- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Infectious Subviral Particle to Membrane Penetration Active Particle (ISVP-to-ISVP*) Conversion Assay for Mammalian Orthoreovirus
Published: Vol 8, Iss 2, Jan 20, 2018 DOI: 10.21769/BioProtoc.2700 Views: 6635
Reviewed by: Yannick DebingKristin ShinglerSmita Nair

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
General Maintenance and Reactivation of iSLK Cell Lines
Ariana C. Calderón-Zavala [...] Ekaterina E. Heldwein
Jun 5, 2025 1934 Views
Inducible HIV-1 Reservoir Reduction Assay (HIVRRA), a Fast and Sensitive Assay to Test Cytotoxicity and Potency of Cure Strategies to Reduce the Replication-Competent HIV-1 Reservoir in Ex Vivo PBMCs
Jade Jansen [...] Neeltje A. Kootstra
Jul 20, 2025 2490 Views
Assembly and Mutagenesis of Human Coronavirus OC43 Genomes in Yeast via Transformation-Associated Recombination
Brett A. Duguay and Craig McCormick
Aug 20, 2025 3072 Views
Abstract
The mammalian orthoreovirus (reovirus) outer capsid undergoes a series of conformational changes prior to or during viral entry. These transitions are necessary for delivering the genome-containing core across host cell membranes. This protocol describes an in vitro assay for monitoring the transition into a membrane penetration-active form (i.e., ISVP*).
Keywords: VirologyBackground
Reoviruses are nonenveloped, double-stranded RNA viruses that are composed of two concentric protein shells: the inner capsid (core) and the outer capsid (Dryden et al., 1993; Zhang et al., 2005; Dermody et al., 2013). Following attachment, virions are endocytosed (Borsa et al., 1979; Ehrlich et al., 2004; Maginnis et al., 2006; Maginnis et al., 2008) and host cathepsin proteases degrade the σ3 outer capsid protein (Chang and Zweerink, 1971; Silverstein et al., 1972; Borsa et al., 1981; Sturzenbecker et al., 1987; Dermody et al., 1993; Baer and Dermody, 1997; Ebert et al., 2002). This process generates a metastable intermediate, called infectious subviral particle (ISVP), in which the cell penetration protein, µ1, is exposed (Dryden et al., 1993). ISVPs are produced in vitro by treating purified virions with chymotrypsin (Joklik, 1972; Borsa et al., 1973a). The outer capsid undergoes a second conformational change to deposit the genome-containing core into the host cell cytoplasm. As a consequence, the central δ fragment of µ1 adopts a protease sensitive conformation. The altered particle is called ISVP* (Chandran et al., 2002). ISVP-to-ISVP* conversion can be induced in vitro using heat (Middleton et al., 2002), large monovalent cations (Borsa et al., 1973b), µ1-derived peptides (Agosto et al., 2008), red blood cells (Chandran et al., 2002; Sarkar and Danthi, 2010), or lipids (Snyder, and Danthi, 2015 and 2016). Thus, questions related to reovirus entry (e.g., the relationship between particle stability and infectivity) are studied using biochemical and cell-based approaches. In this protocol, we describe an in vitro assay that recapitulates ISVP-to-ISVP* conversion.
