- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Live Imaging of Apoptotic Extrusion and Quantification of Apical Extrusion in Epithelial Cells
Published: Vol 11, Iss 22, Nov 20, 2021 DOI: 10.21769/BioProtoc.4232 Views: 3779
Reviewed by: Andrea PuharJan HuebingerDana Manuela Savulescu

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Monitoring of Sperm-Independent Calcium Oscillations in Immature Oocytes of Mice
Sae Horiike [...] Hidehiko Ogawa
Feb 5, 2026 246 Views
Time-Lapse Into Immunofluorescence Imaging Using a Gridded Dish
Nick Lang [...] Andrew D. Stephens
Feb 20, 2026 242 Views

How to Train Custom Cell Segmentation Models Using Cell-APP
Anish J. Virdi and Ajit P. Joglekar
Feb 20, 2026 355 Views
Abstract
Apoptotic cell death eliminates unhealthy cells and maintains homeostatic cell numbers within tissues. Epithelia, which serve as fundamental tissue barriers for the body, depend on a physical expulsion of dying cells (apoptotic cell extrusion) to remain sealed and intact. Apoptotic cell extrusion has been widely studied over recent years, with researchers using various approaches to induce apoptotic cell death. Unfortunately, the majority of chemical-based approaches for cell death induction rely on sporadically occurring apoptosis and extrusion, making imagining lengthy, often unsuccessful, and difficult to capture in high-quality images because of the frequent frame sampling needed to visualise the key molecular processes that drive extrusion. Here, we present a protocol that describes steps needed for laser-mediated induction of apoptosis in a cell of choice, followed by imaging of apoptotic extrusion in confluent monolayers of epithelial cells. Moreover, we provide the description of a new approach involving the mixing of labelled and unlabelled cells. In particular, this protocol characterises how cells surrounding apoptotic cells behave, with high spatial and temporal resolution. This can be achieved without the optical interference that apoptotic cells cause as they are physically expelled from the monolayer and move out of focus for imaging. Finally, the protocol is accompanied by detailed procedures describing cell preparation for apoptotic extrusion experiments, as well as post-acquisition analysis required to evaluate rates of successful extrusion.
Keywords: EpitheliaBackground
Epithelial tissues rely on effective extrusion to eliminate apoptotic cells for the preservation of tissue barriers and to prevent inflammation. As homeostatic apoptosis is a relatively sporadic event in healthy cultured epithelia, apoptosis needs to be experimentally induced to reliably visualise the extrusion phenomenon. This may be achieved by a variety of methods, including exposure to UV radiation (Rosenblatt et al., 2001; Slattum et al., 2009; Andrade and Rosenblatt, 2011), treatment with various chemicals [e.g., Etoposide (Andrade and Rosenblatt, 2011; Michael et al., 2016; Teo et al., 2020), Camptothecin (Andrade and Rosenblatt, 2011), or activation of Fas receptors (Andrade and Rosenblatt, 2011)]. However, most commonly used approaches rely on randomly-induced apoptosis, making it challenging to capture high quality images with high temporal resolution to characterise dynamically progressing extrusion events. The protocol presented here describes induction of apoptosis in pre-selected cells of choice, thus allowing continuous high-quality imaging of apoptotic extrusion from induction of cell death to completion of extrusion. The following protocol may be adopted to selectively induce apoptosis in other models [e.g., Zebrafish periderm (Duszyc et al., 2021)] and can also be used for detailed analysis of the behaviour of neighbouring cells during the extrusion process by selectively labelling neighbours with genetically encoded fluorescent proteins (Duszyc et al., 2021).
