- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Development and Implementation of an in vitro Culture System for Intact Detached Grape Berries
Published: Vol 5, Iss 12, Jun 20, 2015 DOI: 10.21769/BioProtoc.1510 Views: 11801
Reviewed by: Arsalan DaudiKabin XieAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Quantification of Ethylene Production in Leaf and Bud Tissue of the Subtropical Tree Crop Litchi (Litchi chinensis Sonn.) Using Gas Chromatography and Flame Ionization Detection
Regina B. Cronje and Arnoldus J. Jonker
Mar 20, 2023 1588 Views
Bi-directional Dual-flow-RootChip for Physiological Analysis of Plant Primary Roots Under Asymmetric Perfusion of Stress Treatments
Claudia Allan [...] Claudia-Nicole Meisrimler
Aug 5, 2023 1986 Views

Enzymatic Starch Quantification in Developing Flower Primordia of Sweet Cherry
Nestor Santolaria [...] Afif Hedhly
Apr 5, 2025 1934 Views
Abstract
Grape composition depends on the metabolites accumulated and synthesized during grape development. It is of paramount importance for grape growers because of its major role in shaping wine quality. Therefore, understanding the regulation mechanisms that control the accumulation of quality-related metabolites in grape is of both scientific and agronomical interests. The composition of grape berry at harvest is under complex regulation and can be affected by many factors (Conde et al., 2007). The study of the effects of these factors on berries still attached to intact plants can be highly challenging because of the large size of the plants, interplant, intercluster and interberry variability; and because it is complicated to precisely control the nutrients and hormones imported by the berries, and the environment. Therefore, in vitro cultured grape berries are a good model system, which better represents berry anatomy structure (skin and flesh) than grape cell suspensions and nevertheless largely reduces the system complexity compared to whole plant (Bravdo et al., 1990; Pérez et al., 2000; Gambetta et al., 2010). To this end, an in vitro culture system of intact detached grape berries has been developed by coupling greenhouse fruiting-cuttings production and in vitro organ culture techniques (Dai et al., 2014). The cultured berries are able to actively absorb and utilize carbon and nitrogen from the culture medium, and exhibit fruit ripening features such as color changing and softening. This in vitro system may serve to investigate the response of berry composition to environmental and nutrient factors.
Keywords: VitisMaterials and Reagents
- Grapevine berries from greenhouse-grown fruiting-cuttings of Vitis vinifera L. cv. Cabernet Sauvignon at various developmental stages (e.g. pea size, green berry, veraison, or later stages). The fruiting-cuttings (i.e. only one primary shoot axis with a single cluster per plant) were prepared as described in Mullins and Rajasekaran (1981) and grown in a naturally illuminated and semi-regulated greenhouse (mean seasonal temperature amplitude 20-35 °C) with fungicide treatments every two weeks (Figure 1).
Note: Efficient fungicide treatments are essential to maximally exclude pathogen infection to grape berries and minimize the sterilization step for the in vitro culture. For example, we continuously provide sulfur contained in sulfur diffusion hotbox (Nivola Sulphur fl 220 v 3 m, Figure 1B) in the greenhouse and treated the vines with armicarb® at 5 g/L every two weeks.

Figure 1. Fruit-bearing cuttings grown in a semi-controlled greenhouse (A) and the sulfur diffusion hotbox (B)
- 70% ethanol
- NaClO with available chlorine 2% (Sigma-Aldrich, catalog number: 425044-1L )
- MS (Murashige & Skoog) medium (Duchefa Biochemie, catalog number: M 0221 )
- Sucrose (Duchefa Biochemie, catalog number: S0809 )
- N-Z-Amine A (Sigma-Aldrich, catalog number: C7290 )
- Vitamines
Myo-inositol 100 mg/L (Sigma-Aldrich, catalog number: I5125 )
Nicotinic acid 1 mg/L (Sigma-Aldrich, catalog number: N0765 )
Pantothenic acid 1 mg/L (Duchefa Biochemie, catalog number: C0604 )
Biotin 0.01 mg/L (Sigma-Aldrich, catalog number: B4639 )
Pyridoxine HCl 1 mg/L (Sigma-Aldrich, catalog number: P9755 )
Thiamine HCl 1 mg/L (Sigma-Aldrich, catalog number: T4625 )
- 0.5 M NaOH
- Agar (Kalys, catalog number: HP 696 )
- EDTA (Sigma-Aldrich, catalog number: E5134 )
- Sterile water
- MS medium with sucrose and vitamins (see Recipes)
- 200x vitamin (see Recipes)
- 20 mM EDTA (see Recipes)
Equipment
- Laminar flow cabinet (Steril-Helios, Figure 2A)
- Shaker (Dragon lab, sk-330-pro, Figure 2A)
- Autoclave machine
- Growth room with constant temperature of 26 ± 0.5 °C, light period 16 h/8 h day/night, and light ~50 μmol m-2 s-1 (Figure 4H)
- Sterilized 6-well plates (Dutscher, catalog number: 353046 , Figure 5B)
- Sterilized plastic boxes with filter (Dutscher, catalog number: E 1650 , Figures 3F, 4G, and 5A)
- Tip plate (Figure 3A)
- Polystyrene (Figure 3C)
- Culture dish (145/20 mm, Greiner Bio-One GmbH, Figure 4E)
- Sterilization solution container (Figure 2C-D)
- Colander, forceps, scissors, and blade (Figure 2B)