Materials and Reagents
- Pipette tips
- PCR 8-well tube strips (VWR, catalog number: 20170-004 )
- 50 ml centrifuge tube (VWR, catalog number: 89039-660 )
- 1.7 ml microcentrifuge tubes (MIDSCI, catalog number: AVSS1700 )
- Purified reovirus stocks (see [Berard and Coombs, 2009; Kobayashi et al., 2010] for propagation and purification procedure)
- Crushed ice
- Standard SDS-PAGE materials and reagents (e.g., 10% SDS-polyacrylamide mini gels)
- Coomassie Brilliant Blue stain and destain solutions (Bio-Rad Laboratories, catalog number: 1610435 )
- Bleach (Biz4USA, Janitorial Supplies, catalog number: CLO30966CT )
- 2-Amino-2-(hydroxymethyl)-1,3-propanediol (Tris) (MP Biomedicals, catalog number: 02103133 )
- Magnesium chloride hexahydrate (MgCl2·6H2O) (Sigma-Aldrich, catalog number: M9272 )
- Sodium chloride (NaCl) (Merck, catalog number: SX0420-3 )
- 0.1 N hydrochloric acid (Sigma-Aldrich, catalog number: 2104 )
- N sodium hydroxide (Sigma-Aldrich, catalog number: 2105 )
- 4x Laemmli sample buffer (Bio-Rad Laboratories, catalog number: 1610747 )
- Nα-p-tosyl-L-lysine chloromethyl ketone (TLCK)-treated chymotrypsin (Worthington Biochemical, catalog number: LS001432 )
- Trypsin (Sigma-Aldrich, catalog number: T6567 )
- Phenylmethylsulfonyl fluoride (PMSF) (Sigma-Aldrich, catalog number: P7626 )
- Ultrapure DNase/RNase-free distilled H2O (Thermo Fisher Scientific, InvitrogenTM, catalog number: 10977015 )
- Isopropyl alcohol (Avantor Performance Materials, Macron, catalog number: 3032-02 )
- 50% bleach (see Recipes)
- Virus storage buffer (VB) (see Recipes)
- 2 mg/ml Nα-p-tosyl-L-lysine chloromethyl ketone (TLCK)-treated chymotrypsin (see Recipes)
- 100 mM phenylmethylsulfonyl fluoride (PMSF) (see Recipes)
- 1 mg/ml trypsin (see Recipes)
Equipment
- Personal protective equipment (PPE)
- Laboratory coat
- Gloves
- Eye protection
- Laboratory coat
- Biosafety level 2 (BSL-2) laboratory facility
- BSL-2 certified tissue culture hood
- Solid and liquid waste containers
- Autoclave
- Ice bucket
- -20 °C freezer
- Micropipettes
- 0.1-2.5 µl capacity (Eppendorf, catalog number: 3123000012 )
- 2-20 µl capacity (Eppendorf, catalog number: 3123000039 )
- 20-200 µl capacity (Eppendorf, catalog number: 3123000055 )
- 100-1,000 µl capacity (Eppendorf, catalog number: 3123000063 )
- 0.1-2.5 µl capacity (Eppendorf, catalog number: 3123000012 )
- Digital pH meter (VWR, model: SB70P )
- Digital laboratory balance (Mettler Toledo, model: PB1502-S )
- NanoDrop spectrophotometer (Thermo Fisher Scientific, Thermo ScientificTM, model: ND-1000 )
- Hot plate stirrer (VWR, catalog number: 12365-382 )
- Magnetic stir bar (VWR, catalog number: 58948-273 )
- Thermal cycler (Bio-Rad Laboratories, model: S1000TM )
- Analog dry block heater (VWR, catalog number: 12621-110 )
- Thermometer (VWR, catalog number: 89095-566 )
- Gel imaging system (LI-COR, model: Odyssey® Classic )
- 1,000 ml glass beaker (VWR, catalog number: 89000-212 )
- 1,000 ml graduated cylinder (VWR, catalog number: 65000-012 )
- 1,000 ml storage bottle (VWR, catalog number: 89000-240 )
Note: This product has been discontinued.
Software
- Image Studio Lite (LI-COR)
Procedure
- Generation of infectious subviral particles (ISVPs)
- Propagate and purify reovirus virions as previously described (Berard and Coombs, 2009; Kobayashi et al., 2010). Using a NanoDrop spectrophotometer, determine particle concentration by measuring the optical density of the purified virus stocks at 260 nm (OD260; 1 unit at OD260 = 2.1 x 1012 particles/ml) (Smith et al., 1969).
- In 1 tube of an 8-well tube strip, dilute 2 x 1011 virions into 90 µl of ice cold VB (see Recipes).
- Add 10 µl of ice cold 2 mg/ml TLCK-treated chymotrypsin (see Recipes) to the diluted virus. Mix by pipetting up and down 3-4 times.
Note: For an undigested control, substitute 10 µl of ice cold VB for 10 µl of TLCK-treated chymotrypsin. - Incubate the reaction for 20 min at 32 °C in a thermal cycler.
Note: Under these conditions, σ3 is degraded (Joklik, 1972; Borsa et al., 1973a) and µ1 is cleaved (Nibert and Fields, 1992; Chandran et al., 1999). - Following digestion, quench chymotrypsin activity by the addition of 1 µl of 100 mM PMSF (see Recipes). Mix by pipetting up and down 3-4 times.
