- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Cryo-focused Ion Beam Sample Preparation for Imaging Vitreous Cells by Cryo-electron Tomography
Published: Vol 5, Iss 17, Sep 5, 2015 DOI: 10.21769/BioProtoc.1575 Views: 23166
Reviewed by: Tie LiuPengpeng LiAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Targeting Ultrastructural Events at the Graft Interface of Arabidopsis thaliana by A Correlative Light Electron Microscopy Approach
Clément Chambaud [...] Lysiane Brocard
Jan 20, 2023 3001 Views
Fast and High-Resolution Imaging of Pollinated Stigmatic Cells by Tabletop Scanning Electron Microscopy
Lucie Riglet and Isabelle Fobis-Loisy
Nov 20, 2024 1811 Views
Cryo-SEM Investigation of Chlorella Using Filter Paper as Substrate
Peng Wan [...] Jinghan Wang
Dec 20, 2024 1475 Views
Abstract
Cryo-electron tomography (CET) is a well-established technique for imaging cellular and molecular structures at sub-nanometer resolution. As the method is limited to samples that are thinner than 500 nm, suitable sample preparation is required to attain CET data from larger cell volumes. Recently, cryo-focused ion beam (cryo-FIB) milling of plunge-frozen biological material has been shown to reproducibly yield large, homogeneously thin, distortion-free vitreous cross-sections for state-of-the-art CET. All eukaryotic and prokaryotic cells that can be plunge-frozen can be thinned with the cryo-FIB technique. Together with advances in low-dose microscopy, this has shifted the frontiers of in situ structural biology. In this protocol we describe the typical steps of the cryo-FIB technique, starting with fully grown cell cultures. Three recently investigated biological samples are given as examples.
Keywords: Focused ion beamMaterials and Reagents
- Biological material
Note: The FIB-milling procedure is compatible with a wide range of cell types, including single-celled eukaryotes, prokaryotes, and mammalian cells such as HeLa and neuronal cultures. The following examples are used in this protocol:- A Chlamydomonas reinhardtii (C. reinhardtii) wild-type strain CC-124 (137c) (Chlamydomonas Resource Center, University of Minnesota, Minneapolis, MN)
- B Chlamydomonas reinhardtii strain mat3-4 (Umen and Goodenough, 2001)
- C Yeast strain Saccharomyces cerevisiae (S. cerevisiae)
- A Chlamydomonas reinhardtii (C. reinhardtii) wild-type strain CC-124 (137c) (Chlamydomonas Resource Center, University of Minnesota, Minneapolis, MN)
- Tris-acetate-phosphate (TAP) medium (Harris et al., 2009; Culture media recipe is available at http://www.chlamy.org/media.html)
- Lugol’s iodine solution (Sigma-Aldrich, catalog number: 62650-100ML-F )
- Liquid nitrogen
- Ethane/propane high purity gas mixture (37% ethane) (Linde)
- Filter paper
- YPD liquid medium (see Recipes)