Materials and Reagents
40 µm cell strainers (Biologix, catalog number: 15-1040)
Glass bottom dishes, No. 1.5 Coverslip (35 mm diameter, MATTEK, catalog number: P35G-1.5-20-C) or an equivalent (e.g., ibidi, catalog number: 81158)
75 cm2 tissue culture treated flasks with filter caps (Thermo Fisher Scientific, NuncTM EasYFlaskTM catalog number: NUN156499) or an equivalent (e.g., Corning, catalog number: CLS430641U)
Disposable serological pipettes (Corning® Costar® Stripette®, catalog numbers: CLS4487 [5 ml]; CLS4488 [10 ml])
Sterile filter tips (ZAPTM, catalog numbers: 1051-965-038 [0.1-10 µl]; 1055-965-018 [1-40 µl]; 1059-965-008 [1-200 µl]; 1057-965-018 [1,000 µl])
1.5 ml Eppendorf tubes (Eppendorf South Pacific Pty Ltd, catalog number: 0030120086)
Centrifuge Falcon Tubes (BD FalconTM, catalog numbers: BDAA352096 [15 ml], BDAA352070 [50 ml])
Epithelial cell lines with well-defined cell-cell junctions such as MCF-7 or Caco-2: wild type and expressing fluorescently tagged proteins that can mark cell-cell junctions (e.g., E-cadherin or ZO-1), the cytoskeletal cortex at cell-cell junctions (e.g., Myosin regulatory light chain, MRLC) or biosensors of junctional proteins (e.g., active RhoA biosensor AHPH). Green (e.g., GFP) or red (e.g., mCherry) fluorescent proteins should be used as fluorescent tags to distinguish from far-red Annexin V staining (Alexa FluorTM 647)
Fetal bovine serum (FBS) (HyCloneTM, catalog number: SH30084.03)
Annexin V, Alexa FluorTM 647 conjugate (Thermo Fisher Scientific, catalog number: A23204)
Opti-MEM media (Thermo Fisher Scientific, catalog number: 31985070)
LipofectamineTM RNAiMAX transfection Reagent (Thermo Fisher Scientific, catalog number: 13778150)
MEM Non-Essential Amino Acids 100× (MEM NEAA; Thermo Fisher Scientific, GibcoTM, catalog number: 11140050)
L-glutamine 100× (Thermo Fisher Scientific, GibcoTM, catalog number: 25030149)
Penicillin-Streptomycin (5,000 U/ml) (Thermo Fisher Scientific, GibcoTM, catalog number: 15070063)
DMEM for MCF-7 cells (Thermo Fisher Scientific, GibcoTM, catalog number: 11995073) or RPMI for Caco-2 cells (Thermo Fisher Scientific, GibcoTM, catalog number: 21870076)
Hank’s balanced salt solution (Sigma-Aldrich, catalog number: H8264)
Complete cell culture medium (see Recipes)
Imaging medium (see Recipes)
Equipment
Pipet gun (e.g., Thermo Fisher ScientificTM, catalog number: 9501)
Set of pipettes (e.g., Gilson PIPETMAN®, catalog numbers: FA10001M [0.2-2 µl]; FA10003M [2-20 µl]; FA10005M [20-200 µl]; FA10006M [100-1,000 µl])
Bright field microscope (e.g., Nikon Eclipse TS100 equipped with Plan Fluor 4×/0.13 phase contrast objective)
Hemocytometer counting chamber (e.g., Livingstone, catalog number: HCCB)
Cell culture CO2 incubator (e.g., PHCbi catalog number: MCO-230AICUVL-PA)
Class II biological safety cabinet (e.g., Gel Aire, model: 20 BHEN 2004D)
Benchtop centrifuge (e.g., Hettich Lab TechnologyTM ROTIFIX 32A)
Laser scanning confocal microscope minimally equipped with:
Dichroic and emission filters for the use of the 488 nm and 633nm laser lines and detection of GFP and Alexa FluorTM 647 fluorescence, respectively
488nm and 633 nm laser lines (e.g., Argon, HeNe or solid-state lasers)
Acoustic-optic tunable filters (AOTF) for irradiation of a specific region of interest (i.e., bleaching of selected areas)
2W or more tunable two-photon laser (adjustable to 790nm laser line) (e.g., MaiTai or Chameleon)
Heated chamber (37°C) for live cell imaging
40× IR-VIS compatible objective (ideally water immersion to minimise chromatic aberrations)
The experiments shown in this protocol were performed on a Zeiss LSM 710 Meta Confocal Scanner equipped with a fully tunable Mai Tai eHP DeepSee 760-1040 nm laser, heated chamber (37°C for cell imaging), and Plan Apochromat 40× 1.3 N.A. water objective.
Software
ImageJ software (https://imagej.nih.gov/ij/)
GraphPad PRISM software (http://www.graphpad.com)
Procedure
Cell preparation
Generation of epithelial cells lines expressing fluorescently tagged junctional proteins can be achieved by virus transduction, transient transfection of a plasmid encoding fluoro-tagged proteins of interest, or by tagging a gene of interest using CRISPR/Cas9 genome editing. The below protocol describes steps needed to generate mixed cultures of unlabelled and fluorescently labelled cells (Figure 1A).
Note: For simplicity, a fluorescently tagged cell line will be referred to as “GFP-positive line”. However, GFP may be replaced with any bright fluorophore from the green-red spectra.
