- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
RNA Strand Displacement Assay for Hepatitis E Virus Helicase
Published: Vol 7, Iss 7, Apr 5, 2017 DOI: 10.21769/BioProtoc.2198 Views: 8748
Reviewed by: Yannick DebingVamseedhar RayaproluDavid Paul

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Mapping RNA Sequences that Contact Viral Capsid Proteins in Virions
C. Cheng Kao [...] Doug B. Rusch
Jul 20, 2017 8886 Views
Real-time Fluorescence Measurement of Enterovirus Uncoating
Visa Ruokolainen [...] Varpu Marjomäki
Apr 5, 2020 4653 Views

Viral Double-Stranded RNA Detection by DNase I and Nuclease S1 digestions in Leishmania parasites
Nathalie Isorce and Nicolas Fasel
May 5, 2020 5911 Views
Abstract
The hepatitis E virus (HEV) helicase uses ATP to unwind the RNA duplexes. This is an essential step for viral replication. This protocol aims to measure the double strand RNA unwinding activity of the HEV helicase.
Keywords: Hepatitis E virusBackground
The RNA unwinding activity of HEV helicase has been measured using radiolabeled double stranded RNA (dsRNA, Karpe et al., 2010). We have established a non-radioactive assay protocol for measuring the dsRNA unwinding activity of the HEV helicase. This assay utilizes a fluorescently tagged RNA to measure the activity of the HEV helicase protein purified from human hepatoma cell, thus eliminating the need to handle radioactive material.
Materials and Reagents
- 1.5 ml microcentrifuge tube
- 60 mm plates
- 1.6 ml RNase free microcentrifuge tube
- PVDF (Polyvinylidene fluoride) membrane (Pall, catalog number: BSP0149 )
- Mammalian expression plasmids (details in Nair et al., 2016):
- pCDNA5 Helicase-flag [HEV helicase sequence was PCR amplified using primers: pCDNA5 HEL-flag FP, pCDNA5 HEL-flag RP; digested with HindIII and ligated into pCDNA5 vector digested with HindIII and EcoRV]
- pUNO ORF2-flag [ORF2 with C-terminal Flag tag was PCR amplified with primers: pUNO ORF2-flag FP, pUNO ORF2-flag RP; digested with BglII and ligated into pUNO vector digested with NheI [blunted] and BamHI]
- Huh7 human hepatoma cells (obtained from Dr. C. M. Rice; Blight et al., 2000)
- RNA oligos: HRNA1 5’-UUUUUUUUUUUUCGCUGAUGUCGCCUGG-3’ and HRNA2 5’-[6FAM] CCAGGCGACAUCAGCG-3’ (Sigma-Aldrich, USA)
- Nuclease-free water (Thermo Fisher Scientific, AmbionTM, catalog number: AM9937 )
- Ethidium bromide solution (Sigma-Aldrich, catalog number: E1510 )
- Glycogen (Sigma-Aldrich, catalog number: G0885 )
- Ethanol (EMD Millipore, catalog number: 100983 )
- Lipofectamine 2000 (Thermo Fisher Scientific, InvitrogenTM, catalog number: 11668019 )
- DMEM
- 10% fetal bovine serum (FBS)
- Phosphate-buffered saline (PBS) (Bio Basic, catalog number: PD0100 )
- Flag M2 agarose resin (Sigma-Aldrich, catalog number: A2220 )
- Flag peptide (Sigma-Aldrich, catalog number: F4799 )
- DEPC-treated water (Thermo Fisher Scientific, AmbionTM, catalog number: AM9922 )
- Octa-probe antibody (Santa Cruz Biotechnology, catalog number: sc-807 )
- Skimmed milk (Sigma-Aldrich, catalog number: 70166 )
Note: This product has been discontinued. - Anti-rabbit IgG Horseradish peroxidase(HRPO) (Santa Cruz Biotechnology, catalog number: sc-2004 )
- Clarity Western ECL blotting substrate (Bio-Rad Laboratories, catalog number: 1705061 )
- Silver stain kit (Thermo Fisher Scientific, Thermo ScientificTM, catalog number: 24612 )
- Bradford assay reagent (Bio-Rad Laboratories, catalog number: 5000002 )
- RNAsin
- ATP
- Proteinase K (Sigma-Aldrich, catalog number: P2308 )
- Glycogen (Sigma-Aldrich, catalog number: G0885 )
- Tris (Sigma-Aldrich, catalog number: T6066 )