Equipment
- R 2/1 Holey Carbon-coated 200-mesh copper grids (Quantifoil Micro Tools GmbH, catalog number: Q2100CR1 )
- Tweezers
- Screwdrivers
- Light microscope
- Teflon sheets (custom made, cut from larger sheets in the exact shape and size as the filter paper used for plunge-freezing)
- Glass petri dish (9-12 cm)
- Glass microscopy slides (Thermo Fisher Scientific, catalog number: 10144633B )
- Cryo grid boxes (custom made, or FEI, Eindhoven, The Netherlands)
- Cryo AutoGrid boxes (custom made, or FEI, Eindhoven, The Netherlands)
- AutoGrids (custom made and modified for FIB work, see Figure 1a)
- Clip rings (FEI, Eindhoven, The Netherlands, see Figure 1b)
- Liquid nitrogen Dewars
- Personal protection equipment for work with liquid nitrogen (safety glasses, face shield, and cold-resistant gloves)
- Hemocytometer (Neubauer)
- Pipets and tips
- Clipping tools (custom made, or FEI, Eindhoven, The Netherlands)
- Personal protective equipment for work with liquid nitrogen and liquid ethane/propane
- Plasma cleaner (Harrick, model: PDC-3XG )
- Vitrobot Mark 4 (FEI)
- Focused ion beam microscope (FEI, model: DB Quanta 3D FEG ), Cryo-system (Quorum Technologies, model: PP3000T ), Cryo-stage (custom made, Max Planck Institute)
- Transmission electron microscope I. (FEI, model: Tecnai G2 Polara , FEG 300kV), Post-column energy-filter (Gatan, model: HR-GIF 2002 ), Direct detection camera (Gatan, model: K2 Summit ), SerialEM software (Mastronarde, 2005)
- Transmission electron microscope II. (FEI, model: Titan Krios, FEG 300kV), post-column energy-filter (Gatan, model: 968 Quantum K2 ) Direct detection camera (Gatan, model: K2 Summit), SerialEM software (Mastronarde, 2005)
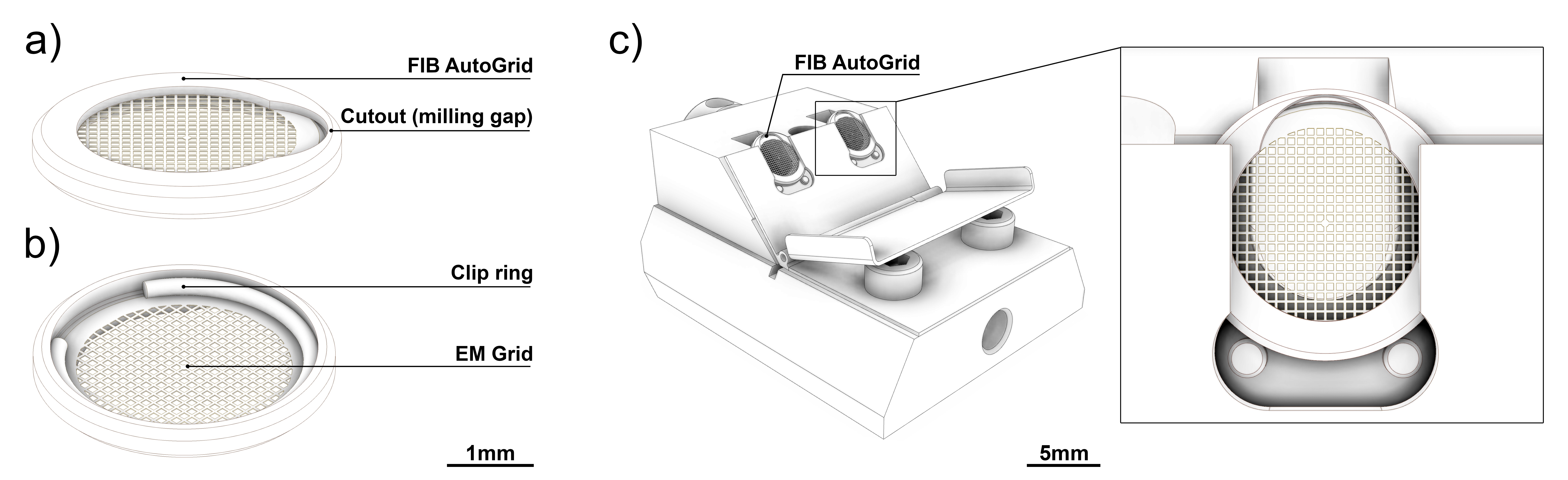
Figure 1. AutoGrids modified for cryo-FIB. a) Top and b) bottom side of an AutoGrid holding a clipped TEM grid. The flat top side shows the cutout required for cryo-FIB milling. c) The FIB shuttle loaded with two AutoGrids. Detailed view showing a properly oriented AutoGrid loaded into the shuttle. Note that the AutoGrid cutout faces up.
Software
- FIB software
Procedure
Note: Working with liquid nitrogen and liquid ethane/propane mixture is potentially dangerous. Personal protective equipment should be used.
- Vitrification by plunge-freezing
Note: Standard protocols can be used for plunge-freezing cells (e.g. Iancu et al., 2006). For proper vitrification (freezing in non-crystalline ice) and cell density, parameters must be optimized for each specific cell type.- Cell concentration is determined by counting using a hemocytometer. (C. reinhardtii cells are fixed with Lugol’s iodine solution for the counting process in order to stop cell motility. A Lugol’s solution to cell culture of ratio 1:10 is sufficient.)
