- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Integration of Human Induced Pluripotent Stem Cell (hiPSC)-Derived Neurons into Rat Brain
Published: Vol 10, Iss 17, Sep 5, 2020 DOI: 10.21769/BioProtoc.3746 Views: 6267
Reviewed by: Patrick Ovando-RocheAnthony FlamierThirupugal GovindarajanAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Reprogramming White Fat Cells for Adipose Manipulation Transplantation (AMT) Therapy
Kelly An [...] Nadav Ahituv
Aug 5, 2025 2279 Views
A One-Step Mouse Model of Parkinson’s Disease Combining rAAV-α-Synuclein and Preformed Fibrils of α-Synuclein
Santhosh Kumar Subramanya [...] Poonam Thakur
Dec 5, 2025 1742 Views
Vascularization of Human Pancreatic Islets With Adaptive Endothelial Cells for In Vitro Analysis and In Vivo Transplantation
Ge Li [...] Shahin Rafii
Dec 20, 2025 819 Views
Abstract
Human neuron transplantation offers novel opportunities for modeling human neurologic diseases and potentially replacement therapies. However, the complex structure of the human cerebral cortex, which is organized in six layers with tightly interconnected excitatory and inhibitory neuronal networks, presents significant challenges for in vivo transplantation techniques to obtain a balanced, functional and homeostatically stable neuronal network. Here, we present a protocol to introduce human induced pluripotent stem cell (hiPSC)-derived neural progenitors to rat brains. Using this approach, hiPSC-derived neurons structurally integrate into the rat forebrain, exhibit electrophysiological characteristics, including firing, excitatory and inhibitory synaptic activity, and establish neuronal connectivity with the host circuitry.
Keywords: Human neuron transplantationBackground
The human cerebral cortex is a complex cellular mosaic containing diversified neuronal subtypes in distinct cortical layers (I-VI) that establish specific patterns of axonal output and dendritic input, providing the essential substrate of cortical circuitry (Rakic, 2009; Lodato et al., 2011; Lui et al., 2011). In particular, a balance of excitatory and inhibitory neurotransmission is necessary for proper brain function (Turrigiano and Nelson, 2004). Human induced pluripotent stem cells (hiPSCs) allow modeling human neurological diseases in a human genetic context (Dolmetsch and Geschwind, 2011; Brennand et al., 2015; Vera and Studer, 2015). Considerable advances have been made in establishing in vitro systems to differentiate hiPSCs into neurons, including generating excitatory (glutamatergic) projection neurons or inhibitory (GABAergic) interneurons (Pasca et al., 2011; Shi et al., 2012; Liu et al., 2013; Maroof et al., 2013; Nicholas et al., 2013; Espuny-Camacho et al., 2017). In vivo studies also addressed that hiPSC-derived neurons exhibit neuronal morphology and possess synaptic activity after grafting to the rodent brain (Weick et al., 2011; Nicholas et al., 2013; Espuny-Camacho et al., 2017). However, development of a balanced network of both excitatory and inhibitory neurons representative of the complex constitution of human cortex had not been achieved until our recent reports (Xu et al., 2016; Yin et al., 2019).
The protocol described here generates an in vivo model which provides a more accurate representation of the human cortex, allowing the study of the interplay between excitatory and inhibitory networks in both normal and pathological conditions. In addition, in this system, transplanted human neurons display functional neuron behaviors and show mature electrophysiological profiles. Together, using this method, hiPSC-derived neural progenitors develop into functional neurons after grafting in the rat neonatal brain, with physiological properties including excitatory and inhibitory postsynaptic currents, which are indicative of the copresence of excitatory and inhibitory networks. This model enables the study of human cortical development and diseases such as autism, schizophrenia, and Alzheimer’s disease, which are thought to involve imbalances in excitatory and inhibitory neural transmission.
Materials and Reagents
- Animals
NIH nude rat pups (Charles River Laboratories, RRID: RGD_2312499) (postnatal Day 1, P1) are used for transplant recipients. These athymic nude rats are T-cell deficient and show depleted cell populations in thymus-dependent areas of peripheral lymphoid organs. Animals are housed and maintained in accordance with the National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals and Institutional Animal Care and Use Committees. - Culture products
- Regular 6-well plates (Thermo Fisher Scientific, catalog number: 140675 )
- Ultra-Low attachment 6-well plates (Corning, catalog number: 3471 )
- Laminin/poly-D-lysine coated plates (Corning, catalog number: 354595 )
- hiPSC dsRED-SC1014 cell line (Johns Hopkins Medical Institutions) (SC1014 cell line is a characterized human iPSC line which was reprogrammed by non-integrating Sendai virus and lentivirus. The stable dsRED-SC1014 cell line was established by nucleofection with piggybac-dsRED transposon and piggyback transposase)
- Mouse embryonic fibroblasts (MEFs, Gibco, catalog number: S1520-100 )
- Dulbecco’s modified Eagle’s medium (DMEM, Thermo Fisher Scientific, catalog number: 10569010 )
- Fetal Bovine Serum (FBS), ESC-qualified (Thermo Fisher Scientific, catalog number: 10439024 )
- Dulbecco’s modified Eagle’s medium/nutrient mixture F-12 (DMEM/F12, Thermo Fisher Scientific, catalog number: 11320033 )