Figure 2. Equipment used in grape berry culture. A. Laminar flow cabinet with a shaker. B. colander, forceps, scissors, and blade. C. solution container, culture boxes, and small materials just after autoclave. D. Sterilized containers for ethanol and NaClO.
Procedure
- Preparation of the floater for liquid medium
- Collect tip plates and cut them into suitable size for the culture box (Figure 3A and B).
- Prepare polystyrene cuboids corresponding to the size of tip plates (Figure 3C).
- Put the polystyrene cuboids on the two sides of the tip plates to form a floater (Figure 3D-F).
- Sterilize floaters with 10% NaClO and 90% ethanol during 20 min each following three rinses with sterile water and then install them in the tissue culture box containing liquid medium.

Figure 3. Preparation of the floater for liquid medium. A. Boxes of micropipette tips. B. Cut tip plates to a suitable size for the culture box. C. Prepare two polystyrene cuboids. D. Assemble polystyrene cuboids with tip plate to form the floater. E. Vertical view of the floater inside of a culture box. F. Side view of the floater inside of a culture box.
- Collect tip plates and cut them into suitable size for the culture box (Figure 3A and B).
- Berry in vitro culture
- Fruit-bearing cuttings were selected and brought to culture room after removing all leaves (Figure 4A).
- Grape clusters were excised from the mother plant, and berries were subsequently excised from peduncle with berry pedicel (about 3 mm) and dropped immediately into tap water (Figure 4B).
- Keep tap water running for 15 min to clean the berries.
- Put berries into 70% ethanol for 2 sec and transfer immediately into NaClO with 2% available chlorine with a sterilized colander (Figure 4D).
Note: The volume ratio between berry and sterilization solution is important for an efficient sterilization, and a minimum ratio 1:3 (berry/solution) is recommended.
- Shaking the container of NaClO with a shaker at 350-450 rpm for 2 min (Figure 4C).
Note: Dependent on the status of the greenhouse-grown berries, this step can be extended to 3-5 min. Longer sterilization will hurt berry skin.
- Transfer berries into sterilized de-ionized water with colander (Figure 4D) and shake at 350-450 rpm for 2 min.
- Repeat step B6 three times.
- Transfer fully rinsed berries into 20 mM EDTA solution, which is contained in a big culture dish (Figure 4E).
- Cut berry pedicel again to about 2 mm under EDTA solution (Figure 4E), in order to exclude cavitations and to prevent plugging of sieve tubes by callose synthase, a strictly calcium-dependent enzyme.
- Gently open the culture boxes and quickly put the berries on the solid or liquid culture medium with berry pedicel rooting into culture medium (see Recipes, Figure 4F and Figure 5).
- Gently put the cover of culture box (Figure 4G) and transport all boxes to culture room with constant temperature of 26 ± 0.5 °C, light period 16 h/8 h day/night, and light ~50 μmol m-2 s-1 (Figure 4H).