- The concentration of the cells is diluted to 200-750 cells/μl in fresh TAP for the C. reinhardtii wild-type strain, 500-4,000 cells/μl in fresh TAP for the C. reinhardtii mat3-4 strain, and 10,000-15,000 cells/μl for S. cerevisiae cells in YPD medium.
- The carbon-coated 200-mesh copper TEM grids placed carbon side up on a glass slide are glow discharged by 30 sec plasma cleaning. The slide is then stored in a glass petri dish until plunging, which should proceed immediately.
- The cells are plunge-frozen using the Vitrobot:
- The Vitrobot is set to 90% humidity, blot force 10, 7 to 10 sec blot time, room temperature (C. reinhardtii cells) or 27 °C (S. cerevisiae cells).
- 3.5 μl of the diluted cell culture is pipetted onto TEM grids inside the Vitrobot.
- The grids are blotted from the reverse side using Teflon sheets on both sides and filter paper on the backside. Then they are immediately plunged into the liquid ethane/propane mixture at liquid nitrogen temperature.
- The plunge-frozen grids are then stored in sealed boxes under liquid nitrogen until used.
- The Vitrobot is set to 90% humidity, blot force 10, 7 to 10 sec blot time, room temperature (C. reinhardtii cells) or 27 °C (S. cerevisiae cells).
- Cell concentration is determined by counting using a hemocytometer. (C. reinhardtii cells are fixed with Lugol’s iodine solution for the counting process in order to stop cell motility. A Lugol’s solution to cell culture of ratio 1:10 is sufficient.)
- Sample thinning using cryo-FIB milling
- Cooling the FIB system from room temperature to cryo conditions.
- Before starting the cooling, the vacuum in the FIB chamber and in the prep-chamber must be lower than 4 x 10-6 mbar to avoid contamination from water vapor.
- The N2 gas pipeline between the liquid nitrogen Dewar and the FIB chamber (vacuum isolated line) is pumped to 2 x 10-2 mbar.
- The nitrogen gas flow for the stage and anti-contaminator are adjusted to 3.8 L/min and 4.2 L/min, respectively. These values ensure that temperatures of 182 °C and 192 °C are reached after cooling, respectively.
- The cooling is started by slowly inserting the cooling rod into the liquid nitrogen Dewar.
- Before starting the cooling, the vacuum in the FIB chamber and in the prep-chamber must be lower than 4 x 10-6 mbar to avoid contamination from water vapor.
- Sample clipping and loading the AutoGrids into the FIB shuttle.
Note: Clipping and loading is started when the cryo-stage and anti-contaminator reach temperatures below -160 °C. Using a surgical mask can minimize water vapor contamination of the work area.- The shuttle and the clipping support base are placed into the loading box (Figure 2).
- The loading box is filled with liquid nitrogen.
- After boiling has finished, the double wall tank is filled with liquid nitrogen by slightly tilting the loading box (Figure 2).
- The sample boxes from the storage Dewar are transferred into the loading box and opened there. Transfer time should be minimized to avoid contamination of the sample.
- All tools that will contact the TEM grids are pre-cooled by liquid nitrogen in the loading box immediately before use.
