- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Wholemount in situ Hybridization for Spatial-temporal Visualization of Gene Expression in Early Post-implantation Mouse Embryos
Published: Vol 11, Iss 22, Nov 20, 2021 DOI: 10.21769/BioProtoc.4229 Views: 3414
Reviewed by: Pilar Villacampa AlcubierreAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Culture and Characterization of Differentiated Airway Organoids from Fetal Mouse Lung Proximal Progenitors
Zhonghui Zhang [...] Qiuling Li
Dec 5, 2024 1830 Views
Efficient circRNA Detection Using the Processive Reverse Transcriptase uMRT
Ruben Warkentin and Anna Marie Pyle
Oct 20, 2025 1433 Views
Simple and Rapid Model to Generate Differentiated Endometrial Floating Organoids
Adriana Bajetto [...] Tullio Florio
Feb 5, 2026 234 Views
Abstract
Regionalized distribution of genes plays crucial roles in the formation of the spatial pattern in tissues and embryos during development. In situ hybridization has been one of the most widely used methods to screen, identify, and validate the spatial distribution of genes in tissues and embryos, due to its relative simplicity and low cost. However, acquisition of high-quality hybridization signals remains a challenge while maintaining good tissue morphology, especially for small tissues such as early post-implantation mouse embryos. In this protocol, we present a detailed RNA in situ hybridization protocol suitable for wholemount early post-implantation mouse embryos and other small tissue samples. This protocol uses digoxigenin (DIG) labeled riboprobes to hybridize with target transcripts, alkaline phosphatase-conjugated anti-DIG antibodies to recognize DIG-labeled nucleotides, and nitroblue tetrazolium (NBT)/5-bromo-4-chloro-3-indolyl-phosphate (BCIP) chromogenic substrates for color development. Specific steps and notes on riboprobe preparation, embryo collection, probe hybridization, and color development are all included in the following protocol.
Graphic abstract:
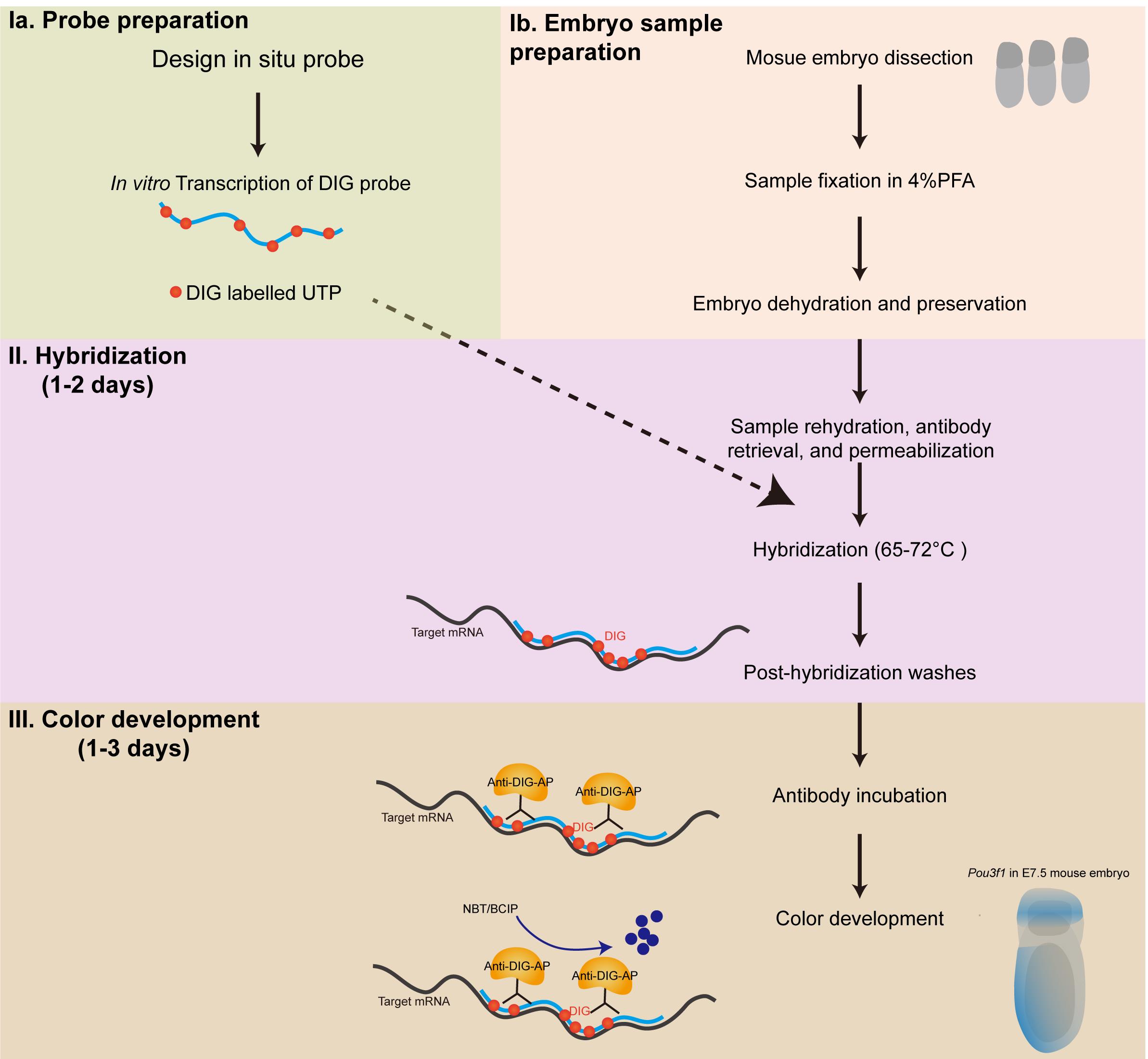
Overview of Wholemount in situ Hybridization in Early Mouse Embryos.
Background
Wholemount in situ hybridization has been widely used to explore gene expression distribution in both tissues and sections (Hauptmann and Gerster, 1994; Nieto et al., 1996). In the field of developmental biology, information on the spatial and temporal distribution of gene expression revealed by in situ hybridization has facilitated the identification of master regulators of embryogenesis. In our recent study, we reported that Pou3f1 is an important regulator of mouse neuroectoderm development by combining wholemount in situ hybridization and