- Incubate the reaction for 20 min on ice.
- To confirm that ISVPs are generated, run 2 x 1010 particles per lane on a 10% SDS-polyacrylamide mini gel. Run the gel for 40-45 min at 200 V constant.
- Visualize the protein bands by Coomassie Brilliant Blue staining (see Data analysis, Figure 1).
- Store ISVPs on ice, and use within 2-3 h for ISVP-to-ISVP* conversion experiments.
- Propagate and purify reovirus virions as previously described (Berard and Coombs, 2009; Kobayashi et al., 2010). Using a NanoDrop spectrophotometer, determine particle concentration by measuring the optical density of the purified virus stocks at 260 nm (OD260; 1 unit at OD260 = 2.1 x 1012 particles/ml) (Smith et al., 1969).
- ISVP-to-ISVP* conversion assay
- Add 10 µl of ISVPs to each tube of an 8-well tube strip on ice.
- Incubate the ISVPs for 1 h on a temperature gradient in a thermal cycler (i.e., each tube is incubated at a different temperature).
Note: Appropriate temperature ranges will vary based on the incubation time (5 min to 1 h) and the reovirus strain under investigation. Each step in the temperature gradient should be no less than 1 °C. ISVP-to-ISVP* conversion typically occurs between 30 °C and 50 °C. - Incubate the heat treated ISVPs for 5 min on ice.
- Add 0.9 µl of 1 mg/ml trypsin (see Recipes) to each tube of heat treated ISVPs. Mix by pipetting up and down 3-4 times.
- Incubate the reactions for 30 min on ice.
- Following digestion, add 3.3 µl of 4x Laemmli sample buffer to each reaction. Mix by pipetting up and down 3-4 times.
- Boil each reaction for 5 min at 95 °C.
- To determine the temperature at which ISVP-to-ISVP* conversion occurs, run 2 x 1010 particles (i.e., the entire ISVP-to-ISVP* conversion reaction at a given temperature) per lane on a 10% SDS-polyacrylamide mini gel. Run the gel for 40-45 min at 200 V constant.
- Visualize the protein bands by Coomassie Brilliant Blue staining (see Data analysis, Figure 2).
- Add 10 µl of ISVPs to each tube of an 8-well tube strip on ice.
Data analysis
- Generation of infectious subviral particles (ISVPs)
- Record and analyze the results using a gel imaging system and Image Studio Lite software (Figure 1).
- Virions contain λ1,2,3, µ1C, σ2, and σ3.
- ISVPs contain λ1,2,3, µ1C, δ, and σ2.
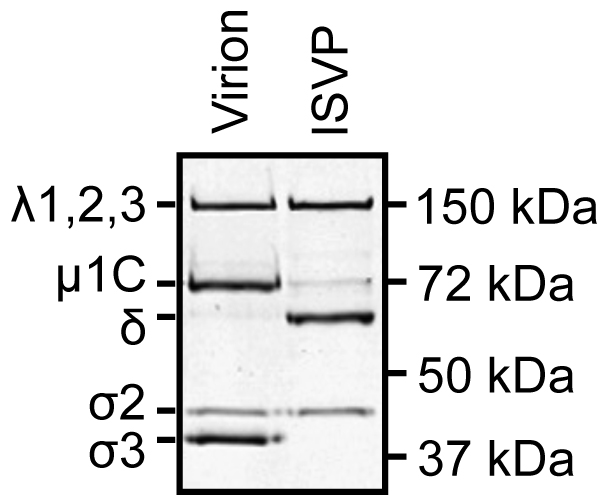
Figure 1. SDS-PAGE gel of reovirus virions and ISVPs - Virions contain λ1,2,3, µ1C, σ2, and σ3.
- ISVP-to-ISVP* conversion assay
- All ISVP-to-ISVP* experiments should be repeated for at least three independent replicates.
- Record and analyze the results using a gel imaging system and Image Studio Lite software (Figure 2).
- Trypsin treated ISVPs contain λ1,2,3, µ1C, δ, and σ2.
- Trypsin treated ISVP*s contain λ1,2,3 and σ2.
- Trypsin treated ISVPs contain λ1,2,3, µ1C, δ, and σ2.