- Sodium chloride (NaCl) (Sigma-Aldrich, catalog number: S7653 )
- Ethylenediaminetetraacetic acid (EDTA) (Sigma-Aldrich, catalog number: E6758 )
- Ammonium acetate (Sigma-Aldrich, catalog number: A1542 )
- SDS (Sigma-Aldrich, catalog number: L3771 )
- Ethylene glycol-bis(2-aminoethylether)-N,N,N’,N’,-tetraacetic acid (EGTA) (Sigma-Aldrich, catalog number: 03777 )
- Triton X-100 (Sigma-Aldrich, catalog number: 93418 )
Note: This product has been discontinued. - Sodium pyrophosphate tetrabasic decahydrate (Na4P2O4·10H2O) (Sigma-Aldrich, catalog number: S6422 )
- β-glycerol phosphate (Sigma-Aldrich, catalog number: 50020 )
- Sodium orthovanadate (Na3VO4) (Sigma-Aldrich, catalog number: S6508 )
- Protease inhibitor cocktail tablet (Roche Diagnostics, catalog number: 04693132001 )
- HEPES (Sigma-Aldrich, catalog number: H8651 )
- Magnesium chloride (MgCl2) (Sigma-Aldrich, catalog number: M8266 )
- Potassium chloride (KCl) (Sigma-Aldrich, catalog number: P9333 )
- BSA
- DL-dithiothreitol (DTT) (Sigma-Aldrich, catalog number: D0632 )
- Tween 20
- Bromophenol blue (Sigma-Aldrich, catalog number: B0126 )
- Xylene cyanol FF (Sigma-Aldrich, catalog number: X4126 )
- Annealing buffer (see Recipes)
- Soaking buffer (see Recipes)
- IP buffer (see Recipes)
- Strand displacement buffer (see Recipes)
- 5x termination buffer (see Recipes)
- 2x proteinase K buffer (see Recipes)
- Phosphate buffered saline with 0.1% Tween 20 (PBST) (see Recipes)
- 50 mg/ml proteinase K (see Recipes)
- 10 mg/ml glycogen (see Recipes)
- RNA loading dye (see Recipes)
Equipment
- Vertical electrophoresis mini apparatus
- Gel documentation system (Bio-Rad Laboratories, model: ChemiDocTM MP )
- NanoDrop (Thermo Fischer Scientific, USA)
- Heat block
- Water bath
- Refrigerated table top centrifuge
- -80 °C deep freezer
- Flip flop rocker
- 5% CO2 incubator
Procedure
- Preparation of fluorescently labeled dsRNA
- All steps should be performed using nuclease free reagents.
- Resuspend the RNA oligos to 100 pmol/μl in nuclease free water. Resolve 1 μl on 20% non-denaturing PAGE (Polyacrylamide gel electrophoresis), stain with ethidium bromide (2 μl of 10 mg/ml stock in 50 ml running buffer) for 15 min at room temperature and visualize the signal using gel documentation system under UV set.
Note: This step is necessary to rule out occasional degradation of the oligo from the source itself. - Mix 100 pmole (100 pmole/μl stock) of each oligo in 40 μl annealing buffer, incubate at 95 °C for 30 sec and cool gradually to room temperature.
- Resolve the annealed oligos in 20% non-denaturing PAGE and excise the band corresponding to the RNA duplex.
- Transfer the excised bands to a fresh 1.5 ml microcentrifuge tube, mince and soak in 500 µl soaking buffer overnight (16 h) in a flip flop rocker at room temperature.
- Next day, centrifuge the tubes at 13,000 x g for 5 min at room temperature.
- Collect the supernatant into a fresh microcentrifuge tube, add 5 μg glycogen (5 mg/ml stock in water) and 1 ml ice cold absolute ethanol and incubate at -80 °C for 45 min.
- Centrifuge at 14,000 x g for 30 min at 4 °C.
- Wash the pellet with 500 μl 75% ethanol, centrifuge at 14,000 x g for 10 min at 4 °C.
- Remove 75% ethanol and dry the pellet at room temperature.
Note: Be careful while removing the 75% ethanol, pellet may be lost. - Dissolve the RNA pellet in 30 µl nuclease-free water and incubate at 55 °C for 10 min.
- Run 1 μl dsRNA in 20% non-denaturing PAGE. Visualize the fluorescent RNA using the gel documentation system using appropriate setting (6-FAM fluorescent dye setting, excitation: 492 nm, emission: 518 nm).