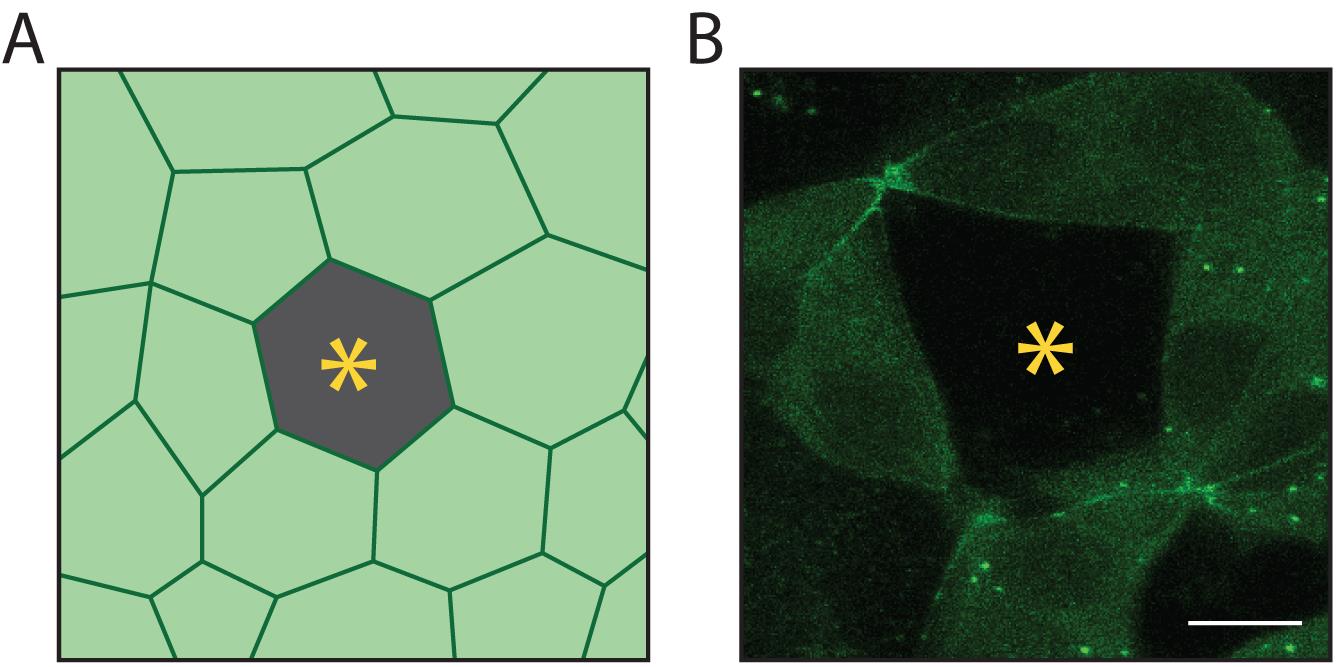
Figure 1. Target of laser injury. Cartoon (A) and a sample image (B) of a cell of interest (yellow asterisk). The target of laser-injury should be composed of one or two unlabelled cells (yellow asterisk) surrounded by fluorophore-positive neighbouring cells. Here, neighbouring cells express MRLC-GFP. Mixing of wild-type and fluorescently-labelled cells prevents optical interference, which is caused by apoptotic cells as they are physically expelled from the monolayer and move out of focus for imaging. Scale bar: 15 µm.Day 1
Harvest approximately 80-90% confluent T75 flasks of GFP-positive and wild-type (WT) cell lines by trypsinisation, spin them down (1,500 rpm, 3 min), and resuspend in 10 ml of fresh full media (see Recipes). Pass the cell suspensions through separate cell strainers.
Combine the GFP-positive and WT cells in a 20:1 ratio by gently pipetting up and down to ensure that cells are thoroughly mixed.
Seed mixed cells on glass bottom dishes at 30% confluency (Caco-2 cells: 3 × 105 cells resuspended in 2 ml of growth medium) or 50% confluency (MCF-7 cells: 5 × 105 cells resuspended in 2 ml of growth medium). Seeding confluency should be adjusted depending on how individual cell lines behave, to allow at least 48 h between seeding and reaching 100% confluency so that mature junctions form between cells.
Day 2
If knock-down of a protein of choice is required for experiments, transfect cells with siRNA according to established procedure or follow siRNA transfection protocol using Lipofectamine RNAiMAX (Appendix I).
Note: If cells are resistant to transfection using Lipofectamine RNAiMAX, use lentiviral transduction to knock-down proteins of choice [for a protocol, see Priya and Gomez (2013)] or use CRISPR/Cas9 gene knock-out lines (for generation of these, see Ran et al., 2013).
Day 3
Continue to culture cells in a cell incubator.
Day 4
Confirm that cells are fully confluent using a bright field microscope.
Warm up 1.5 ml of imaging medium to 37°C.
Directly before imaging, add 3 µl of Annexin V, Alexa FluorTM 647 conjugate to the imaging medium. Gently remove the growth medium using a 100-1,000 µl pipette (Gilson PIPETMAN® or an equivalent), and replace it with 1.5 ml of the imaging medium.
