- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Measurement of TACE Activity in Extracts from Cultured Cells
Published: Vol 4, Iss 20, Oct 20, 2014 DOI: 10.21769/BioProtoc.1264 Views: 9439
Reviewed by: Elizabeth V. ClarkeGuillaume Bompard HongLok Lung

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Novel Experimental Approach to Investigate Immune Control of Vascular Function: Co-culture of Murine Aortas With T Lymphocytes or Macrophages
Taylor C. Kress [...] Eric J. Belin de Chantemèle
Sep 5, 2025 3584 Views
Dual Phospho-CyTOF Workflows for Comparative JAK/STAT Signaling Analysis in Human Cryopreserved PBMCs and Whole Blood
Ilyssa E. Ramos [...] James M. Cherry
Nov 20, 2025 2390 Views
Detecting the Activation of Endogenous Small GTPases via Fluorescent Signals Utilizing a Split mNeonGreen: Small GTPase ActIvitY ANalyzing (SAIYAN) System
Miharu Maeda and Kota Saito
Jan 5, 2026 534 Views
Abstract
In cigarette smoke–induced and inflammation-associated lung cancer development, cigarette smoke extract (CSE) activates tumor necrosis factor-alpha (TNF-α) secretion from macrophages. TNF-α converting enzyme (TACE), also known as α-Secretase or ADAM17 (A Disintegrin and Metalloprotease), is a member of the ADAM family of metalloproteases. TACE mediated ectodomain shedding leads to the conversion of the inactive TNF-α precursor into the active mature pro-inflammatory cytokine. The SensoLyte 520 TACE (α-Secretase) Activity Assay Kit was used to detect TACE activity in CSE-activated macrophages. This assay is reliable, reproducible and easy to carry out in 96 well plate format.
Keywords: TACEMaterials and Reagents
- Cultured cells such as the macrophage line THP1
- SensoLyte 520 TACE (α-Secretase) Activity Assay Kit *Fluorimetric* (AnaSpec, catalog number: 72085 )
Components used from the kit:- Component A QXL™ 520/5-FAM (TACE substrate, Ex/Em = 490 nm/520 nm)
- Component B 5-FAM, fluorescence reference standard (Ex/Em = 490 nm/520 nm)
- Component C assay buffer
- Component A QXL™ 520/5-FAM (TACE substrate, Ex/Em = 490 nm/520 nm)
- Triton-X 100 (Sigma-Aldrich, catalog number: T8532 )
- Phosphate buffered saline (PBS) (see Recipes)
Equipment
- 96 well black microplate (for fluorescence analysis) (Greiner Bio-One GmbH, catalog number: 655077 )
- Tube/vial unidirectional rotator (Glas-Col, catalog number: 099A RD4512 )
- Cell lifter (polyethylene, sterile) (Corning Incorporated Costar, catalog number: 3008 )
- 1.5 ml microcentrifuge tubes (polypropylene) (USA Scientific, catalog number: 1615-5599 )
- Microplate fluorometer with detection range (excitation 490 nm and emission 520 nm) (settings recommended by SensoLyte 520 TACE Activity Assay Kit) (Thermo Fisher Scientific, Fluoroskan AscentTM) (used for TACE activity assay)
Procedure
Following Protocol B from the manufacturer’s manual and described the details below:
- Prepare cell lysates
- Amount of adherent cells needed for this assay are approximately 3 x 105 or 70 - 80% confluent cells per experimental sample in one well of 12 well plate.
- Add 0.1% (v/v) Triton-X 100 to Component C assay buffer.
- After induction of TACE activation with treatment of choice, remove media; wash cells with PBS; lift cells, collect into 1.5 ml microcentrifuge tube, and lyse with 200 µl Component C assay buffer. (The amount of assay buffer needs to be determined empirically for the experimental samples used.)
- Incubate the cell suspension at 4 °C for 10 min on unidirectional rotator.
- Centrifuge the lysed cells at 2,500 x g at 4 °C.
- Collect supernatant containing protein sample into a new 1.5 ml microcentrifuge tube.
- Amount of adherent cells needed for this assay are approximately 3 x 105 or 70 - 80% confluent cells per experimental sample in one well of 12 well plate.