multiple functional analyses (Zhu et al., 2014). We optimized a wholemount RNA in situ hybridization protocol that uses digoxigenin labeled RNA probes and an anti-digoxigenin antibody conjugated with alkaline phosphatase to detect the enrichment of Pou3f1 in the anterior embryonic region of the mouse gastrula, which indicated potential biological functions of Pou3f1 in embryonic ectoderm development. Thereafter, more lineage regulators of the mouse gastrulation have been revealed and validated using this optimized protocol (Yang et al., 2018 and 2019; Peng et al., 2016 and 2019). The current protocol exhibits strong experimental robustness and displays application potential in a wide range of biological studies. Thus, we summarize the protocol here, in the hope its application can facilitate the study of gene expression.
The wholemount RNA in situ hybridization assay starts with the preparation of digoxigenin labeled RNA probes corresponding to target gene transcripts by using an in vitro transcription system and digoxigenin labeled dNTP mix. Pre-fixed embryo samples are treated with H2O2 and protease K for antigen retrieval and permeabilization. Embryos are then incubated with RNA probes and hybridized overnight. Several rounds of stringent wash are performed to remove unbound RNA probes. Subsequently, an antibody that recognizes digoxigenin is added to the reaction system and incubated overnight. Color development is performed to visualize the signal, and samples can be stored in a 50% Glycerol/PBS solution.
Further extensions based on the current protocol can be explored in the future, including, but not limited to, replacing digoxigenin labeled RNA probes with multiple fluorescent RNA probes, replacing the AP-conjugated DIG antibody with fluorescent conjugated antibodies, and even combining this method with protein immunofluorescence staining. However, for that to occur, essential optimization and adjustment of experimental conditions should be carefully performed. Noticeably, multiple alternative methods have been established these days, such as RNAscope (Wang, 2012). We recognize that the current protocol may exhibit a relative low detection sensitivity in comparison with RNAscope. Nevertheless, the outstanding properties of experimental robustness, no requirement for specialized instruments, and extremely low economic cost undoubtedly make our protocol an excellent option for the rapid screening and validation of gene expression in multiple fields of biological research.
Materials and Reagents
Note: All materials and reagents should be prepared in a DNase and RNase free environment unless otherwise described.