- Knockout serum replacement (KSR, Thermo Fisher Scientific, catalog number: 10828028 )
- Fibroblast growth factor 2 (FGF2; 4 ng/ml; PeproTech, catalog number: 100-18B )
- 1 mM Glutamax (Thermo Fisher Scientific, catalog number: 35050061 )
- 100 μM non-essential amino acids (Thermo Fisher Scientific, catalog number: 11140050 )
- 100 μM 2-mercaptoethanol (Thermo Fisher Scientific, catalog number: 21985023 )
- Noggin (50 ng/ml, R&D system, catalog number: 6057-NG-025 )
- Dorsomorphin (1 μM, Tocris, catalog number: 3093 )
- SB431542 (10 μM, Tocris, catalog number: 1614 )
- 1% N2 supplement (Thermo Fisher Scientific, catalog number: 17502001 )
- Heparin (2 μg/ml, Millipore Sigma, catalog number: H3149 )
- Neurobasal medium (Thermo Fisher Scientific, catalog number: 21103049 )
- B27 minus VitA (Thermo Fisher Scientific, catalog number: 12587001 )
- Retinoic acid (RA, Millipore Sigma, catalog number: R2625 )
- Sonic Hedgehog (SHH, 50 ng/ml, R&D system, catalog number: 1845-SH-025 )
- Purmorphamine (2 μM, R&D system, catalog number: 4551/10 )
- Brain-derived neurotrophic factor (BDNF, 20 ng/ml, PeproTech, catalog number: 450-02 )
- Glial cell line-derived neurotrophic factor (GDNF, 20 ng/ml, PeproTech, catalog number: 450-10 )
- Ascorbic acid (0.2 mM, Millipore Sigma, catalog number: 1043003 )
- Dibutyryl cAMP (0.5 mM, Millipore Sigma, catalog number: D0260 )
- Collagenase (1 mg/ml, Millipore Sigma, catalog number: C9722 )
- Matrigel (Corning, catalog number: 354230 )
- Accutase (Corning, catalog number: 25-058-Cl )
- Dulbecco’s PBS (DPBS) without calcium and magnesium (Thermo Fisher Scientific, catalog number: 14190144 )
- Human embryonic stem cell (ESC) medium (see Recipes)
- Mouse embryonic fibroblast (MEF) medium (see Recipes)
- N2-induction medium (NIM) (see Recipes)
- Neural differentiation medium (see Recipes)
- Immunostaining
- Ketamine (Henry Schein, catalog number: 1049007 )
- Xylazine (Rompun, catalog number: 321350RX )
- Tissue-Tek OCT Compound (SAKURA, catalog number: 4583 )
- 2-methylbutane (Fisher Scientific, catalog number: 03551-4 )
- Donkey serum (Abcam, catalog number: ab7475 )
- Triton X-100 (Millipore Sigma, catalog number: T8787 )
- Anti-human-specific NES (Millipore, MAB5326, RRID: AB_11211837)
- Anti-TBR1 (Abcam, catalog number: ab31940 , RRID: AB_2200219)
- Anti-CTIP2 (Abcam, catalog number: ab18465 , RRID: AB_2064130)
- Anti-BRN2 (Santa Cruz Biotechnology, catalog number: sc-6029 , RRID: AB_2167385)
- Anti-SATB2 (Abcam, catalog number: ab51502 , RRID: AB_882455)
- Anti-PROX1 (Abcam, catalog number: ab37128 , RRID: AB_882189)
- Anti-TUJ1 (Millipore, catalog number: MAB1637 , RRID: AB_2210524)
- Anti-MAP2 (Millipore Sigma, catalog number: M2320 , RRID: AB_609904; Millipore, catalog number: AB5622 , RRID: AB_91939)
- Phosphate-buffered saline (PBS), 1x, pH 7.4 (see Recipes)
- 4% paraformaldehyde (PFA) (see Recipes)
- Ketamine/xylazine solution (see Recipes)
- Electrophysiological Recordings
- Isoflurane (Henry Schein, catalog number: 1182097 )
- NaCl (Millipore Sigma, catalog number: 71380 )
- KCl (Millipore Sigma, catalog number: 31248 )
- MgSO4 (Millipore Sigma, catalog number: 63138 )
- NaH2PO4 (Millipore Sigma, catalog number: S5011 )
- NaHCO3 (Millipore Sigma, catalog number: S5761 )
- CaCl2 (Millipore Sigma, catalog number: C7902 )
- D-glucose (Millipore Sigma, catalog number: G6152 )
- HEPES (Millipore Sigma, catalog number: H6147 )
- EGTA (Millipore Sigma, catalog number: E8145 )
- MgATP (Millipore Sigma, catalog number: A9187 )
- Na3GTP (Millipore Sigma, catalog number: G8877 )
- K-gluconate (Millipore Sigma, catalog number: G4500 )
- CsCl (Millipore Sigma, catalog number: 203025 )
- Tetrodoxin (TTX, 1 μM, Tocris, catalog number: 10-691 )
- Tetraethylammonium (TEA, 10 mM, Tocris, catalog number: 30-685-0 )
- Picrotoxin (10 μM, Tocris, catalog number: 11-281 )
- Bicuculline (Bic, 10 μM, Tocris, catalog number: 25-031-0 )
- 6-Cyano-7-nitroquinoxaline-2, 3-dione (CNQX, 20 μM, Tocris, catalog number: 10-451-0 )
- D-2-amino-5-phosphonovaleric acid (D-AP5, 50 μM, Tocris, catalog number: 01-061-0 )
- ACSF (see Recipes)
- Internal solution (see Recipes)
Equipment
- CO2 incubator (Eppendorf, Hauppauge, NY, USA)
- 37 °C water bath
- Centrifuge (Thermo Fisher Scientific, Waltham, MA, USA)
- Auto-nanoliter injector (Drummond, model: Nanoinject II )
- Cryostat (Leica, model: CM3050 )
- LSM880 confocal laser-scanning microscope (Carl Zeiss, Germany)
- Vibratome (Leica, model: VT1200S )
- Flaming-Brown micropipette puller (Sutter Instruments, model: P-1000 )
- HEKA EPC10 amplifier (HEKA Elektronik, Lambrech, Germany)
Software
- ZEN lite (Carl Zeiss, https://www.zeiss.com/microscopy/us/products/microscope-software/zen.html)
- PatchMaster (HEKA Elektronik, https://www.heka.com/downloads/downloads_main.html)
- Clampfit (Molecular devices, https://www.moleculardevices.com/products/axon-patch-clamp-system/acquisition-and-analysis-software/pclamp-software-suite#gref)
- MiniAnalysis (Synaptosoft, http://www.synaptosoft.com/MiniAnalysis/)
Procedure
- hiPSCs culture and passage
- Culture hiPSC dsRED-SC1014 cells on inactivated MEFs in human embryonic stem cell (ESC) medium (see Recipes) in regular 6-well plates. Change medium every day.
- Passage cells at a ratio 1:6 to 1:12 when cells reach 80-90% confluency.
- Prepare MEF plates following Steps A4-A6 one or two days before passaging hiPSCs culture.
- Cover the whole surface of culture vessels with 2% Matrigel (in DMEM/F12) and incubate the vessels at room temperature for at least 1 hour.
