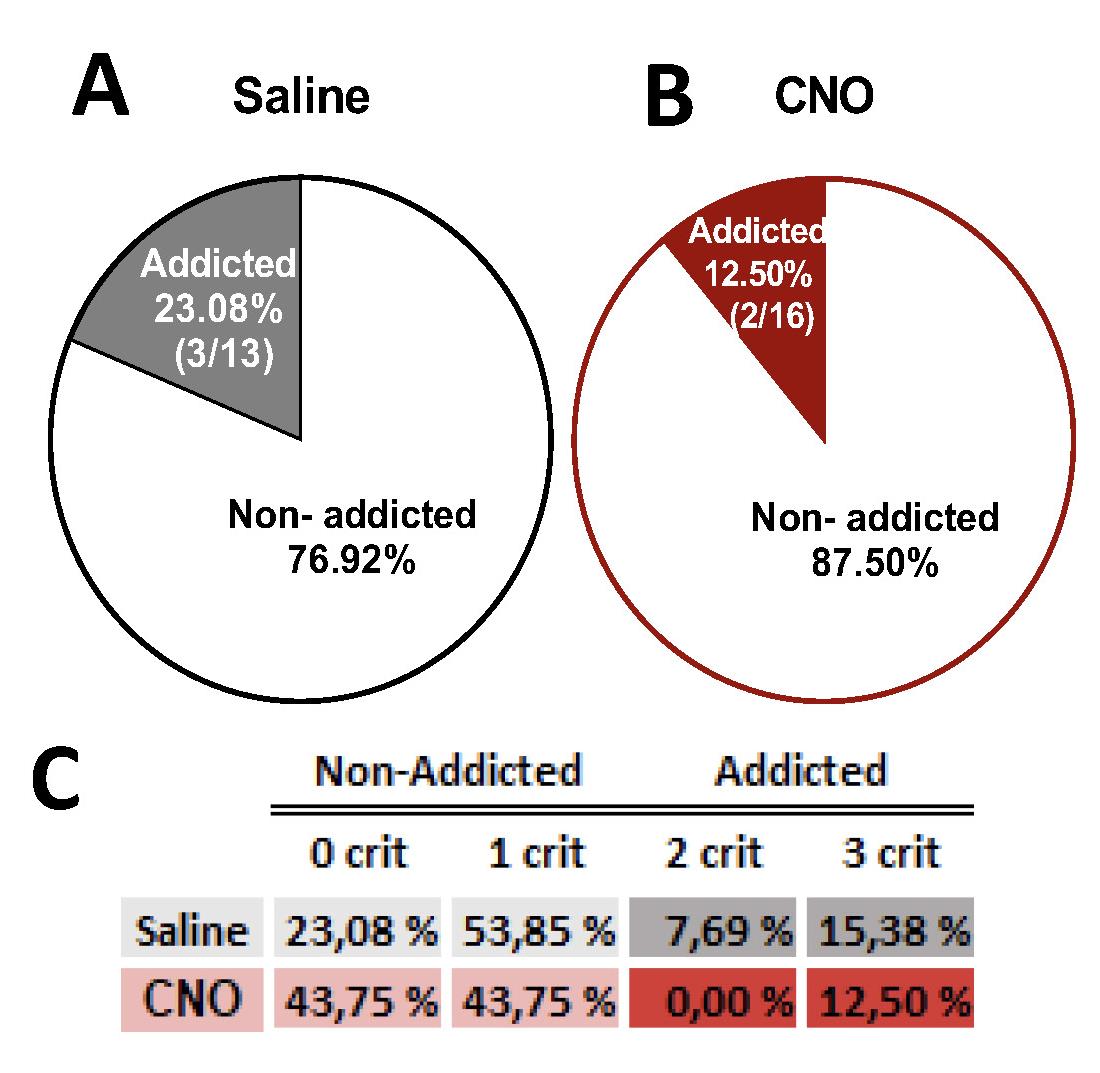
Figure 13. Percentage of clozapine-N-Oxide (CNO)-treated mice with an inhibition of the prelimbic to nucleus accumbens (PL-NAc) core pathway classified as cannabinoid-addicted and corresponding saline controls. Chi-square, ***P < 0.001, n = 13 for saline-treated mice (A) and n = 16 for CNO-treated mice (B). (C) Table with percentages per criteria. Data derived from Cajiao et al. [7].