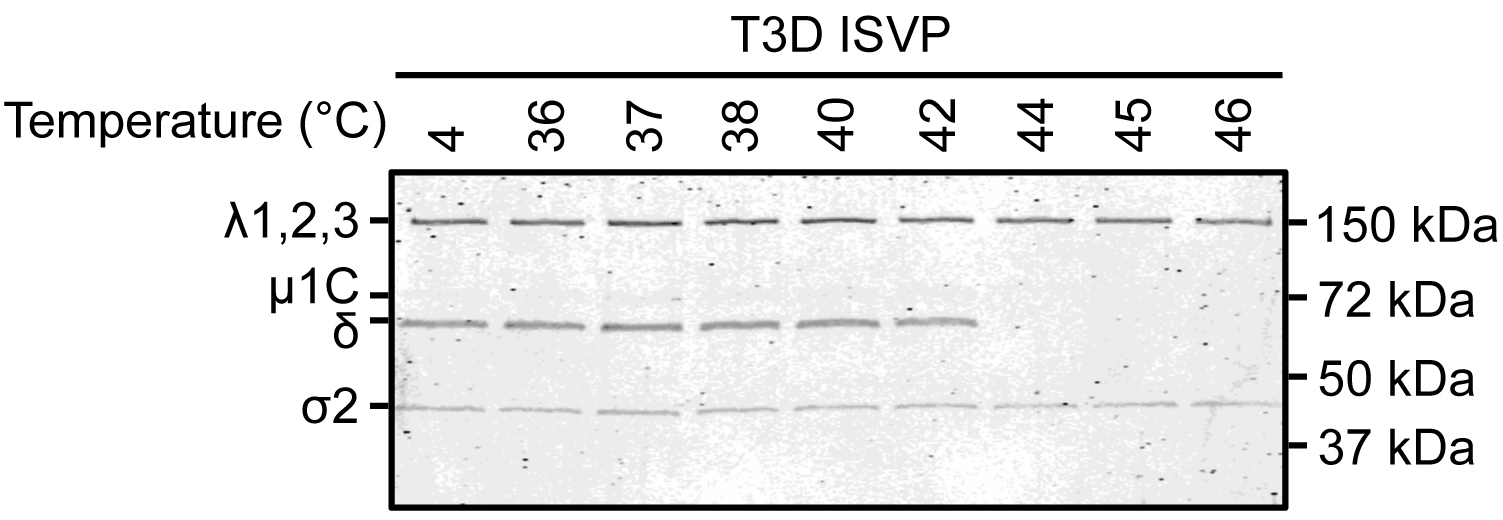
Figure 2. SDS-PAGE gel of heat and trypsin treated reovirus ISVPs. In the experiment shown here, ISVP-to-ISVP* conversion occurred at 44, 45, and 46 °C. - All ISVP-to-ISVP* experiments should be repeated for at least three independent replicates.
- Record and analyze the results using a gel imaging system and Image Studio Lite software (Figure 1).
Notes
- When possible, all procedures are performed in a BSL-2 certified tissue culture hood.
- Laboratory personnel should use appropriate PPE.
- All solid waste is autoclaved prior to disposal.
- All liquid waste is inactivated with 50% bleach prior to disposal.