Notes:
As Annexin V, Alexa FluorTM 647 conjugate is photosensitive, aliquot the original tube into one-use aluminium-foil wrapped tubes. Store aliquots at 4°C up to the expiration date.
To confirm the knock down of a protein of interest, straight after imagining is completed, lyse the cells and extract protein for a western blot.
Image acquisition – laser injury and acquisition of apoptotic extrusion
Note: The following protocol is based on the Zeiss LSM 710 Meta Confocal Scanner (equipped with a fully tunable Mai Tai eHP DeepSee 760-1,040 nm laser, 2.4 W average laser power), but it may be optimised for use with other comparable systems [e.g., Michael et al. (2016) successfully used a Zeiss LSM 510 Meta Confocal microscope equipped with a tunable 700-1,100 nm Chameleon Laser, >2W power].
Set up the imaging to detect GFP, Alexa FluorTM 647 and Differential Interference Contrast (DIC).
Note: As AnnexinV Alexa FluorTM 647 does not mark living cells, adjust the channel gain to see the unbound Alexa FluorTM 647 positive residue in the imaging media (Figure 3A, yellow arrowheads).
In the acquisition panel, activate the following options: “Z-Stack”, “Time Series”, “Bleaching”, and “Regions” (Figure 2A).
Note: We use the ‘Bleaching’ tool in the ZEN software to irradiate a region of interest and induce apoptosis in the target cell. Similar tools are available for other commercial microscopes like Leica and Olympus, although they may be named differently.
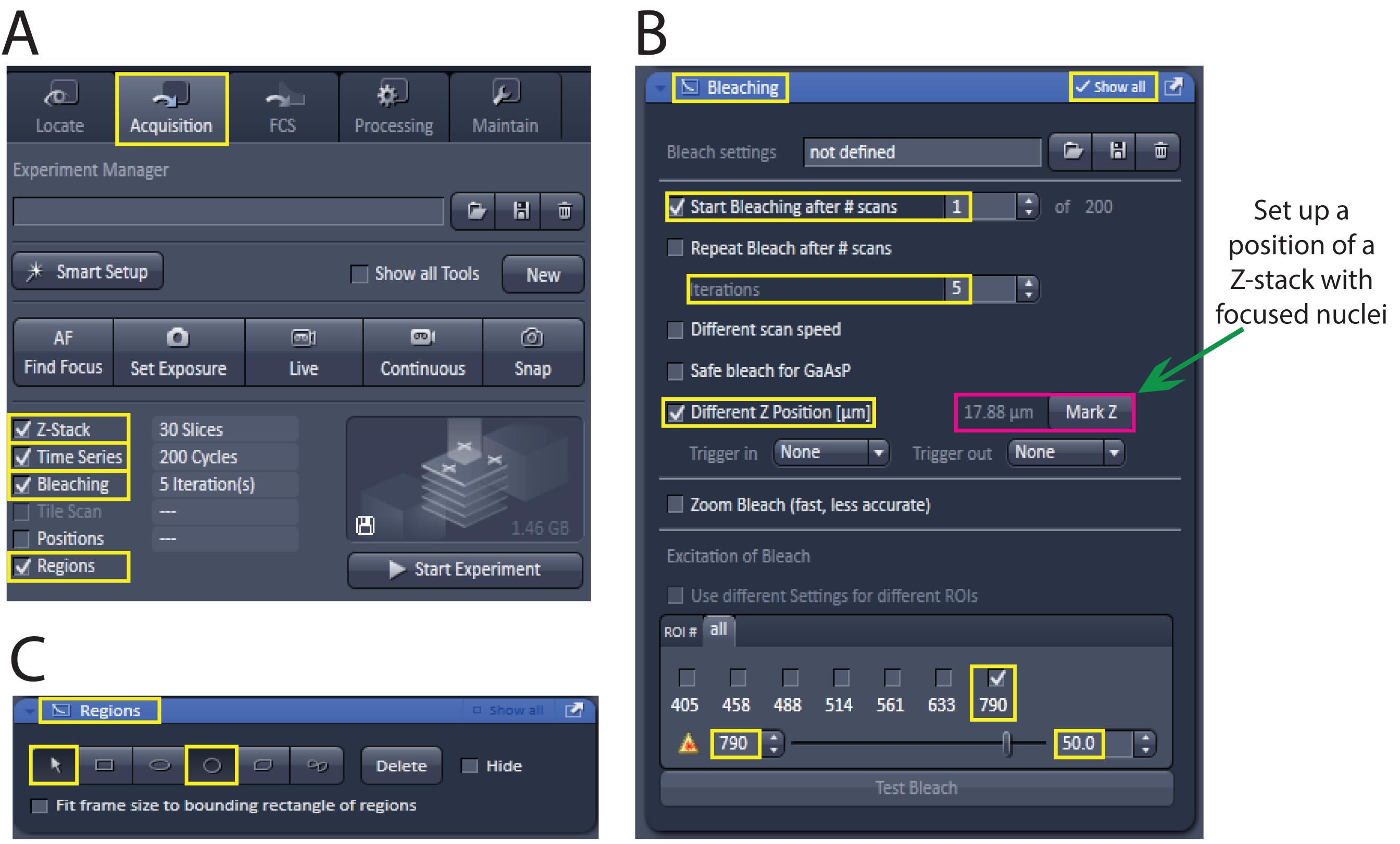
Figure 2. Snapshot of the ZEISS software including settings optimised for induction of apoptosis using the 790 nm wavelength of the MAI TAI laser.Expand the “Bleaching” window by selecting “Show All”. Activate “Start Bleaching after #scans” and enter “1”. Enter “5” in “Iterations” and select “Different Z Position (µm)”. Choose 790 nm wavelength and 50% transmission (transmission may be further adjusted to approximately 60% if apoptosis of the irradiated cells is not efficiently induced, see Step B7) (Figure 2B).