Validation of protocol
The protocol was validated in the following publications:
Cajiao-Manrique et al. [7]. THC exposure during adolescence increases impulsivity-like behavior in adulthood in a WIN 55,212-2 self-administration mouse model. Front psychiatry.
Cajiao-Manrique et al. [8]. A male mouse model of WIN 55,212–2 self-administration to study cannabinoid addiction. Front Pharmacol. 1–14.
The present protocol was validated in male mice; application to female mice may require additional validation to account for potential sex-specific variability.
General notes and troubleshooting
General notes
1. Mice should be monitored at least once daily for a minimum of 3 days after surgery. If a mouse removes its skin sutures, anesthetize it with isoflurane (2.5% v/v for induction and maintenance), clean the incision with sterile saline, close the skin using 6/0 black silk square knots, and disinfect the area with povidone–iodine.
2. Postoperative welfare should be evaluated following standardized procedures adapted from Morton and Griffiths’ guidelines for the recognition of pain, distress, and discomfort. This scoring system includes four variables, each rated from 0 to 3, as follows:
a. Weight loss
0 (normal): No weight loss or normal growth.
1: Weight loss < 10%.
2: Weight loss 10%–20% and/or changes in stool appearance or amount.
3: Weight loss >20%; the animal does not consume food or water.
b. Coat appearance
0: Normal.
1: Hair in mildly poor condition.
2: Hair in poor condition with ocular or nasal secretions.
3: Marked piloerection.
c. Movement/posture/behavior
0: Normal.
1: Mild changes.
2: Moderate changes.
3: Severe alterations such as inactivity, aggressiveness, self-mutilation, or vocalizations.
d. Incision site
0: Normal.
1: Mild redness.
2: Local edema.
3: Signs of infection or tissue darkening (necrosis).
The final score is the sum of the four subscores and guides intervention, as follows:
0–3: No corrective action required.
4–7: Apply corrective measures.
≥8: Humane euthanasia is required.
Euthanasia should also be performed if any single variable scores 3. Corrective measures may include wound cleaning with disinfectant and topical antibiotic, providing external heat (heating pad under the cage), and offering nutrient-enriched hydrating gel or softened food pellets to support intake.
3. Throughout the experimental protocol, animals should be regularly handled by the experimenter to promote habituation and reduce stress, thereby improving data reliability. Handling consists of allowing each mouse to freely explore the experimenter’s arm for 2 min per day for at least 7 days before the start of behavioral testing.
Troubleshooting
Body weight and food intake are measured weekly throughout the cannabis addiction mouse protocol. These measurements are especially crucial because they are used with osmotic minipumps filled with CNO. We demonstrated that under our conditions, CNO had no effects on body weight, food intake, or locomotor activity [17].
Particular attention should be paid to experimental steps in which technical failure may compromise data validity, including catheter patency, minipump implantation, and punishment calibration. Catheter patency should be routinely monitored throughout the experiment and confirmed at the end of the self-administration phase, after the shock, using a thiopental test; animals exhibiting signs of catheter malfunction should be excluded. Correct subcutaneous placement of osmotic minipumps should be ensured, with post-mortem verification performed when necessary. During punishment testing, shock intensity must be calibrated and maintained constant, and the grid floor should be thoroughly cleaned to ensure optimal electrical conductivity.
In cases of failure to acquire self-administration, verify proper operant chamber functioning, ensure the infusion system is unobstructed, confirm the animal is correctly connected to the infusion line, and check that the tubing length permits free and unrestricted movement. Potential anxiety- or stress-related interference with task engagement should also be considered. In cases of excessive inactive responding, confirm proper nose-poke functioning and accurate response registration to distinguish technical malfunction from behavioral factors.