Pipette tips: 10 µl, 20 µl, 200 µl, 1,000 µl Microvolume tips (Axygen®, catalog numbers: TF-300-R-S, TF-20-R-S, TF-200-R-S, TF-1000-R-S)
Eppendorf tubes (Axygen®, catalog number: MCT-150-C)
35 mm × 10 mm dish (Corning, catalog number: CLS430165)
24-well plate (Corning, catalog number: 3524)
Paraformaldehyde (PFA; Sigma-Aldrich, catalog number: P6148-1kg)
DPBS (Gibco, catalog number: 14190144)
Tween-20 (Sigma-Aldrich, catalog number: P9416-100ML)
Invitrogen UltraPureTM SSC, 20× (Thermo Fisher Scientific, catalog number: 15557044)
Yeast RNA (Sigma-Aldrich, catalog number: 10109223001)
Heparin (Sigma-Aldrich, catalog number: H3149-500ku)
RiboLock RNase Inhibitor (Thermo Fisher Scientific, catalog number: Eo0382)
ScriptMAX Thermo T7 Transcription Kit (Toyobo, catalog number: TYB-TSK-101)
Methanol (e.g., Sensi Chemical)
Formamide (e.g., Sensi Chemical)
Proteinase K solution (Invitrogen, catalog number: AM2548)
Glutaraldehyde (Sinopharm Chemical, catalog number: 30092436)
DIG RNA Labeling Mix (Sigma-Aldrich, catalog number: 11277073910)
Anti-Digoxigenin AP antibody (Roche, catalog number: 11093274910)
NBT/BCIP stock solution (Sigma-Aldrich, catalog number: 11681451001)
Glycerol (Sigma-Aldrich, catalog number: G9012-100 ml)
QIAquick Gel extraction kit (QIAGEN, catalog number: 28704)
MEGAclearTM Kit (Ambion, catalog number: AM1908)
DNase I (RNase-free) (New England Biolabs, catalog number: M0303S)
30% H2O2 (w/w) in H2O (Sigma-Aldrich, catalog number: H1009-100ML)
UltraPure 0.5 M EDTA, pH 8.0 (Invitrogen, catalog number: 15575020)
Albumin, Bovine Serum, Fraction V, Crystalline (Sigma-Aldrich, catalog number: 9048-46-8)
NaCl (Sigma-Aldrich, catalog number: S5886-1KG)
Tris base (Sigma-Aldrich, catalog number: TRIS-RO)
Magnesium chloride (MgCl2; 1.00 M ± 0.01 M; Sigma-Aldrich, catalog number: M1028-100ML)
KOD FX neo (Toyobo, catalog number: KFX-201)
UltraPureTM DNase/RNase-Free Distilled Water (Invitrogen, catalog number: 10977015)
4% PFA (see Recipes)
PTW buffer (see Recipes)
20 mg/ml Yeast RNA (see Recipes)
50 mg/ml Heparin (see Recipes)
Hybridization solution (see Recipes)
10× TBST stock (see Recipes)
Blocking buffer (see Recipes)
NTMT buffer (see Recipes)
4% PFA/0.1% glutaraldehyde (see Recipes)
6% H2O2/PTW solution (see Recipes)
Equipment
Thermal cycler (Applied Biosystems, model: 9700)
Thin-walled PCR tubes with caps (Axygen®, catalog number: PCR-02-L-C)
NanoDrop 2000 (Thermal Scientific)
Hybridization incubator (SciGene, model: 2000)
Olympus SZX10/16 microscope
Procedure
Note: All steps should be performed in a DNase and RNase free environment unless otherwise described.
Collection of Sample/Embryo
Carefully collect tissue samples/mouse embryos (Figure 1) in a 35 mm dish or 24-well plate with DPBS (Downs and Davies, 1993; Piliszek et al., 2011; Pereira et al., 2011).
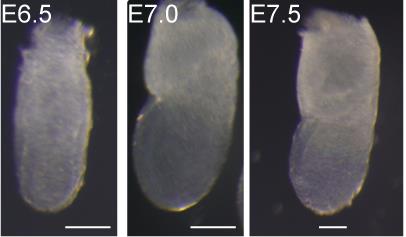
Figure 1. Representative images of the unstained collected mouse gastrula at E6.5, E7.0, and E7.5 stages. Images were acquired with a Olympus SZX10/16 microscope. Scale bars: 100 μm.Fix embryos in 4% PFA (see Recipes) at 4°C overnight.
Transfer the embryos into a graded series of methanol (25% Methanol/DPBS; 50% Methanol/DPBS; 75% Methanol/DPBS; 100% methanol) at room temperature (RT). Embryos are dehydrated for 5 min in each condition. Sufficient volume should be applied to completely submerge the embryo samples.
