- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Development of a Chemical Reproductive Aging Model in Female Rats
Published: Vol 11, Iss 8, Apr 20, 2021 DOI: 10.21769/BioProtoc.3994 Views: 7341
Reviewed by: Victor Rodrigues SantosGilliard LachAnonymous reviewer(s)
Abstract
Women are born with an abundant but finite pool of ovarian follicles, which naturally and progressively decreased during their reproductive years until menstrual periods stop permanently (menopause). Perimenopause represents the transition from reproductive to non-reproductive life. It is usually characterized by neuroendocrine, metabolic and behavioral changes, which result from a follicular depletion and reduced number of ovarian follicles. During this period, around 45-50 years old, women are more likely to express mood disorders, anxiety, irritability and vasomotor symptoms. The current animal models of reproductive aging do not successfully replicate human perimenopause and the gradual changes that occur in this phase. While the traditional rat model of menopause involves ovariectomy or surgical menopause consisting of the rapid and definitive removal of the ovaries resulting in a complete loss of all ovarian hormones, natural or transitional menopause is achieved by the selective loss of ovarian follicles (perimenopause period). However, the natural aging rodent (around 18-24 months) model fails to reach very low estrogen concentrations and overlaps the processes of somatic and reproductive aging. The chronic exposure of young rodents to 4-vinylcyclohexene diepoxide (VCD) is a well-established experimental model for perimenopause and menopause studies. VCD induces loss of ovarian small follicles (primary and primordial) in mice and rats by accelerating the natural process of atresia (apoptosis). The VCD, ovary-intact or accelerated ovarian failure (AOF) model is the experimental model that most closely matches natural human progression to menopause mimicking both hormonal and behavioral changes typically manifested by women in perimenopause.
Graphical abstract: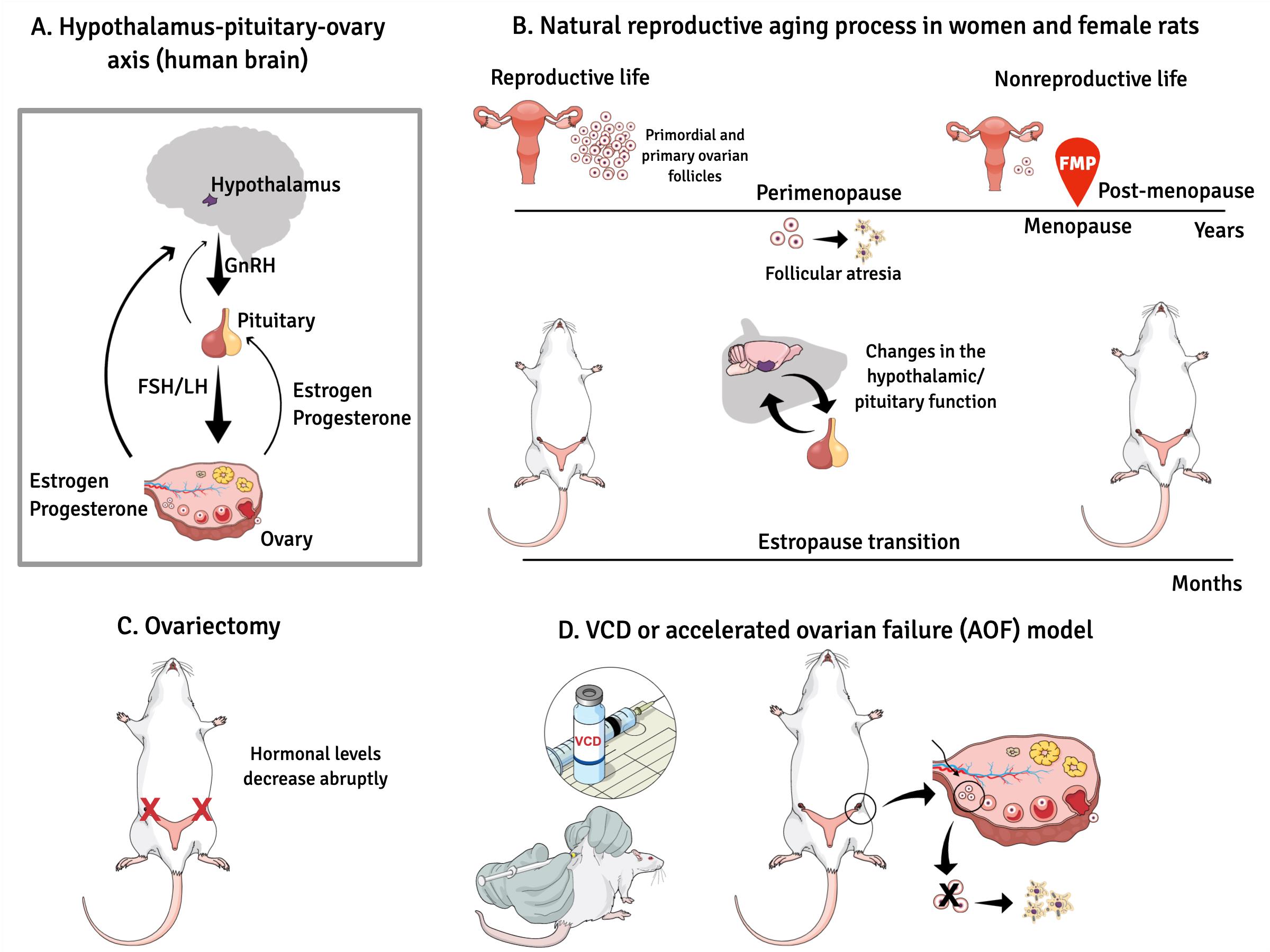
The female reproductive system is regulated by a series of neuroendocrine events controlled by central and peripheral components. (A). The mechanisms involved in this control are extremely complex and have not yet been fully clarified. In female mammals whose ovulation (the most important event in a reproductive cycle) occurs spontaneously, reproductive success is achieved through the precise functional and temporal integration of the hypothalamus-pituitary-ovary (HPO) axis. (B). In women, loss of fertility appears to be primarily associated with exhaustion of ovarian follicles, and this process occurs progressively until complete follicular exhaustion marked by the final menstrual period (FMP). (C). While in female rodents, reproductive aging seems to begin as a neuroendocrine process, in which changes in hypothalamic/pituitary function appear independently of follicular atresia. The traditional rat model of menopause, ovariectomy or surgical menopause consists of the rapid and definitive removal of the ovaries resulting in a complete loss of all ovarian hormones. (D). The chronic exposure (15-30 days) to the chemical compound 4-vinylcyclehexene diepoxide (VCD) in young rodents accelerates gradual failure of ovarian function by progressive depletion of primordial and primary follicles, but retains residual ovarian tissue before brain alterations that occurs in women in perimenopause. Low doses of VCD cause the selective destruction of the small preantral follicles of the ovary without affecting other peripheral tissues.
Background
Perimenopause, the transition period from reproductive to non-reproductive life, is defined as the period immediately before menopause. This period is marked by the onset of endocrine and biological changes, as well as clinical symptoms suggestive of the approach of menopause and can extend up to twelve months after the final menstruation, with an average duration of five years (WHO, 1996; Soules et al., 2001; Bacon, 2017; Wang et al., 2019). In addition to menopause or definitive cessation of menstrual cycling, perimenopause is a uniquely human process, but it can be mimicked by experimental models, especially in rodents. According to Prior and Hitchcock, perimenopause, previously seen as a period of hypoestrogenism, can be characterized by three main hormonal changes in women whose menstrual cycle remains regular: 1) normal or erratically high concentrations of estradiol; 2) decline in plasma progesterone concentrations and 3) changes at all levels of the reproductive axis (Prior and Hitchcock, 2011). During perimenopause a high percentage of women manifest typical symptoms of this period, which include: vasomotor changes, variations in menstrual cycle duration, sleep disorders, worsening cognitive functions, behavioral and mood changes (irritability, nervousness, anxiety and depression), in addition to metabolic and physiological changes (Mitchell and Woods, 1996; Brinton et al., 2015; Chalouhi, 2017). Considering the brain changes during this period, Brinton et al. (2015) defined perimenopause as a “state of neurological transition”.