- Aspirate Matrigel and plate 30,000 cells/cm2 of mitotically inactivated MEFs in the vessels in MEF medium (see Recipes).
- Keep MEF plates in a 37 °C, 5% CO2 incubator.
- Aspirate the MEF medium from a plate containing inactivated MEFs and add pre-warmed human ESC medium 3-4 h before plating hiPSCs.
- Remove hiPSC colonies which need to be passaged under a dissection microscope.
- Add collagenase (1 mg/ml in DMEM/F12) solution to the plate containing hiPSCs, and incubate the plate in a 37 °C, 5% CO2 incubator for 30-60 min.
- Stop the incubation when the edges of the colonies start to pull away from the plate.
- Remove the collagenase carefully to avoid disturbing the attached cell layer.
- Add human ESC medium to the plate, gently blow hiPSCs off, and collect hiPSCs in a new conical tube. Make sure to minimize foaming.
- Centrifuge at 200 x g for 5 min, and then remove the supernatant from the hiPSC pellet.
- Resuspend the pellet with human ESC medium.
- Add appropriate volume of cell suspension to each MEF plate, and return the plate to the incubator. Replace medium daily.
- Neural differentiation of hiPSCs
- Differentiate hiPSCs to neurons based on rosette neural aggregates (termed RONAs) method (Xu et al., 2016) as below.
- Incubate hiPSC colonies with collagenase (1 mg/ml in DMEM/F12) in the incubator for about 5-10 min, and then gently wash off the collagenase with medium.
- Grow detached hiPSC colonies in suspension as embyroid bodies (EBs) (100-200 μm in diameter, ~10 EBs/ml medium) in human ESC medium without FGF2 for 2 days in Ultra-Low attachment 6-well plates (Corning).
- From day 2 to day 6, add Noggin (50 ng/ml, R&D system) or Dorsomorphin (1 μm, Tocris) and SB431542 (10 μM, Tocris) in human ESC medium (without FGF2).
- On day 7, transfer free-floating EBs to Matrigel precoated culture plates (as described above) to allow the complete attachment of EB aggregates with the supplement of N2-induction medium (NIM, see Recipes).
- Continuously feed cultures with N2-medium every other day from days 7 to 12.
- Change N2-induction medium every day from day 12 onward.
- Attached EB aggregates break down to form a monolayer colony on day 8 to 9 with typical neural-specific rosette formation. With the extension of neural induction, highly compact three-dimensional column-like neural aggregates RONAs form in the center of attached colonies. Microisolate RONAs manually, taking special care to minimize contaminating the peripheral monolayer of flat cells and cells underneath RONAs.
- At day 17, collect and maintain RONA clusters as neurospheres in Neurobasal medium (Thermo Fisher Scientific) containing B27 minus VitA (Thermo Fisher Scientific), 1 mM Glutamax (Thermo Fisher Scientific) for one day. A typical RONA is shown in Figure 1.
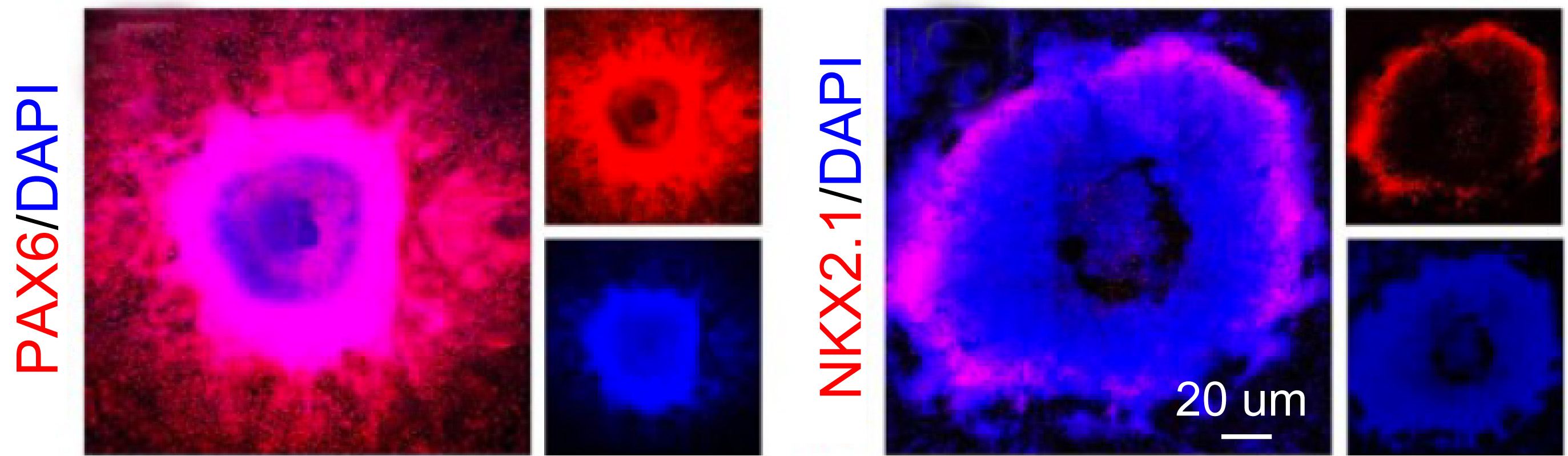
Figure 1. Typical RONAs on day 17. Immunostaining of PAX6 and NKX2.1 showing cortical patterning related transcriptional factors in neural progenitors (reproduced from Xu et al., 2016). - At day 18, dissociate neurospheres into single cells following Steps B11 to B19.
- Remove neurosphere suspension from culture plate and transfer to a 15 ml conical tube.
- Let neurospheres settle down in the tube 2-5 min, and then remove the medium carefully leaving ~100 μl medium remaining with the neurospheres.
- Resuspend neurospheres in 5 ml DPBS (Thermo Fisher Scientific).
- Let neurospheres settle down in the tube 2-5 min, and remove DPBS carefully leaving ~100 μl DPBS remaining with the neurospheres.
- Add 1 ml Accutase (Corning) to the neurospheres and incubate for 10 min at room temperature.
- Pipet up and down until all the neurospheres are in a single cell suspension.
- Add 4 ml neural differentiation medium (see Recipes) to the tube, and centrifuge the cells at 200 x g for 5 min.
- Remove the supernatant gently.
