- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Open-book Preparations from Chick Embryos and DiI Labeling of Commissural Axons
Published: Vol 4, Iss 13, Jul 5, 2014 DOI: 10.21769/BioProtoc.1176 Views: 13191
Reviewed by: Xuecai Ge

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Implantation of Dkk-1-soaked Beads into the Neural Tube of Chicken Embryos
Almudena Martinez-Ferre [...] Salvador Martinez
Nov 5, 2013 13013 Views
Live Cell Imaging to Monitor Axonal Pruning in Drosophila Motor Neurons
Keyao Long [...] Menglong Rui
Jul 5, 2025 2073 Views

A Novel Composite Method of Post-stroke Epilepsy Induction
Yiting Guo and Raymond Tak Fai Cheung
Jul 20, 2025 1998 Views
Abstract
Successful neural circuit formation relies on the accurate navigation of axons towards their targets during development. Axons are guided by a combination of short-range and long-range, attractive and repulsive cues. The commissural axons of the developing spinal cord have provided an informative in vivo model for the identification of multiple axon guidance molecules and mechanisms. These axons extend ventrally from the dorsal spinal cord and cross the midline at the floor plate, before making a sharp rostral turn towards the head. This simple trajectory has facilitated the identification of many axon guidance molecules, because perturbation of the stereotypical guidance decisions as a result of genetic manipulations can be easily identified. The open-book assay is a method to assess the trajectory of spinal commissural axons. The spinal cord is dissected out, opened at the roof plate and pinned flat. Punctate injections of the lipophilic fluorescent dye, DiI, are used to trace commissural axon trajectories prior to microscopy and analysis.
Keywords: Axon guidanceMaterials and Reagents
- Fertilized eggs containing chicken embryos (incubated until Hamburger and Hamilton stage 25-26)
- PBS (add 50 ml of 20x PBS to 950 ml of ddH2O)
- Sylgard silicone elastomer (World Precision Instruments, catalog number: SYLG184 )
- Fast DiI (Life Technologies, Molecular Probes®, catalog number: D-7756 ) (dissolved at 5 mg/ml in ethanol)
- Vacuum grease (Dow Corning Corporation, catalog number: 976V )
- 20x sterile phosphate buffered saline (PBS) (see Recipes)
- 4% paraformaldehyde (4% PFA) (see Recipes)
- Phosphate (PB) buffer (1 M, 1 L, pH 7.4) (see Recipes)
Equipment
- Incubator set at 38.5 °C and 45% humidity (e.g. FIEM, Juppiter 576 Setter+Hatcher; Kendro Laboratory Products, Heraeus/Kendro, model: B12 )
- Spring scissors (Fine Science Tools, catalog number: 15003-08 )
- Dumont #5 forceps (Fine Science Tools, catalog number: 11252-20 )
- 90 mm Petri dishes (e.g. Thermo Fisher Scientific, Sterilin®, catalog number: 101VR20/C )
- 0.20 mm insect pins (Fine Science Tools, catalog number: 26002-20 )
- 0.10 mm insect pins (Fine Science Tools, catalog number: 26002-10 )
- Microscope with adjustable transmitted light source (Leica MZ6 stereomicroscope with Leica CLS 150x )
- Fine surgical scalpel (Grieshaber Logistic Group, catalog number: 68101 )
- Tungsten wire (0.075 mm) (World Precision Instruments, catalog number: TGW0325 )
- Metal flat spatula/spoon (e.g. Sigma-Aldrich, catalog number: S4022 , S3397 )
- Borosilicate glass capillaries (outer Ø/inner Ø: 1.2 mm/0.68 mm) (World Precision Instruments, catalog number: 1B120F-4 )
- Glass needle puller (Narishige Group, catalog number: PC-10 )
- Polyethylene tubing (Ø 1.24 mm) (e.g. Angst and Pfister AG, catalog number: FT0110350018 )
- Plastic transfer pipettes (e.g. Sarstedt, catalog number: 86.1171 )
- 18 G x 1.5” needle (e.g. Braun, catalog number: 4665120 )
- 1 ml syringe (e.g. Braun, catalog number: 9166017V )
- 20 ml syringe (e.g. Braun, catalog number : 4606205V )
- 24 mm x 24 mm glass coverslips (VWR International, catalog number: 631-0127 )
- Microscope suitable for brightfield and fluorescent microscopy (e.g. Olympus, model: BX51 )
Procedure
- Preparation of open-books
- PBS is added to a Petri dish lined with ~5 mm of Sylgard elastomer (prepared previously, following the manufacturer’s instructions).
