- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Mouse Model of Dextran Sodium Sulfate (DSS)-induced Colitis
Published: Vol 7, Iss 16, Aug 20, 2017 DOI: 10.21769/BioProtoc.2515 Views: 20691
Reviewed by: Pasquale PellegriniAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Systematic Analysis of Smooth Muscle and Cartilage Ring Formation during Mouse Tracheal Tubulogenesis
Haoyu Wu [...] Wenguang Yin
Jul 5, 2023 1534 Views

Protocol for the Implantation of Scaffolds in a Humanized Mouse Cutaneous Excisional Wound Healing Model
Dina Gadalla [...] David G. Lott
Sep 20, 2024 1452 Views
Dissection and Whole-Mount Immunofluorescent Staining of Mouse Hind Paw Muscles for Neuromuscular Junction Analysis
Rebecca L. Simkin [...] James N. Sleigh
May 20, 2025 3912 Views
Abstract
Inflammatory bowel disease (IBD) is a chronic inflammatory disease of the intestinal tract and is primarily comprised of Crohn’s disease (CD) and ulcerative colitis (UC). Several murine models that include both chemical induced and genetic models have been developed that mimic some aspects of either CD or UC. These models have been instrumental in our understanding of IBD. Of the chemical induced colitis models, dextran sodium sulfate (DSS) induced colitis model is a relatively simple and very widely used model of experimental colitis.
Keywords: Dextran sodium sulfate (DSS)Background
Inflammatory bowel disease (IBD) is a complex and multifactorial disease of unknown etiology (Sartor, 2006). However, multiple factors are considered to be critical in conferring IBD susceptibility, e.g., defects in host genetics, environmental triggers, aberrant immune response against microbial and dietary antigens (Sartor, 2006). Several murine models that address specific aspects of the disease process are available (Mizoguchi, 2012). DSS induced experimental colitis is a rapid and widely used model of intestinal inflammation (Perse and Cerar, 2012). Although the exact mechanism of DSS induced colitis is not well understood, it is widely accepted that disruption of the epithelial monolayer resulting in exposure of the underlying immune system to the intestinal contents containing microbiota and microbial products (Perse and Cerar, 2012). DSS induced colonic inflammation can be adapted as acute, chronic or relapsing model of intestinal inflammation by changing the concentration, duration and cycles of administration of DSS in drinking water. In this protocol, we provide a detailed description of procedures, important considerations when performing the protocol. We have used this protocol to address the role of Muc4 in DSS induced colitis in our previous work (Das et al., 2015).
Materials and Reagents
- Sterile Eppendorf tubes (National Scientific, catalog number: CN1700-BP )
- Tissue Path Macrosette processing cassettes (Fisher Scientific, catalog number: 15-182-706 )
- Glass slides
- 8-10 week old mice generated in house or obtained from the commercial vendors
Note: Mice used in the study of Das et al., (2015) were of 129/Sv and C57BL/6J mixed background. Mice in this study were littermates and included both males and females housed under specific pathogen free (SPF) conditions. - Dextran sodium sulfate (DSS) (TdB Consultancy, catalog number: DB001 , molecular weight: 35-55 kDa)
Note: Stored in a dry place at room temperature. - Liquid nitrogen
- Autoclaved drinking water
- Anesthesia (Isofluorane or CO2)
- 10% buffered formalin (Fisher Scientific, catalog number: SF100-4 )
- Ethanol
- Xylene
- Paraffin
- mirVana miRNA isolation kit (Thermo Fisher Scientific, InvitrogenTM, catalog number: AM1560 )
- Bioanalyzer (Agilent Technologies, Waldbronn, Germany)
- DNase I (QIAGEN, catalog number: 79254 )
- Oligo-dT or random hexamer primers (Thermo Fisher Scientific, InvitrogenTM, catalog number: 100023441 )
- SuperScript reverse transcriptase II (Thermo Fisher Scientific, InvitrogenTM, catalog number: 18064014 )
- Light Cycler 480 SYBR Green mix (Roche Molecular Systems, catalog number: 04707516001 )
- Hematoxylin and eosin (H & E)
Equipment
- Measuring scale/Ruler or Vernier calipers
- Animal weighing balance
- Feeding bottles
- Tissue-Tek VIP processing machine
- Dissection equipment
- Mouse microisolator chambers
- Pestle and mortar
Procedure
- On day 0, tag all the mice in both the control and experimental groups according to the appropriate method (ear tagging, ear punching etc.) approved by the institute. It is important to tag mice individually so that body weight, stool consistency, blood in the stool etc. after DSS administration can be monitored. Weigh all the mice before treating them (ideally the weight of mice should be approximately around 20 g) with either autoclaved water or DSS dissolved in autoclaved drinking water. Collect stool samples if required.