- Resuspend the cells in neural differentiation medium to desired cell density, and plate the cells on Laminin/poly-D-lysine coated plates (Corning).
- From days 24 to 30, apply RA (2 μM, Millipore Sigma), SHH (50 ng/ml, R&D system), and Purmorphamine (2 μM, R&D system) into neural differentiation medium to maintain the identity of neural progenitors, which provides an appropriate percentage of excitatory (~80-85%) and inhibitory (~15-20%) neurons representative of the human cerebral cortex.
- hiPSC-derived neural progenitors at days 31-32 are injected into neonatal rat cortices as described below (Procedure C), and characterization are performed at 10 weeks after transplantation.
- In culture conditions, hiPSC-derived neurons show mature human cortical identity after 10 weeks (Figure 2).
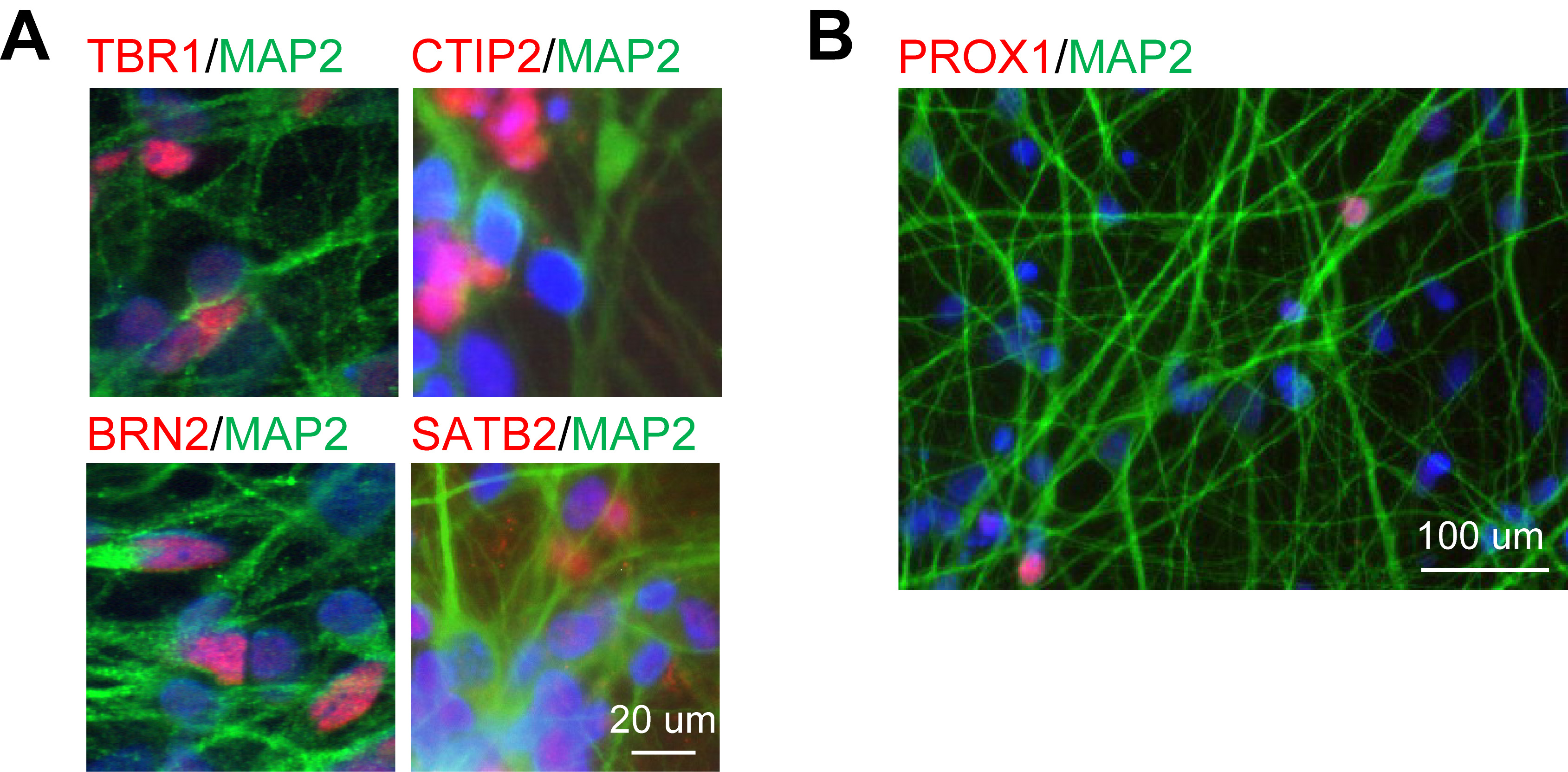
Figure 2. hiPSC-derived neurons show human cortical identity in culture after 10 weeks of differentiation. A. Cultures express the cortical layer-specific markers TBR1, CTIP2, BRN2, and STAB2. B. Some cells express the hippocampal marker PROX1 (reproduced from Yin et al., 2019).
- Animal transplantation
- The whole procedure from the cell culture to transplantation is shown in Figure 3.