Note: Sylgard is required as a substrate for the insect pins during the dissection. Petri dishes lined with Sylgard can be made in advance (in batches) and reused for several years.
- At HH25-26 (day 5 of development), the chicken embryo is removed from the egg using forceps and a small spatula and placed in the Petri dish (Figure 1a).
Note: For windowed eggs (experimentally manipulated), the embryo can be carefully lifted out of the egg through the window by inserting a closed pair of forceps under the neck. Non-windowed eggs should be carefully cracked into a dish. The embryo can be separated from the blood vessels using forceps and spring scissors, rinsed in PBS, then transferred to the dissection dish.
- The extraembryonic membranes are removed with forceps and the head is cut off.
Note: The embryo can be stabilized during this step if necessary, by inserting a 0.2 mm insect pin through the eye. Remove the membranes by grasping them with forceps under the neck, then gently pull them over the head and down towards the tail.
- The embryo is placed on its back and pinned (using 0.2 mm insect pins) through the neck and tail, with gentle stretching.
- The embryo is pinned (using 0.2 mm insect pins) through the wing buds and limb buds (Figure 1b).
Note: All pins should be inserted at an angle away from the embryo so that they do not interfere with the next steps of the dissection.
- The heart and internal organs are removed using spring scissors and gently scraping with forceps.
Note: The embryo should be illuminated from below to enable tissue density to be perceived. The segmented vertebrae should be visible if all the organs have been removed completely (Figure 1c).
- Using spring scissors, a shallow cut is made through the vertebrae overlying the spinal cord at the neck, and longitudinally for a few vertebral segments along each side, towards the tail (Figure 1c).
Note: Do not cut too deeply or too close to the spinal cord. Rotate the dish to make the cutting angle easier.
- Forceps are used to grasp and lift the flap of vertebrae away from the spinal cord. With gentle pulling towards the tail, the vertebrae overlying the spinal cord are removed in a single strip of tissue (Figure 1d).
Note: Be careful not to damage the spinal cord.
- The embryo is gently stretched and re-pinned through the tail and limbs.
- A fine microsurgical scalpel (or a tungsten needle) is used to cut away the meninges overlying the spinal cord.
Note: Look for a denser line of tissue between the spinal cord and the dorsal root ganglia. Adjust the illumination angle, if necessary (Figure 1e). Gently cut longitudinally along this line on each side of the spinal cord, from the neck to the tail. The meninges should separate from the spinal cord due to the gentle stretching of the pinned embryo (Figure 1f).
- Using spring scissors, the spinal cord is cut at the level of the wing buds and caudal to the limb buds.
- Using forceps, the isolated spinal cord is grasped at the rostral end and removed from the embryo in a single piece, by gently pulling it in a smooth motion towards the tail (Figure 1g).
Note: The spinal cord is kept immersed in PBS during this step. In chickens, a small amount of the dorsal spinal cord (the roof plate) is left behind. The isolated spinal cord will therefore resemble a sheet rather than a tube.
- The isolated spinal cord is gently lifted onto a flat metal spatula (for support) and transferred to a new Sylgard-lined Petri dish containing 4% paraformaldehyde in PBS.
Note: The spinal cord is very delicate and should be gently immersed in the 4% PFA using closed forceps (or another blunt tool). Avoid touching the spinal cord at all, if possible. The 4% PFA solution should be at room temperature, as the spinal cords tend to float if the 4% PFA is cold. To break the surface tension, it is also possible to place 1-2 of the previously dissected heads in the dish of 4% PFA, prior to transferring the spinal cords.
- A flat-mount preparation (Figure 2) is produced by carefully pinning the spinal cord in six positions (rostrally, medially and caudally on each side), using 0.10 mm insect pins (Figure 1h). The medial surface of the spinal cord (which previously lined the central canal of the closed neural tube) should be facing upwards.
Note: Angle the pins away from the spinal cord to prevent them interfering with the subsequent DiI injections. We label each open-book using a little ‘flag’ (made of tape wrapped around a 0.2 mm insect pin) that not only identifies the embryo from which the open-book was derived, but also indicates the anterior end of the preparation.
- The open-book preparations are fixed in 4% PFA for 30 min to 1 h at room temperature.
Note: Open-books should not be over-fixed as this reduces the efficiency of DiI diffusion and increases background.
- The fixative is carefully poured out of the dish and replaced with PBS.
Note: Preparations should be refrigerated until ready to inject with DiI or mount for microscopic inspection. DiI should be injected within 2 weeks.