- Prepare working solutions
Note: All kit components should be thawed to room temperature before starting the experiment. Protect light sensitive materials from exposure to ambient light.
For TACE substrate solution, dilute TACE substrate (Component A) 1:100 in assay buffer (Component C).
Note: For each experiment, prepare fresh substrate solution.
- Set up TACE enzymatic reaction
- Set up at least triplicate enzymatic reactions.
- Add 50 µl of protein sample (supernatant collected in step 1f) into black 96 well plate.
- Set up a 50 µl blank control containing assay buffer (Component C) only. The blank control well fluorescence reading accounts for the background fluorescence contributed by the assay buffer. In this case, the blank control functions as the reagent blank consisting only of the assay buffer used to lyse the cells; in general, the absorbance reading for this solution is typically subtracted from each sample absorbance reading in order to only consider the substance you are trying to analyze or measure.
- In the dark, set up a 50 µl positive control with the 5-FAM fluorescence reference standard: Dilute 1 mM 5-FAM (Component B) to 4 μM in assay buffer (Component C). Do 2-fold serial dilutions to get concentrations of 2, 1, 0.5, 0.25, 0.12, and 0.06 μM, include an assay buffer blank. Add 50 μl per well of these serially diluted 5-FAM reference solutions. Protect tubes and plate from ambient light.
- On lab bench, pre-incubate the plate for 10 min at room temperature protected from ambient light.
- Set up at least triplicate enzymatic reactions.
- Measure TACE activity
Use TACE substrate solution equilibrated to room temperature. Add 50 µl of TACE substrate solution into each well (each well now contains a total volume of 100 µl). Mix reagents in plate by gently shaking by hand for 30 sec.
Immediately quantify TACE enzymatic activity by measuring fluorescence intensity at Ex/Em=485 nm/538 nm.
- Detect protein concentration
Use a protein quantification method, such as the Bradford assay, to measure total protein concentration in each experimental sample from the cell lysates prepared in steps 1c-f (He, 2011).
- Data analysis
