- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
A Simple and Straightforward Approach for Generating Small, Stable, Homogeneous, Unilamellar 1-Palmitoyl 2-Oleoyl Phosphatidylcholine (POPC) Bilayer Vesicles
Published: Vol 11, Iss 24, Dec 20, 2021 DOI: 10.21769/BioProtoc.4271 Views: 3283
Reviewed by: Philipp A.M. Schmidpetersujan kumar mondalAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Visualizing Loss of Plasma Membrane Lipid Asymmetry Using Annexin V Staining
Julia F. Baum [...] Thomas Günther Pomorski
Jul 20, 2023 2538 Views
Fluorescence Resonance Energy Transfer to Detect Plasma Membrane Perturbations in Giant Plasma Membrane Vesicles
Mathew Sebastiao [...] Steve Bourgault
Oct 5, 2023 2100 Views
PI(4,5)P2 Imaging Using a GFP Reporter in Living Cells
Mariam Alkandari [...] Mahtab Tavasoli
Jun 5, 2025 1954 Views
Abstract
Various methods have been developed to generate phosphoglyceride liposomes. Approaches resulting in homogeneous populations of unilamellar bilayer vesicles are generally preferred to mimic various cell membrane situations, as well as to optimize aqueous solute trapping efficiency using the least amount of lipid for biotechnological purposes. Most are time-consuming, often tedious, or require specialized equipment, and produce vesicles with limited shelf-life at room temperature or in cold storage. Herein, we describe a straightforward approach that avoids the preceding complications and streamlines the construction of unilamellar bilayer vesicles from 1-palmitoyl-2-oleoyl phosphatidylcholine (POPC)/dihexanoyl phosphatidylcholine (DHPC) bicelle mixtures at room temperature. The resulting vesicles are small (32-36 nm diameter), unilamellar, bilayer vesicles that are homogeneous, stable, and resistant to freeze-thaw alterations.
Graphic abstract:

Cryo-EM of POPC vesicles formed by dilution of 0.5 q-value POPC/DHPC bicelle mix.
Background
The fundamental building blocks for biomembranes are phosphoglycerides. Their low solubility and amphiphilic nature enable these lipids to self-assemble into various structural mesophases including bilayers, the basic barriers that compartmentalize cells. Cells take advantage of membrane bilayers to form internal and external barriers, as well as to carry out transport of materials within and between cells. The latter process involves generation of bilayer vesicles to act as delivery shuttles, by budding from and fusing with different membranes. Such functionality is critically important during periods of cell growth, development, and proliferation for recycling and re-utilization of biomolecules during programmed cell death processes, and for neurotransmission. To gain insights into the physicochemical properties that enable cell membrane functionality, studies of model bilayer membranes produced in vitro, i.e., liposomes, have proven invaluable. The studies have also led to biotechnological advances that include the engineering of liposomes for delivery of drugs, genes, and vaccines, as well as for applications in medical diagnostics, cosmetics, and food industry-related nutraceutics and dietetics (Lichtenberg and Barenholz, 1988; Laouini et al., 2012; Akbarzadeh et al., 2013; Keller et al., 2013; Patil and Jadha, 2014; Nkanga et al., 2019; Maja et al., 2020; Ajeeshkumar et al., 2021).
Over the years, a variety of methodologies have been used to generate bilayer liposomes.Generally, approaches that result in homogeneous populations of single-wall, i.e. unilamellar, bilayer vesicles are preferred to mimic the situation generally encountered in cells, and to optimize trapping efficiency using the least amount of lipid. In biomedical research, approaches involving ultrasonication, reverse-phase evaporation, and extrusion have been widely used. Less widely used are ethanol injection, detergent dialysis/removal, and supercritical fluid approaches. Nearly all of the approaches are either tedious, time-consuming, require specialized equipment, and produce vesicles with limited shelf-life at either room temperature or in cold storage.
We sought a straightforward approach for fast and easy construction of homogeneous unilamellar bilayer vesicles comprised of 1-palmitoyl-2-oleoyl phosphatidylcholine (POPC). We turned to bicelle-based approaches because of insights provided by theoretical and molecular dynamics modeling of mechanistic pathways, involving bicelle disc transformation into unilamellar bilayer vesicles (Fromherz, 1983; Marrink and Mark, 2003; Chng, 2013). These studies suggest that loss of rim-stabilizing short-chain PC (e.g., dihexanoyl PC; DHPC) or detergents from bicelles will lead to their coalescence into larger-diameter discs, followed by the formation of cup-shaped intermediates to reduce long-chain PC hydrocarbon chain exposure to water. With further departure of short-chain DHPC or detergent from the rim areas, the cup-shaped hemi-vesicle intermediates are predicted to undergo rim closure to form unilamellar vesicles. From a thermodynamic standpoint, the loss of DHPC or detergent from the bicelle rims alters the energetic balance between the rim perimeter line tension and the bicelle disc elastic bending energy, with the perimeter line tension becoming more dominant, as DHPC or detergent is lost from the bicelle rim. This situation promotes formation of the cup-shaped hemi-vesicle intermediates that further transition to unilamellar vesicles when nearly complete loss of rim-stabilizing DHPC occurs.