- PBS is added to a Petri dish lined with ~5 mm of Sylgard elastomer (prepared previously, following the manufacturer’s instructions).
- Injection of open-book preparations with DiI
- A needle puller is used to make glass micropipettes from borosilicate glass capillaries.
- A glass micropipette is inserted into polyethylene tubing.
- Using forceps, the end of the micropipette is broken off, to obtain a very small diameter opening (~5 μm) at the tip.
- Fast DiI (5 mg/ml in ethanol) is drawn into the micropipette using gentle mouth suction.
Note: Do not draw up too much DiI, in case the tip of the needle breaks during the injection procedure, causing the DiI to spill and ruin your samples. Insert the needle into a dish of PBS and gently blow some out. Check that the DiI does not continue to leak from the needle after you have finished blowing. If the DiI leaks, the needle diameter is too big. In that case, prepare a new needle.
- The open-book preparations are illuminated from below, and the region to be injected is identified.
Note: The cell bodies of the commissural neurons are located just ventral to the roof plate and just dorsal to the dorsal funiculus. The dorsal funiculus can be identified in the open-books due to its different density compared to the surrounding tissue. Look for a denser longitudinal stripe of tissue, located approximately 1/5 of the width of a hemibook from the lateral edge of the preparation (Figure 1i). The commissural neurons are located immediately lateral to this stripe (i.e. immediately dorsal to the dorsal funiculus). Make your injections along the lateral border of the dorsal funiculus.
- Starting at one end of the open-book, the glass needle is inserted into the tissue at an angle of >45° and, as the needle is withdrawn, a small amount of DiI is gently blown in.
Note: Because ethanol is less dense than PBS, the DiI will float away from the injection site, which may impede your view. Try not to inject excess DiI. You may need to practise mouth-pipetting to get this right.
- Working quickly, DiI injections are made along the length of the open-book at regular intervals of approximately 0.5 mm (Figure 1j).
Note: If the needle becomes clogged, carefully clear it with forceps. If the tip becomes too big (and DiI leaks), replace the needle.
- After completing the injections of each open-book, a transfer pipette is used to suck away and discard any excess DiI.
Note: Failure to do this will result in high background.
- The injected open-book preparations are refridgerated for 3-4 days to allow the DiI to diffuse along the axons.
Note: If necessary, top up the Petri dish with PBS to ensure that the preparations remain immersed during the DiI diffusion.
- A needle puller is used to make glass micropipettes from borosilicate glass capillaries.
- Mounting and microscopic analysis
- A 1 ml syringe is filled with vacuum grease and fitted with an 18 G needle.
- The syringe is used to spread a thin, uninterrupted border of vacuum grease around the edges of a 24 mm x 24 mm glass coverslip.
Note: The grease layer should not be too thick, otherwise it will be difficult to visualize the preparations under microscopy. Make sure there are no gaps in the grease border, or the preparations will dry out. The type of grease used is also important. Vacuum grease of low viscosity may mix with the PBS (in subsequent steps) and result in high background.
- Several drops of sterile PBS are added to the middle of the well.
Note: Mounting medium containing glycerol with n-propyl gallate may not be compatible with DiI.
- The pins are removed from the open-book and it is transferred into the PBS droplet using forceps.
- The open-book is immersed in the PBS and positioned in the middle of the well.
- Another 24 mm x 24 mm coverslip is gently placed on top, making sure the open-book stays open and there are no significant air bubbles.
Note: If necessary, the coverslip can be removed, and the open-book repositioned.
- Gentle pressure is applied to the edges of the coverslips to create a complete seal of grease.
Note: Excess PBS will be squeezed out during this step. Again, avoid air bubbles.
- The preparations are kept refrigerated in the dark until ready for inspection and documentation by fluorescent microscopy (Figure 1k-l). Mounted preparations are placed on top of a glass microscope slide for imaging and analysis.
Note: The PBS will eventually dry out. Analysis should be done within 1-2 weeks, to ensure that the preparations do not dry out before being assessed.