- Quantitate the dsRNA by spectrophotometry using NanoDrop and store in aliquots in RNase free microcentrifuge tubes at -80 °C.
- All steps should be performed using nuclease free reagents.
- Preparation of flag-affinity purified proteins
- Transfect 3 μg each of pUNO RdRp-flag and pUNO ORF2-flag plasmids into ten 60 mm plates (each plasmid transfected into 10 plates, not cotransfected), containing Huh7 human hepatoma cells, using Lipofectamine 2000 in 1:1 ratio, following manufacturer’s instructions. Incubate cells at 37 °C in 5% CO2 incubator. 24 h post-transfection, replace media with 2 ml DMEM supplemented with 10% fetal bovine serum.
- Forty eight hours post transfection, wash the cells with 1x PBS and resuspend in 800 μl IP buffer.
- Generate a homogenous suspension by repeated pipetting and vortexing, and incubate overnight (16 h) on ice at 4 °C (cold room or fridge).
- Next day, clarify the lysate by centrifugation at 13,000 x g, 4 °C, 10 min and collect the supernatant into a fresh 1.6 ml RNase free microcentrifuge tube.
- Add 100 μl of flag agarose beads, incubate on a rocker at 4 °C for 4 h, wash 3 x in 1 ml IP buffer each by centrifuging at 800 x g, 1 min, at 4 °C. Add 200 μl flag peptide (0.2 mg/ml in PBS) and incubate on a rocker for 15 min at 4 °C. Centrifuge at 800 x g, 1 min, at 4 °C and collect the supernatant, which contains the eluted protein.
- Check an aliquot of the purified protein by Western blotting using octa-probe antibody and silver staining, as mentioned below.
- Resolve the proteins by 10% SDS-PAGE and transfer onto a PVDF (Polyvinylidene fluoride) membrane.
- Block the membrane using 5% skimmed milk in 1x PBS for 45 min at room temperature, incubate overnight (16 h) with 1:1,000 octa-probe primary antibody diluted using 5% skimmed milk in 1x PBST at 4 °C, wash 3 x in PBST, incubate with 1:5,000 anti-rabbit IgG HRPO secondary antibody diluted using 5% skimmed milk in 1x PBST at room temperature, followed by detection of the signal by enhanced chemiluminescence using ‘clarity Western ECL blotting substrate’.
- Acquire the Images and analyze using a gel documentation system. A single band of approximately 27 kDa should be obtained, as illustrated in Nair et al. (2016).
- For silver staining, resolve the proteins in 10% SDS-PAGE.
- Proceed for silver staining using silver stain kit, following the manufacturer’s protocol (Thermo Fisher Scientific, USA). A major band of approximately 27 kDa (corresponding to the size of the band obtained in Western) should be obtained, as illustrated in Nair et al. (2016). Few additional bands are also detected above 27 kDa, which does not affect the efficiency of the assay in our experience. If expected bands are obtained in Western and silver staining, estimate the respective protein concentration by serial dilutions by Bradford assay and store the protein in single use aliquots at -80 °C.
- Transfect 3 μg each of pUNO RdRp-flag and pUNO ORF2-flag plasmids into ten 60 mm plates (each plasmid transfected into 10 plates, not cotransfected), containing Huh7 human hepatoma cells, using Lipofectamine 2000 in 1:1 ratio, following manufacturer’s instructions. Incubate cells at 37 °C in 5% CO2 incubator. 24 h post-transfection, replace media with 2 ml DMEM supplemented with 10% fetal bovine serum.
- RNA strand displacement assay
- Incubate 1 pmol dsRNA and 2 µg flag affinity purified HEV helicase protein or ORF2 protein in strand displacement buffer at 37 °C for 2 h. Following order may be followed.
Water 24 µl 10x buffer 4 µl RNAsin 1 µl ATP (10 mM) 4 µl Protein 5 µl dsRNA 2 µl Total volume 40 µl - Use appropriate negative controls such as dsRNA without protein or dsRNA with an unrelated protein (ORF2).
- Terminate the reaction by adding 10 µl 5x termination buffer.
- Add 50 µl 2x proteinase K buffer and 1 µl proteinase K (50 mg/ml stock solution in water) and incubate for 10 min at 37 °C.
- Add 1 µl glycogen (10 mg/ml stock solution in water) and 1 ml of ice-cold absolute ethanol to the samples; incubate at -80 °C for 45 min.