The process of reproductive senescence in mammal species is complex and poorly understood, especially in humans (Brinton et al., 2009; Brinton, 2010). Consequently, animal models of menopause and perimenopause function as windows into the complex mechanisms involved in reproductive biology senescence at different levels (systemic, cellular, molecular, and genomic), which are not possible to perform in humans (Brinton, 2012). Nonetheless, current animal models of menopause do not successfully replicate human perimenopause and the gradual changes that occur in this phase. While the traditional rat model of menopause, ovariectomy or surgical menopause that consist of the rapid and definitive removal of the ovaries resulting in a complete loss of all ovarian hormones, natural or transitional menopause is achieved by the selective loss of ovarian follicle (perimenopause period). However, natural aging models fail to reach very low estrogen concentrations and overlapping changes related to somatic aging and those of reproductive aging (Kermath and Gore, 2012; Frye et al., 2012; Kirshner et al., 2020). Additionally, these models do not reproduce what occurs in women, since the primary causes of reproductive aging between species diverge significantly. In women, loss of fertility appears to be primarily associated with exhaustion of ovarian follicles (Faddy et al., 1992; Rubin, 2000), while in female rodents, reproductive aging seems to begin as a neuroendocrine process, with the changes in hypothalamic/pituitary function appearing independently of the follicular atresia (Gore et al., 2000).
A well-established experimental model in the literature for studying perimenopause and menopause is exposure of rodents to the chemical 4-vinylcyclohexene diepoxide (VCD), which leads a gradual failure of ovarian function by progressive depletion of primordial and primary follicles, but retains residual ovarian tissue similar to women in perimenopause (Springer et al., 1996; Kao et al., 1999; Hoyer et al., 2001). Importantly, the VCD model or model of Accelerated Ovarian Failure (AOF, Brooks et al., 2016) mimic both hormonal (Reis et al., 2014; Pestana-Oliveira et al., 2018; Carolino et al., 2019) and behavioral changes such as anxiety (Reis et al., 2014), impaired memory (Koebele et al., 2016), depression (Kalil et al., 2020) and aggressiveness (Dalpogeto et al., 2016; Scafuto et al., 2017) typically manifested by women in perimenopause. Low doses of VCD specifically cause selective destruction of ovarian small pre-antral follicles without affecting other peripheral tissues. Furthermore this occupational chemical doesn’t cross the blood-brain barrier (Lukefahr et al., 2012).
Therefore, the VCD-induced follicular depletion model, followed by ovarian failure has been widely used in experimental research on perimenopause and menopause (Reis et al., 2014; Liu et al., 2015; Brooks et al., 2016; Koebele et al., 2016; Pestana-Oliveira et al., 2018; Wang et al., 2019; Carolino et al., 2019; Kirshner et al., 2020) and is the experimental model that most closely matches the natural human progression to menopause, since the majority of women enter menopause through a gradual and irreversible process of reduction in ovarian function, while retaining the residual tissue of the ovary (Brooks et al., 2016). Thus, considering that it is a critical number of ovarian follicles and not the woman's age that determines the onset of menopause (Faddy et al., 1992), VCD-induced perimenopause is a translational model that presents analogy, predictability and homology, and allows plausible inferences to be made about the dynamics of follicular loss and its effects on the neurochemistry of women in perimenopause and menopause, periods in which affective disorders, vasomotor alterations and several other symptoms compromise the quality of life of middle-aged women.
Recently (2015-2017) the AOF model was applied to the streets and subways of large North American cities such as Chicago, New York, San Francisco, and Los Angeles with the aim of reducing the population of rats that has infested those cities (https://www.chicagomag.com/Chicago-Magazine/March-2015/birth-control-for-rats/).
Materials and Reagents
Polyethylene tubing (Thermo ScientificTM Immuno Tubes and Stoppers, catalog number: 12-565-150), stored at room temperature (RT)
Plastic funnel
Pipet tips (Eppendorf®, catalog numbers: 1300 RN [1-100 μl, yellow]; 1400 [101-1,000 μl, blue]), stored at room temperature
Glass slides for immunofluorescence (dimensions: size 25.4 x 76. 2 mm; thickness: 1.0 x 1.2 mm), twelve transparent circles (Perfecta, catalog number: 214-6), stored at RT
Flexible, translucent silicone elastomer tubing (Silastic, Dow CorningTM, 7.8 mm × 12.7 mm × 2.38, catalog number: 11-189-13B), stored at RT
Syringe (1 ml Tuberculin Syringe Regular Tip) (MonojectTM, catalog number: 8881501400), stored at RT
Sterile Gloves (Synthetical Surgical Gloves Powder-Free, Confiderm® SPT), stored at RT
Gauze sponges (Non-woven Gauze Sponges, 2 in. × 2 in. [5.08 cm × 5.08 cm]) (AVANT GAUZE®, catalog number: 25223), stored at RT
Needles (Standard Hypodermic Needles, MonojectTM, catalog numbers: 8881250255 [23G], 8881250149 [21G]), stored at RT
Females Wistar rats (age 28 days) from the animal facilities of the University of São Paulo, campus Ribeirão Preto, Brazil
4-vinylcyclohexene diepoxide (VCD [C8H12O2] Sigma-Aldrich, catalog number: 94956-250ML), stored at RT
17-β-estradiol (Sigma-Aldrich, catalog number: E8875-1G), stored at RT
Corn oil (Liza-900ML), stored at RT
Ketamine HCl Injection, USP (Ketaset®, 100 mg/kg; NDC: 0856-2013-1), RT
Xylazine (Schering-Plough, Coopers of Brazil, Cotia, São Paulo, 14 mg/kg), RT
Small Veterinary Pentabiotic® (Zoetis, 1.7 g/3 ml, catalog number: 232092),RT
Banamine Solution Injectable (Schering-Plough Animal Health, 2.5 mg/kg; catalog number: 12080097), stored at room temperature Sodium Cloride (NaCl) 0.9% (Samtec Biotecnology, 10 ml), RT
5% Povidone-iodine antiseptic microbicide for animal use (Betadine® Solution, catalog number: 12265), stored at RT
70% Isopropyl Alcohol (473 ml) (Medline Industries, catalog number: 53329-800-06), stored at RT
ELISA estradiol kit (DRG® Instruments GmbH DRG, EIA 2693), storage: 2 °C-8 °C
Progesterone double antibody RIA kit (P4) (MP Biomedicals, catalog number: SKU 07-170105 CF), storage: 2 °C-8 °C
Standard Rat Chow (23.2% Protein Rodent Diet, LabDiet®,catalog number: 5012), stored at RT
Heparin 50 units/ml (Heparin Sodium Injection USP, 50 units USP per ml) (B. Braun Medical Inc., catalog number: 0264-9577-10), stored at RT
4-vinylcyclohexene diepoxide dilution (VCD) (see Recipes)
Equipment
Standard cages (Bonther, 40 × 33 × 17 cm)
Guillotine (Bonther, model: Inox 420)
Surgical scissors
Forceps
Needle holder
Microscope (Zeiss Axioskop 2 Plus Ergonomic Trinocular, catalog number: 452342)
Centrifuge (Eppendorf, model: 5425, catalog number: 5405000042; 24 × 1.5/2.0 ml Capacity, up to 21, 330 × g (15,060 rpm), includes FA-24x2 rotor with aerosol-tight QuickLock lid, keypad control, 120 V)
Magnetic Stirrer Magnetic Stirrer with Heating (Thermo Scientific, catalog number: N2400-3010)
Shaker/Vortexer with racks (BenchMixer XLQ, catalog number: BV 1010-TST)
Detector Gamma Counter (Wizard2TM, PerkinElmer®, catalog number: 2470-0020)
Absorbance Reader 800 TS (BioTek)
Pipette single channel (EppendorfTM Research [0.5-10 µl, EP: 3123000020 yellow; 20-200 µl, EP: 312300055 yellow; 100-1,000 μl, EP: 312300063 blue]
Electronic Pipette Multichannel (EppendorfTM Research, 100 μl, SKU #184287407894)
Software
GraphPad Prism 7 software (GraphPad Software, La Jolla, CA)
Adobe Photoshop (Adobe Photoshop Lightroom, version 5.3; Adobe Systems, Inc.)