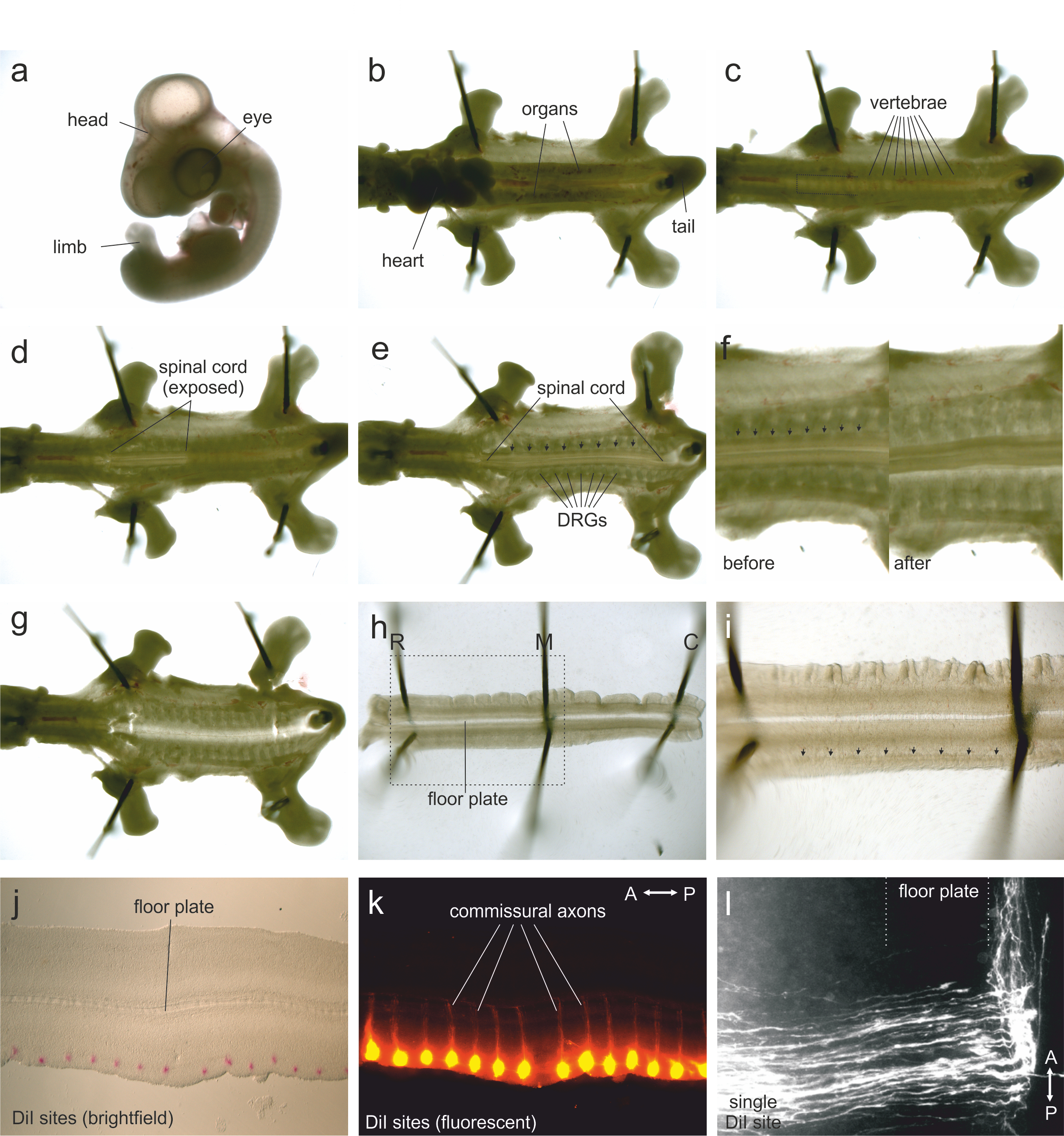
Figure 1. Steps in open-book preparation
- HH25-26 chicken embryo in a dish of PBS. The extraembryonic membranes have been removed.
- Embryo (with head removed) is pinned through the neck, tail, wings and limbs. Pins are angled away from the body. Anterior is to the left.
- Embryo with the heart and organs removed. The segmented vertebrae are now visible. The area to be cut by the spring scissors to create a flap of vertebral tissue is shown by the dotted lines.
- Some of the vertebrae at the anterior end have been removed, exposing the spinal cord. At the posterior end, the vertebrae are still overlying the spinal cord.
- Exposed spinal cord with all vertebrae removed. The DRGs are visible. Arrows indicate the dense line of tissue where longitudinal cuts should be made to separate the meninges from the spinal cord.
- Zoomed views of preparation before and after the spinal meninges (arrows) have been cut.
- Preparation after the spinal cord has been lifted out of the embryo.
- Isolated spinal cord, pinned into the flat ‘open-book’ configuration (see Figure 2). The floor plate lies medially and the dorsal part of the spinal cord is now located laterally. Pins located rostrally (R) medially (M) and caudally (C) are angled away from the preparation.
