- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Measuring UV-induced Mutagenesis at the CAN1 Locus in Saccharomyces cerevisiae
Published: Vol 4, Iss 20, Oct 20, 2014 DOI: 10.21769/BioProtoc.1272 Views: 12783
Reviewed by: Kanika GeraEmilia Krypotou Anonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
MNase Digestion for Nucleosome Mapping in Neurospora
Cigdem Sancar [...] Michael Brunner
Jun 5, 2016 10332 Views

Using CRISPR/Cas9 for Large Fragment Deletions in Saccharomyces cerevisiae
Huanhuan Hao [...] Liping Zhang
Jul 20, 2017 14282 Views
Method for Multiplexing CRISPR/Cas9 in Saccharomyces cerevisiae Using Artificial Target DNA Sequences
Rachael M. Giersch and Gregory C. Finnigan
Sep 20, 2017 13400 Views
Abstract
There are several methods to measure the capacity of yeast cell to respond to environmental impacts on their genome by mutating it. One frequently used method involves the detection of forward mutations in the CAN1 gene. The CAN1 gene encodes for an arginine permease that is responsible for the uptake of arginine and it can also transport the toxic analog of arginine, canavanine (Whelan et al., 1979). When CAN1 cells are grown on a media containing canavanine but lacking arginine, the cells die because of the uptake of the toxic canavanine. However, if a mutation in the CAN1 gene inactivates the permease, that cell survives and forms a colony on the plate.
The following protocol describes the measurement of UV-induced mutagenesis at the CAN1 locus.
Materials and Reagents
- Strains that have the wild type CAN1 gene (e.g., BY4741, EMY747)
- Yeast nitrogen base [w/o amino acids and w (NH4)2SO4] (Difco)
- Adenine (Sigma-Aldrich)
- Arginine (Sigma-Aldrich)
- Canavanine (Sigma-Aldrich)
- Histidine (Sigma-Aldrich)
- Isoleucine (Sigma-Aldrich)
- Leucine(Sigma-Aldrich)
- Lysine (Sigma-Aldrich)
- Methionine (Sigma-Aldrich)
- Phenylalanine (Sigma-Aldrich)
- Tryptophan (Sigma-Aldrich)
- Tyrosine (Sigma-Aldrich)
- Uracil (Sigma-Aldrich)
- Valine (Sigma-Aldrich)
- Yeast extract
- Pepton
- D-glucose
- Bacto agar
- Yeast extract-pepton-D-glucose (YPD) media (see Recipes) (or other media needed for the strain in use)
- Synthetic complete media (SC) plates (see Recipes)
- SC-arginine plates containing canavanine (+can) (see Recipes)
Equipment
- 30 °C incubator-shaker (180-200 round per minute)
- 30 °C incubator
- Glass slide
- Centrifuge
- Microscope
- Vortex
- UV-irradiation machine
- Box
- Spreader
- Culture tube
- Waterbath sonicator
- Hemocytometer (Burker counting chamber)
Procedure
- By inoculating a single colony from a fresh plate, grow up 10 ml overnight cultures in YPD (one reference, or wild type strain, and the strains to be examined).
- Put the culture tube into a waterbath sonicator and sonicate the cells at room temperature for 3 min to disrupt clumps (40 Hz). Check for the presence of clumps under the microscope.
- Make 200x dilutions of each strain in water (5 µl cell culture into 995 µl water) and put 10 µl onto a Burker-chamber.
- Count the cells under the microscope in a big square of the Burker-chamber (bordered by three lines) (Figure 1). One cell in a big square means 1 x 104 cells/ml.
- Calculate the density of the original cultures (multiply the counted cell number by 200).
- Make 10x serial dilutions of each strain, starting from 108 to 103 cells/ml. If necessary concentrate cells to get 108 cells/ml. When calculating the volume of a given dilution step, take into consideration how many plates you will be plating from that dilution (see steps 8 and 9 below).
- Label the SC and the +can plates with the strain names and the UV doses you want to apply. For each UV dose, including zero, label 2 SC and 2 +can plates for each strain. The SC plates will be used to calculate survival at different UV doses, the +can plates will be used to calculate mutagenesis at different UV doses.
- Plate 200 µl on the control, 0 J/m2 SC plates from the 103 cells/ml dilution. For wild type, EMY747 or BY4741 cells that are quite resistant to UV showing 20% survival at 80 J/m2, for up to that dose the 103 cells/ml dilution should be used for plating. In case of more sensitive strains the expected survival rate should be taken into consideration when determining which dilution to use for plating for given UV doses.