Mind the graph (www.mindthegraph.com)
Microsofit Excel
Procedure
Although many women in perimenopause have normal or erratically high estradiol plasma concentrations (Santoro et al., 1996), estrogenic therapy is very common in clinical practice. Interestingly, improvement was observed regarding the typical symptoms of this transition period (Schmidt et al., 2000). The study that inspired this protocol (Pestana-Oliveira et al., 2018) had as one of its objectives to understand the possible effects of estradiol in the model of accelerated ovarian failure (Figure 1).
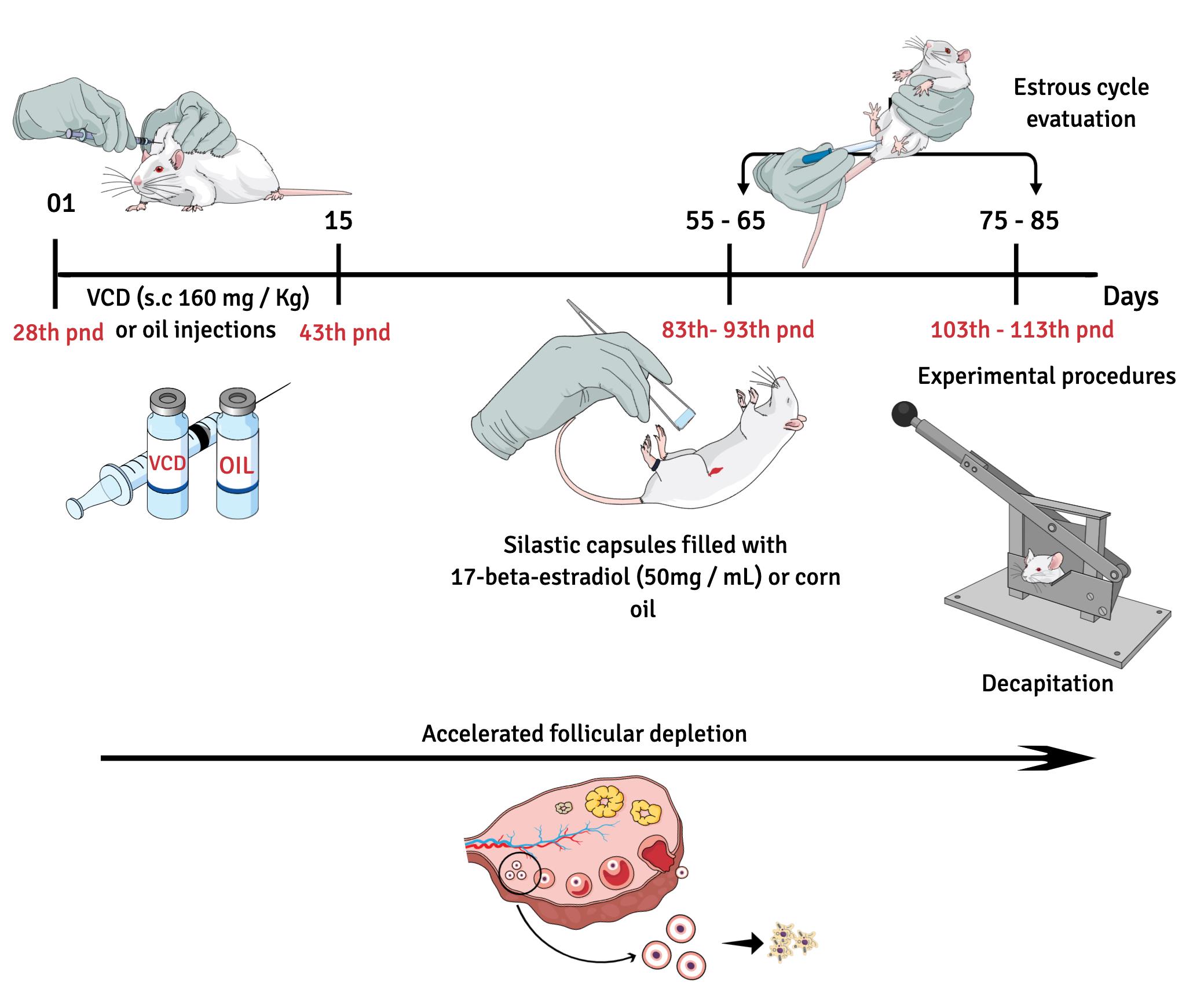
Figure 1. Schematic diagram showing the timeline of the experimental protocol. Female Wistar rats (28 post-natal days) must be daily injected with VCD (160 mg/Kg) or corn oil (O; 1.25 ml/kg of body weigh) for 15 days. Approximately 55 days after the first VCD or O injection, insert implant pellets of 17β-estradiol or O s.c in the dorso-lateral region (Groups O + O; VCD + O; VCD + E). 21 days after oil or estradiol pellets implantation rats must be decapitated in the morning of diestrus.
VCD or corn oil injections (Figure 2)
Weigh and identify the female rats (28th post-natal day) individually and calculate the volume of VCD or corn oil that each will receive. It is important that this procedure is performed every two days. Then prepare and arrange the environment to give the injections. Cover the table or bench with paper and fill the syringes with the volume of VCD or oil previously calculated.
Take one animal and apply the subcutaneous injection on the back near to the neck region. Repeat the same procedure for all animals. Perform the applications slowly as the oil is viscous and the syringe may burst from the needle with pressure. In the case of animals that will receive injections containing VCD, avoid wasting liquid after the injection.
After finishing all the injections, return the animals to the specific animal facility. Repeat the entire procedure for 15 consecutive days. To avoid cutaneous and subcutaneous injuries we strongly suggest that injections be applied to different parts of the animal's body.
Note: The contact of the skin and mucous membranes with the VCD should be avoided as much as possible, especially if it is handled by female individuals. Therefore, the use of individual safety equipment such as gloves, masks and glasses is strongly recommended.