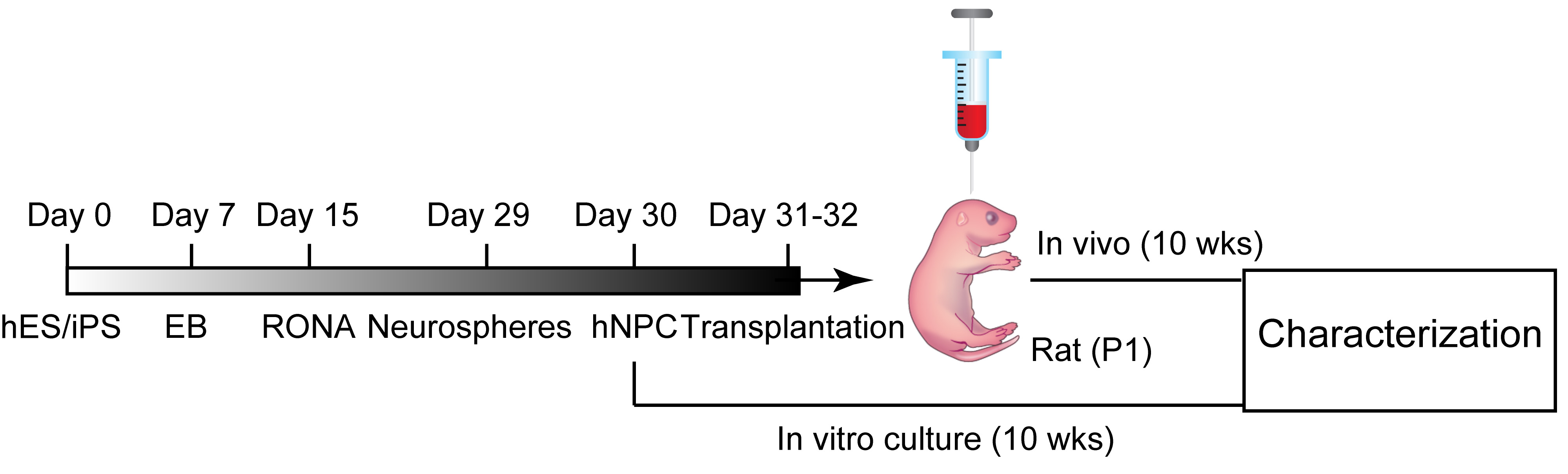
Figure 3. An outline showing transplantation of forebrain progenitors from hiPSCs (reproduced from Yin et al., 2019). - Trypsinize hiPSC-derived neural progenitors at days 31-32 in culture and thoroughly dissociate with a pipette. Then, centrifuge the cells at 300 x g for 3 min.
- Carefully remove the supernatant, and resuspend the pellet in ice cold 1x PBS with a final density of 2 x 106 cells/μl.
- Fill 1.0 μl of cells into a pulled glass micropipette (0.5 mm I.D., 1 mm O.D.) by suction, and then fit glass micropipette to an auto-nanoliter injector ( Nanoinject II , Drummond). Using nanoinjector allows to minimize tissue damage and inject small volume at a relatively slow rate.
- Gently and deeply anesthesize neonatal rat pups.
- Inject the cells into the right cortex (2.0 mm posterior and 1.9 mm lateral to bregma, 2.6 mm below the dura) over 1 min. Keep the micropipette tip in place for a further 2 min before withdrawal.
- The whole procedure from the cell culture to transplantation is shown in Figure 3.
- Brain sections immunostaining
- Anesthetize rats (10-week-old, also 10 weeks after transplantation) with ketamine/xylazine solution (0.5 ml/kg, see Recipes).
- Perfuse rats through the left ventricle of the heart with 1x PBS until the liver is bleached.
- Continue perfusion with freshly prepared 4% PFA.
- Dissect the brains and post-fix them in 4% PFA for 2-3 days at 4 °C.
- Transfer the brains to 30% sucrose until they sink to the bottom when they get saturated with sucrose solution.
- Remove the brains from sucrose solution and put them in 2-methylbutane (Fisher Scientific) in a container already chilled in liquid nitrogen for 30 s to 1 min.
- Drop some OCT compound (SAKURA) on the cryostat tissue holder already placed on dry ice.
- Place the brain tissue in the center of the holder and add more OCT compound surrounding the brain until it becomes solidified.
- Store the OCT brain blocks at -80 °C until being processed for serial sections.
- Prepare brain coronal sections (25 μm) using a cryostat ( CM3050 , Leica).
- Antigen retrieval step is optional and should be standardized depending upon the guidelines for each antibody. For the antibodies used in the present study, antigen retrieval is not necessary.
- Proceed sections for blocking with 10% (v/v) donkey serum and 0.2% (v/v) Triton X-100 in 1x PBS for one hour at room temperature.
- Incubate sections overnight at 4 °C with primary antibodies of interest.
- Wash sections with PBS three times of 5 min each, followed by incubations with appropriate, fluorescently labeled secondary antibodies at room temperature for one hour.
- After incubation for one hour at room temperature, wash sections with PBS three times for 5 min each and mount them on a clean slide. Dry the slide completely, and then proceed for imaging with a confocal microscope (Figure 4 and Video 1).