In the majority of previous experimental studies involving bicelles, the long-chain phosphoglyceride component contains saturated acyl chains, e.g., dimyristoyl phosphatidylcholine (DMPC) or DMPC/dimyristoyl phosphatidylglycerol mixtures (DMPG). These lipid species are generally absent or very minor components of eukaryotic biomembranes. Typically, biomembrane phosphoglyceride acyl composition is asymmetric, with the sn1-chain being long and saturated (C ≥ 16), while the sn2-chain is long (C ≥ 18) and contains cis unsaturation, as occurs in POPC (Figure 1). This arrangement has interesting biological consequences, including more efficient membrane vesiculation, and lower membrane permeability compared to phosphoglycerides containing two saturated or polyunsaturated chains (Manni et al., 2018).
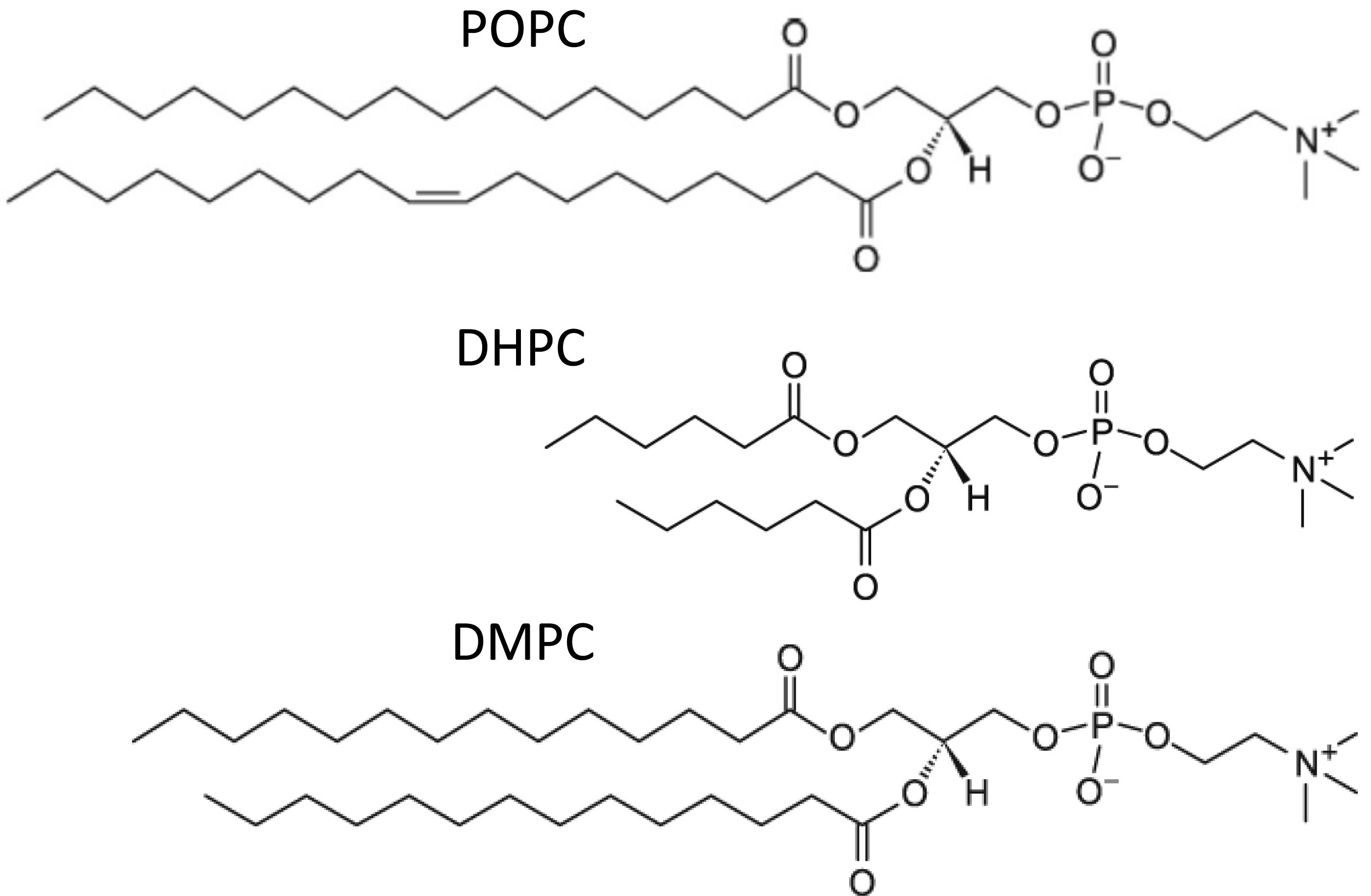
Figure 1. Chemical structures of phosphatidylcholines used or discussed in this study.