Recipes
- 50% bleach
In a storage bottle, dilute 50 ml of 100% bleach into 50 ml of ultrapure H2O - Virus storage buffer (VB) (10 mM Tris, pH 7.4, 15 mM MgCl2, and 150 mM NaCl)
- In a glass beaker, dissolve the following into 900 ml of ultrapure H2O:
1.21 g Tris
3.05 g MgCl2·6H2O
8.77 g NaCl - Mix at room temperature using a magnetic stir bar on a stir plate
- Adjust to pH 7.4 with 0.1 N hydrochloric acid
- In a graduated cylinder, bring the final volume up to 1,000 ml with ultrapure water
- Transfer the solution to a storage bottle
- Sterilize by autoclaving
- Store at room temperature
- In a glass beaker, dissolve the following into 900 ml of ultrapure H2O:
- 2 mg/ml Nα-p-tosyl-L-lysine chloromethyl ketone (TLCK)-treated chymotrypsin
- In a centrifuge tube, dissolve 100 mg of TLCK-treated chymotrypsin into 50 ml of ultrapure H2O
- Mix at room temperature by gently inverting the tube until the solution becomes clear
- Transfer 1 ml aliquots to microcentrifuge tubes
- Store at -20 °C
- In a centrifuge tube, dissolve 100 mg of TLCK-treated chymotrypsin into 50 ml of ultrapure H2O
- 100 mM phenylmethylsulfonyl fluoride (PMSF)
- In a microcentrifuge tube, dissolve 17.4 mg of PMSF into 1 ml of isopropyl alcohol
- Mix at room temperature by gently inverting the tube until the solution becomes clear
- Store at -20 °C
- In a microcentrifuge tube, dissolve 17.4 mg of PMSF into 1 ml of isopropyl alcohol
- 1 mg/ml trypsin
- In a microcentrifuge tube, dissolve 1 mg of trypsin into 1 ml of ultrapure H2O
- Mix at room temperature by gently inverting the tube until the solution becomes clear
- Store at -20 °C
Acknowledgments
This protocol was adapted from a previously published study (Chandran et al., 2002). Research reported in this publication was supported by the National Institute of Allergy and Infectious Diseases of the National Institutes of Health under award numbers 1R01AI110637 (to P.D.) and F32AI126643 (to A.J.S.) and by Indiana University Bloomington. The content is solely the responsibility of the authors and does not necessarily represent the official views of the funders. The authors declare no conflict of interest.
References
- Agosto,M. A., Myers, K. S., Ivanovic, T. and Nibert, M. L. (2008). A positive-feedback mechanism promotes reovirus particle conversion tothe intermediate associated with membrane penetration. Proc Natl Acad Sci U S A 105(30):10571-10576.
- Baer,G. S. andDermody, T. S. (1997). Mutationsin reovirus outer-capsid protein sigma3 selected during persistent infectionsof L cells confer resistance to protease inhibitor E64. J Virol 71(7): 4921-4928.
- Berard,A. and Coombs, K. M. (2009). Mammalianreoviruses: propagation, quantification, and storage. Curr Protoc Microbiol Chapter 15:Unit15C 11.
- Borsa,J., Copps, T. P., Sargent, M. D., Long, D. G. and Chapman, J. D. (1973a). Newintermediate subviral particles in the invitro uncoating of reovirus virions by chymotrypsin. J Virol 11(4): 552-564.
- Borsa,J., Morash, B. D., Sargent, M. D., Copps, T. P., Lievaart, P. A. and Szekely,J. G. (1979). Twomodes of entry of reovirus particles into L cells. J Gen Virol 45(1): 161-170.
- Borsa,J., Sargent, M. D., Lievaart, P. A. and Copps, T. P. (1981). Reovirus: evidence for a second step in the intracellular uncoating andtranscriptase activation process. Virology 111(1): 191-200.
- Borsa,J., Sargent, M. D., Long, D. G. and Chapman, J. D. (1973b). Extraordinaryeffects of specific monovalent cations on activation of reovirus transcriptaseby chymotrypsin in vitro. J Virol 11(2): 207-217.
- Chandran,K., Farsetta, D. L. and Nibert, M. L. (2002). Strategyfor nonenveloped virus entry: a hydrophobic conformer of the reovirus membranepenetration protein micro 1 mediates membrane disruption. J Virol 76(19): 9920-9933.
- Chandran,K., Walker, S. B., Chen, Y., Contreras, C. M., Schiff, L. A., Baker, T. S. andNibert, M. L. (1999). In vitro recoatingof reovirus cores with baculovirus-expressed outer-capsid proteins mu1 andsigma3. J Virol 73(5): 3941-3950.
- Chang,C. T. andZweerink, H. J. (1971). Fate of parental reovirus ininfected cell. Virology 46(3): 544-555.
- Dermody,T. S., Nibert, M. L., Wetzel, J. D., Tong, X. and Fields, B. N. (1993). Cells and viruses with mutations affecting viral entry are selected during persistent infections of Lcells with mammalian reoviruses. J Virol 67(4): 2055-2063.
- Dermody, T. S., Parker, J. S. L. and Sherry, B. (2013).Orthoreoviruses. In: Knipe, D. M. and Howley, P. M. (Eds.). Fields Virology 6th edn. LippincottWilliams & Wilkins: 1304-1346.