Identify target cells by finding a single or double WT, GFP-negative cell(s) surrounded by GFP-positive cells (Figure 1).
Set up approximately 30 1 µm Z-stacks, where the middle stack is focused on the junctional marker.
Using the DIC channel, go to a Z-stack where the nucleus of targeted cell is in focus (Figure 3C, 3D) and choose this Z-stack for bleaching, by updating the position in “Different Z Position (µm), Mark Z” in the “Bleaching” options (Figure 2B). Select a circular tool from “Regions” (Figure 2C) and draw a circle over a nucleolus of the selected cell(s) (Figure 3D, 3E).
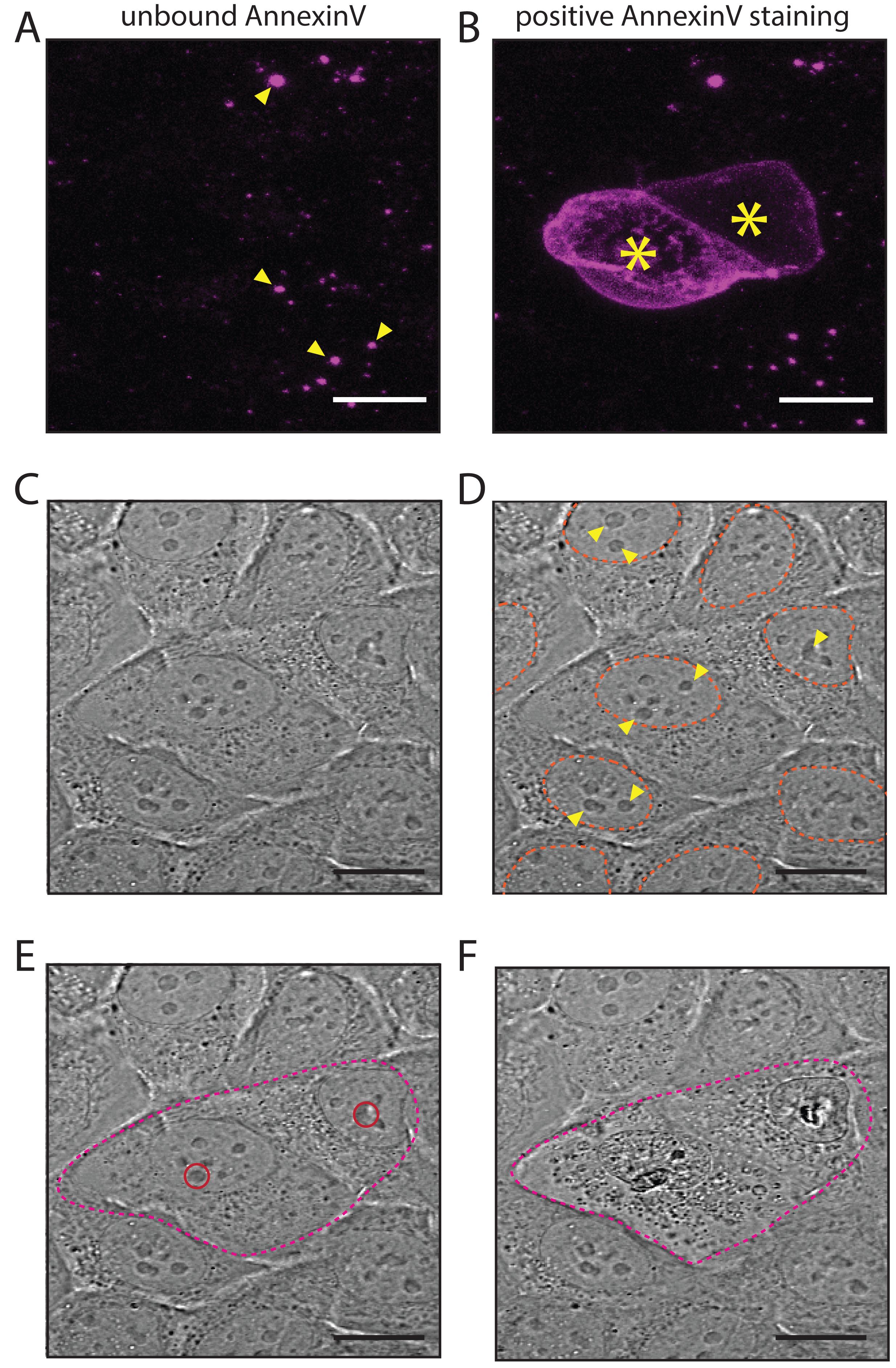
Figure 3. Optimisation of laser-injury setup. A. No apoptotic cells are present before laser injury; yellow arrowheads point at background residue of unbound Annexin V, Alexa FluorTM 647 conjugate in the cell media; B. Positive Annexin V staining of two cells following laser-induced apoptosis. Alexa FluorTM 647 conjugate was added to the imagining media at 1:500 dilution; C-D. Representative image of the DIC view focused on cell nuclei. For reference, cell nuclei are marked with orange dashed lines, and examples of nuclear condensations (nucleoli) are marked with yellow arrowheads. E-F. DIC view of laser injury. The approximate area of targeted cells is marked with a magenta dashed line, and circular regions of laser injury (E) are marked with red circles. Note the morphology of targeted cells before laser injury (E) and approximately 1 min after laser injury (F). Scale bars: 15 µm.Note: Both Alexa FluorTM 647 images (A, B) were captured using the same laser settings. To ensure that positive AnnexinV staining will be visible, ensure that the unbound residue of AnnexinV is visible before laser injury (A).
Start the imaging using the above settings. Between acquisition of the first and second frames, the MAI TAI laser will be automatically turned on and irradiate the selected region with high-power infrared light multiple times, thus injuring the targeted cell(s).