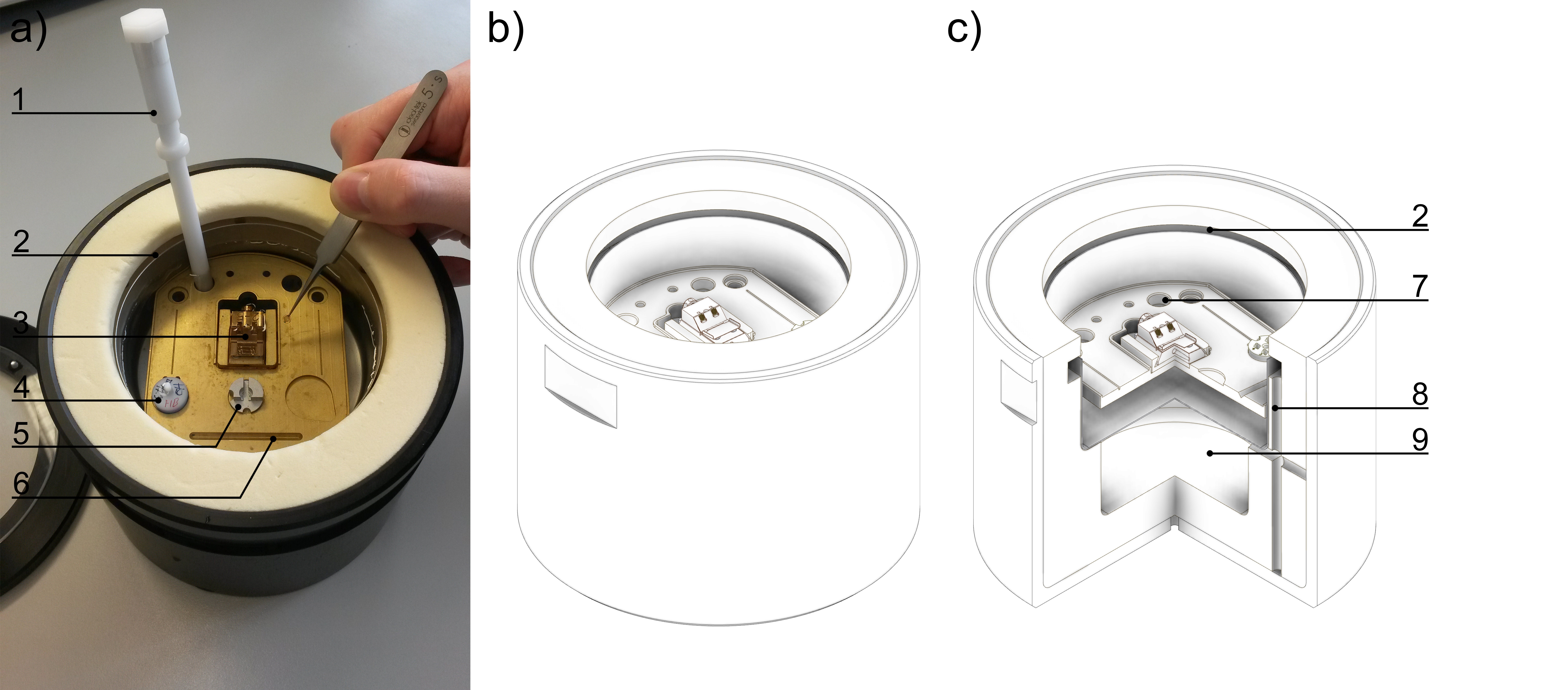
Figure 2. Sample clipping and loading. a-c) The sample loading box with double-walled reservoir (9). The second reservoir is filled with liquid nitrogen through the filling gaps (2, 8). The AutoGrid notch (6) aids the fast and easy handling of Autogrids under liquid nitrogen. The clipping tool (1) and tweezers can be cooled in the round bore (7). The shuttle (3) (in loading position), sample storage box (4) and clipping metal support (5) are immersed under liquid nitrogen.- The AutoGrid is placed into the clipping metal support with the flat side facing down. It must be centered properly. The TEM grid is removed from the sample box and placed into the AutoGrid with cells facing down. The grid is then clipped into the AutoGrid with a clip ring using the clipping tool (Figure 1b).
- As the shuttle has space for two AutoGrids, step B2f) is repeated for the second AutoGrid.
- The AutoGrids are loaded into the shuttle and oriented such that the cutouts are facing up (Figure 1c). The shuttle is then flipped back into the transfer position.
- The shuttle and the clipping support base are placed into the loading box (Figure 2).
- Shuttle transfer onto the FIB cryo-stage.
- The temperatures of the cryo-stage and anti-contaminator should be between -194 °C and -165 °C. The temperature of the anti-contaminator should be lower than the temperature of the cryo-stage.
- The stage is moved to the transfer position.
- The loading box (Figure 3c) is placed onto the pumping station (Figure 3a) and covered.
- The transfer unit (Figure 3b) is placed onto the cover of the loading box, and the transfer unit’s insertion rod is attached to the shuttle.
- The pumping of the transfer unit and loading box is started. After one minute, the shuttle is lifted out of the loading box into the transfer unit. The transfer unit valve is closed, the pumping is stopped and the loading station is vented (Figure 3d).