From previous studies, it is known that the structural stability of DMPC/DHPC bicelles is critically dependent upon the total concentration of long-chain PC+DHPC/detergent (Sanders and Schwonek, 1992; Sanders and Prosser, 1998; Harroun et al., 2005; Lu et al., 2012; Beaugrand et al., 2014). Dilution of bicelles after formation can alter the long-chain PC-to-DHPC/detergent ratio (q-value) and drive mesomorphic structural change, including transformation to liposome bilayer vesicles (Nieh et al., 2004, 2009 and 2011; Yue et al., 2005; Mahabir et al., 2013). The dilution-driven structural transformation is a consequence of the vastly differing aqueous solubilities of the long-chain PC versus the DHPC/detergent lipids forming the bicelle. In the case of DMPC/DHPC mixtures, the aqueous solubilities of DMPC (~6 nM) and DHPC (~16 mM) differ by 2.7 million-fold. Thus, when the bicelle concentration is suddenly decreased and becomes too low (as can occur with aqueous dilution), DHPC moves from the bicelle into aqueous phase. This situation changes the q-value and alters the structural form of the bicelle lipid mixture. This response is accounted for by q*, through provision of the effective DMPC/DHPC molar ratio for bicelle mixtures at various lipid concentrations (Beaugrand et al., 2014). For example, dilution of 1.0 q-value DMPC/DHPC mixtures from 75 to 25 mM results in q increasing from 1.0 up to a q* of 1.8, where isotropic bicelles still prevail. Upon dilution to 16 mM total PC, a 3.2 q*-value results in the prevalence of large bicelles. Further dilution to 2 mM total PC leads to an extremely high q*, where small DMPC liposome vesicles become detectable by 31P-NMR.
From a practical standpoint, whether the preceding modeled and experimental behavior that characterizes DMPC/DHPC and DMPC/DMPG/DHPC bicelle lipid mixtures can yield a simple and easy method for producing homogeneous populations of POPC unilamellar bilayer vesicles has remained unclear. Recently, a straightforward approach that streamlines production of POPC vesicles from POPC/DHPC bicelle mixtures has been used in applications for monitoring protein-mediated intervesicular lipid transfer activity (Gao et al., 2020). Characterization has revealed these POPC vesicles to be small (32-36 nm diameter), unilamellar, bilayer vesicles that are surprisingly homogeneous, stable, resistant to freeze-thaw alteration, and usable in assays involving cell cytosolic fractions. The pathway for vesicle formation is illustrated in Figure 2 and production details follow:
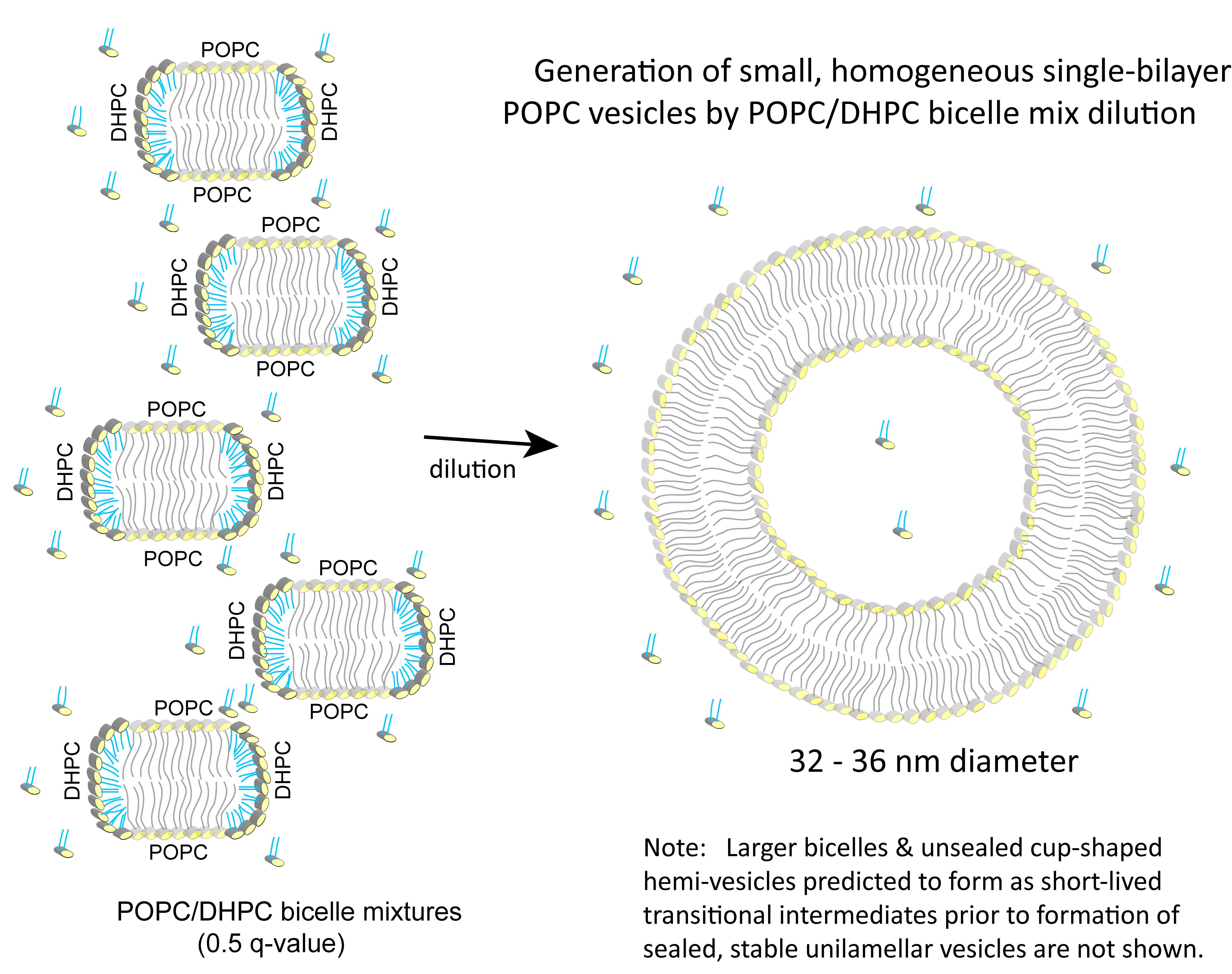
Figure 2. Generation of small, homogeneous single-bilayer POPC vesicles by dilution of POPC/DHPC bicelle mixtures. The phosphorylcholine polar head groups are shown as yellow ellipsoids with the long acyl chains of POPC depicted in gray (stick fashion) and the short acyl chains of DHPC depicted in cyan (stick fashion). As noted, dilution (>125-fold) of the 0.5 q-value POPC/DHPC bicelle mixture with aqueous buffer initially leads to larger bicelle formation (not shown), followed by transition to unsealed cup-shaped hemi-vesicles (not shown), that seal to form stable unilamellar vesicles. Illustrations of these short-lived transitional intermediates are depicted in Fromherz (1983), Chng (2013), or Marrink and Mark (2003). The bicelles (left) and vesicle (right) are shown in two-dimensional cross-section.