Note: Use the DIC channel to observe the morphological changes happening in the targeted cell(s) – nuclear/cytoplasmic condensation should be visible within 1-2 min after the injury (Figure 3E, 3F). If no morphological changes/AnnexinV staining are visible within 5 min after the injury (Figure 3B), adjust the transmission settings of the MAI TAI laser (Step B3) until apoptosis successfully occurs.
Set up suitable time intervals for imaging – if the goal of the experiment is to capture the behaviour of the neighbouring cells, the interval should be frequent (20-30 s); if the aim is to quantify extrusion, time intervals may be less frequent (5-10 min). Acquire time-lapse images until extrusion is completed (20-30 min) or for 1 h if extrusion is not progressing. See below for assessment of apical apoptotic extrusion.
Quantification of apoptotic extrusion
Open an acquired time-lapse image in ImageJ software, choose “colorized” as a colour mode.
Use the attached macro (Attachment 1: Max projection and Orthogonal View.ijm) to generate orthogonal views and a maximum projection of the time lapse image.
Note: Alternatively, follow the step-by-step protocol (Appendix 2) to generate the orthogonal views and maximum projection of the time lapse images.
Attachment 1: Macro generating orthogonal views and a maximum projection from time laps images.
Orthogonal views: using the bottom slider, select a last frame of the time-lapse; position the Z-view-marking lines according to Figure 4A: the lines should cross at the point where neighbouring cells merged underneath the apoptotic cell (Figure 4A, middle panels), or in the centre of the apoptotic cell, if extrusion failed (Figure 4A, right panels). Go back to the first frame of the movie to confirm that the GFP-tagged junctional protein is clearly visible in the Z-views (Figure 4A, left panels).
Note: Rotate the image and slightly adjust the position of the Z-views to clearly capture both the junctional protein and the AnnexinV-tagged apoptotic cell.
