- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Metabolic Assays for Detection of Neutral Fat Stores
Published: Vol 5, Iss 12, Jun 20, 2015 DOI: 10.21769/BioProtoc.1511 Views: 15710
Reviewed by: HongLok LungJustine MarsolierKate Hannan

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Measurement of the Intracellular Calcium Concentration with Fura-2 AM Using a Fluorescence Plate Reader
Magdiel Martínez [...] Walter I. Silva
Jul 20, 2017 34016 Views
Abstract
Lipid droplets (LDs) are ubiquitous intracellular structures whose formation, growth, and maintenance are highly regulated (Wang et al., 2013; Ranall et al., 2011; Goodman, 2009). Lipid metabolism and droplet dynamics are of considerable interest to agriculture, biofuel production, viral pathology, nutrition, and cancer biology (Walther and Farese, 2009; Liu et al., 2010). Accumulation of fatty acids and neutral lipids in nonadipose tissues is cytotoxic (Kourtidis et al., 2009). BODIPY 493/503 (4,4-Difluoro-1,3,5,7,8-Pentamethyl-4-Bora-3a,4a-Diaza-s-Indacene) is the standard dye to study LDs within adipocytes. BODIPY 493/503 contains a nonpolar structure that, upon binding to neutral lipid, emits a green fluorescence signal with a narrow wavelength range, making it an ideal fluorophore for multi-labeling experiments. The hydrophobic nature of the dye molecules promotes rapid entry into the nonpolar environment of LDs (Listenberge and Brown, 2007). Gocze and Freeman showed that the lipid fluorescent variability is significantly lower when using BODIPY493/503 compared to Nile Red, suggesting that it may be more specific for the LD (Gocze and Freeman, 1994). Here, we describe a BODIPY 493/503 assay for the detection of neural fat stores in cultured cells (Figure 1) (Wang et al., 2013).
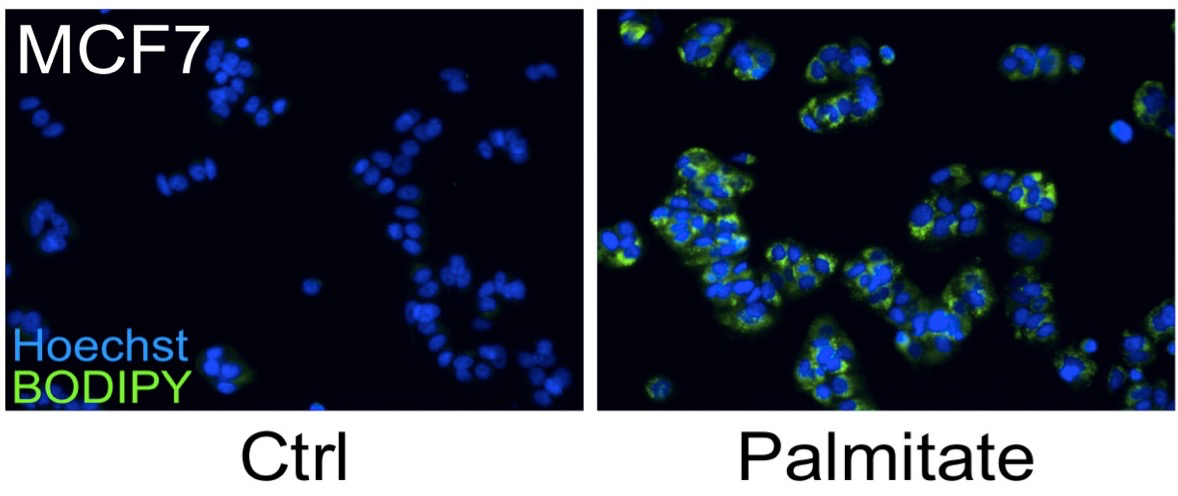
Figure 1. MCF7 cells were treated with 250 μM palmitate or vehicle control for 24 h. A number of breast cancer cells possess a lipogenic metabolic phenotype that makes them especially sensitive to the addition of physiological concentrations of exogenous saturated fatty acids, such as palmitate. Although palmitate supplementation induces cell death in HER2/neu-positive cells, other breast cancer sub-types, including MCF-7 cells, accumulate the fatty acid which leads to significant increases in intracellular triglyceride fat stores. Cells were fixed and stained for fat stores with BODIPY 493/503 (green). Hoechst 33342 (blue) was used for nuclei staining.
Materials and Reagents
- Cell line suitable for testing, MCF7 (ATCC, catalog number: HTB-22 TM) cells perform well as a staining control
- Dulbecco’s Modified Eagle’s Medium (DMEM) (high glucose with L-glutamine) (Thermo Fisher Scientific, catalog number: SH3024301 ) or other media appropriate for MCF7 cell culture
- Fetal bovine serum (FBS) (Sigma-Aldrich, catalog number: F4135 )
- Phosphate buffered saline (PBS) (e.g. HyClone, catalog number: SH30258-02 ) or Dulbecco's phosphate buffered saline (with Ca2+ and Mg2+) (DPBS) (e.g. Sigma-Aldrich, catalog number: D1283 )
- Sodium palmitate, (used as positive control) (Sigma-Aldrich, catalog number: P9767 )
- Dimethyl sulfoxide (Sigma-Aldrich, catalog number: D8418 )
- BODIPY 493/503 (4,4-Difluoro-1,3,5,7,8-Pentamethyl-4-Bora-3a,4a-Diaza-s-Indacene) (Life Technologies, catalog number: D-3922 )
- 37% formaldehyde (Thermo Fisher Scientific, catalog number: F79 )
- Hoechst 33342 (Life Technologies, catalog number: H21492 )
- 500x BODIPY 493/503 stock solution (see Recipes)
- 10,000x Hoechst 33342 stock solution (see Recipes)
Equipment
- 96-well tissue culture plate suitable for imaging, (e.g. Corning, Costar®, catalog number: 3603 )
- Cell culture incubator at 37 °C with 5% CO2
- Fluorescence microscope or automated imaging system (e.g. IN Cell Analyzer, GE Healthcare)
Procedure
- Plate 10,000-20,000 cells per well in 100 μl DMEM high glucose with L-glutamine, 10% FBS, in the 96-well plate. Doing a serial dilution can be helpful to determine the effect of cell number on BODIPY intensity.