Acknowledgments
This work was supported by the Spanish Ministerio de Ciencia e Innovación (MICIN), Agencia Estatal de Investigación (AEI) (PID2023- 120029GB-I00/MICIN/AEI/10.13039/501100011033, RD21/0009/0019), the Spanish Instituto de Salud Carlos III, RETICS-RTA (RD12/0028/0023), the Generalitat de Catalunya, AGAUR (2017 SGR-669), ICREA-Acadèmia (2025) and the Spanish Ministerio de Sanidad, Servicios Sociales e Igualdad, Plan Nacional Sobre Drogas of the Spanish Ministry of Health (PNSD-2017I068) to RM, Plan Nacional Sobre Drogas of the Spanish Ministry of Health (PNSD-2023I040), Spanish Ministerio de Ciencia e Innovación (ERA-NET) PCI2021-122073-2A and Caixa Health HR22-00737 to E.M.-G. The methodology described was previously used in [8,17]. Figures with drawings were created with BioRender.com.
The following figures were created using BioRender: Graphical overview, https://biorender.com/aa7hzec; Figure 8, https://biorender.com/31sg9vj; Figure 9, https://biorender.com/2d6irgq; Figure 5, https://biorender.com/3ovfx9v; Figure 3, https://biorender.com/4jlfzwd; Figure 2: https://biorender.com/y1h6ixv; Figure 9: https://biorender.com/mr9m8z1.
Competing interests
The authors have no conflicts of interest.
Ethical considerations
All experimental protocols were performed in accordance with the guidelines of the European Communities Council Directive 2010/63/EU and approved by the local ethical committee (Comitè Ètic dExperimentació Animal, Parc de Recerca Biomèdica de Barcelona, CEEA-PRBB, agreement N 9687).
References
- Hall, W., Manthey, J. and Stjepanović, D. (2024). Cannabis use and cannabis use disorders and their treatment in the Europe. Eur Arch Psychiatry Clin Neurosci. 275(2): 307–313. https://doi.org/10.1007/s00406-024-01776-1
- Weiss, S. R. B. and Volkow, N. D. (2021). Coordinating cannabis data collection globally: Policy implications. Addiction 117(6): 1520–1521. https://doi.org/10.1111/add.15751
- Koob, G. F. and Volkow, N. D. (2016). Neurobiology of addiction: a neurocircuitry analysis. The Lancet Psychiatry 3(8): 760–773. https://doi.org/10.1016/s2215-0366(16)00104-8
- Maldonado, R., Calvé, P., García-Blanco, A., Domingo-Rodriguez, L., Senabre, E. and Martín-García, E. (2021). Vulnerability to addiction. Neuropharmacology. 186: 108466. https://doi.org/10.1016/j.neuropharm.2021.108466
- Maldonado, R., Calvé, P., García‐Blanco, A., Domingo‐Rodriguez, L., Senabre, E. and Martín‐García, E. (2021). Genomics and epigenomics of addiction. Am J Med Genet Part B: Neuropsychiatr Genet. 186(3): 128–139. https://doi.org/10.1002/ajmg.b.32843
- Piazza, P. V. and Deroche-Gamonet, V. (2013). A multistep general theory of transition to addiction. Psychopharmacology (Berl). 229(3): 387–413. https://doi.org/10.1007/s00213-013-3224-4
- Cajiao-Manrique, M. d. M., Casadó-Anguera, V., García-Blanco, A., Maldonado, R. and Martín-García, E. (2023). THC exposure during adolescence increases impulsivity-like behavior in adulthood in a WIN 55,212-2 self-administration mouse model. Front Psychiatry. 14: e1148993. https://doi.org/10.3389/fpsyt.2023.1148993
- Cajiao-Manrique, M. d. M., Maldonado, R. and Martín-García, E. (2023). A male mouse model of WIN 55,212–2 self-administration to study cannabinoid addiction. Front Pharmacol. 14: e1143365. https://doi.org/10.3389/fphar.2023.1143365