Figure 3. Sample transfer system. a) The Quorum cryo-system with the transfer unit (1). The pumping station (2) with the loading box inside. b) The sample transfer unit with the small transfer chamber (3) and the shuttle (4) attached to the transfer rod. c) The loading box (5) inside the pumping station (7). Here, the shuttle is in the transfer position (6). d) The transfer unit attached to the loading box for shuttle pick-up/drop-off. The small transfer chamber (3) is closed/opened using the transfer unit valve (8).- The transfer unit is attached to the airlock of the FIB (“prep chamber” connected to the turbo pump of the Quorum system).
- The airlock is pumped to the vacuum level required to safely open the airlock valves and the transfer unit valve.
- The shuttle is transferred onto the cryo-stage with the insertion rod.
- The insertion rod is detached from the shuttle and retracted, then the airlock valves and the transfer unit valve are closed. The airlock is vented and the transfer unit is removed.
- The temperatures of the cryo-stage and anti-contaminator should be between -194 °C and -165 °C. The temperature of the anti-contaminator should be lower than the temperature of the cryo-stage.
- Lamella milling.
Note: See Video 1 for an overview of the milling procedure. For help with basic FIB operation, press the F1 key in the FIB software.- Before starting the milling, the vacuum in the FIB chamber at cryo condition must be lower than 2 x 10-6 mbar to avoid contamination from water vapor.
- The FIB user interface of the FIB microscope is active.
- The column valves are opened and ion beam emission is started (“Beam on” or “Wake up” buttons are pressed).
- Electron and ion beams are in the operational state.
Note: The following parameters enable rapid milling of the bulk cellular material, followed by fine milling and polishing of the lamella. - The scanning parameters for the electron beam are set to: 5.0 kV beam energy, 12 pA beam current, 1,024 x, 884 or 1,536 x 1,024 scan resolution, 1 µs dwell time. Scanning is started.
- Scan rotation for both beams is set to 180 degrees.
- Low magnification (70x) is set and the stage is moved to one of the two AutoGrids.
- The grid bar is scanned at increased zoom to adjust focus, astigmatism and other required alignments for the electron beam.
- The stage is linked to a focused working distance (“Link Z to FWD” button).
- The stage is moved to the eucentric height (coincidence point) of the microscope (ion and electron beams display the same location).
- The stage rotation is adjusted to align the AutoGrid cutout with the incident direction of the ion beam. This is achieved by facing the cutout towards the bottom of the electron image (Figure 4a).
- Organometallic platinum deposition is performed on both AutoGrids using the in situ gas injection system to protect the lamella surface and reduce curtaining effects (protocol adapted from Hayles et al., 2007):
- Stage parameters are set to 0° tilt and 3 mm below eucentric height.
- The stage is adjusted such that the AutoGrid faces the gas injection system (GIS). For this, a relative stage rotation of 180° is performed using the compucentric rotation. The AutoGrid is then centered at the GIS position in the electron image (Figure 4b).
- The GIS temperature is set to 26 °C.
- The GIS needle is inserted and opened for 4 to 8 sec.
- The GIS needle is retracted.
- The stage is moved back to the initial position where it was before the procedure.
- Stage parameters are set to 0° tilt and 3 mm below eucentric height.

Figure 4. Lamella milling. a). Scanning electron microscope (SEM) image of an AutoGrid aligned with the incident direction of the ion beam. b). The AutoGrid is centered at the GIS position in the SEM image and the GIS needle is inserted. c). SEM image of a TEM grid with Chlamydomonas cells. d). SEM image of two target Chlamydomonas cells before FIB milling. e). FIB-induced secondary electron (FIB SE) image of the two target cells with two rectangle standard milling patterns as a starting point for the milling procedure. f). FIB SE image showing the milling step at 50pA where patterns on both sides are made slimmer (but pattern width remains constant) and shifted closer to the lamella edge. g, i). FIB SE and h) SEM images of the finished lamella after cryo-FIB milling. See Video 1 for an overview of cryo-FIB milling.Video 1. Cryo-FIB milling of a vitreous Chlamydomonas cell- The stage is tilted to the milling position with the ion beam almost parallel (typically 6 to 12°) to the grid surface. For a shuttle that is 7° pre-tilted, and a desired milling angle of 10° with respect to TEM grid surface, the stage is tilted to 17°.
- The ion beam parameters are set to: 30.0 kV beam energy, 10 pA beam current, 1,024 x 884 or 1,536 x 1,024 scan resolution, 1 µs dwell time.
- The focus and astigmatism of the ion beam are adjusted while scanning the grid bar at increased zoom.