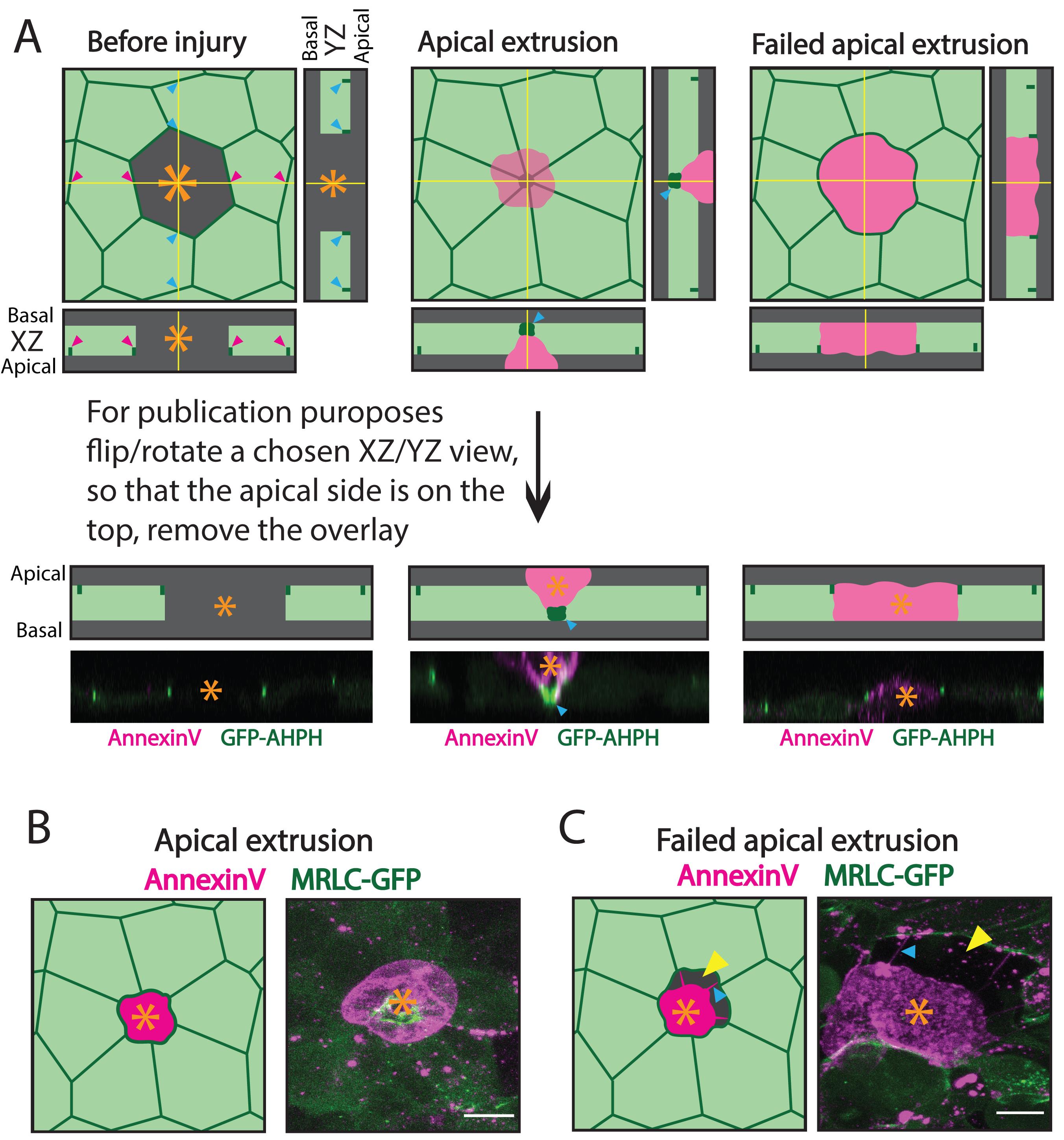
Figure 4. Assessment of apoptotic apical extrusion. A. A cartoon (top) and a sample example (bottom; magenta – AnnexinV staining, green – AHPH RhoA biosensor expressed in the neighbouring cells) of orthogonal views. Left panels: views before laser injury, middle panels: after completion of apical extrusion (blue arrowheads – accumulation of a junctional maker of the neighbouring cells underneath the apoptotic cell), right panels: failed extrusion (an apoptotic cell is retained in a monolayer 60 min after laser injury). Orange asterisks mark apoptotic cells (middle and right) or a targeted cell before the laser injury (left). B. A cartoon (left) and an example (right) of a successful apical extrusion, during which the epithelial monolayer maintained its integrity. C. A cartoon (left) and an example (right) of failed apical extrusion, during which the epithelial monolayer did not maintain its integrity – an apoptotic cell (orange asterisk) detaches from the neighbouring cells, leading to the formation of empty gaps (yellow arrowheads) between the dying cell and its neighbours. Blue arrowheads point at “detachment fibres”. Scale bars: 15 µm.Asses apical extrusion of apoptotic cells by noting whether the junctional-GFP signal from the neighbouring cells came together underneath the AnnexinV-positive apoptotic cell. If the apoptotic cell was still present between the junctional marker 60 min after the laser-injury, classify extrusion as failed (Figure 4A). If the neighbouring cells came together underneath the apoptotic cell, move on to the next step.