- Video 1 is produced using Z-Stack images. Set the first and last positions for the Z-Stack, i.e., the depth of the section. 1.01 µm is used for the Interval in the current study. Alternatively, choosing interval can be done by clicking on Optimal to set the number of slices.
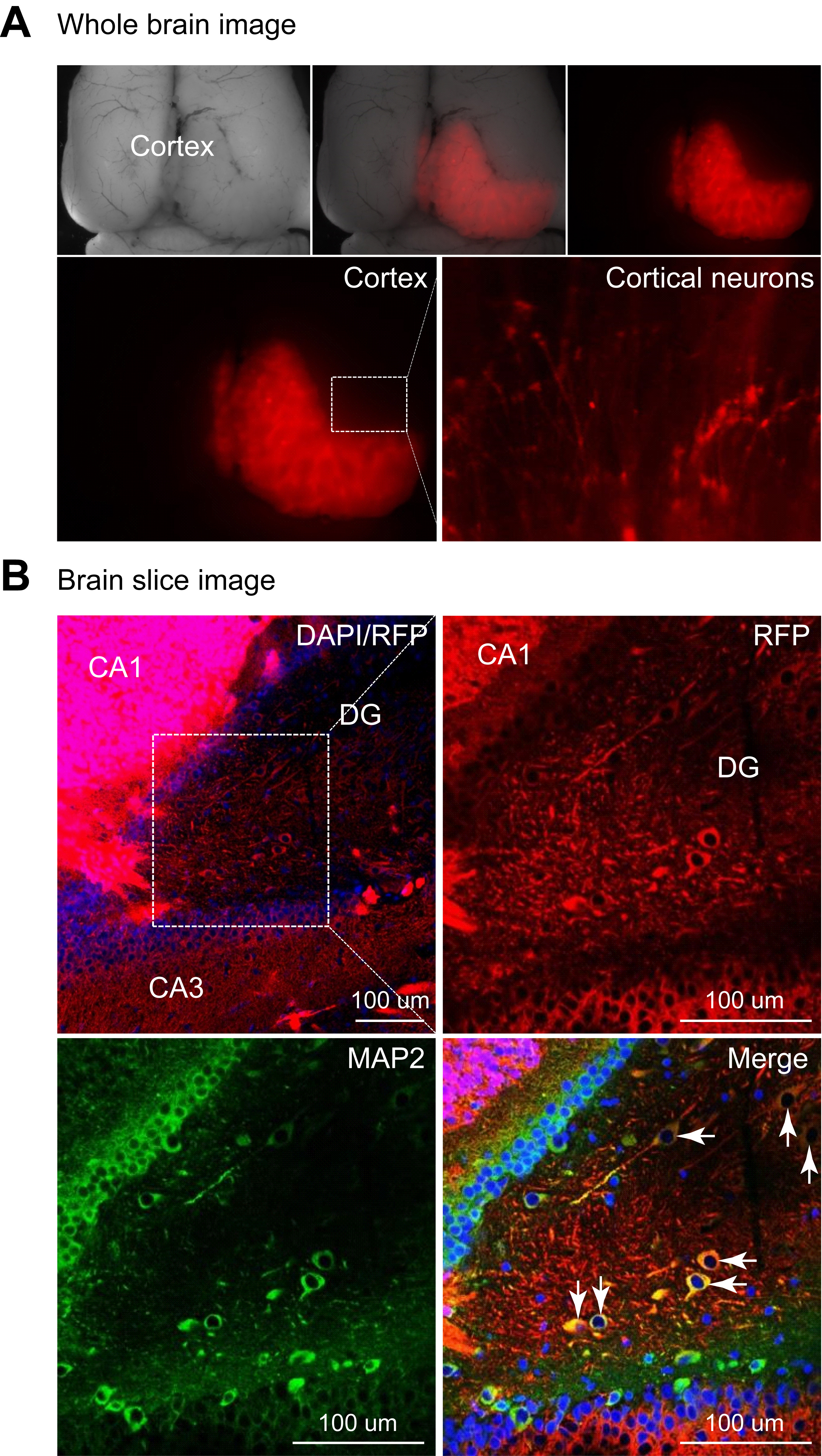
Figure 4. Human neurons expressing RFP colocalize with the mature neuronal marker MAP2, suggesting hiPSC-derived neurons integrate into the rat brain. A. A low magnification image showing RFP positive human cells in rat brain. B. Brain sections immunostained with MAP2 and DAPI (reproduced from Yin et al., 2019).Video 1. hiPSC-derived neurons in rat brain as shown by RFP/MAP2 double positive cells (reproduced from Yin et al., 2019)
- Electrophysiological recordings of acute brain slices
- Anesthetize rat (10-week-old, also 10 weeks following transplantation) with isoflurane (300 μl in a 500 ml container for drop jar anesthesia) and quickly remove the brain.
- Prepare transverse brain slices of 350 μm thickness using a vibratome (Leica VT1200S ).
- Incubate slices in ACSF solution (see Recipe 4) continuously bubbled with 95% O2 and 5% CO2, first at 34 °C for 30 min, and then at room temperature.
- Transfer a single slice into a submerged recording chamber constantly perfused with carbogen-equilibrated ACSF.
- Visualize injected human neurons expressing RFP under a 40x water immersion objective by fluorescence and DIC optics (Carl Zeiss).
- Perform whole-cell patch clamp recordings using a glass pipettes (3-5 MΩ) filled with internal solution (see Recipes). Randomly select RFP-positive human neurons for recording 300 μm to 1,000 μm from graft site.
- Induce action potentials by a series of hyperpolarizing and depolarizing step currents.
- Evoke sodium and potassium currents by a series of voltage steps (from –100 mV to +60 mV in 20-mV steps).
- Record spontaneous excitatory postsynaptic currents (sEPSCs) and spontaneous inhibitory postsynaptic currents (sIPSCs) in voltage-clamp configuration at –70 mV. Obtain sEPSCs in the presence of picrotoxin (100 μM), and sIPSCs in the presence of CNQX (20 μM) and D-AP5 (50 μM).
- The recording results show that grafting hiPSC-derived neurons functionally integrate to the rat brain, and they are able to fire and display excitatory and inhibitory synaptic activity (Figure 5).
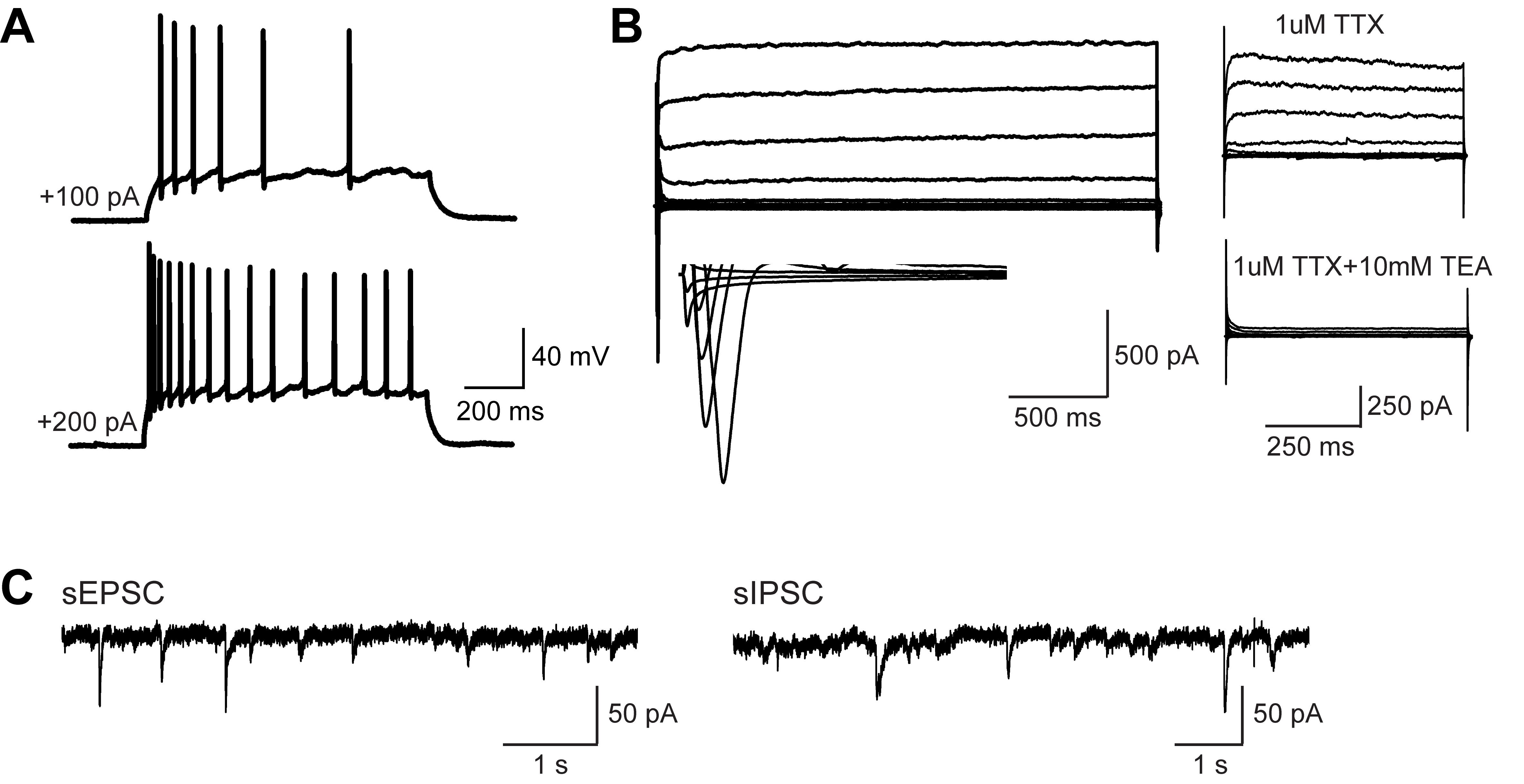
Figure 5. hiPSC-derived neurons functionally integrate into the synaptic circuitry of the rat brain. A. AP firing patterns of hiPSC-derived neurons. B. Representative traces of whole-cell Na+ (inwards) and K+ (outwards) currents recorded from grafted cells. C. Representative traces of sEPSCs and sIPSCs (reproduced from Yin et al., 2019).