- A target cell is localized in both electron and ion beam images (Figure 4c-e).
Note: The three meshes closest to the edge of the TEM grid are often not accessible in the TEM microscope. Generally, central meshes are preferred. - The eucentric height is adjusted using stage movement in the z-direction so that the ion beam and the electron beam show the same sample surface.
Note: The eucentric height must be separately adjusted for each milling position. - Two parallel rectangular standard milling patterns are drawn (Figure 4e). With pre-tilt correction disabled, the size of the pattern is approximately 10 x 6 µm and the distance between the patterns is more than 5 µm. The milling direction is set to top-to-bottom for one pattern and to bottom-to-top for the other, so that the milling direction of the pattern is always towards the lamella edge. The milling parameters are set to: Ice material (or Si), 1 µs dwell time, 60% overlap.
- The ion beam current is changed to 0.3 nA, and a fast single scan is taken (50 ns dwell time) to verify the proper milling position.
- The milling process is started. The milling is stopped as soon as the material is completely removed, as observed by live imaging of the scanned area.
- Iteratively, the beam current is reduced, pattern heights on both sides are made slimmer (while keeping pattern widths constant) and patterns are shifted closer to the lamella edge (Figure 4f and Video 1). Typical steps for the beam current are 0.1 nA, 50 pA and then 30 pA.
- At a sample thickness of about 500 nm, the final milling is performed using 30 pA and optionally the cleaning-cross-section pattern. The target lamella thickness is between 300 and 100 nm (Figure 4g). During this step, thickness is estimated using electron images (Figure 4h-i).
- It is possible to prepare multiple lamellas (~10) during one FIB session. For each new lamella, first set the ion beam current back to 10 pA and then repeat steps B4p-v).
Note: To minimize time-dependent ice contamination, it is recommended to perform steps B4p-U at all sample locations first, then afterwards perform step B4v for each lamella.
- Before starting the milling, the vacuum in the FIB chamber at cryo condition must be lower than 2 x 10-6 mbar to avoid contamination from water vapor.
- Shuttle transfer out of the FIB
- The column valves are closed (“Beam on\off” buttons, or “Sleep” button to end the FIB session).
- The stage is moved to the transfer position.
- The loading box is filled with liquid nitrogen and then placed onto the pumping station.
- The transfer unit is docked to the airlock of the FIB.
- The airlock is pumped to the vacuum level required to safely open the airlock valves. The airlock valves and the transfer unit valve are then opened.
- The shuttle is retrieved from the cryo-stage with the insertion rod and retracted out of the FIB chamber into the transfer unit.
- The airlock valves and the transfer unit valve are closed.
- The airlock is vented.
- The transfer unit is attached to the loading box.
- The loading station is pumped for 50 sec. Then the transfer unit valve is opened, and the shuttle is inserted into the loading box with the liquid nitrogen. The pumping is stopped and the loading station is vented.
- The insertion rod is detached from the shuttle and the transfer unit is removed.
- AutoGrids are removed from the shuttle and stored under liquid nitrogen in storage boxes suitable for AutoGrids.
- The column valves are closed (“Beam on\off” buttons, or “Sleep” button to end the FIB session).
- Warming the FIB system
- The cooling rod is taken out of the liquid nitrogen Dewar.
- When the cryo-stage and anti-contaminator have reached a temperature of about 20 °C, the gas flow is decreased to 0.5 L/min.
- The cooling rod is taken out of the liquid nitrogen Dewar.
- Cooling the FIB system from room temperature to cryo conditions.
- Cryo-ET
- The AutoGrid sample is loaded into the cryo-TEM microscope under cryo conditions using the standard procedure (see FEI user manual). When loading the AutoGrid, the AutoGrid cutout (and thus the milling direction of the lamellas) must be aligned perpendicular to the tilt axis of the stage.
- The lamella positions are localized using low magnification imaging.
- Tilt-series acquisition is performed using SerialEM software under low-dose conditions (<100 e/Å2 cumulative dose). K2 images are recorded at 2° tilt increments, with −4 μm to −8 μm defocus, typically with pixel sizes of 3 to 5 Å.
- The AutoGrid sample is loaded into the cryo-TEM microscope under cryo conditions using the standard procedure (see FEI user manual). When loading the AutoGrid, the AutoGrid cutout (and thus the milling direction of the lamellas) must be aligned perpendicular to the tilt axis of the stage.