- Centrifuge the samples at 14,000 x g, 15 min, 4 °C.
- Wash the pellet with 1 ml 75% ethanol and resuspend in 10 µl nuclease free water.
Note: Be careful while removing the 75% ethanol, pellet may be lost. Although not necessarily a better alternative, it may be possible to minimize pellet loss by gently pipetting out 75% ethanol instead of directly decanting. Irrespective of whatever handling technique preferred, attention should be given not to disturb the pellet. - Heat denature an equal amount of dsRNA containing sample at 95 °C for 10 min.
- Mix the samples with 10 µl RNA loading dye and resolve in 20% non-denaturing PAGE to differentiate the migration between single strand (ss) RNA and dsRNA.
- Visualize the fluorescent bands using a gel documentation system.
- Incubate 1 pmol dsRNA and 2 µg flag affinity purified HEV helicase protein or ORF2 protein in strand displacement buffer at 37 °C for 2 h. Following order may be followed.
Data analysis
While performing the assay, appropriate controls should be included in order to interpret the data and rule out non-specific signals. Negative controls should include omission of helicase protein and nucleotides from the reaction mixture. A titration experiment using increasing quantities of helicase protein in the assay should also be performed to rule out non-specific signals. An assay using an unrelated protein instead of helicase will also rule out a possible non-specific signal. A sample assay has been illustrated in Figure 1. The effect of other factors or compounds on helicase activity may be evaluated by adding them to the reaction mixture.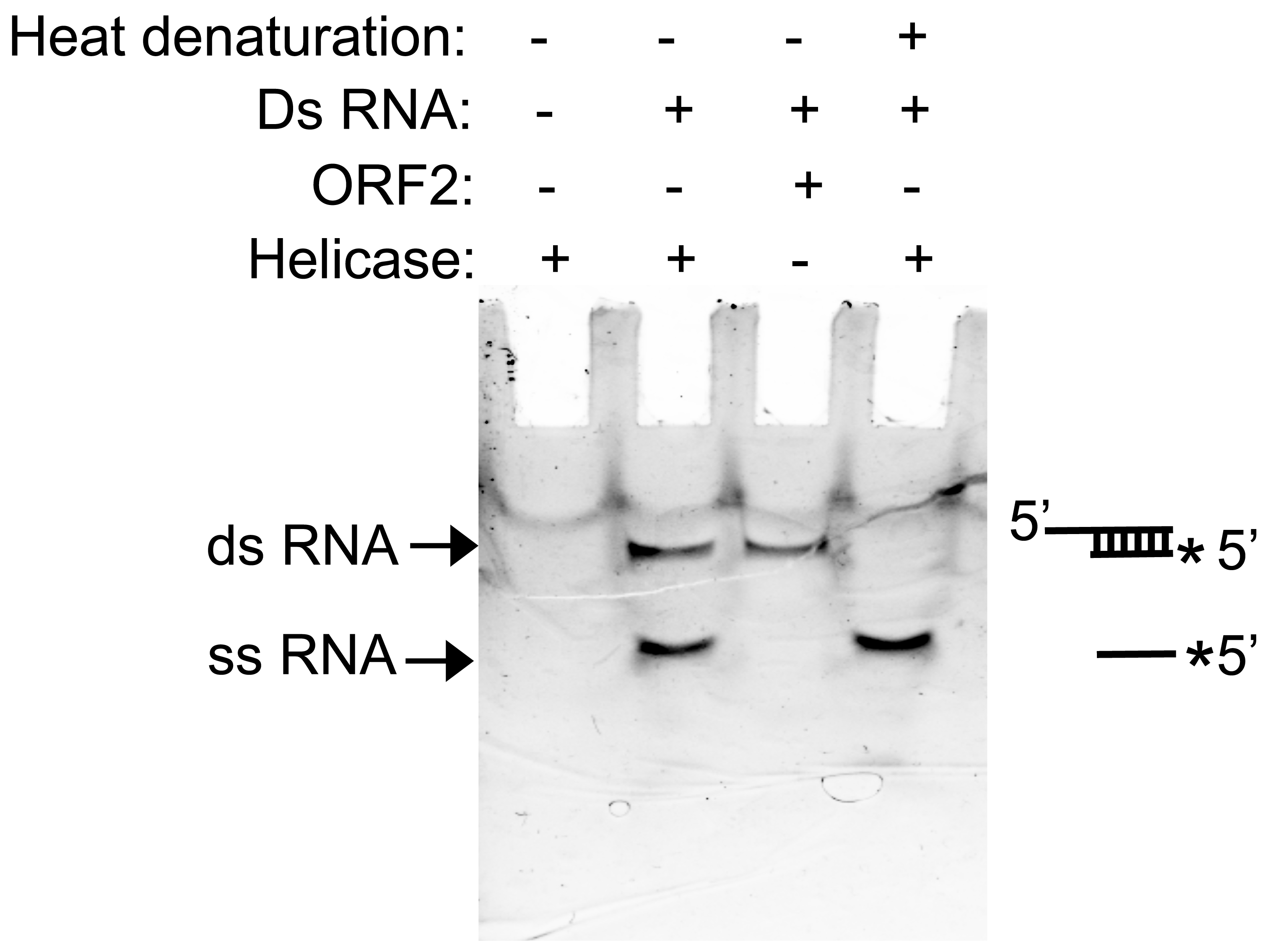
Figure 1. Assay of RNA strand displacement by helicase. dsRNA: double strand RNA, ssRNA: single strand RNA, Heat denaturation: the sample was incubated at 95 °C for 10 min before loading into the gel.