- Prepare an appropriate concentration of DSS (2-3% based on the question in study, please see step 3 below) in autoclaved drinking water by dissolving DSS powder until a clear solution is reached. Fill the water bottles in the cages with approximately 100 ml for 2 days per cage with 4-5 mice. Discard the remaining water after 2 days and replenish with freshly prepared DSS containing water.
- For an injury repair model, 2% DSS (i.e., 2 g in 100 ml) in the drinking water is administered for the first 7 days followed by regular drinking water for another 7 days. However, the number of days on DSS followed by regular water can be changed based on the question in study. For example, if the question in study does not involve the recovery or the repair phase, then the DSS administration can be terminated either on day 7 or can be continued if prolonged inflammatory phase is desired. In order to determine the survival rate, 3% DSS in the drinking water can be administered until all the mice in either the control or the experimental group reach a humane endpoint. Ideally, the humane endpoint is when the mice have arrived the stipulated body weight loss approved by the ethical committee. In addition, severe and bloody diarrhea accompanied by hunched position, ruffled fur and reduced mobility are considered to be humane endpoint and the mice should be sacrificed by approved methods.
- Mice treated with DSS will lose a significant amount of body weight and therefore, should be weighed daily to monitor the health and to ensure that mice are sacrificed at a humane endpoint based on the approved ethical standards. Additionally, the mice should be monitored daily for the stool consistency (0 = well formed stool pellet, 2 = semi-formed stool, 4 = liquid stool that adhere to anal region) and blood in the stool (0 = no blood, 2 = blood trace in the stool clearly visible, 4 = gross rectal bleeding). The individual recording this information should be blinded to the study. Loss of body weight, diarrhea and fecal bleeding score can be plotted to determine the effect of DSS induced colitis between mice of different genotypes and/or treatment groups. As a general guideline, mice start to lose body weight around day 6 of DSS treatment and continue to lose weight approximately till day 10 (3 days after DSS removal) that is approximately 15-20% less than the initial body weight. During this time, the mice display behavioral changes such as reduced appetite, hunched back posture, ruffled fur etc. Following day 10, mice usually start to gain body weight and reaches the initial body weight by around day 14. However, this can vary depending on microbiota composition, animal facility and therefore should be carefully interpreted.
- Following the cycles of DSS (7 days) and regular water (7 days), on day 14, mice can be euthanized according to the appropriate method approved by the ethical committee (IACUC). Colon length should be measured using a ruler or Vernier calipers as DSS administration leads to swelling and shortening of colon. Pieces of colon (~0.5 cm) can be snap frozen in liquid nitrogen to be further processed for RNA extraction and gene expression analysis. For histology, the rest of the tissues can be Swiss rolled and stored in 10% buffered formalin using Tissue Path Macrosette processing cassettes. Following 48-72 h of fixation in formalin, the tissue samples should be removed from formalin and placed in 70% ethanol until further tissue processing and embedding. Normally, tissue processing can be carried out overnight (Table 1) using a Tissue-Tek VIP processing machine followed by paraffin embedding.
