- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Insect Feeding Assays with Spodoptera exigua on Arabidopsis thaliana
(*contributed equally to this work) Published: Vol 10, Iss 5, Mar 5, 2020 DOI: 10.21769/BioProtoc.3538 Views: 5357
Reviewed by: Zhibing LaiDemosthenis ChronisAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

A Detailed Guide to Recording and Analyzing Arabidopsis thaliana Leaf Surface Potential Dynamics Elicited by Mechanical Wounding
Fatiha Atanjaoui [...] Michael M. Wudick
Apr 5, 2025 1499 Views
Image-Based Lignin Detection in Nematode-Induced Feeding Sites in Arabidopsis Roots
Muhammad Amjad Ali and Krzysztof Wieczorek
May 5, 2025 1851 Views
Abstract
Plant-insect interaction is an important field for studying plant immunity. The beet armyworm, Spodoptera exigua, is one of the best-known agricultural pest insects and is usually used to study plant interactions with chewing insects. Here, we describe a protocol for insect feeding assays with Spodoptera exigua lavae using model host plant Arabidopsis thaliana, which is simple and easy to conduct, and can be used to evaluate the effect of host genes on insect growth and thus to study plant resistance to chewing insects.
Background
Plants face a variety of biotic stresses throughout their lives, such as herbivore attack and pathogen infection. The beet armyworm, Spodoptera exigua, is a worldwide phytophagous pest with a broad host range, damaging various vegetable crops and causing considerable economic agricultural losses (Howe and Jander, 2008; Hu et al., 2013). Spodoptera exigua larvae usually feed on both foliage and fruit, and are extremely destructive. Arabidopsis thaliana is a host plant of Spodoptera exigua, and is also a classic model plant to study plant resistance to herbivores. Here, we describe a method adapted from our previous work (You et al., 2019) to conduct the feeding assays with Spodoptera exigua larvae on Arabidopsis thaliana rosette leaves. By counting the weight of the larvae after feeding leaves from different genotypes, we were able to evaluate plant resistance to herbivore attacks in the laboratory settings.
Materials and Reagents
- Square Petri dishes (100 mm × 100 mm, Beijing Ruiaizhengte Biological Technology Co., Ltd., catalog number: YC-HC99050 )
- Petri dishes (150 mm, Corning, catalog number: 430599 )
- 1.5 ml microtubes (AXYGEN, catalog number: MCT-150-C )
- Pipet tips (AXYGEN, catalog number: T-200-Y and T-1000-B )
- Micropore tape (3M, MicroporeTM, catalog number: 1530C-0 )
- Parafilm ( Bemis, catalog number: PM-996 )
- Toothpick (Suncha, catalog number: YQ1250 )
- Black cloth (Beiyang, catalog number: 13000133 )
- Arabidopsis thaliana
- Spodoptera exigua (KEYUN) (https://item.taobao.com/item.htm?spm=a1z09.2.0.0.10672e8dnlXuwi&id=567208183626&_u=pmk8luh1123)
- Artificial diet (Ingredients: wheat germ, yeast, carrageenan, konjac powder, sorbic acid, vitamin C, corn oil, and linoleic acid; KEYUN)
(https://item.taobao.com/item.htm?spm=a1z09.2.0.0.10672e8dnlXuwi&id=43498077050&_u=pmk8luh74e5) - Murashige & Skoog basal medium with vitamins (Phyto Technology Laboratories, catalog number: M519 )
- Bacto-agar (BD, BactoTM, catalog number: 214010 )
- 10% Bleach (KAO, 600 ml)
- 1 M KOH solution (Aladdin, catalog number: P112281 )
- Sucrose (Sinopharm Chemical Reagent Co., Ltd., catalog number: 10021418 )
- Diethyl ether (Sinopharm Chemical Reagent Co., Ltd., catalog number: 10009328 )
- Sterile distilled water
- Nutritional soil (moss peat:vermiculite = 2:1, PINDSTRUP, type: 0-10 mm)
- ½ MS medium (see Recipes)
- 0.8% agar medium (see Recipes)
Equipment
- Pipettes (Gilson, Pipetman® G)
- Graduated cylinder
- Reagent bottle
- Refrigerator or a cold room
- Tweezers
- Plant growth chamber
- Square pot
- Autoclave
- Laminar flow hood
- Balance
- Canon camera
- Ruler
Software
- Microsoft Excel
Procedure
- Cultivation of Arabidopsis thaliana plants
- Prepare the ½ MS solid medium, and pour enough media into square Petri dishes (100 mm × 100 mm) to cover approximately half of the depth of the dish (40 ml).