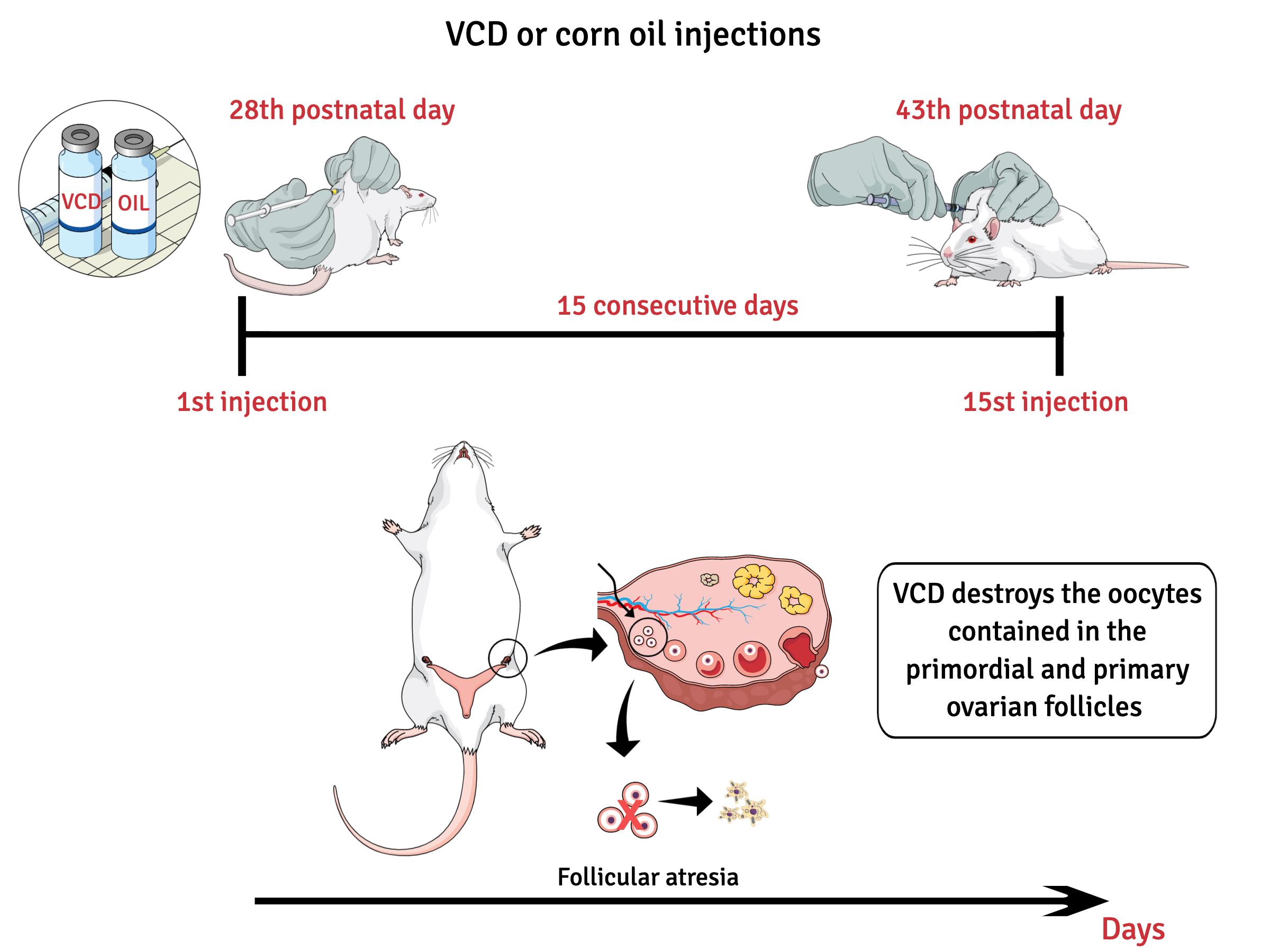
Figure 2. Schematic diagram showing the preparation of the accelerated ovarian failure model and control groups
Estrogen therapy
Silastic capsules preparation (Figure 3)
Extend the silastic tubing on a table or bench, with the aid of a ruler, divide it into 15 mm long pieces. Then cut the pieces. Insert the tip of one end of the silastic into a drop of silicone glue, press the opposite end so that the glue enters into tube. With the aid of hemostatic scissors, clamp the end filled with glue. Repeat this procedure with the remaining pieces of silastic. Place the tubes on a flat surface for 24 h so that the glue can dry and thus seal the tube end (Figure 3-1).
Use a pipette and a plastic tip (0.5-10 µl) to fill the silastic capsules with 8 µl of 17-β-estradiol (50 mg/ml, previously diluted in corn oil). The estradiol suspension must be placed on a magnetic stirrer to ensure homogeneity throughout the process of filling the pellets. In addition, on the previous day it is important to subject the solution to ultrasound for 3 h followed by overnight stirring. When ready, it should appear as a homogeneous milky solution. At rest, the suspension settles, and it is possible to observe two phases: the upper one (vehicle) represented by the oil is transparent while the lower one is whitish. The oil pellets must be filled with 8 µl of corn oil. The sealing process of the open end of the capsules is the same as described above except that it requires greater care considering that the capsules are filled with liquid. After complete sealing, it is important to trim the ends using small scissors (Figure 3-2).
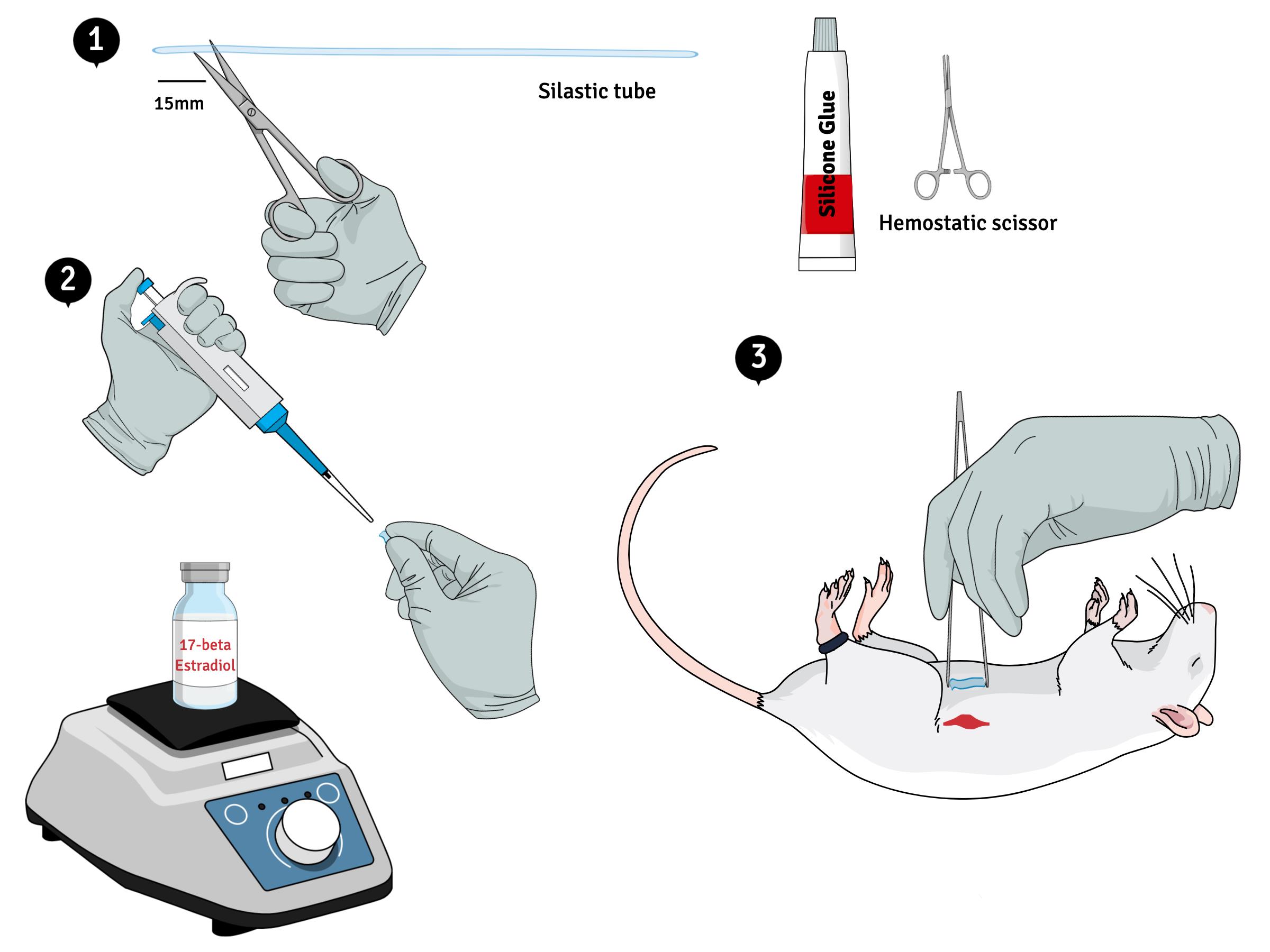
Figure 3. Schematic diagram showing the silastic capsules preparation and surgery implantation
Pellets implantation surgery
All Instruments and materials used for surgery must be previously sterilized. We recommend steam sterilization (autoclaving). Furthermore, female rats should be weighed for the doses of anesthetics and medications be given according to individual body weight.