Figure 4. Berry in vitro culture procedure. A. Bring fruit-bearing cuttings to the lab after removing all leaves. B. Excise berries from grape cluster and drop them immediately into tap water. C. Sterilize berries with ethanol and bleach solutions with a shaker. D. Transform the sterilized berries to water for rinsing. E. Recut berry pedicel in 20 mM EDTA. F. Install berries into culture medium. G. Put the cover of tissue culture box. H. Install tissue culture boxes into culture room.
- Alternative culture methods depending on the desired experiment might be chosen (Figure 5).
- The berry culture can be maintained as long as 3 months. Since the medium volume is much greater than the berries, there is no need for change of medium. However, it will be very easy to change the medium composition of liquid medium with syringe injection.
Note: Berries are not very tightly fixed on the culture medium, so any movement and transport should be done gently to avoid berry slanting.
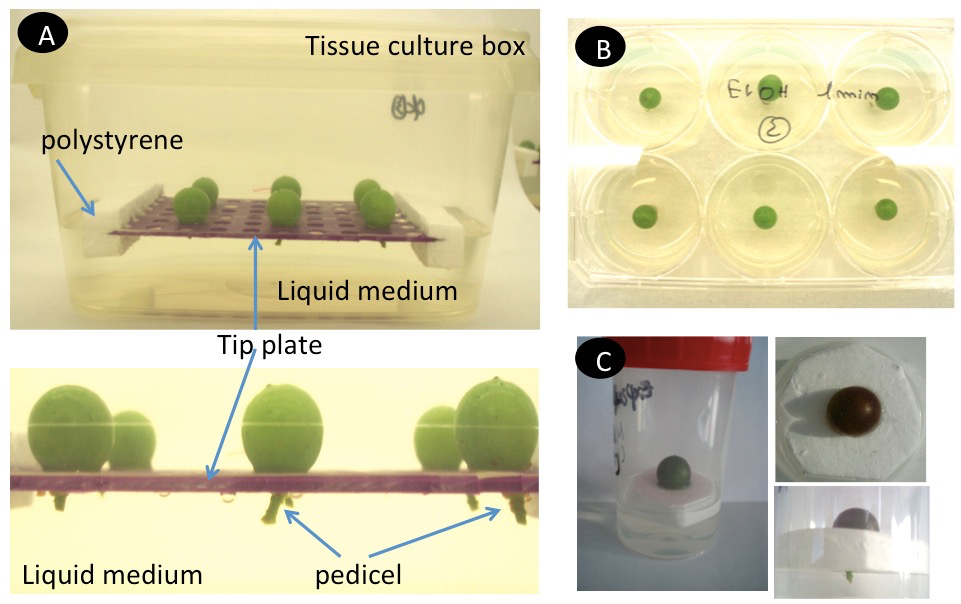
Figure 5. Different types of berry in vitro culture. A. Culture with liquid medium and floater. B. Culture with solid medium in a 6-well plate. C. Culture with liquid medium and floater in a small container.
- Fruit-bearing cuttings were selected and brought to culture room after removing all leaves (Figure 4A).
Recipes
- MS medium with sucrose and vitamins
4.3 g M0221
20 g sucrose
0.25 g N-Z-AmineA
Dissolve in 800 ml deionized water with vortex
5 ml vitamin 200x (see below)
Adjust pH to 5.8 with 0.5 M NaOH
Complete to 1 L with deionized water
Add 9 g agar for solid medium
Autoclave (120 °C, 20min)
For medium containing agar, pour 4 ml medium to each well of the 6-well plate (Figure 5B); or 50 ml medium to the tissue culture box with special filters allowing gas exchange (Figure 4G), before medium concretion and inside of the laminar flow cabinet.
For liquid medium, pour 150 ml medium the tissue culture box with special filters (Figure 5A).
Note: A minimum 2% of sucrose is needed to prevent berry crack for green Cabernet Sauvignon berries. Different cultivars may have different sensitivity and therefore a suitable sucrose concentration has to be established for other cultivars.
- 200x vitamin
Dissolve 8 mg biotin into 1 ml de-ionized water with one drop of KOH
25 µl biotin solution
2 g myo-inositol
20 mg nicotinic acid
20 mg pantothenic acid
20 mg pyridoxine HCl
20 mg thiamine HCl
Dissolve and complete to 100 ml with deionized water
Stored at -20 °C
- 20 mM EDTA
Dissolve 7.44 g EDTA in 1 L de-ionized water
Autoclave (120 °C, 20 min)
Stored at room temperature
Acknowledgments
This work was supported by a specific grant from the Environment and Agronomy division (EA) of the Institut National de la Recherche Agronomique (INRA), France. This protocol was developed based on our previous paper (Dai et al., 2014).
References
- Bravdo, B., Shoseyov, O., Ikan, R. and Altman, A. (1990). Monoterpene glycoside biosynthesis in detached grape berries grown in vitro. Physiol Plant 78(1): 93-99.
- Conde, C., Silva, P., Fontes, N., Dias, A. C. P., Tavares, R. M., Sousa, M. J., Agasse, A., Delrot, S. and Gerós, H. (2007). Biochemical changes throughout grape berry development and fruit and wine quality. Food 1, 1-22.
- Dai, Z. W., Meddar, M., Renaud, C., Merlin, I., Hilbert, G., Delrot, S. and Gomes, E. (2014). Long-term in vitro culture of grape berries and its application to assess the effects of sugar supply on anthocyanin accumulation. J Exp Bot 65(16): 4665-4677.
- Gambetta, G. A., Matthews, M. A., Shaghasi, T. H., McElrone, A. J. and Castellarin, S. D. (2010). Sugar and abscisic acid signaling orthologs are activated at the onset of ripening in grape. Planta 232(1): 219-234.
- Mullins, M. G., and Rajasekaran, K. (1981). Fruiting cuttings: revised method for producing test plants of grapevine cultivars. Am J Enol Vitic, 32: 35-40.
- Pérez, F. J., Meza, P., Berti, M. and Pinto, M. (2000). Effect of carbon source and sucrose concentration on growth and hexose accumulation of grape berries cultured in vitro. Plant Cell Tiss Org 61(1): 37-40.
Article Information
Copyright
© 2015 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Dai, Z., Meddar, M., Delrot, S. and Gomès, E. (2015). Development and Implementation of an in vitro Culture System for Intact Detached Grape Berries. Bio-protocol 5(12): e1510. DOI: 10.21769/BioProtoc.1510.
Category
Plant Science > Plant physiology > Plant growth
Plant Science > Plant physiology > Tissue analysis
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link