Maximum projection: Asses the integrity of the monolayer at the site of apoptosis – in a successful extrusion, the integrity of the monolayer should be maintained (the apoptotic cell should not detach from the neighbouring cells before extrusion is completed) (Figure 4B). If an empty space is present between the neighbouring cells and the apoptotic cells, classify extrusion as failed (Figure 4C). Classify extrusion as successful if the integrity of the monolayer was not perturbed during the extrusion process (the apoptotic cell did not detach from the neighbouring cells at any point while extrusion was progressing) and the apoptotic cell was expelled in an apical direction (see Step C4).
Note: If the apoptotic cell detaches from the neighbouring cells, you may observe characteristic “detachment fibres” (Figure 4C, blue arrowheads).
Score the extrusion for all of the remaining time-lapse images (n ≥ 5 apoptotic cells per condition per experiment). Calculate the percentage of successfully extruded cells/all of the apoptotic cells in the condition. Enter the obtained numbers acquired from at least three independent experiments into a Prism Column Graph. If comparing various conditions, calculate significance and generate a graph.
Note: A raw file of a sample time-lapse imaging of induction of apoptosis and subsequent apoptotic extrusion is attached (Attachment 2).
Attachment 2: A sample raw file of a sample time-lapse imaging of induction of apoptosis and subsequent apoptotic extrusion. Neighbouring cells express a RhoA biosensor (AHPH-GFP, green channel), apoptosis of two GFP-negative cells was induced using laser microirradiation according to the protocol presented here. Apoptosis of the injured cells was confirmed by Annexin-V staining (magenta channel).
Recipes
Complete cell culture medium
DMEM or RPMI supplemented with 10% FBS
1% Non-Essential Amino Acids (NEAA)
1% L-glutamine
100 units/ml Penicillin and 100 units/ml Streptomycin
Imaging medium
Hank’s balanced salt solution supplemented with 10 mM HEPES pH 7.4
5 mM CaCl2 and 10% FBS
Acknowledgments
This work was supported by grants from the National Health and Medical Research Council of Australia (1123816, 1164462, 1136592) and from the Australian Research Council (DP19010287).
Competing interests
The authors declare no competing interests.
References
- Andrade, D. and Rosenblatt, J. (2011). Apoptotic regulation of epithelial cellular extrusion. Apoptosis 16(5): 491-501.
- Duszyc, K., Gomez, G. A., Lagendijk, A. K., Yau, M. K., Nanavati, B. N., Gliddon, B. L., Hall, T. E., Verma, S., Hogan, B. M., Pitson, S. M., Fairlie, D. P., Parton, R. G. and Yap, A. S. (2021). Mechanotransduction activates RhoA in the neighbors of apoptotic epithelial cells to engage apical extrusion. Curr Biol 31(6): 1326-1336 e1325.
- Michael, M., Meiring, J. C. M., Acharya, B. R., Matthews, D. R., Verma, S., Han, S. P., Hill, M. M., Parton, R. G., Gomez, G. A. and Yap, A. S. (2016). Coronin 1B Reorganizes the Architecture of F-Actin Networks for Contractility at Steady-State and Apoptotic Adherens Junctions. Dev Cell 37(1): 58-71.
- Priya, R. and Gomez, G. A. (2013). Measurement of Junctional Protein Dynamics Using Fluorescence Recovery After Photobleaching (FRAP). Bio-protocol 3(20): e937.
- Ran, F. A., Hsu, P. D., Wright, J. Agarwala, V., Scott, D. A. and Zhang, F. (2013). Genome engineering using the CRISPR-Cas9 system. Nat Protoc 8: 2281-2308.
- Rosenblatt, J., Raff, M. C. and Cramer, L. P. (2001). An epithelial cell destined for apoptosis signals its neighbors to extrude it by an actin- and myosin-dependent mechanism. Curr Biol 11(23): 1847-1857.
- Slattum, G., McGee, K. M. and Rosenblatt, J. (2009). P115 RhoGEF and microtubules decide the direction apoptotic cells extrude from an epithelium. J Cell Biol 186(5): 693-702.
- Teo, J. L., Tomatis, V. M., Coburn, L., Lagendijk, A. K., Schouwenaar, I. M., Budnar, S., Hall, T. E., Verma, S., McLachlan, R. W., Hogan, B. M., et al. (2020). Src kinases relax adherens junctions between the neighbors of apoptotic cells to permit apical extrusion. Mol Biol Cell 31(23): 2557-2569.
Article Information
Copyright
© 2021 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Duszyc, K., Noordstra, I., Yap, A. S. and Gomez, G. A. (2021). Live Imaging of Apoptotic Extrusion and Quantification of Apical Extrusion in Epithelial Cells. Bio-protocol 11(22): e4232. DOI: 10.21769/BioProtoc.4232.
Category
Cell Biology > Cell imaging > Live-cell imaging
Cell Biology > Cell viability > Cell death
Developmental Biology > Cell growth and fate
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link