The blank control well fluorescence reading accounts for the background fluorescence contributed by the assay buffer. Subtract the background fluorescence reading of the blank from each sample in order to only consider the TACE activity measured. Normalize the relative fluorescence units (RFU) of each experimental sample to its total protein concentration. Plot the data as TACE activity RFU/ µg protein as shown in Figure 1.
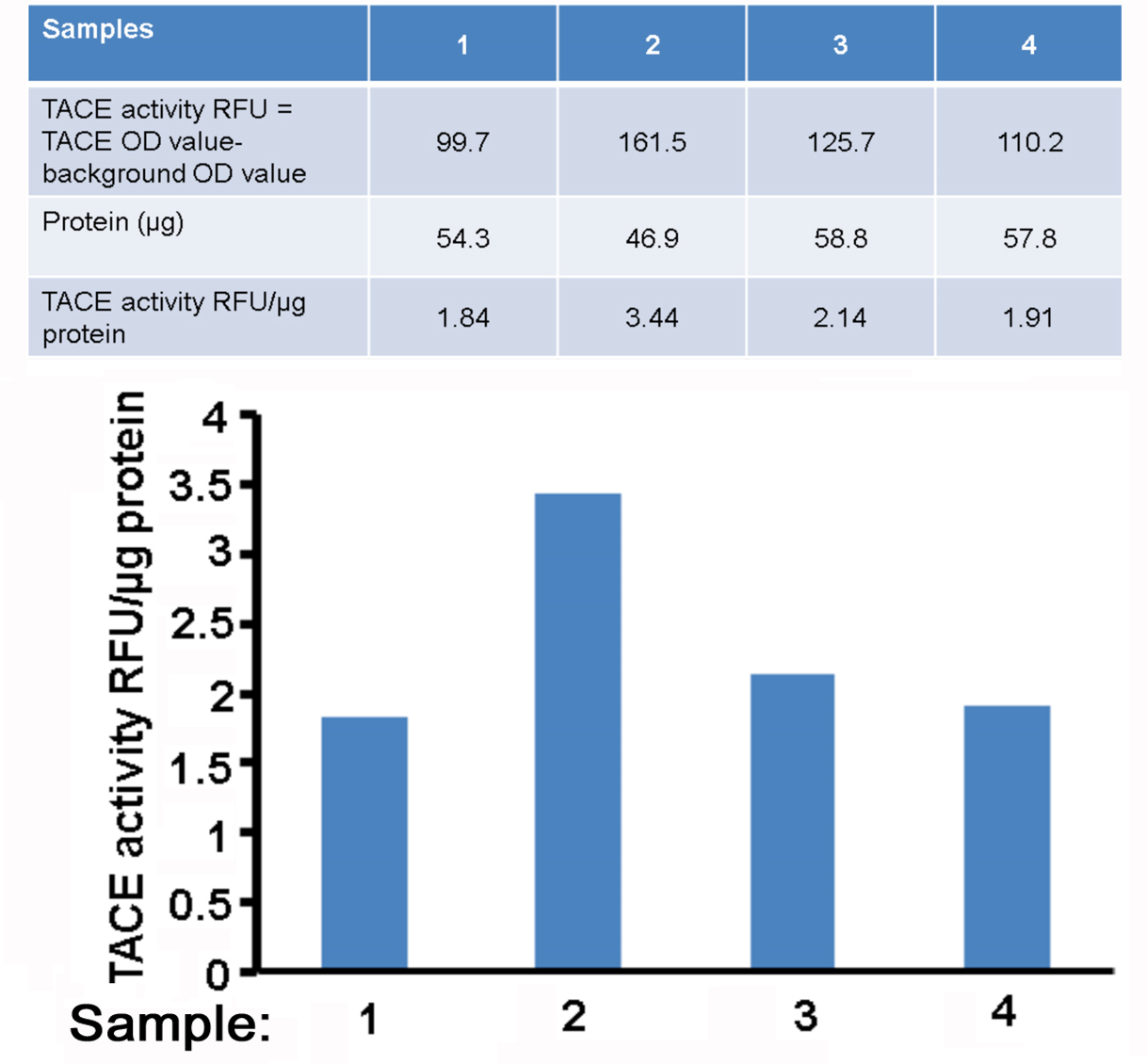
Figure 1. An example of plot of TACE activity (RFU/μg protein)
Notes
- Ex/Em=485 nm/538 nm was used to detect fluorescence in cell lysates. Settings were modified from those recommended by SensoLyte 520 TACE Activity Assay Kit (excitation 490 nm and emission 520 nm) because the Fluoroskan AscentTM fluorescence plate reader used in this experiment is limited to this pair of excitation/emission settings.
- Measuring TACE activity may be repeated several times from 0 to 15 min to capture an optimal reading. Kinetic reading of TACE activity should be empirically determined for your experimental system.
Recipes
- Phosphate buffered saline [PBS, 0.9% (w/v) sodium chloride in 10 mM phosphate buffer] Deionized water 800 ml
NaCl
8.00 g
KCl
0.20 g
Na2HPO4
1.44 g
KH2PO4
0.24 g
Adjust pH to 7.4 with HCl or NaOH
Deionized water to 1 L
Acknowledgments
This protocol was adapted from a previously published paper Xu et al. (2014). This study was partly supported by grants from NIEHS/NIH (R01ES017328), NCI/NIH (R01CA142649), and the Office of Science (BER), U.S. Department of Energy (DE-FG02-09ER64783).
References
- He, F. (2011). Bradford Protein Assay. Bio-protocol 1(6): e45.
- Xu, X., Padilla, M. T., Li, B., Wells, A., Kato, K., Tellez, C., Belinsky, S. A., Kim, K. C. and Lin, Y. (2014). MUC1 in macrophage: contributions to cigarette smoke-induced lung cancer. Cancer Res 74(2): 460-470.
Article Information
Copyright
© 2014 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Xu, X., Padilla, M. T. and Lin, Y. (2014). Measurement of TACE Activity in Extracts from Cultured Cells. Bio-protocol 4(20): e1264. DOI: 10.21769/BioProtoc.1264.
Category
Immunology > Immune cell function > Cytokine
Immunology > Immune cell function > Macrophage
Biochemistry > Protein > Activity
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link