- Dryden,K. A., Wang, G., Yeager, M., Nibert, M. L., Coombs, K. M., Furlong, D. B.,Fields, B. N. and Baker, T. S. (1993). Earlysteps in reovirus infection are associatedwith dramatic changes in supramolecular structure and protein conformation:analysis of virions and subviral particles by cryoelectron microscopy and imagereconstruction. JCell Biol 122(5): 1023-1041.
- Ebert,D. H., Deussing, J., Peters, C. and Dermody, T. S. (2002). Cathepsin L and cathepsin B mediate reovirus disassembly in murinefibroblast cells. JBiol Chem 277(27): 24609-24617.
- Ehrlich,M., Boll, W., Van Oijen, A., Hariharan, R., Chandran, K., Nibert, M. L. andKirchhausen, T. (2004). Endocytosis by randominitiation and stabilization of clathrin-coated pits. Cell 118(5): 591-605.
- Joklik,W. K. (1972). Studies on the effect ofchymotrypsin on reovirions. Virology 49(3): 700-715.
- Kobayashi,T., Ooms, L. S., Ikizler, M., Chappell, J. D. and Dermody, T. S. (2010). An improved reverse genetics system for mammalian orthoreoviruses. Virology 398(2): 194-200.
- Maginnis,M. S., Forrest, J. C., Kopecky-Bromberg, S. A., Dickeson, S. K., Santoro, S.A., Zutter, M. M., Nemerow, G. R., Bergelson, J. M. and Dermody, T. S. (2006). β1 integrin mediates internalization of mammalian reovirus. J Virol 80(6): 2760-2770.
- Maginnis,M. S., Mainou, B. A., Derdowski, A., Johnson, E. M., Zent, R. and Dermody, T.S. (2008). NPXYmotifs in the beta1 integrin cytoplasmic tail arerequired for functional reovirus entry. J Virol 82(7): 3181-3191.
- Middleton,J. K., Severson, T. F., Chandran, K., Gillian, A. L., Yin, J. and Nibert, M. L.(2002). Thermostabilityof reovirus disassembly intermediates (ISVPs) correlates with genetic,biochemical, and thermodynamic properties of major surface protein mu1. J Virol 76(3): 1051-1061.
- Nibert,M. L. andFields, B. N. (1992). Acarboxy-terminal fragment of protein mu 1/mu 1C is present in infectious subvirion particles of mammalian reoviruses andis proposed to have a role in penetration. J Virol 66(11): 6408-6418.
- Sarkar,P. and Danthi, P. (2010). Determinants of strain-specificdifferences in efficiency of reovirus entry. J Virol 84(24): 12723-12732.
- Silverstein,S. C., Astell, C., Levin, D. H., Schonberg, M. and Acs, G. (1972). The mechanisms of reovirus uncoating and gene activation in vivo. Virology 47(3): 797-806.
- Smith,R. E., Zweerink, H. J. and Joklik, W. K. (1969). Polypeptidecomponents of virions, top component and cores of reovirus type 3. Virology 39(4): 791-810.
- Snyder,A. J. andDanthi, P. (2015). Lipidmembranes facilitate conformational changes required for reovirus cell entry. J Virol 90(5): 2628-2638.
- Snyder,A. J. andDanthi, P. (2016). Lipidscooperate with the reovirus membrane penetration peptide to facilitate particleuncoating. JBiol Chem 291(52): 26773-26785.
- Sturzenbecker,L. J., Nibert, M., Furlong, D. and Fields, B. N. (1987). Intracellular digestion of reovirus particles requires a low pH and isan essential step in the viral infectious cycle. J Virol 61(8): 2351-2361.
- Zhang,X., Ji, Y., Zhang, L., Harrison, S. C., Marinescu, D. C., Nibert, M. L. andBaker, T. S. (2005). Featuresof reovirus outer capsid protein mu1 revealed by electron cryomicroscopy andimage reconstruction of the virion at 7.0 Angstrom resolution. Structure 13(10): 1545-1557.
Article Information
Copyright
© 2018 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Snyder, A. J. and Danthi, P. (2018). Infectious Subviral Particle to Membrane Penetration Active Particle (ISVP-to-ISVP*) Conversion Assay for Mammalian Orthoreovirus. Bio-protocol 8(2): e2700. DOI: 10.21769/BioProtoc.2700.
Category
Microbiology > Microbe-host interactions > Virus
Biochemistry > Protein > Activity
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link