Note: Prepare graded series of methanol buffer right before use.
Pause point: The dehydrated embryos could be stored in 100% methanol at -20°C for up to 1 week.
Preparation of digoxigenin labeled RNA Probes
Primer design for target cDNA sequence cloning:
For direct transcription of the PCR product in vitro, a minimal T7 promoter sequence (5-TAATACGACTCACTATAGGGAGA-3) should be added to the 5’ terminal of the primer. The length of probe sequence should be 250-1,500 bases; probes with 600-900 bases exhibit the highest sensitivity and specificity.
PCR amplification and purification of probe DNA:
A cDNA pool with high enrichment of target transcripts was used as PCR template for amplification of probe DNA. DNA polymerases such as KOD FX Neo with high fidelity characteristics are recommended. The exact PCR conditions should be adjusted according to the selected probe. After agarose gel separation, excise target DNA fragments precisely and perform gel extraction following the manufacturer’s instructions (Figure 2). Determine the concentration of acquired DNA using NanoDrop 2000.
Note: For in vitro transcription from plasmid, the plasmid should be linearized with appropriate restriction enzyme digestion and purified with a commercial kit.
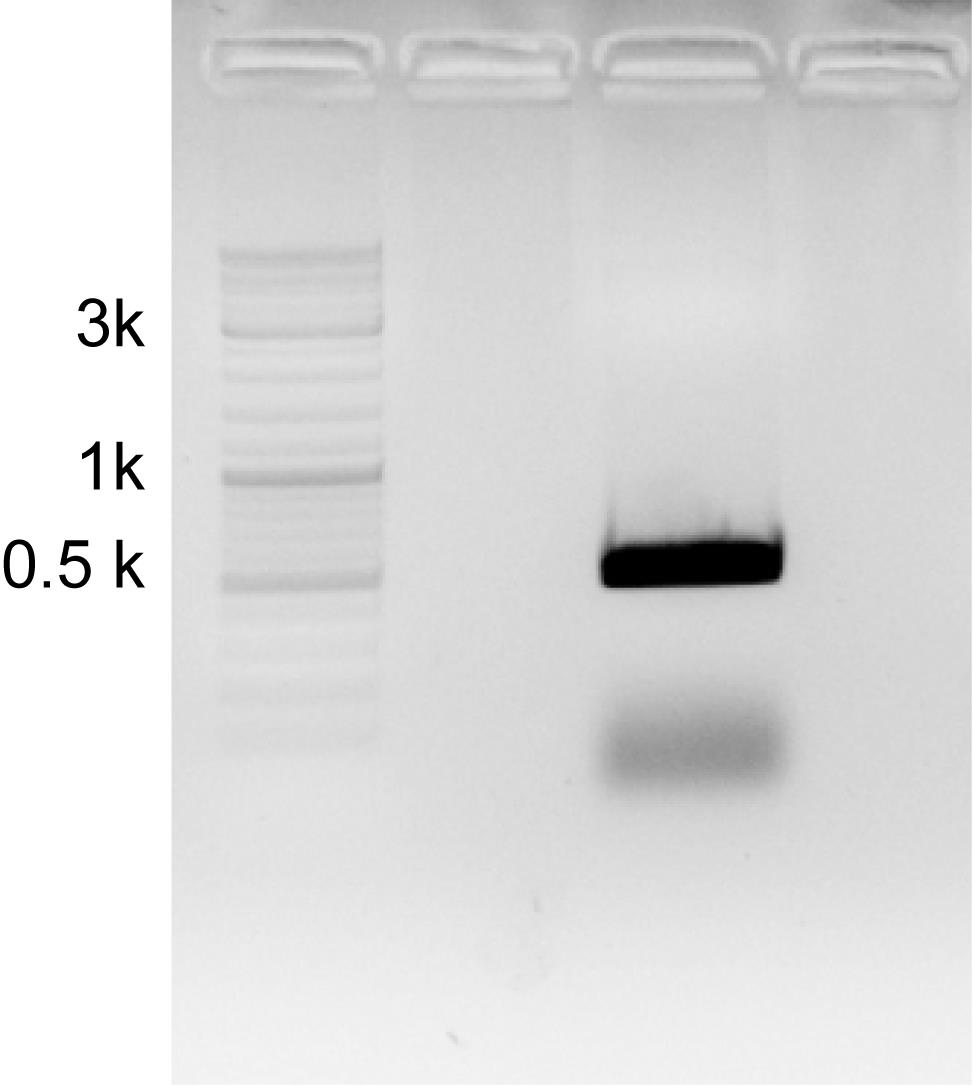
Figure 2. Specific probe DNA (here for Tal1 gene) amplified through PCR. To determine PCR specificity, the PCR product is subjected to agarose gel electrophoresis. A specific DNA band can be observed, excised, and purified for further usage.Transcription of the DIG probe:
Prepare the following reaction system:
Component For 1 µg DNA DNA 1 µg 10× transcription buffer 3 µl DIG-nucleotide mix 2 µl RiboLock RNase Inhibitor 1 µl T7 RNA polymerase 2 µl Water to 30 µl Total 30 µl Incubate the reaction at 37°C for 3 h in the thermocycler, with lid temperature no higher than 55°C.