The animals must be anesthetized with ketamine (55 mg/kg, IP) and xylazine (10 mg/kg, IP) and perform aseptic procedures as follows: shave the rats, scrub the surgical site with antiseptic solution (Betadine).
Note: In this protocol we use injectable anesthesia. However, is possible to use other approach such as inhaled anesthetics.
With the aid of a small scissors, make a discreet incision (0.5 cm) and insert the tip of the scissors to separate the tissue, a subcutaneous pocket is formed to store the pellet. This surgery is minimally invasive and quick. After surgery, the animals must receive prophylactic antibiotics (Pentabiotico 0.2 ml/rat, i.m.) and anti-inflammatory treatment (Banamine, 2.5 mg/kg, s.c).
Vaginal smears collection and estrous cycle evaluation (Figure 4)
The estrous cycle of female rats lasts an average of 4 to 5 days and has 4 distinct phases: proestrus, estrus, metestrus (or diestrus I) and diestrus (or diestrus II), which can be easily identified by the cell types observed in the vaginal smear (Marcondes et al., 2002).
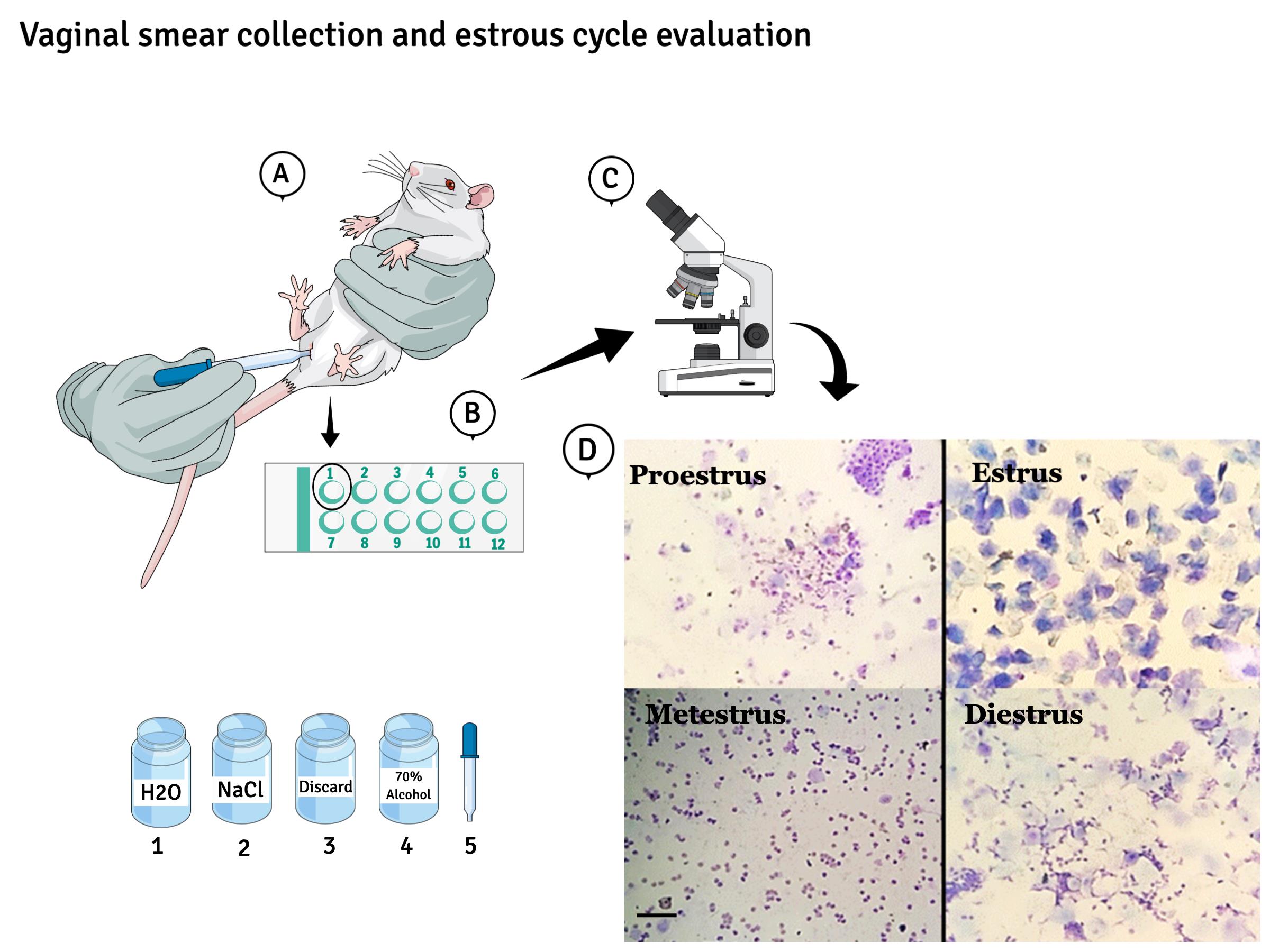
Figure 4. Schematic diagram showing the vaginal smear collection and estrous cycle evaluation. Photomicrography (10×) kindly provided by Thalita de Oliveira Gonçalves and Guilherme de Souza Gagliano.With one hand, hold the animal firmly so that the vagina is visible. With the other hand, gently insert the tip of a plastic pipette previously filled with 0.9% NaCl (approx. 10 μl) into the vagina of the rat, it is not necessary to deepen the pipette. Press the rubber on the surface of the pipette to push the NaCl, repeat this process twice as if washing (Figure 4A-1).
Remove the vaginal fluid and deposit a drop on a glass slide (Figure 4B). Discard the excess of the collected smear in the specific container (Figure 4-3). The pipette must be sanitized between one animal and another, filling it with water (Figure 4-1) and discarding it in container 3. Repeat the wash 3 times, then fill the pipette with 70% alcohol (Figure 4-4) and discard, the pipette will dry immediately and be ready to be reused in another animal. One drop from each rat must to be placed sequentially on the slide (Figure 4B).
After collecting the vaginal smear from all rats, the glass slide is ready to be observed under an optical or light microscope without the need for a lens condenser. 10× and 40× objective lenses are suitable for observing fresh material (Figure 4C).
When observing the slide under the microscope, it will be possible to recognize 3 distinct cell types: 1) the round and nucleated ones that resemble bunches of grape or fried eggs are non-cornified squamous epithelial cells typically observed during the proestrus; 2) irregular cells, in the shape of a dry leaf or corn flakes, are the cornified epithelial cells, characteristic of estrus; 3) small round cells are the leukocytes present in the diestrus (Figure 4D). Animals in metestrus will present a variety of these cells. It is important to emphasize that the proportion between them should be used to determine the estrous cycle phases (Marcondes et al., 2002).