- Plate 200 µl cells on the +can plates from the 108 cells/ml dilutions.
- Wait till plates absorb the moisture, then irradiate the plates without lids, with the required UV doses. Make sure the irradiated plates are not exposed to white light after irradiation (work with yellow light on), and they are placed right away in a box that shields them from light and put in the 30 °C incubator.
- Incubate the plates until colonies grow to 2-3 mm in diameter. For SC plates it takes usually 2-3 days, for +can plates it takes up to 5 days (check them under yellow light).
- Count the colonies on each plate.
- Calculate the percentage of survival on SC plates. Divide the average number of colonies of the two 0 J/m2 plates with the number of cells that were plated on one plate and multiply it by 100. That gives the percentage of cells that survived plating.
- Calculate the survival at each UV doses. Multiply the average colony number of the two parallel plates with the plating survival percentage calculated from 0 J/m2 plates (see step 13 above), and divide it by the number of cells that was plated. (Take into consideration the actual volumes you plated at different doses: 100 µl, or 200 µl!)
- Calculate mutagenesis from +can plates. Multiply with the survival percentage the number of cells plated on a plate at the given UV dose. Calculate the average number of colonies from the two parallel plates of the same UV dose. That gives you the number of mutants/plated cells. Based on that calculate how many mutants would be in 106 cells, because mutagenesis data usually corresponds to 106 cells. (Take into consideration the actual volumes you plated at different doses: 100 µl, or 200 µl!)
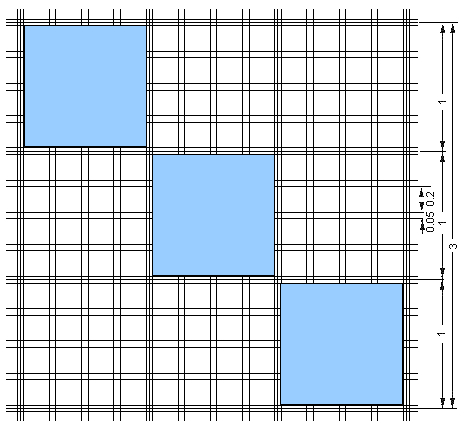
Figure 1. Burker chamber. One cell in one blue area means 1 x 104 cells/ml. (Source: http://openwetware.org/wiki/IGEM:University_of_Debrecen:_transfection)
Representative data
- Since the result of this experiment depends on the number of inactivating mutations in the CAN1 gene inflicted upon by UV, the number of mutants can vary. Because of that average numbers should be calculated based on 3-5 experiments.
Recipes
- YPD liquid
1% yeast extract
2% pepton
2% D-glucose
- SC plates
Bacto-agar: 16.6 g/L
D-glucose: 20 g/L
12 media mix: 7.2 g/L
12 media mixYeast nitrogen base [w/o amino acids and w (NH4)2SO4]
400 g
Adenine
1.8 g
Arginine
1.2 g
Histidine
1.2 g
Isoleucine
1.8 g
Leucine
1.8 g
Lysine
1.8 g
Methionine
1.2 g
Phenylalanine
3.0 g
Tryptophan
1.2 g
Tyrosine
1.8 g
Uracil
1.2 g
Valine
9.0 g
- SC -arginine plates containing canavanine
Same as the SC plates, but the media mix contains 3.6 g canavanine instead of arginine
Acknowledgments
We used this protocol in our work (Daraba et al., 2014). Funding support: Wellcome Trust, 070247/Z/03/A.
References
- Daraba, A., Gali, V. K., Halmai, M., Haracska, L. and Unk, I. (2014). Def1 promotes the degradation of Pol3 for polymerase exchange to occur during DNA-damage--induced mutagenesis in Saccharomyces cerevisiae. PLoS Biol 12(1): e1001771.
- Whelan, W. L., Gocke, E. and Manney, T. R. (1979). The CAN1 locus of Saccharomyces cerevisiae: fine-structure analysis and forward mutation rates. Genetics 91(1): 35-51.
Article Information
Copyright
© 2014 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Unk, I. and Daraba, A. (2014). Measuring UV-induced Mutagenesis at the CAN1 Locus in Saccharomyces cerevisiae. Bio-protocol 4(20): e1272. DOI: 10.21769/BioProtoc.1272.
Category
Microbiology > Microbial genetics > Mutagenesis
Microbiology > Microbial genetics > DNA > Chromosomal
Molecular Biology > DNA > Mutagenesis
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link