To remove the template DNA, add 0.5 µl DNase I to the reaction mix directly and mix well, then incubate the mix at 37°C for 15 min.
Purify the acquired RNA transcript with MEGAclearTM Kit following the manufacturer’s instructions or perform phenol:chloroform extraction followed by alcohol precipitation manually. The RNA probes can be directly dissolved in nuclease free water.
Sample rehydration, antigen retrieval, and permeabilization
Rehydrate the embryos in graded methanol/PTW buffer (see Recipes) (75%, 50%, and 25% methanol in PTW) for 2-4 min in each concentration, allowing embryos to settle down to the bottom between changes. Wash embryos with PTW for 10 min twice.
Note: Prepare graded series of methanol buffer right before use.
Incubate the embryos in 6% H2O2/PTW solution at RT for 10 min, and then wash twice with PTW buffer.
Dilute proteinase K in PTW buffer at a final concentration of 10 µg/ml proteinase K in the reaction mix. Remove PTW buffer thoroughly and incubate embryos in 10 µg/ml proteinase K reaction mix at RT. The reaction duration varies for different embryo stages. To specify, for embryos ranging from E7.0 to E9.0 embryos, the appropriate reaction duration should be 7-20 min, but longer times should be pre-tested for more advanced embryos. A pre-experiment for optimization of the conditions is strongly recommended.
Remove proteinase K buffer carefully and rinse with PTW buffer twice.
Post-fix the digested embryos in 4% PFA/0.1% glutaraldehyde fixation mix (see Recipes). Incubate the embryos for 20-30 min at RT.
Remove the fixation buffer, and carefully wash with PTW buffer twice.
Hybridization of RNA probes to the embryos
Wash the embryos with hybridization solution warmed at 68°C twice; add the hybridization solution (see Recipes) and allow embryos to equilibrate until they sink to the bottom.
Incubate for 2-6 h at 65-72°C. The optimal temperature varies between different RNA probes. Usually, a 68°C hybridization temperature works for most probes we have tested.
Remove the hybridization solution and replace with the probe diluted in hybridization solution (200-500 ng/ml). Incubate the embryos at their corresponding temperature overnight.
Re-collect the probe. Probes in hybridization solution can be re-used 6-8 times. Re-collected probes can be stored at -20°C for up to two months.
Wash embryos with hybridization buffer warmed at 70°C for 30 min three times.
Wash embryos with 50% hybridization buffer/50% TBST buffer at 70°C for 20 min.
Wash embryos with TBST buffer (see Recipes) on a rocker platform at RT three times.
Antibody incubation and digoxygenin detection
Prepare blocking buffer (see Recipes) and incubate with embryos for 2-3 h at RT.
Prepare antibody incubation reaction solution with 1:2,000 diluted anti-digoxigenin AP antibody in blocking buffer. Incubate on a rocker platform at 4°C overnight.
Discard the antibody solution and wash the embryos with TBST buffer for 30 min three times. If required, wash the embryos with an extended overnight wash to reduce background signals.
Wash the embryos twice with freshly made NTMT buffer (see Recipes).
Discard the NTMT buffer and incubate embryos with NBT/BCIP solution (1:50 in NTMT buffer).
Observe the signal frequently during the first two hours of NBT/BCIP solution incubation.
Note: Refresh the NBT/BCIP solution if the solution turns red.
Stop the reaction by rinsing the embryos in TBST approximately three times until an obvious signal appears.