Materials and Reagents
Disposable Pasteur pipets (5.75” borosilicate glass, nonsterile; Fisherbrand, catalog number: 13-678-20A)
Kimble clear glass screw-thread sample vials with PTFE closure (Fisher, catalog numbers: 03-340-60A [4 ml, 17 × 60 mm]; 03-340-60C [12 ml, 19 × 65 mm])
MininertTM syringe valves, leak-tight PTFE closures for screw cap vials and valve open/close action enabling Hamilton syringe access ensures that only the PTFE and the glass come in contact with the lipid sample solution dissolved in nonpolar solvent mixes [e.g., chloroform/methanol (2:1) or hexane/isopropanol/water (7:3:0.25)]
Conical V-bottom vials (clear glass, Reacti-VialTM 1 ml capacity; Thermo Scientific, catalog number: 13221)
Glass syringes, gastight, assorted volumes (Hamilton, catalog numbers:1702 [25 µl]; 1705 [50 µl]; 1710 [100 µl]; 1725 [250 µl]; 1750 [500 µl]; 1001 [10,00 µl])
1-palmitoyl-2-oleoyl phosphatidylcholine (POPC) (Avanti Polar Lipids, Croda Intl., catalog number: 850457)
Dihexanoyl phosphatidylcholine (DHPC) (Avanti Polar Lipids, Croda Intl., catalog number: 850305)
Hexane, HPLC grade (Fisher Chemical, catalog number: H302-4)
Isopropanol, HPLC grade (Fisher Chemical, catalog number: A451-4)
Chloroform, stabilized with 0.75% EtOH (Mallinckrodt, catalog number: 4440-08)
Methanol (Fisher Chemical, catalog number: A412-4)
Nitrogen gas
Cahn style aluminum microbalance weigh pans (Mfr. # 120627) (Thomas Scientific, catalog number: 1162D64)
Equipment
100 ml beaker
Nitrogen tank equipped with gas pressure regulator
Lab support stand (8.3” × 5.5”) with rod (19.7”), lab stand clamp holder, and 3-prong finger-style clamp for holding disposal glass Pasteur pipette linked to N2 tank with Tygon tubing, for evaporating organic solvent from lipid sample (< 2 ml) using N2 gas in lab exhaust hood.
Note: N-EVAPTM Analytical Evaporator (Organomation, Model 111 or 112 24 slot system) is useful when simultaneous evaporation of organic solvents from several small volume (< 2 ml) lipid samples using N2 gas is needed.
Lab chemical fume exhaust hood, e.g., Supreme Air (Keewaunee Sci. Corp.)
Rotary evaporator (Buchi R-300 Rotavapor model 11R300152V002) (Fisher Scientific, catalog number: 05-000-485) [needed if lipids are dissolved in large solvent volumes (e.g., >5-10 ml)]
Duo-Seal Vacuum Pump (Welch, model: 1400B-01); generates vacuum of ~0.001 Torr w/ pumping speed of 25 L/min
Vortex Genie 2 (Daigger Scientific)
Bath Sonicator (Branson, Model 2200 or Model 22-4)
KIMAX glass vacuum dessicator, 15 cm plate size (Cole Parmer, catalog number: EW-06536-11)
Ohaus Analytical Plus Microbalance (Precision Weighing Balances, Model AP250D)
Procedure
Note: Steps 1 through 6 describe general approaches for properly working with lipids in preparation for production of unilamellar bilayer vesicles using a bicelle lipid mixture dilution.
Lipids (received from Avanti Polar Lipids, shipped on dry-ice) are stored at -85°C until use.
Note: The -85°C temperature is best for long-term storage to avoid oxidation of lipids containing unsaturation. For day-to-day short term use, -20°C storage is sufficient.
Transfer the total lipid contents (typically 25 or 100 mg) from the Avanti glass ampule into Kimble glass screw-thread sample vials, which have been previously rinsed with hexane/isopropanol (7:3), or chloroform/methanol (2:1), and then dried under a stream of N2.
Notes:
Solvents for working with lipids should be HPLC grade quality.
Non-chlorinated solvent mixtures such as hexane/isopropanol/water (7:3:0.25) are preferred for dissolving lipids for health and safety reasons.