Note: The collection of the vaginal smear and evaluation of the estrous cycle must be carried out in the early morning, between 7:30 and 9:30 am to prevent the cells present in the vaginal fluid from moving from one phase to the next. Since until close to 90 days after the onset of VCD treatment the proportion of rats that cycle regularly (about 20%) is the same in control rats and those treated with VCD (Carolino et al., 2019). Only control and VCD treated rats cycling regularly be used in the experiments performed before 90 days after starting VCD injections.
Euthanasia and blood samples collection
Between 75 and 85 days after the start of the VCD/oil injections, the animals should be euthanized in the morning, by decapitation or anesthetic overdose (ketamine [110 mg/kg, IP] and xylazine [20 mg/kg, IP].
Before starting the decapitation, make sure the guillotine is sharp enough. Quickly and firmly hold the animal and position the head at the height of the trunk between the guillotine blades, once positioned, lower the lever.
With the aid of a plastic funnel previously heparinized and identified attached to the polyethylene tube, position the animal's body downwards, facilitating the exit of the blood. Shake the tube gently, facilitating the mixing of blood and heparin to prevent clotting. Store the tube containing blood in a refrigerated environment (2 °C-8 °C).
Remove the brain quickly and gently from the skull and freeze it immediately either on dry ice or liquid nitrogen. Store the brain at -70 °C.
Centrifuge the blood samples in a refrigerated centrifuge at 1,200 × g for 20 min at 4 °C. Separate the plasma and store at -70 °C until the assay.
Note: The decapitation must be done the most humane way possible, guaranteeing the animal's well-being as well as the safety of the experimenter. Avoid noise, odors and crowding in the experimental room. The environment should be well lit with circulating air. We recommend using exhaust fans to help dispel specific odors such as blood. We recommend that the guillotine, the sink and the counter be cleaned with water and 70% alcohol to avoid the odor of blood between becapitations.
Data analysis
All comparisons were performed using one-way ANOVA followed by Newman-Keuls post hoc test. Data are presented as the mean ± SEM. Significance was accepted at P > 0.05. All statistical analyses and graphs were performed using GraphPad Prism 7 software.
Notes
An important question regarding the accelerated ovarian failure model is: how to assess whether the VCD injections were effective in promoting the depletion of primordial and primary follicles?
We suggest performing at least 1 of the three positive control tests:
Ovarian histology. This procedure is highly effective, since significant qualitative and quantitative changes are expected in ovarian follicles (Reis et al., 2014). The disadvantage of this method is the time to obtain the results. Depending on the demand of each laboratory, between the collection of samples and obtaining the slides ready for microscopy, it will require at least 1 week.
After euthanasia (decapitation or perfusion), remove the ovaries, separate them from the connective tissue and fat and fix them in 10% formaldehyde for 24 h. Perform the histological processing that includes the dehydration processes (subject the material successively in increasing concentrations of alcohol to 70%, 90%, 100%), clearing with xylol, assembling the paraffin blocks and obtaining sections with a microtome of 8 μm thickness. To assemble the slides, we recommend semi-serial sections and stained with hemotoxilin and eosin (HE). With the aid of an optical microscope, count and classify the ovarian follicles. The Leica Biosystems website provides the detailed guidance on procedures (Overview of the steps in tissue processing for paraffin sections; https://www.leicabiosystems.com/knowledge-pathway/an-introduction-to-specimen-processing/).
Measurement of estradiol and progesterone plasma concentrations. Most perimenopause women have a typical hormonal profile, with reduction in progesterone plasma concentrations while estradiol concentrations are normal or erratically high (Santoro et al., 1996; Prior and Hitchcock, 2011). The same pattern was observed in rats submitted to chronic VCD injections (Reis et al., 2014; Pestana-Oliveira et al., 2018; Carolino et al., 2019). Therefore, we consider this an effective way to assess whether perimenopause was induced in female rats.
Immediately after the decapitation collect the blood samples and centrifuge at 1,200 × g for 20 min at 4 °C. The plasma must then be separated and be stored at -70 °C until the assay. All samples must be measured in the same assay to avoid intraassay variation.
Progesterone assay: To measure progesterone plasma concentrations we recommend specific radioimmunoassay (RIA) kits for humans provided by MP Biomedicals. Polyethylene tubes must be numbered with a permanent pen in ascending order. For a more accurate assay we recommend that samples be measured in duplicate or triplicate. However, before the test it is necessary to perform a dilution test in order to identify the volume of plasma needed so that the value obtained from the samples plasma concentrations is in accordance with the kit standard curve. The MP Biomedicals website provides the step-by-step instructions on how to perform the assay.
Estradiol assay: To measure estradiol plasma concentrations we recommend specific kit for an enzyme-linked immunosorbent assay provided by DRG.
Measurement of anti-Mullerian plasma concentrations. It is well established in the literature that the anti-Mullerian hormone concentrations (AMH) have a direct correlation with the number of ovarian follicles, being thus considered a marker of ovarian follicular reserve whose levels decrease with increasing age, and therefore practically undetectable after menopause (Cui et al., 2015). Also, the AOF model in both monkeys and mice has demonstrated a linear correlation between reduced levels of AMH and reduced number of developing follicles (Sahambi et al., 2008). In a recently published study using the AOF model in female rats, Dr. Anselmo-Franci's laboratory (Carolino et al., 2019) demonstrated that in fact plasma AMH concentrations can be considered a good index of ovarian follicular reserve as well as effectiveness of VCD injections in follicular depletion. To measure AMH plasma concentrations, use a specific enzyme-linked immunosorbent assay kit for mice and rat provided by Ansh Labs (Webster, TX, USA).
Recipes
4-vinylcyclohexene diepoxide dilution (VCD, Figure 5)
The chemical compound VCD is obtained through epoxidation catalyzed by cytochrome P450 from 4-vinylcyclohexene (HCV), an occupational ovarian toxin produced from the dimerization of 1,3-butadiene during the manufacture of synthetic rubber, flame retardants, insecticides and plasticizers (Mayer et al., 2002). The occupational chemical 4-vinylcyclohexene diepoxide (VCD) has been shown to cause selective destruction of ovarian small pre-antral (primordial and primary) follicles in rats and mice by accelerating the natural, apoptotic process of atresia (Kappeler and Hoyer, 2012).
To obtain the dose of 160mg/Kg (1.25 μl/g body weight) dilute the VCD reagent in corn oil at a concentration of 12%, in other words, if you want to prepare 100 ml of stock solution, measure 12 ml of VCD in a graduated cylinder and make up to 100 ml with corn oil. With the aid of a magnetic stirrer, homogenize the solution. The ultrasound is not necessary to improve the dilution.
Notes:
The amount of solution can be prepared according to the demand of each laboratory. On average every 6 months it is possible to prepare 1 L of stock solution that should be stored at RT and kept in an amber bottle or wrapped in aluminum foil to avoid exposure to light. We recommend distributing the stock solution in smaller bottles that will be used routinely.
In all processes involving the handling of the chemical compound VCD, the use of personal protective equipment is essential. In addition, dilution of the VCD must be performed under ventilation and properly cleaned with 70% alcohol. Avoid skin contact and inhalation.