Recipes
- Annealing buffer
10 mM Tris-Cl (pH 7.4)
100 mM NaCl
1 mM EDTA - Soaking buffer
0.5 M ammonium acetate
10 mM EDTA
1% SDS - IP buffer
20 mM Tris-Cl (pH 7.4)
150 mM NaCl
1 mM EDTA (pH 8.0)
1 mM EGTA (pH 8.0)
1% Triton X-100
2.5 mM sodium pyrophosphate
1 mM β-glycerol phosphate
1 mM sodium orthovanadate
1x protease inhibitor cocktail (25x stock prepared by dissolving one tablet in 2 ml 1x PBS) - Strand displacement buffer
50 mM HEPES (pH 7)
2 mM MgCl2
10 mM KCl
0.05 mg BSA/ml
2 mM DTT
1 mM ATP - 5x termination buffer
1% SDS
100 mM EDTA - 2x proteinase K buffer
300 mM NaCl
100 mM Tris-Cl (pH 7.5)
1% SDS - PBST
1x phosphate-buffered saline
0.1% Tween 20 - 50 mg/ml proteinase K
50 mg/ml stock solution prepared in nuclease free water - 10 mg/ml glycogen
10 mg/ml stock solution prepared in nuclease free water - RNA loading dye
0.5 mM EDTA
0.025% bromophenol blue
0.025% xylene cyanol FF
Acknowledgments
The work was funded by Ramalingaswamy fellowship and THSTI core grant to MS. VN is supported by a grant from the Department of Science and Technology, Government of India. The protocol has been adapted from Karpe et al. (2010).
References
- Blight, K. J., Kolykhalov, A. A. and Rice, C. M. (2000). Efficient initiation of HCV RNA replication in cell culture. Science 290(5498): 1972-1974.
- Karpe, Y. A. and Lole, S. K. (2010). NTPase and 5’ to 3’ RNA duplex-unwinding activities of the hepatitis E virus helicase domain. J Virol 84(7): 3595-602.
- Nair, V. P., Anang, S., Subramani, C., Madhvi, A., Bakshi, K., Srivastava, A., Shalimar, Nayak, B., Ct, R. K. and Surjit, M. (2016). Endoplasmic reticulum stress induced synthesis of a novel viral factor mediates efficient replication of genotype-1 hepatitis E virus. PLoS Pathog 12(4): e1005521.
Article Information
Copyright
© 2017 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Readers should cite both the Bio-protocol article and the original research article where this protocol was used:
- Nair, V. P. and Surjit, M. (2017). RNA Strand Displacement Assay for Hepatitis E Virus Helicase. Bio-protocol 7(7): e2198. DOI: 10.21769/BioProtoc.2198.
- Nair, V. P., Anang, S., Subramani, C., Madhvi, A., Bakshi, K., Srivastava, A., Shalimar, Nayak, B., Ct, R. K. and Surjit, M. (2016). Endoplasmic reticulum stress induced synthesis of a novel viral factor mediates efficient replication of genotype-1 hepatitis E virus. PLoS Pathog 12(4): e1005521.
Category
Microbiology > Microbial biochemistry > RNA
Biochemistry > RNA > RNA-protein interaction
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link