The use of plastic storage vessels (e.g., plastic serum tubes; Eppendorf tubes®, etc.) for lipid solutions in solvents is not advisable because, over time, the organic solvents can extract materials from the plastics and contaminate the lipids. To avoid such problems, lipids dissolved in organic solvents should be stored in clean glass screw-type vials with the screw-threads sealed with PTFE tape and the screw caps containing PTFE-liners, or better yet, sealed with MininertTM syringe valves (see below).
Evaporate the solvent from the glass screw-thread vials in a chemical exhaust fume hood using a gentle stream of N2.
Place a lab support stand (8.3” × 5.5”) containing a metal rod (19.7”) and two 3-prong finger-style clamps (secured by lab-stand clamp holders) in the exhaust hood.
Clamp the vial so that its lower half is submerged into a 100 ml beaker containing room temperature water.
Clamp a disposable glass Pasteur pipette above the screw-cap vial containing lipid + solvent.
Connect the disposable glass Pasteur pipette to the N2 tank with Tygon® tubing. Align the Pasteur pipette above the screw-cap vial and then lower it so that the pipette tip remains above the solvent containing the dissolved lipid.
Evaporate the solvent by purging the sample with a gentle stream of N2 gas. Submerging the lower half of the screw-cap vial into the beaker containing room temperature water is not essential, but will reduce the solvent evaporation time by counteracting the evaporative cooling effect of the solvent removal from the glass vial.
To remove all traces of any remaining solvent, place the vial containing the lipid in a heavy duty desiccator jar and subject to vacuum for 2 h.
Add the desired amount of solvent mix (hexane/isopropanol/water [7:3:0.25]) to the vial containing the dried lipid using clean Hamilton syringes.
Notes:
A typical working stock lipid solution (~10 mg/ml) will require a 25 mg aliquot of dried lipid that is then dissolved in 2.5 ml of solvent mix.
Adding a trace of H2O to the hexane/isopropanol solvent helps ensure solubility of polar lipids such as phosphoglycerides and glycosphingolipids
Seal the vials using PTFE-lined screw caps to avoid potential contaminants present in rubber-lined caps. Better yet, seal with MininertTM syringe valves that enable easy access via a Hamilton syringe, to avoid solvent evaporation that will alter the lipid stock concentration.
Note: Do not use Pipetman®-style pipetters with plastic tips to aliquot the lipids dissolved in organic solvents. The dispensed volumes are calibrated based on the surface tension of water and leached plastics can contaminate the lipid sample.
Determine the lipid concentrations in the stock vials in one of two ways:
Using a Hamilton gastight syringe, deposit aliquots of the stock solutions on dry, pre-weighed aluminum microbalance weigh pans (Cahn Style, 25 mm). Completely dry by warming to 60°C for 15-30 min and/or placing under vacuum, and weigh triplicate samples immediately using an analytical microbalance (Ali et al., 1991). A variation of this method is that of Tejera-Garcia et al. (2012) .
Alternatively, quantify the stock concentrations of phosphoglycerides by chemical assay for phosphate (Bartlett, 1959). Associated issues are discussed in Tejera-Garcia et al. (2012) .
Notes:
PCs with short saturated chains (e.g., DHPC) are hygroscopic.
To avoid solvent evaporation that will alter lipid concentration during cold storage, vials containing lipid stocks can be further sealed by wrapping the cap/vial threaded joint with PTFE-tape and then overwrapping the cap-glass vial joint with a Parafilm strip.
When stored in the freezers in organic solvents, most lipids will tend to precipitate from solution. However, when brought back to room temperature, the lipids will dissolve again upon vortexing and/or brief bath sonication in 30-40°C water.
Prepare POPC unilamellar bilayer vesicles from 0.5 q-value POPC/DHPC stock bicelle mixtures as follows:
Combine POPC (0.4 µmol) and DHPC (0.8 µmol) in a clear glass conical V-bottom vial.
Evaporate the solvent under a gentle stream of N2 gas.
Subject to vacuum as described in step 3.
Hydrate the lipid mixture by adding 200 µl of buffer at room temperature, to yield a concentration of 6 mM total phospholipid (equals 0.3335 wt% [w/v]).
Vortex vigorously for 5 min to achieve optical clarity and incubate overnight at room temperature.
For 5× POPC vesicle concentrations, combine POPC (2 µmol) and DHPC (4 µmol) in a clear glass conical V-bottom vial.
Perform steps b-d.
Hydrate the lipid mixture by adding 200 µl of buffer at room temperature to yield a concentration of 30 mM total lipid (equals 1.6675 wt% [w/v]).
Vortex vigorously for 5 min to achieve optical clarity and incubate overnight at room temperature.
Notes:
The buffer used here is phosphate buffered saline (pH 7.2) at physiological ionic strength (145 mM NaCl). Other extreme conditions such as high ionic strength (e.g., 1.5 M NaCl), which can affect the critical micelle concentration of certain amphiphiles (e.g., Chattopadhyay and Harikumar, 1996; Palladino and Ragone, 2011), have not yet been analyzed.
After vortexing, if needed briefly bath sonicate to achieve clarity.
Figure 2 depicts the structural transition that occurs in response to the dilute conditions encountered by 0.5 q-value POPC/DHPC bicelle lipid mixtures. For simplicity, transitional intermediate structures (e.g., larger bicelle discs formed by coalescence and cup-shaped hemi-vesicle intermediates that form prior to rim closure) are not shown.