Fix the embryos in 4% PFA buffer overnight.
Transfer the embryos into 50% glycerol/PBS, and record representative images of embryos samples (Figure 3).
The post-fixed embryos can be store at 4°C for more than one year.
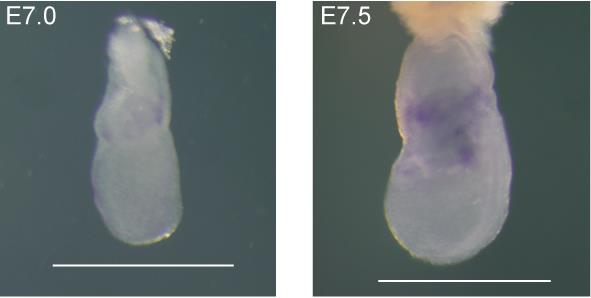
Figure 3. Representative images of wholemount mouse early embryo in situ hybridization results of Tal1 gene. The images list embryos at E7.0 and E7.5 stages, from which Tal1 starts to be expressed in extraembryonic mesoderm cells, as indicated by the triangle at E7.0, and peaking at E7.5. Both embryos were stained with the same probe against the Tal1 transcript. The images were taken using an Olympus SZX10/16 microscope. Scale bars: 500 μm.
Recipes
4% PFA
4 g of paraformaldehyde in 100 ml of DPBS, thoroughly dissolve.
Adjust pH to 7.4-7.6 using 1 M NaOH solution and store in 4°C for up to one week.
Note: Take care to avoid direct contact with PFA powder and solution.
PTW buffer
Calcium and magnesium free DPBS with 0.1% Tween-20.
Store at room temperature for up to one week.
Note: Take care to avoid direct contact with Tween-20 solution due to potential harm to skin.
20 mg/ml Yeast RNA
Dissolve 20 mg of Yeast RNA in 1 ml of nuclease free water and mix thoroughly.
Store at -20°C for up to one month.
50 mg/ml Heparin
Dissolve 50 mg of Heparin in 1 ml of nuclease free water and mix thoroughly.
Store at -20°C for up to one month.
Hybridization solution
Store at -20°C for up to one month.
Component (stock conc.) Final conc. Volume to add
Formamide 50% 25 ml
SSC (20×, pH 5.3 adjusted with citric acid) 1.3× SSC 3.25 ml
EDTA (0.5 M, pH 8.0) 5 mM 0.5 ml
Yeast RNA (20 mg/ml in H2O) 50 µg/ml 125 µl
Tween-20 0.002 100 µl
Heparin (50 mg/ml in H2O) 100 µg/ml 100 µl
UltraPureTM DNase/RNase-Free Distilled Water Replenish to 50 ml
Total 50 ml
10× TBST stock
Store at 4°C for up to one month.
Component Mass
NaCl 4 g
KCl 0.1 g
1 M Tris-HCl pH 7.5 12.5 ml
Tween-20 5.5 g
H2O Replenish to 50 ml
Total 50 ml
Blocking buffer
1 mg/ml BSA in 1× TBST
Store at 4°C for up to one week
NTMT buffer
Prepare right before use; store at room temperature for up to 2 days.
Component (stock concentration) Final concentration Volume to add
2.5 M NaCl 0.1 M 1 ml
2 M Tris-HCl (pH 9.5) 0.1 M 1.25 ml
1 M MgCl2 0.05 M 1.25 ml
Tween-20 1% 0.25 ml
H2O ~21.25 ml
Total 25 ml
4% PFA/0.1% glutaraldehyde
Dilute 25% glutaraldehyde in freshly prepared 4% PFA to a final concentration of 0.1%. Prepare right before use.
Note: Take care to avoid direct contact with PFA and glutaraldehyde solution.
6% H2O2/PTW solution
Dilute 30% H2O2 stock buffer in freshly prepared PTW buffer to a final concentration of 6%. Prepare right before use.
Note: Take care to avoid direct physical contact with H2O2 solution.
Acknowledgments
This work was supported in part by the National Key Basic Research and Development Program of China (2018YFA0800100, 2019YFA0801402, 2018YFA0108000, 2018YFA0107200, 2017YFA0102700), the Strategic Priority Research Program of the Chinese Academy of Sciences (XDA16020501 and XDA16020404), and the National Natural Science Foundation of China (31900454). This protocol was adapted from Zhu, Q., Song, L., Peng, G., Sun, N., Chen, J., Zhang, T., Sheng, N., Tang, W., Qian, C., Qiao, Y., et al. (2014). The transcription factor Pou3f1 promotes neural fate commitment via activation of neural lineage genes and inhibition of external signaling pathways. Elife 3: e02224.