- Surface-sterilize Arabidopsis seeds in 1.5 ml microtubes by soaking in 10% bleach for 15 min, then remove all bleach residue by rinsing five times with sterile distilled water.
- Sow the seeds in the square Petri dishes containing ½ MS medium (Figure 1A), and seal the dishes with Micropore tape.
- Place the dishes in the dark at 4 °C for 2 days to allow for efficient and synchronous germination.
- Transfer the dishes to the growth chamber set at 22 °C with a 10-h light/14-h dark photoperiod for 10 days.
- Transplant the seedlings into square pots filled with nutritional soil (Figure 1B), and keep them in the growth chamber (22 °C, 10-h light/14-h dark) for another 4 weeks (Figure 1C).
Note: Be careful not to damage the roots of the seedlings. Take care of the plants to prevent them from being affected by herbivores or pathogens.
Figure 1. The cultivation of Arabidopsis plants. A. Sterilized Arabidopsis seeds were sowed on the square dish containing ½ MS medium. B. Ten-day old Arabidopsis seedlings were transplanted into a pot filled with nutritional soil. C. Arabidopsis seedlings grown in pot for 4 weeks are used for insect feeding assays.
- Preparation of Spodoptera exigua larvae
- Place the Spodoptera exigua eggs in a square Petri dish, and seal the dish with Micropore tape (Figure 2A).
- Place the dish in the 27 °C incubator with relative humidity of 40-50% for hatching (about 2-3 days).
- As soon as the eggs hatch, add artificial diet to the dish (Figure 2B) and continue incubation for 5 days.
Note: Cut the artificial diet into small pieces of 1-1.5 cm square and placed them in the Petri dish at a certain interval from the eggs or larvae. Usually, after 5 days of feeding, the larvae become third-instar larvae.
Figure 2. The hatching of Spodoptera exigua eggs. A. Spodoptera exigua eggs. B. Newly hatched larvae and the artificial diet. - Use a toothpick to transfer the larvae into a new Petri dish (Video 1 and Figure 3A) and starve them for 12 h before being used in the experiments (Figure 3B).
Note: This step needs to be careful and gentle. Usually, we use a water-soaked toothpick to let the larvae catch and gently transfer the larvae to prevent them from being injured.
Figure 3. The starvation treatment of Spodoptera exigua larvae. A. A larva clutching a toothpick. B. Third-instar larvae starved for 12 h.Video 1. How to catch a larva with a toothpick?
- Insect feeding assays
- Prepare and pour enough 0.8% agar into plastic Petri dishes (150 mm) to cover approximately half of the depth of the dish.
- Cut mature rosette leaves of similar size from Arabidopsis plants grown in soil for 4 weeks, and place them in a plastic Petri dish (150 mm) containing 0.8% agar (Figure 4A).
Note: If the leaves of different genotypes are similar in shape and size, we usually arrange them in the way shown in Figure 4A. And if the shape and size of the leaves vary greatly, it is required to weigh the leaves each time to ensure that the same weight of leaves are added. - Use a water-soaked toothpick to gently transfer 15 starved third-instar larvae into the Petri dish containing rosette leaves.
Note: This step needs to be careful and gentle. - Seal the Petri dishes with Micropore tape, and put them in the growth chamber (22 °C, 10-h light/14-h dark).
- Feed the larvae for 3 days (Figure 4B), and replace the leaves in each Petri dish by fresh leaves every day.
Note: Add fresh leaves in time to ensure that the larvae have enough food. The feeding days can be adjusted according to the experimental conditions.
As the plant hormone jasmonate (JA) plays a vital role in regulating plant defense response against herbivore attack, we conducted the insect feeding assays using the wild type (WT) plants and the coi1-2 mutant, which harbors a point mutation of the JA receptor gene CORONATINE INSENSITIVE1 (COI1) (Xu et al., 2002), and compared their resistance to the S. exigua larvae (Figures 4C and 4D).
- Collection of experimental results
- After three days of feeding, transfer the larvae into a new Petri dish, and weigh all Spodoptera exigua larvae from each Petri dish.
- Place a cotton ball soaked in ether in the Petri dish, and seal the dish with parafilm.
- When the larvae are unconscious, use a small pair of tweezers to gently place them on a black cloth, arrange them from large to small, and take photos (Figure 4C).