Cryo-EM analyses confirm the spontaneous formation of POPC unilamellar vesicle from the highly diluted (<125-fold) 0.5 qvalue POPC/DHPC bicelle mixtures (see Data analysis, Figure 3).
The best results for measuring protein-mediated lipid intermembrane transfer, tracked by Förster resonance energy transfer using fluorescent lipids (Gao et al., 2020) were observed using the POPC unilamellar bilayer vesicles formed from highly diluted 0.5 q-value POPC/DHPC bicelle mixtures compared to 0.1, 1.0, and 2.0 q-value POPC/DHPC mixtures diluted similarly.
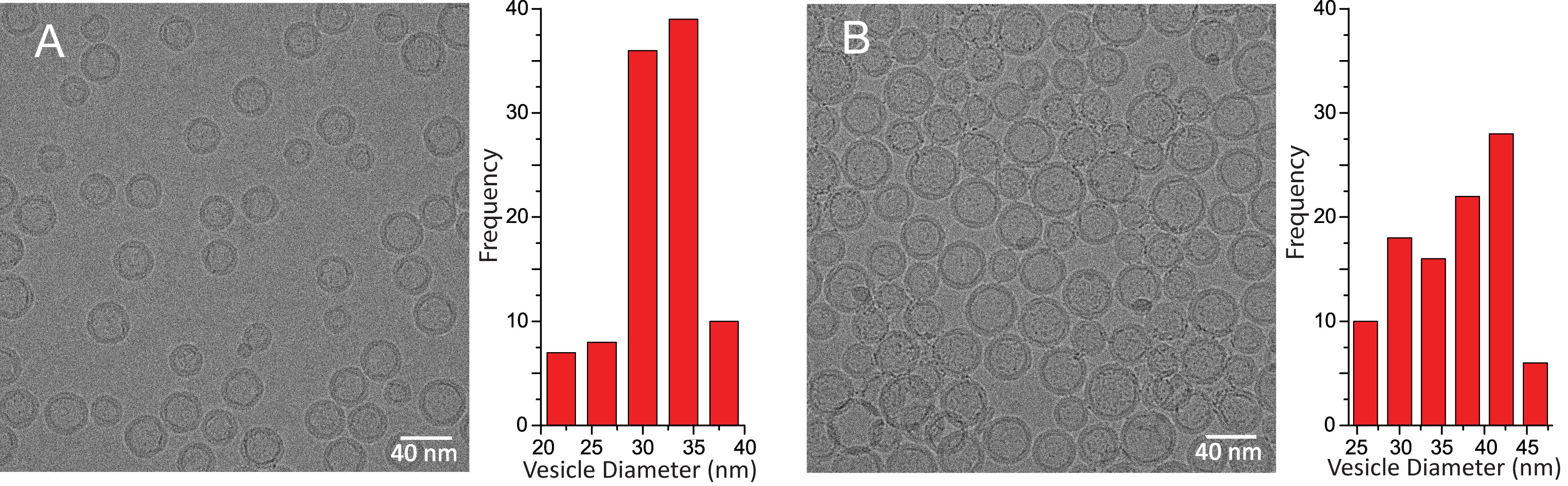
Figure 3. Cryo-EM analyses showing the size, homogeneity, and unilamellar nature of the POPC vesicles formed from highly diluted POPC/DHPC bicelle mixtures. A. Cryo-EM showing POPC unilamellar bilayer vesicles (31.5 ± 3.8 nm outer diameter) resulting from 0.5 q-value POPC/DHPC bicelle mixtures (6 mM; 0.335 wt% [w/v]) kept overnight at room temperature. The bar graph shows vesicle size distribution. B. Cryo-EM showing POPC unilamellar bilayer vesicles (36.3 ± 6.1 nm outer diameter) resulting from 0.5 q-value POPC/DHPC bicelle mixtures (30 mM; 1.67 wt% [w/v]) kept overnight at room temperature. The bar graph depicts the distribution of vesicle size (adapted from Gao et al., 2020).
If more dilute concentrations of POPC unilamellar bilayer vesicles formed from highly diluted 0.5 q-value POPC/DHPC bicelle mixtures are needed, simply add and mix the desired amount of aqueous buffer at room temperature (Gao et al., 2020).
Note: As documented by Gao et al. (2020), the ‘shelf-life’ of the bicelle mix dilution vesicles is exceptionally good with respect to stability and storage capacity. In intervesicular lipid transfer assays, bicelle mix dilution vesicles, which were subjected to freeze-thawing, or kept up to 6 days at room temperature, showed no negative effects on lipid transfer kinetics.
Data analysis
Cryo-EM data was collected, as previously described (Gao et al., 2020), using a Titan Krios electron microscope operating at 300kV and equipped with a Falcon 3EC direct electron detector (Thermo Fisher Scientific). Cryo-EM specimens were prepared by applying samples of 4 μl of the phosphatidylcholine suspensions to glow-discharged Quantifoil Cu R1.2/1.3 grids (Electron Microscopy Sciences). The grids were blotted for 4 s at room temperature under 95% humidity and plunge frozen in liquid ethane using a Vitrobot Mark IV (Thermo Fisher Scientific). Micrograph movies were recorded in counting mode, using the EPU automated data collection software at nominal magnification of 96,000×, which corresponds to a calibrated pixel size of 0.895 Angstroms. The single-bilayer (unilamellar) nature of the POPC vesicles was determined by observation of cryo-EM micrographs. POPC vesicle homogeneity was based on diameter measurement. Standard errors for vesicle size were calculated from the diameters of ~200 vesicles.