Data analysis
The hiPSC cultures and human neurons obtained using these protocols were investigated and compared for their human cortical identity using a set of cortical-specific markers through a series of assays (Xu et al., 2016; Yin et al., 2019). Electrophysiological recording data were analyzed using Clampfit 10.5 software (Molecular devices) and MiniAnalysis software (Synaptosoft) as published (Yin et al., 2019). Therefore, the data analysis has not been discussed here.
Notes
- At Step A4, the collagenase should be gently washed off the plate with growth medium after the colony borders begin to peel away from the plate while the colony center remains attached, then the colonies will be selectively detached undisturbed.
- At Step A8, please note that attached aggregates usually break down to form a monolayer colony on day 8 to 9 with typical neural specific rosette formation. With the extension of neural induction, highly compact 3-dimensional column-like neural aggregates RONAs will form in the center of attached colonies.
- At Step A10, when collect the RONAs, the isolation should be done under microscope, taking special care to minimize contaminating the peripheral monolayer of flat cells and cells underneath RONAs.
- When preparing acute brain slices, the brain should be removed as soon as possible.
- ACSF solution should be prepared freshly every day before experiment. Also, it is very important to keep the slices in ACSF solution continuously bubbled with 95% O2 and 5% CO2 during recording. These will help maintain the brain slices in a healthily physiological condition.
Recipes
- 1x PBS, pH 7.4
NaCl 8 g (0.137 M)
KCl 200 mg (0.0027 M)
Na2HPO4 1.44 g (0.01 M)
KH2PO4 240 mg (0.0018 M)
Make up the volume to 1 L with MilliQ water, adjust the pH to 7.4 - 4% PFA
Dissolve 2 g PFA in 50 ml 1x PBS - Human embryonic stem cell (ESC) medium
DMEM/F12 (Thermo Fisher Scientific)
20% knockout serum replacement (KSR, Thermo Fisher Scientific)
4 ng/ml FGF2 (PeproTech)
1 mM Glutamax (Thermo Fisher Scientific)
100 μM non-essential amino acids (Thermo Fisher Scientific)
100 μM 2-mercaptoethanol (Thermo Fisher Scientific) - Mouse embryonic fibroblast (MEF) medium
DMEM (Thermo Fisher Scientific)
10% FBS, ESC-qualified (Thermo Fisher Scientific)
100 μM non-essential amino acid solution (Thermo Fisher Scientific)
100 μM 2-mercaptoethanol (Thermo Fisher Scientific) - N2-induction medium (NIM)
DMEM/F12
1% N2 supplement
100 μM MEM non-essential amino acids solution
1 mM Glutamax
2 μg/ml Heparin - Neural differentiation medium
Neurobasal/B27 (Thermo Fisher Scientific)
BDNF (20 ng/ml, PeproTech)
GDNF (20 ng/ml, PeproTech)
Ascorbic acid (0.2 mM, Millipore Sigma)
Dibutyryl cAMP (0.5 mM, Millipore Sigma) - Ketamine/xylazine solution
Ketamine (100 mg/ml)
Xylazine (20 mg/ml)
Mix them in sterile saline (0.9% NaCl) for anesthesia - ACSF
NaCl 125 mM
KCl 2.5 mM
MgSO4 1 mM
NaH2PO4 1.2 mM
NaHCO3 26 mM
CaCl2 2 mM
D-glucose 10 mM - Internal solution
K-gluconate 126 mM
KCl 8 mM
HEPES 20 mM
EGTA 0.2 mM
NaCl 2 mM
MgATP 3 mM
Na3GTP 0.5 mM
Adjust the pH to 7.3, and the osmolality to 290-300 mOsm
Acknowledgments
The protocols were developed with the grants from MSCRF and NIH. We thank Dr. Jin-Chong Xu, Dr. Gun-sik Cho and Dr. Chulan Kown for helping to standardize the protocols. The protocols were used in two separate studies published in 2016 and 2019 (Xu et al., 2016; Yin et al., 2019).
Competing interests
The authors declare no competing financial or non-financial interests.
Ethics
All experiments using hiPSCs were conducted in accordance with the policy of the Johns Hopkins University (JHU) School of Medicine (SOM) that research involving hiPSCs being conducted by JHU faculty, staff or students or involving the use of JHU facilities or resources shall be subject to oversight by the JHU Institutional Stem Cell Research Oversight (ISCRO) Committee.