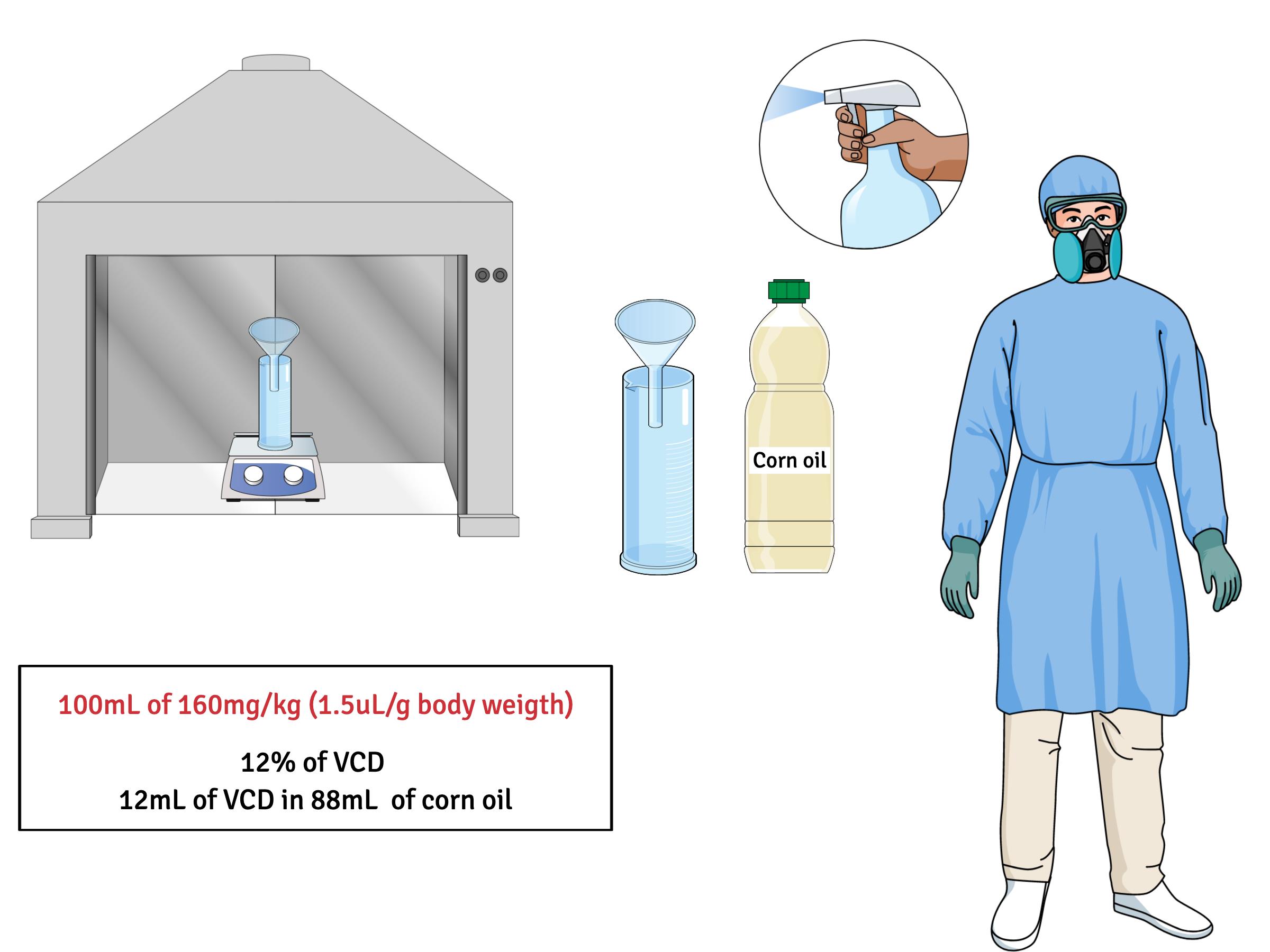
Figure 5. Schematic diagram showing the 4-vinylcyclohexene diepoxide (VCD) dilution
Acknowledgments
The original study that supported this paper, “Effects of Estrogen Therapy on the Serotonergic System in an Animal Model of Perimenopause Induced by 4-Vinylcyclohexen Diepoxide (VCD)” was funded by Brazilian National Council for Scientific and Technological Development (CNPq, Portuguese: Conselho Nacional de Desenvolvimento Científico e Tecnológico) and São Paulo Research Foundation (FAPESP, Portuguese: Fundação de Amparo à Pesquisa do Estado de São Paulo). This study was developed in the Laboratory of Neuroendocrinology of Female Reproduction under the guidance of Dr. Janete A Anselmo-Franci, Department of Basic and Oral Science, School of Dentistry of Ribeirao Preto, University of Sao Paulo, Ribeirao Preto, Sao Paulo, Brazil.
Competing interests
The authors declare that they have no conflict of interest.
Ethics
All procedures were approved by the Committee for Animal Care and Use (2013.1.1412.58.7), University of Sao Paulo.
References
- Bacon, J. L. (2017). The Menopausal Transition. Obstet Gynecol Clin N Am 44(2): 285-296.
- Brinton, R. D., Gore, A. C., Schmidt, P. J. and Morrison, J. H. (2009). Hormones, Brain and Behavior (2nd edition). In: Pfaff, D. W. et al. (Eds.). Elsevier 2199-2222.
- Brinton, R. D. (2010). In: Brockelhurst's Textbook of Geriatric Medicine and Gerontology. Fillit, H., Rockwood, K. and Young, J. (Eds.). Saunders 163-171.
- Brinton, R. D. (2012). Minireview: translational animal models of human menopause: challenges and emerging opportunities. Endocrinology 153(8): 3571-3578.
- Brinton, R. D., Yao, J., Yin, F., Mack, W. J. and Cadenas, E. (2015). Perimenopause as a neurological transition state. Nat Rev Endocrinol 11(7): 393-405.
- Brooks, H. L., Pollow, D. P. and Hoyer, P. B. (2016). The VCD Mouse Model of Menopause and Perimenopause for the Study of Sex Differences in Cardiovascular Disease and the Metabolic Syndrome. Physiology (Bethesda) 31(4): 250-7.
- Carolino, R. O. G., Barros, P. T., Kalil, B. and Anselmo-Franci, J. (2019). Endocrine profile of the VCD-induced perimenopausal model rat. PLoS One 14(12): e0226874.
- Chalouhi, S. (2017). Menopause: A complex and controversial journey. Post Reprod Health 23(3): 128-131.
- Cui, L., Qin, Y., Gao, X., Lu, J., Geng, L., Ding, L., Qu, Z., Zhang, X. and Chen, Z. J. (2016). Antimullerian hormone: correlation with age and androgenic and metabolic factors in women from birth to postmenopause. Fertil Steril 105(2): 481-485 e481.
- Dalpogeto, L. C., Pestana, de Oliveira Nayara, Anselmo-Franci, J. A. (2016). Evaluation of aggressive behavior in an animal model of perimenopause. Annual Meeting Society of Neuroscience, Washington, DC, USA.
- Faddy, M. J., Gosden, R. G., Gougeon, A., Richardson, S. J. and Nelson, J. F. (1992). Accelerated disappearance of ovarian follicles in mid-life: implications for forecasting menopause. Hum Reprod 7(10): 1342-1346.
- Frye, J. B., Lukefahr, A. L., Wright, L. E., Marion, S. L., Hoyer, P. B. and Funk, J. L. (2012). Modeling perimenopause in Sprague-Dawley rats by chemical manipulation of the transition to ovarian failure. Comp Med 62(3): 193-202.
- Gore, A. C., Oung, T., Yung, S., Flagg, R. A. and Woller, M. J. (2000). Neuroendocrine mechanisms for reproductive senescence in the female rat: gonadotropin-releasing hormone neurons. Endocrine 13(3): 315-323.