Notes
Our findings are consistent with studies by Katsaras and colleagues who investigated conditions that promote unilamellar bilayer vesicle (ULV) formation from DMPC/DMPG/DHPC bicelle mixtures (Nieh et al., 2004, 2005, 2009 and 2011; Harroun et al., 2005; Yue et al., 2005; Mahabir et al., 2014). Their phase diagrams indicate that ULVs can exist if temperatures are >35°C, and at lipid concentrations <1.25 wt%. Low temperature monodisperse bicelles could form low-polydispersity ULVs when sufficiently warmed, to undergo a gel-to-liquid crystalline (Lα) phase transition for DMPC (24°C). Notably, dilution of high-concentration lipid mixtures at temperatures >DMPC’s phase transition temperature resulted in polydisperse ULVs. Also, an optimal charge density of DMPG was needed to avoid generation of a mixture of ULVs and multilamellar vesicles. In contrast, with our approach, the much lower POPC liquid crystalline (Lα) phase transition temperature (-2°C), and the 12fold lower critical bilayer concentration of POPC (0.5 nM) versus DMPC (6 mM) appear to be factors enabling the simpler and more straightforward generation of homogeneous POPC ULVs at room temperature.
The protocol reported here is also useful for assessing protein binding affinity to lipid vesicles. In fluorescence resonance energy transfer studies used to compare the binding of wild-type cytosolic phospholipase A2α C2-domain and various point mutants to PC model membranes (Hirano et al., 2019), the POPC/DHPC bicelle mixture dilutions are nearly identical to those described in Steps 7a-e, and used for ‘donor’ vesicle formation reported in the lipid transfer assay study (Gao et al., 2020), consistent with POPC unilamellar bilayer vesicle formation.
For additional details regarding the benefits of applying the current protocol for POPC unilamellar bilayer vesicle production, readers are referred to Gao et al. (2020) . The simplicity of the approach could be useful in high throughput assays, when phosphoglyceride-based unilamellar bilayer vesicles are needed as an assay component.
Recipes
The precise details regarding the ingredients and preparation conditions used to generate small, stable, homogeneous, unilamellar POPC bilayer vesicles from 0.5 q-value POPC/DHPC bicelle mixtures are provided in steps 7 and 8 of the Procedure section.
Acknowledgments
We are grateful for support from the National Institutes of Health via HL125353 and GM45928 and the Hormel Foundation. The approach detailed here was initially summarized in Gao et al. (2020). Anal Chem 92(4): 3417-3425.
Competing interests
The authors declare no competing interests.
Ethics
The current study involved no use or involvement of human or animal subjects.
References
- Akbarzadeh, A., Rezaei-Sadabady, R., Davaran, S., Joo, S. W., Zarghami, N., Hanifehpour, Y., Samiei, M., Kouhi, M. and Nejati-Koshki, K. (2013). Liposome: classification, preparation, and applications. Nanoscale Res Lett 8(1): 102.
- Ajeeshkumar, K. K., Aneesh, P. A., Raju, N., Suseela, M., Ravishankar, C. N. and Benjakul, S. (2021). Advancements in liposome technology: Preparation techniques and applications in food, functional foods, and bioactive delivery: A review. Compr Rev Food Sci Food Saf 20(2): 1280-1306.
- Ali, S., Brockman, H. L. and Brown, R. E. (1991). Structural determinants of miscibility in surface films of galactosylcermamide and phophatidylcholine: effect of unsaturation in the galactosylceramide acyl chain. Biochemistry 30(47): 11198-11205.
- Bartlett, G. R. (1959). Phosphorus assay in column chromatography. J Biol Chem 234(3): 466-468.
- Beaugrand, M., Arnold, A. A., Hénin, J., Warschawski, D. E., Williamson, P. T. and Marcotte, I. (2014). Lipid concentration and molar ratio boundaries for the use of isotropic bicelles. Langmuir 30(21): 6162-6170.
- Chattopadhyay, A. and Harikumar, K. G. (1996). Dependence of critical micelle concentration of a zwitterionic detergent on ionic strength: implications in receptor solubilization. FEBS Lett 391(1-2): 199-202.
- Chng,C. P. (2013). Effect of simulation temperature on phospholipid bilayer–vesicle transition studied by coarse-grained molecular dynamics simulations. Soft Matter 9(30): 7294-7301.
- Fromherz, P. (1983). Lipid-vesicle structure: Size control by edge-active agents. Chemical Physics Letters 94(3): 259-266.
- Gao, Y. G., My Le, L. T., Zhai, X., Boldyrev, I. A., Mishra, S. K., Tischer, A., Murayama, T., Nishida, A., Molotkovsky, J. G., Alam, A. et al. (2020). Measuring Lipid Transfer Protein Activity Using Bicelle-Dilution Model Membranes. Anal Chem 92(4): 3417-3425.
- Harroun, T. A., Koslowsky, M., Nieh, M. P., de Lannoy, C. F., Raghunathan, V. A. and Katsaras, J. (2005). Comprehensive examination of mesophases formed by DMPC and DHPC mixtures. Langmuir 21(12): 5356-5361.