Representative data
Figure 5 shows examples of final cryo-FIB-prepared lamellas of Chlamydomonas and yeast cells. Samples of this quality are reproducibly produced by the described technique (Rigort et al., 2012) and have been used for data collection published elsewhere (Engel et al., 2014; Villa et al., 2013).
Figure 5. TEM images of final lamellas. a) Cross-section of a Chlamydomonas cell, with a lamella thickness of 100 nm. b) Cross-section through several yeast cells, with a lamella thickness of 250 nm.
Recipes
- YPD liquid medium
1% yeast extract
2% peptone
2% D-glucose
Sterilize by autoclaving
Acknowledgments
We thank Alexander Rigort and Felix Bäuerlein for their contributions to developing the cryo-FIB technique (Rigort et al., 2012), and Elizabeth Villa for fruitful scientific discussions. This work was supported by the European Commission's 7th Framework Programme grant agreements ERC-2012-SyG_318987-ToPAG and HEALTH-F4-2008-201648/PROSPECTS, the Deutsche Forschungsgemeinschaft Excellence Clusters CIPSM and SFB 1035, the Federal Ministry of Education and Research (BMBF), an inter-institutional research initiative of the Max Planck Society, a postdoctoral research fellowship from the Alexander von Humboldt Foundation (to BDE), and by EMBO and HFSP postdoctoral research fellowships.
References
- Engel, B. D., Schaffer, M., Kuhn Cuellar, L., Villa, E., Plitzko, J. M. and Baumeister, W. (2015). Native architecture of the Chlamydomonas chloroplast revealed by in situ cryo-electron tomography. Elife 4: e04889.
- Harris, E. H., Stern, D. B. and Witman, G. B. (2009). The chlamydomonas sourcebook. Academic Press, Elsevier, 2000.
- Hayles, M. F., Stokes, D. J., Phifer, D. and Findlay, K. C. (2007). A technique for improved focused ion beam milling of cryo-prepared life science specimens. J Microsc 226(Pt 3): 263-269.
- Iancu, C. V., Tivol, W. F., Schooler, J. B., Dias, D. P., Henderson, G. P., Murphy, G. E., Wright, E. R., Li, Z., Yu, Z., Briegel, A., Gan, L., He, Y. and Jensen, G. J. (2006). Electron cryotomography sample preparation using the Vitrobot. Nat Protoc 1(6): 2813-2819.
- Mastronarde, D. N. (2005). Automated electron microscope tomography using robust prediction of specimen movements. J Struct Biol 152(1): 36-51.
- Rigort, A., Villa, E., Bauerlein, F. J., Engel, B. D. and Plitzko, J. M. (2012). Integrative approaches for cellular cryo-electron tomography: correlative imaging and focused ion beam micromachining. Methods Cell Biol 111: 259-281.
- Umen, J. G. and Goodenough, U. W. (2001). Control of cell division by a retinoblastoma protein homolog in Chlamydomonas. Genes Dev 15(13): 1652-1661.
- Villa, E., Schaffer, M., Plitzko J. M. and Baumeister, W. (2013). Opening windows into the cell: Focused-ion-beam milling for cryo-electron tomography. Curr Opin Struc Biol 23(5), 771–777.
Article Information
Copyright
Schaffer et al. This article is distributed under the terms of the Creative Commons Attribution License (CC BY 4.0).
How to cite
Readers should cite both the Bio-protocol article and the original research article where this protocol was used:
- Schaffer, M., Engel, B. D., Laugks, T., Mahamid, J., Plitzko, J. M. and Baumeister, W. (2015). Cryo-focused Ion Beam Sample Preparation for Imaging Vitreous Cells by Cryo-electron Tomography. Bio-protocol 5(17): e1575. DOI: 10.21769/BioProtoc.1575.
- Engel, B. D., Schaffer, M., Kuhn Cuellar, L., Villa, E., Plitzko, J. M. and Baumeister, W. (2015). Native architecture of the Chlamydomonas chloroplast revealed by in situ cryo-electron tomography. Elife 4: e04889.
Category
Plant Science > Phycology > Cell analysis
Cell Biology > Cell imaging > Electron microscopy
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link