Competing interests
The authors declare no conflicts of interest or competing interests.
References
- Downs, K. M. and Davies, T. (1993). Staging of gastrulating mouse embryos by morphological landmarks in the dissecting microscope. Development 118(4): 1255-1266.
- Hauptmann, G. and Gerster, T. (1994). Two-color whole-mount in situ hybridization to vertebrate and Drosophila embryos. Trends Genet 10(8): 266.
- Nieto, M. A., Patel, K. and Wilkinson, D. G. (1996). In: Methods in Cell Biology. Vol. 51. Bronner-Fraser, M. (Ed.). 219-235, Academic Press.
- Peng, G., Suo, S., Chen, J., Chen, W., Liu, C., Yu, F., Wang, R., Chen, S., Sun, N., Cui, G., et al. (2016). Spatial Transcriptome for the Molecular Annotation of Lineage Fates and Cell Identity in Mid-gastrula Mouse Embryo. Dev Cell 36(6): 681-697.
- Peng, G., Suo, S., Cui, G., Yu, F., Wang, R., Chen, J., Chen, S., Liu, Z., Chen, G., Qian, Y., Tam, P. P. L., Han, J. J. and Jing, N. (2019). Molecular architecture of lineage allocation and tissue organization in early mouse embryo. Nature 572(7770): 528-532.
- Pereira, P. N., Dobreva, M. P., Graham, L., Huylebroeck, D., Lawson, K. A. and Zwijsen, A. N. (2011). Amnion formation in the mouse embryo: the single amniochorionic fold model. BMC Dev Biol 11: 48.
- Piliszek, A., Kwon, G. S. and Hadjantonakis, A. K. (2011). Ex utero culture and live imaging of mouse embryos. Methods Mol Biol 770: 243-257.
- Wang, F., Flanagan, J., Su, N., Wang, L. C., Bui, S., Nielson, A., Wu, X., Vo, H. T., Ma, X. J. and Luo, Y. (2012). RNAscope: a novel in situ RNA analysis platform for formalin-fixed, paraffin-embedded tissues. J Mol Diagn 14(1): 22-29.
- Yang, X., Hu, B., Hou, Y., Qiao, Y., Wang, R., Chen, Y., Qian, Y., Feng, S., Chen, J., Liu, C., et al. (2018). Silencing of developmental genes by H3K27me3 and DNA methylation reflects the discrepant plasticity of embryonic and extraembryonic lineages. Cell Res 28(5): 593-596.
- Yang, X., Hu, B., Liao, J., Qiao, Y., Chen, Y., Qian, Y., Feng, S., Yu, F., Dong, J., Hou, Y., et al. (2019). Distinct enhancer signatures in the mouse gastrula delineate progressive cell fate continuum during embryo development. Cell Res 29(11): 911-926.
- Zhu, Q., Song, L., Peng, G., Sun, N., Chen, J., Zhang, T., Sheng, N., Tang, W., Qian, C., Qiao, Y., et al. (2014). The transcription factor Pou3f1 promotes neural fate commitment via activation of neural lineage genes and inhibition of external signaling pathways. Elife 3: e02224.
Article Information
Copyright
Yang et al. This article is distributed under the terms of the Creative Commons Attribution License (CC BY 4.0).
How to cite
Readers should cite both the Bio-protocol article and the original research article where this protocol was used:
- Yang, X., Chen, Y., Song, L., Zhang, T. and Jing, N. (2021). Wholemount in situ Hybridization for Spatial-temporal Visualization of Gene Expression in Early Post-implantation Mouse Embryos. Bio-protocol 11(22): e4229. DOI: 10.21769/BioProtoc.4229.
- Zhu, Q., Song, L., Peng, G., Sun, N., Chen, J., Zhang, T., Sheng, N., Tang, W., Qian, C., Qiao, Y., et al. (2014). The transcription factor Pou3f1 promotes neural fate commitment via activation of neural lineage genes and inhibition of external signaling pathways. Elife 3: e02224.
Category
Developmental Biology > Cell growth and fate > Differentiation
Molecular Biology > RNA > RNA detection
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link