- Huang, C., Quinn, D., Sadovsky, Y., Suresh, S. and Hsia, K. J. (2017). Formation and size distribution of self-assembled vesicles. Proc Natl Acad Sci U S A 114(11): 2910-2915.
- Hirano, Y., Gao, Y. G., Stephenson, D. J., Vu, N. T., Malinina, L., Simanshu, D. K., Chalfant, C. E., Patel, D. J. and Brown, R. E. (2019). Structural basis of phosphatidylcholine recognition by the C2-domain of cytosolic phospholipase A2α. eLife 8: e44760.
- Keller, H., Worch, R. and Schwille, P. (2013). Model membrane systems. Methods Mol Biol 1008: 417-438.
- Lichtenberg, D. and Barenholz, Y. (1988). Liposomes: preparation, characterization, and preservation. Methods Biochem Anal 33: 337-462.
- Lu, Z., Van Horn, W. D., Chen, J., Mathew, S., Zent, R. and Sanders, C. R. (2012). Bicelles at low concentrations. Mol Pharm 9(4): 752-761.
- Laouini, A., Jaafar-Maalej, C., Limayem-Blouza, I., Sfar, S., Charcosset, C. and Fessi, H. (2012). Preparation, Characterization and Applications of Liposomes: State of the Art. J Colloid Sci Biotech 1(2): 147-168.
- Marrink, S. J. and Mark, A. E. (2003). Molecular dynamics simulation of the formation, structure, and dynamics of small phospholipid vesicles. J Am Chem Soc 125(49): 15233-15242.
- Mahabir, S., Small, D., Li, M., Wan, W., Kučerka, N., Littrell, K., Katsaras, J. and Nieh, M. P. (2013). Growth kinetics of lipid-based nanodiscs to unilamellar vesicles-a time-resolved small angle neutron scattering (SANS) study. Biochim Biophys Acta 1828(3): 1025-1035.
- Manni, M. M., Tiberti, M. L., Pagnotta, S., Barelli, H., Gautier, R. and Antonny, B. (2018). Acyl chain asymmetry and polyunsaturation of brain phospholipids facilitate membrane vesiculation without leakage. eLife 7: e34394.
- Maja, L., Eljko, K. and Mateja, P. (2020). Sustainable technologies for liposome preparation. J Supercrit Fluids 165: 104984.
- Nieh, M. P., Harroun, T. A., Raghunathan, V. A., Glinka, C. J. and Katsaras, J. (2004). Spontaneously formed monodisperse biomimetic unilamellar vesicles: the effect of charge, dilution, and time. Biophys J 86(4): 2615-2629.
- Nieh, M. P., Raghunathan, V. A., Kline, S. R., Harroun, T. A., Huang, C. Y., Pencer, J. and Katsaras, J. (2005). Spontaneously formed unilamellar vesicles with path-dependent size distribution. Langmuir 21(15): 6656-6661.
- Nieh, M. P., Kucerka, N. and Katsaras, J. (2009). Spontaneously formed unilamellar vesicles. Methods Enzymol 465: 3-20.
- Nieh, M. P., Dolinar, P., Kucerka, N., Kline, S. R., Debeer-Schmitt, L. M., Littrell, K. C. and Katsaras, J. (2011). Formation of kinetically trapped nanoscopic unilamellar vesicles from metastable nanodiscs. Langmuir 27(23): 14308-14316.
- Nkanga, C., Bapolisi, A., Ikemefuna, N. and Rui, K. (2019). General Perception of Liposomes: Formation, Manufacturing and Applications. In: Liposomes - Advances and Perspectives. Catala, A. (Ed.). DOI: 10.5772/intechopen.84255.
- Palladino, P. and Ragone, R. (2011). Ionic strengths effects on the critical micellar concentration of ionic and nonionic surfactants: the binding model. Langmuir 27: 14065-14070.
- Patil, Y. P. and Jadhav, S. (2014). Novel methods for liposome preparation. Chem Phys Lipids 177: 8-18.
- Sanders, C. R., 2nd and Schwonek, J. P. (1992). Characterization of magnetically orientable bilayers in mixtures of dihexanoylphosphatidylcholine and dimyristoylphosphatidylcholine by solid-state NMR. Biochemistry 31(37): 8898-8905.
- Sanders, C. R. and Prosser, R. S. (1998). Bicelles: a model membrane system for all seasons? Structure 6(10): 1227-1234.
- Tejera-Garcia, R., Connell, L., Shaw, W. A. and Kinnunen, P. K. (2012). Gravimetric determination of phospholipid concentration. Chem Phys Lipids 165(6): 689-695.
- Yue, B., Huang, C. Y., Nieh, M. P., Glinka, C. J. and Katsaras, J. (2005). Highly stable phospholipid unilamellar vesicles from spontaneous vesiculation: A DLS and SANS study. J Phys Chem B 109(1): 609-616.
Article Information
Copyright
© 2021 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Gao, Y. G., My Le, L. T., Alam, A. and Brown, R. E. (2021). A Simple and Straightforward Approach for Generating Small, Stable, Homogeneous, Unilamellar 1-Palmitoyl 2-Oleoyl Phosphatidylcholine (POPC) Bilayer Vesicles. Bio-protocol 11(24): e4271. DOI: 10.21769/BioProtoc.4271.
Category
Biochemistry > Lipid > Membrane lipid
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link