- Incubate the cells at 37 °C overnight.
- Dilute the sodium palmitate stock solution in pre warmed DMEM high glucose with L-glutamine, 10% FBS. A range of 50-500 μM is useful to demonstrate BODIPY-based lipid content relative quantification. Prepare appropriate vehicle controls.
- Carefully aspirate medium from the 96-well plate and replace with treatment medium (use 100 μl per well).
- Incubate the cells at 37 °C overnight.
- Prepare a 5% formaldehyde solution in PBS. Add 100 μl per well directly into the tissue culture medium to achieve a final formaldehyde concentration of 2.5%.
- Incubate for 15 min at room temperature.
- Prepare the staining solution; dilute the BODIPY and Hoechst stock solutions in PBS to a working concentration of 10 μg/ml BODIPY and 1 μg/ml Hoechst. Calculate 100 μl per well. Both dyes can be added in parallel, there is no need for sequential staining.
- Carefully remove the formaldehyde from the cells and wash twice with 100 μl of PBS per well.
- Add 100 μl staining solution per well.
- Incubate for 30 min at room temperature, in the dark.
- Wash twice with 100 μl of PBS per well.
- Remove PBS, add 130 μl of PBS per well (only required for IN Cell Analyzer).
- BODIPY and nuclei Hoechst fluorescence are imaged and quantified using fluorescence microscopy with appropriate filter sets. Our laboratory makes extensive use of the INCell Analyzer 2200 and INCell Investigator software for these measurements. Fluorescence intensity per cell is proportional to the neutral lipid content in the cell.
Notes
- Solubilize sodium palmitate in DMSO at 75 mM immediately before usage, do not store.
- If many cells are lost from the plate during washing with regular PBS, use DPBS for washing and preparing working solutions of BODIPY and Hoechst.
- Care should be taken to stain at approximately equal cell densities when comparing different cells or culture conditions.
Recipes
- 500x BODIPY 493/503 stock solution
Solubilize BODIPY 493/503 in DMSO at 5 mg/ml
Aliquot and stored at -20 °C in the dark - 10,000x Hoechst 33342 stock solution
Solubilize Hoechst 33342 in DMSO at 10 mg/ml
Aliquot and stored at -20 °C in the dark
Acknowledgments
Supported by U.S. Army Medical Research Acquisition Activity W8IWXH-04-1-0474 and NCI 1R01CA136658 to DSC.
References
- Gocze, P. M. and Freeman, D. A. (1994). Factors underlying the variability of lipid droplet fluorescence in MA-10 Leydig tumor cells. Cytometry 17(2): 151-158.
- Goodman, J. M. (2009). Demonstrated and inferred metabolism associated with cytosolic lipid droplets. J Lipid Res 50(11): 2148-2156.
- Kourtidis, A., Srinivasaiah, R., Carkner, R. D., Brosnan, M. J. and Conklin, D. S. (2009). Peroxisome proliferator-activated receptor-gamma protects ERBB2-positive breast cancer cells from palmitate toxicity. Breast Cancer Res 11(2): R16.
- Listenberger, L. L. and Brown, D. A. (2007). Fluorescent detection of lipid droplets and associated proteins. Curr Protoc Cell Biol Chapter 24: Unit 24 22.
- Liu, H., Liu, J. Y., Wu, X. and Zhang, J. T. (2010). Biochemistry, molecular biology, and pharmacology of fatty acid synthase, an emerging therapeutic target and diagnosis/prognosis marker. Int J Biochem Mol Biol 1(1): 69-89.
- Ranall, M. V., Gabrielli, B. G. and Gonda, T. J. (2011). High-content imaging of neutral lipid droplets with 1,6-diphenylhexatriene. Biotechniques 51(1): 35-36, 38-42.
- Walther, T. C. and Farese, R. V., Jr. (2009). The life of lipid droplets. Biochim Biophys Acta 1791(6): 459-466.
- Wang, X., Sun, Y., Wong, J. and Conklin, D. S. (2013). PPARgamma maintains ERBB2-positive breast cancer stem cells. Oncogene 32(49): 5512-552.
Article Information
Copyright
© 2015 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Baumann, J. M., Kokabee, L., Wang, X., Sun, Y., Wong, J. and Conklin, D. S. (2015). Metabolic Assays for Detection of Neutral Fat Stores. Bio-protocol 5(12): e1511. DOI: 10.21769/BioProtoc.1511.
Category
Cancer Biology > General technique > Cell biology assays > Metabolism
Biochemistry > Lipid > Lipid measurement
Cell Biology > Cell staining > Lipid
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link