- Hoyer P. B., Devine P. J., Hu, X., Thompson, K. E., Sipes, I. G. (2001). Ovarian toxicity of 4-vinylcyclohexene diepoxide: a mechanistic model. Toxicol Pathol 29(1): 91-9.
- https://www.chicagomag.com/Chicago-Magazine/March-2015/birth-control-for-rats/.
- https://www.leicabiosystems.com/knowledge-pathway/an-introduction-to-specimen-processing/
- Kalil, B. Pestana-Oliveira, N., Santos, I. R., Carolino, R. O. G. and Anselmo-Franci, J. A. (2020). Effect of Estradiol Therapy on Depressive like Behavior in an Ovarian Intact Rat Model of Perimenopause. J Endocrine Society MON-017. https://doi.org/10.1210/jendso/bvaa046.1298.
- Kao, S. W., Sipes, I. G. and Hoyer, P. B. (1999). Early effects of ovotoxicity induced by 4-vinylcyclohexene diepoxide in rats and mice. Reprod Toxicol 13(1): 67-75.
- Kappeler, C. J. and Hoyer, P. B. (2012). 4-vinylcyclohexene diepoxide: a model chemical for ovotoxicity. Syst Biol Reprod Med 58(1): 57-62.
- Kermath, B. A. and Gore, A. C. (2012). Neuroendocrine control of the transition to reproductive senescence: lessons learned from the female rodent model. Neuroendocrinology 96(1): 1-12.
- Kirshner, Z. Z., Yao, J. K., Li, J., Long, T., Nelson, D. and Gibbs, R. B. (2020). Impact of estrogen receptor agonists and model of menopause on enzymes involved in brain metabolism, acetyl-CoA production and cholinergic function. Life Sci 256: 117975.
- Koebele, S. V. and Bimonte-Nelson, H. A. (2016). Modeling menopause: The utility of rodents in translational behavioral endocrinology research. Maturitas 87: 5-17.
- Liu, W., Wang, L. Y., Xing, X. X. and Fan, G. W. (2015). Conditions and possible mechanisms of VCD-induced ovarian failure. Altern Lab Anim 43(6): 385-392.
- Lukefahr, A. L., Frye, J. B., Wright, L. E., Marion, S. L., Hoyer, P. B. and Funk, J. L. (2012). Decreased bone mineral density in rats rendered follicle-deplete by an ovotoxic chemical correlates with changes in follicle-stimulating hormone and inhibin A. Calcif Tissue Int 90(3): 239-249.
- Marcondes, F. K., Bianchi, F. J. and Tanno, A. P. (2002). Determination of the estrous cycle phases of rats: some helpful considerations. Braz J Biol 62(4A): 609-614.
- Mayer, L. P., Pearsall, N. A., Christian, P. J., Devine, P. J., Payne, C. M., McCuskey, M. K., Marion, S. L., Sipes, I. G. and Hoyer, P. B. (2002). Long-term effects of ovarian follicular depletion in rats by 4-vinylcyclohexene diepoxide. Reprod Toxicol 16(6): 775-781.
- Mitchell, E. S. and Woods, N. F. (1996). Symptom experiences of midlife women: observations from the Seattle Midlife Women's Health Study. Maturitas 25(1): 1-10.
- Pestana-Oliveira, N., Kalil, B., Leite, C. M., Carolino, R. O. G., Debarba, L. K., Elias, L. L. K., Antunes-Rodrigues, J. and Anselmo-Franci, J. A. (2018). Effects of Estrogen Therapy on the Serotonergic System in an Animal Model of Perimenopause Induced by 4-Vinylcyclohexen Diepoxide (VCD). eNeuro 5(1): ENEURO.0247-17.2017. doi: 10.1523/ENEURO.0247-17.2017.
- Prior, J. C. and Hitchcock, C. L. (2011). The endocrinology of perimenopause: need for a paradigm shift. Front Biosci (Schol Ed) 3: 474-486.
- Reis, F. M., Pestana-Oliveira, N., Leite, C. M., Lima, F. B., Brandao, M. L., Graeff, F. G., Del-Ben, C. M. and Anselmo-Franci, J. A. (2014). Hormonal changes and increased anxiety-like behavior in a perimenopause-animal model induced by 4-vinylcyclohexene diepoxide (VCD) in female rats. Psychoneuroendocrinology 49: 130-140.
- Rubin, B. S. (2000). Hypothalamic alterations and reproductive aging in female rats: evidence of altered luteinizing hormone-releasing hormone neuronal function. Biol Reprod 63(4): 968-976.
- Sahambi, S. K., Visser, J. A., Themmen, A. P., Mayer, L. P. and Devine, P. J. (2008). Correlation of serum anti-Mullerian hormone with accelerated follicle loss following 4-vinylcyclohexene diepoxide-induced follicle loss in mice. Reprod Toxicol 26(2): 116-122.
- Santoro, N., Brown, J. R., Adel, T. and Skurnick, J. H. (1996). Characterization of reproductive hormonal dynamics in the perimenopause. J Clin Endocrinol Metab 81(4): 1495-1501.
- Scafuto, M., Pestana-Oliveria, N., Almeida, R. M., Carolino, R. O., Anselmo-Franci, J. A. (2017).Effects of Hormonal Therapy on Aggressive Behavior and Central Nervous System in an Animal Model of Perimenopause. Annual Meeting Society of Neuroscience, DC, USA.
- Schmidt, P. J., Nieman, L., Danaceau, M. A., Tobin, M.B., Roca, C. A., Murphy J. H., Rubinow, D. R. (2000) Estrogen replacement in perimenopause-related depression: a preliminary report. Am J Obstet Gynecol 183:414-420.
- Soules, M. R., Sherman, S., Parrott, E., Rebar, R., Santoro, N., Utian, W. and Woods, N. (2001). Executive summary: Stages of Reproductive Aging Workshop (STRAW). Fertil Steril 76(5): 874-878.
- Springer, L. N., Flaws, J. A., Sipes, I. G. and Hoyer, P. B. (1996). Follicular mechanisms associated with 4-vinylcyclohexene diepoxide-induced ovotoxicity in rats. Reprod Toxicol 10(2): 137-143.
- Wang, Y., Liu, Y., Xiong, J., Di, T., Yuan, Z., Wu, J. and Chen, L. (2019). Reduced serotonin impairs long-term depression in basolateral amygdala complex and causes anxiety-like behaviors in a mouse model of perimenopause. Exp Neurol 321: 113030.
- WHO Scientific Group. (1996). Research on the menopause in the 1990s. Geneva: World Health Organization: 1-79.
Article Information
Copyright
© 2021 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Readers should cite both the Bio-protocol article and the original research article where this protocol was used:
- Pestana-Oliveira, N., Carolino, R. O. G., Kalil, B., Mota Leite, C., Dalpogeto, L. C., De Paula, B. B., Collister, J. P. and Anselmo-Franci, J. A. (2021). Development of a Chemical Reproductive Aging Model in Female Rats. Bio-protocol 11(8): e3994. DOI: 10.21769/BioProtoc.3994.
- Pestana-Oliveira, N., Kalil, B., Leite, C. M., Carolino, R. O. G., Debarba, L. K., Elias, L. L. K., Antunes-Rodrigues, J. and Anselmo-Franci, J. A. (2018). Effects of Estrogen Therapy on the Serotonergic System in an Animal Model of Perimenopause Induced by 4-Vinylcyclohexen Diepoxide (VCD). eNeuro 5(1): ENEURO.0247-17.2017. doi: 10.1523/ENEURO.0247-17.2017.
Category
Developmental Biology > Reproduction
Neuroscience > Development
Biological Sciences > Biological techniques
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link