Note: Steps D1 and D3 should be gentle to prevent the larvae from being injured and spit out green oral secretions.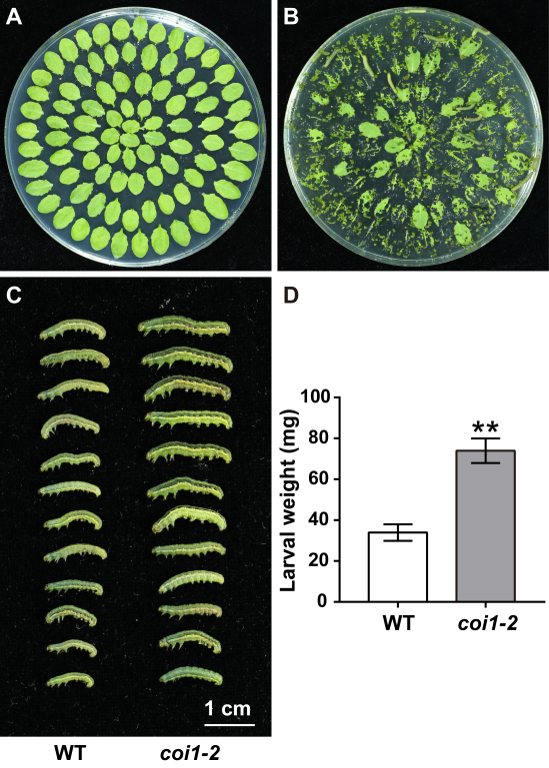
Figure 4. Insect feeding assays with Spodoptera exigua larvae. A. Mature rosette leaves were cut and placed in the plastic Petri dish (150 mm) containing 0.8% agar. B. The leaves after feeding by Spodoptera exigua larvae for 1 day. C. Spodoptera exigua larvae after feeding on rosette leaves of WT and coi1-2 plants for 3 days. Scale Bar = 1 cm. D. Average weight of the larvae fed with rosette leaves of WT and coi1-2 plants for 3 days. Data represent means ± SD (n = 3). Asterisks indicate significant differences from the WT according to Student’s t-test at **, P < 0.01.
Data analysis
Count the weight of each larva in each dish, and calculate the average. Statistical analysis should be done by calculating the average of three independent experiments and standard deviation using Microsoft Excel or any other statistical analysis software. Significance of the difference between two samples can be obtained by performing the Student’s t-test (Figure 4D). Usually, the larvae grow well after eating the leaves. In some cases, one or two larvae will die, and avoid using the dead larvae for analysis. Otherwise, collect all larvae for data analysis.
Recipes
- ½ MS medium
2.215 g Murashige & Skoog basal medium with vitamins
10 g sucrose
8 g Bacto-agar
Add ddH2O to 1,000 ml
Use KOH to adjust pH to 5.8
Autoclave at 15 psi, 121 °C for 15 min - 0.8% agar medium
8 g Agar
Add ddH2O to 1,000 ml
Autoclave at 121 °C for 15 min
Acknowledgments
This work was supported by the Strategic Priority Research Program of the Chinese Academy of Sciences (Grant XDB11030200), the Ministry of Science and Technology of China (Grant 2015CB942900), and the National Natural Science Foundation of China (Grants 31730010 and 31770303). This protocol was adapted from the methods described in You et al. (2019).
Competing interests
The authors declare that they have no competing interests.
References
- Howe, G. A. and Jander, G. (2008). Plant immunity to insect herbivores. Annu Rev Plant Biol 59: 41-66.
- Hu, P., Zhou, W., Cheng, Z., Fan, M., Wang, L. and Xie, D. (2013). JAV1 controls jasmonate-regulated plant defense. Mol Cell 50(4): 504-515.
- Xu, L., Liu, F., Lechner, E., Genschik, P., Crosby, W.L., Ma, H., Peng, W., Huang, D. and Xie, D. (2002). The SCFCOI1 ubiquitin-ligase complexes are required for jasmonate response in Arabidopsis. Plant Cell 14(8): 1919-1935.
- You, Y., Zhai, Q., An, C. and Li, C. (2019). LEUNIG_HOMOLOG mediates MYC2-dependent transcriptional activation in cooperation with the coactivators HAC1 and MED25. Plant Cell 31(9): 2187-2205.
Article Information
Copyright
© 2020 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
You, Y., An, C. and Li, C. (2020). Insect Feeding Assays with Spodoptera exigua on Arabidopsis thaliana. Bio-protocol 10(5): e3538. DOI: 10.21769/BioProtoc.3538.
Category
Plant Science > Plant immunity > Plant-insect interaction
Cell Biology > Cell signaling > Stress response
Plant Science > Plant physiology > Biotic stress
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link