- In higher magnification, the dense longitudinal stripe of tissue corresponding to the dorsal funiculus is indicated by arrows. DiI injections should be made along the lateral border of the dorsal funiculus.
- Brightfield image of a mounted open-book preparation with DiI injection sites (red).
- Fluorescent image of an open-book preparation with DiI injection sites (red). The commissural axons, which have grown ventrally and crossed the floor plate, can be seen.
- Higher magnification of a single DiI injection site. Commissural axons grow towards and across the floor plate before turning anteriorly.
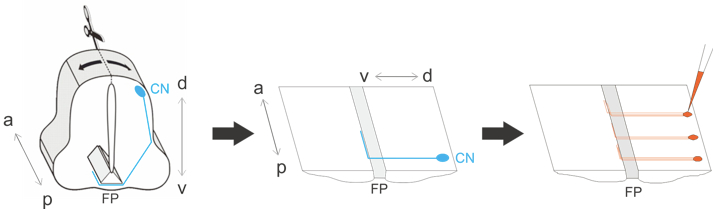
Figure 2. Schematic overview of procedure. The spinal cord is dissected out, opened dorsally at the roofplate and laid flat. The commissural neurons (CN, shown in blue) extend axons that grow ventrally and cross the floor plate (FP) before turning anteriorly. Commissural axons are labeled by injections of DiI (red). a, anterior; p, posterior; d, dorsal; v, ventral.
- HH25-26 chicken embryo in a dish of PBS. The extraembryonic membranes have been removed.
- A 1 ml syringe is filled with vacuum grease and fitted with an 18 G needle.
Notes
- The isolated spinal cords are very fragile. Avoid touching them as much as possible and minimize/avoid changes of solution. When transferring the spinal cords from dish to dish, or when mounting the DiI-injected open-books for analysis, always grasp them at the anterior end using forceps. That way, only a small part of the spinal cord will be damaged during the handling.
- We have previously published a video protocol demonstrating electroporations, open-book dissections and DiI injection steps, which may be useful (Wilson and Stoeckli, 2012).
- For further details of chicken embryo incubation and handling, see Reference 1 or Reference 4.
- Please refer to Labeling of Precursor Granule Cells in the Cerebellum by ex vivo Electroporation (Ito-Ishida, 2013) for mouth pipetting approaches.
Recipes
- 20x sterile PBS (1 L)
160 g of NaCl
4 g of KCl
28.84 g of Na2HPO4.2H2O
4.14 g of NaH2PO4.H2O
Dissolved in 1 L ddH2O, autoclave.
- 4% paraformaldehyde (4% PFA) (200 ml)
150 ml of ddH2O
0.8 ml of 2 M NaOH
8 g of Paraformaldehyde
Heat at 70 °C for 30 min, cool, add 20 ml of 1 M PB buffer.
Adjust pH to 7.4, make up to 200 ml with ddH2O.
Stored at 4 °C
- Phosphate (PB) buffer (1 M, 1 L, pH 7.4)
206.6 g of Na2HPO4.7H2O
32 g of NaH2PO4.H2O
Dissolved in 1 L ddH2O
Acknowledgments
This protocol was adapted from the previously published papers Wilson and Stoeckli (2012) and Andermatt et al. (2014). This work was supported by a grant from the Swiss National Science Foundation to E.S.
References
- Andermatt, I., Wilson, N. and Stoeckli, E. T. (2014). In ovo electroporation of miRNA-based-plasmids to investigate gene function in the developing neural tube. Methods Mol Biol 1101: 353-368.
- Ito-Ishida, A. (2013). Labeling of precursor granule cells in the cerebellum by ex vivo electroporation. Bio-protocol 3(12): e778.
- Wilson, N. H. and Stoeckli, E. T. (2013). Sonic hedgehog regulates its own receptor on postcrossing commissural axons in a glypican1-dependent manner. Neuron 79(3): 478-491.
- Wilson, N.H. and Stoeckli, E. T. (2012). In ovo electroporation of miRNA-based plasmids in the developing neural tube and assessment of phenotypes by dil injection in open-book preparations. J Vis Exp (68), e4384, doi:10.3791/4384.
Article Information
Copyright
© 2014 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Wilson, N. H. and Stoeckli, E. T. (2014). Open-book Preparations from Chick Embryos and DiI Labeling of Commissural Axons. Bio-protocol 4(13): e1176. DOI: 10.21769/BioProtoc.1176.
Category
Neuroscience > Development > Neuron > Membrane protein detection
Neuroscience > Neuroanatomy and circuitry > Animal model
Developmental Biology > Cell growth and fate > Neuron
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link