- Haney, M., Vallée, M., Fabre, S., Collins Reed, S., Zanese, M., Campistron, G., Arout, C. A., Foltin, R. W., Cooper, Z. D., Kearney-Ramos, T., et al. (2023). Signaling-specific inhibition of the CB1 receptor for cannabis use disorder: phase 1 and phase 2a randomized trials. Nat Med. 29(6): 1487–1499. https://doi.org/10.1038/s41591-023-02381-w
- Vallée, M., Vitiello, S., Bellocchio, L., Hébert-Chatelain, E., Monlezun, S., Martin-Garcia, E., Kasanetz, F., Baillie, G. L., Panin, F., Cathala, A., et al. (2014). Pregnenolone Can Protect the Brain from Cannabis Intoxication. Science. 343(6166): 94–98. https://doi.org/10.1126/science.1243985
- Flores, Ã., Maldonado, R. and Berrendero, F. (2019). THC exposure during adolescence does not modify nicotine reinforcing effects and relapse in adult male mice. Psychopharmacology (Berl). 237(3): 801–809. https://doi.org/10.1007/s00213-019-05416-8
- Mendizábal, V., Zimmer, A. and Maldonado, R. (2005). Involvement of κ/Dynorphin System in WIN 55,212-2 Self-Administration in Mice. Neuropsychopharmacology 31(9): 1957–1966. https://doi.org/10.1038/sj.npp.1300957
- Maldonado, R. and Rodrı́guez de Fonseca, F. (2002). Cannabinoid Addiction: Behavioral Models and Neural Correlates. J Neurosci. 22(9): 3326–3331. https://doi.org/10.1523/jneurosci.22-09-03326.2002
- Cabañero, D., Álvarez Pérez, B., Martín-García, E. and Maldonado, R. (2022). Operant Self-medication for Assessment of Spontaneous Pain Relief and Drug Abuse Liability in Mouse Models of Chronic Pain. Bio Protoc. 12(3): e4348. https://doi.org/10.21769/bioprotoc.4348
- Martín-García, E., Domingo-Rodriguez, L. and Maldonado, R. (2020). An Operant Conditioning Model Combined with a Chemogenetic Approach to Study the Neurobiology of Food Addiction in Mice. Bio Protoc. 10(19): e3777. https://doi.org/10.21769/bioprotoc.3777
- Martín-García, E., Bourgoin, L., Cathala, A., Kasanetz, F., Mondesir, M., Gutiérrez-Rodriguez, A., Reguero, L., Fiancette, J., Grandes, P., Spampinato, U., et al. (2015). Differential Control of Cocaine Self-Administration by GABAergic and Glutamatergic CB1 Cannabinoid Receptors. Neuropsychopharmacology. 41(9): 2192–2205. https://doi.org/10.1038/npp.2015.351
- Domingo-Rodriguez, L., Ruiz de Azua, I., Dominguez, E., Senabre, E., Serra, I., Kummer, S., Navandar, M., Baddenhausen, S., Hofmann, C., Andero, R., et al. (2020). A specific prelimbic-nucleus accumbens pathway controls resilience versus vulnerability to food addiction. Nat Commun. 11(1): e1038/s41467–020–14458–y. https://doi.org/10.1038/s41467-020-14458-y
- Valjent, E. and Maldonado, R. (2000). A behavioural model to reveal place preference to Δ9-tetrahydrocannabinol in mice. Psychopharmacology (Berl). 147(4): 436–438. https://doi.org/10.1007/s002130050013
- Deroche-Gamonet, V., Belin, D. and Piazza, P. V. (2004). Evidence for Addiction-like Behavior in the Rat. Science. 305(5686): 1014–1017. https://doi.org/10.1126/science.1099020
Article Information
Publication history
Received: Dec 30, 2025
Accepted: Mar 3, 2026
Available online: Mar 13, 2026
Published: Apr 5, 2026
Copyright
© 2026 The Author(s); This is an open access article under the CC BY-NC license (https://creativecommons.org/licenses/by-nc/4.0/).
How to cite
Martín-García, E., Ponce-Beti, M. F., Gusinskaia, T., López-Moraga, A., Capellán, R. and Maldonado, R. (2026). A Male Mouse Model of WIN 55,212–2 Self-Administration to Study Cannabinoid Addiction. Bio-protocol 16(7): e5652. DOI: 10.21769/BioProtoc.5652.
Category
Neuroscience > Behavioral neuroscience > Addiction
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link