All the animal procedures used in establishing the protocols were in accordance with the National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals and Institutional Animal Care and Use Committees. The approval ID of the animal experiment in this protocol was M017M288, which is validated from 10/1/2017 to 9/30/2020.
References
- Brennand, K. J., Marchetto, M. C., Benvenisty, N., Brustle, O., Ebert, A., Izpisua Belmonte, J. C., Kaykas, A., Lancaster, M. A., Livesey, F. J., McConnell, M. J., McKay, R. D., Morrow, E. M., Muotri, A. R., Panchision, D. M., Rubin, L. L., Sawa, A., Soldner, F., Song, H., Studer, L., Temple, S., Vaccarino, F. M., Wu, J., Vanderhaeghen, P., Gage, F. H. and Jaenisch, R. (2015). Creating patient-specific neural cells for the in vitro study of brain disorders. Stem Cell Reports 5(6): 933-945.
- Dolmetsch, R. and Geschwind, D. H. (2011). The human brain in a dish: the promise of iPSC-derived neurons. Cell 145(6): 831-834.
- Espuny-Camacho, I., Arranz, A. M., Fiers, M., Snellinx, A., Ando, K., Munck, S., Bonnefont, J., Lambot, L., Corthout, N., Omodho, L., Vanden Eynden, E., Radaelli, E., Tesseur, I., Wray, S., Ebneth, A., Hardy, J., Leroy, K., Brion, J. P., Vanderhaeghen, P. and De Strooper, B. (2017). Hallmarks of Alzheimer's disease in stem-cell-derived human neurons transplanted into mouse brain. Neuron 93(5): 1066-1081 e1068.
- Liu, Y., Weick, J. P., Liu, H., Krencik, R., Zhang, X., Ma, L., Zhou, G. M., Ayala, M. and Zhang, S. C. (2013). Medial ganglionic eminence-like cells derived from human embryonic stem cells correct learning and memory deficits. Nat Biotechnol 31(5): 440-447.
- Lodato, S., Rouaux, C., Quast, K. B., Jantrachotechatchawan, C., Studer, M., Hensch, T. K. and Arlotta, P. (2011). Excitatory projection neuron subtypes control the distribution of local inhibitory interneurons in the cerebral cortex. Neuron 69(4): 763-779.
- Lui, J. H., Hansen, D. V. and Kriegstein, A. R. (2011). Development and evolution of the human neocortex. Cell 146(1): 18-36.
- Maroof, A. M., Keros, S., Tyson, J. A., Ying, S. W., Ganat, Y. M., Merkle, F. T., Liu, B., Goulburn, A., Stanley, E. G., Elefanty, A. G., Widmer, H. R., Eggan, K., Goldstein, P. A., Anderson, S. A. and Studer, L. (2013). Directed differentiation and functional maturation of cortical interneurons from human embryonic stem cells. Cell Stem Cell 12(5): 559-572.
- Nicholas, C. R., Chen, J., Tang, Y., Southwell, D. G., Chalmers, N., Vogt, D., Arnold, C. M., Chen, Y. J., Stanley, E. G., Elefanty, A. G., Sasai, Y., Alvarez-Buylla, A., Rubenstein, J. L. and Kriegstein, A. R. (2013). Functional maturation of hPSC-derived forebrain interneurons requires an extended timeline and mimics human neural development. Cell Stem Cell 12(5): 573-586.
- Pasca, S. P., Portmann, T., Voineagu, I., Yazawa, M., Shcheglovitov, A., Pasca, A. M., Cord, B., Palmer, T. D., Chikahisa, S., Nishino, S., Bernstein, J. A., Hallmayer, J., Geschwind, D. H. and Dolmetsch, R. E. (2011). Using iPSC-derived neurons to uncover cellular phenotypes associated with Timothy syndrome. Nat Med 17(12): 1657-1662.
- Rakic, P. (2009). Evolution of the neocortex: a perspective from developmental biology. Nat Rev Neurosci 10(10): 724-735.
- Shi, Y., Kirwan, P., Smith, J., Robinson, H. P. and Livesey, F. J. (2012). Human cerebral cortex development from pluripotent stem cells to functional excitatory synapses. Nat Neurosci 15(3): 477-486, S471.
- Turrigiano, G. G. and Nelson, S. B. (2004). Homeostatic plasticity in the developing nervous system. Nat Rev Neurosci 5(2): 97-107.
- Vera, E. and Studer, L. (2015). When rejuvenation is a problem: challenges of modeling late-onset neurodegenerative disease. Development 142(18): 3085-3089.
- Weick, J. P., Liu, Y. and Zhang, S. C. (2011). Human embryonic stem cell-derived neurons adopt and regulate the activity of an established neural network. Proc Natl Acad Sci U S A 108(50): 20189-20194.
- Xu, J. C., Fan, J., Wang, X., Eacker, S. M., Kam, T. I., Chen, L., Yin, X., Zhu, J., Chi, Z., Jiang, H., Chen, R., Dawson, T. M. and Dawson, V. L. (2016). Cultured networks of excitatory projection neurons and inhibitory interneurons for studying human cortical neurotoxicity. Sci Transl Med 8(333): 333ra348.
- Yin, X., Xu, J. C., Cho, G. S., Kwon, C., Dawson, T. M. and Dawson, V. L. (2019). Neurons derived from human induced pluripotent stem cells integrate into rat brain circuits and maintain both excitatory and inhibitory synaptic activities. eNeuro 6(4).
Article Information
Copyright
© 2020 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Readers should cite both the Bio-protocol article and the original research article where this protocol was used:
- Yin, X., Dawson, T. M. and Dawson, V. L. (2020). Integration of Human Induced Pluripotent Stem Cell (hiPSC)-Derived Neurons into Rat Brain . Bio-protocol 10(17): e3746. DOI: 10.21769/BioProtoc.3746.
- Yin, X., Xu, J. C., Cho, G. S., Kwon, C., Dawson, T. M. and Dawson, V. L. (2019). Neurons derived from human induced pluripotent stem cells integrate into rat brain circuits and maintain both excitatory and inhibitory synaptic activities. eNeuro 6(4).
Category
Stem Cell > Pluripotent stem cell > Cell differentiation
Neuroscience > Nervous system disorders > Animal model
Cell Biology > Cell Transplantation > Xenograft
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link