Table 1. Tissue processing protocol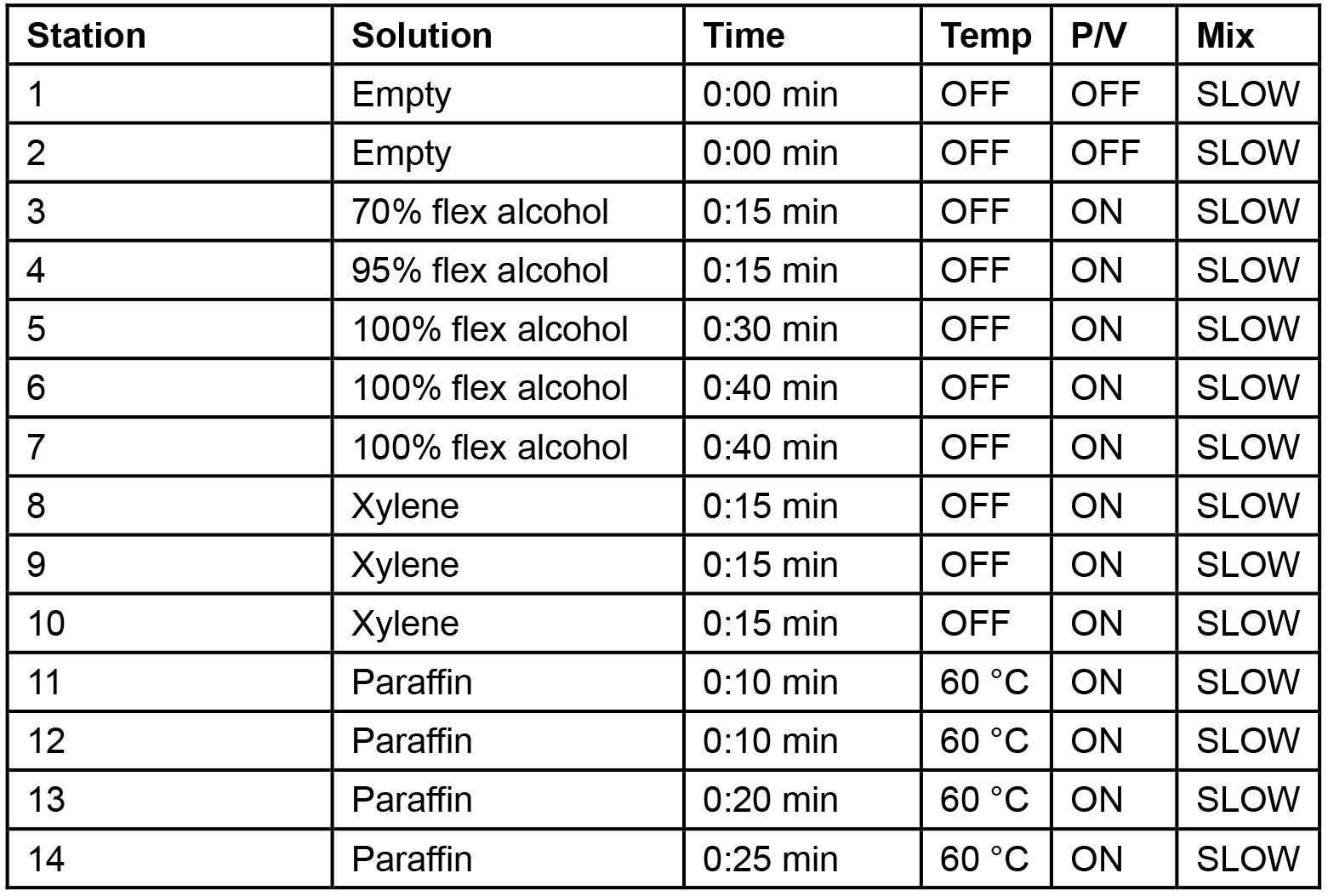
- Snap frozen tissues in liquid nitrogen can be used for RNA extraction following homogenization (using sterile pestle and mortar) using mirVana miRNA isolation kit according to manufacturer’s instructions, which can be used to isolate total RNA as well as small RNAs. Further treatment of the extracted RNA with DNase I (QIAGEN) according to manufacturer’s instructions is advised to obtain pure RNA without DNA contamination. RNA is further converted to cDNA using either oligo-dT or random hexamer primers and SuperScript reverse transcriptase II (Thermo Fisher Scientific) according to the manufacturer’s instructions. Expression of pro inflammatory cytokines such as TNF-α and IL1-β that are upregulated during DSS colitis can be used as useful markers of DSS induced colitis.
- Fomalin fixed tissues can be further paraffin embedded, sectioned and mounted on glass slides for histological analysis. For histological score analysis tissue sections can be stained with hematoxylin and eosin (H & E) and scored by a mouse pathologist in a blinded fashion. Briefly, a score from 0-3 is assigned for infiltration of inflammatory cells and 0-3 is assigned for tissue damage. Finally, a combined score of ‘0’ would indicate healthy intestine compared to a score of 6 which will indicate extensive infiltration and tissue damage. Immuno-histochemical staining for immune cells such as Macrophages (F4/80), apoptotic cells (Caspase 3), loss of epithelial architecture (E-cadherin) are reliable indicators of DSS induced colitis.
- Since the effect of DSS is more pronounced in the distal compared to the proximal colon, a histological scoring of proximal and distal colon can be performed separately.
Data analysis
- Appropriate statistical methods should be applied for data analysis.
- For representative data including survival curves, fecal blood score, gene expression analysis and histologic images, we refer readers to Das et al., (2015) Figures 2, 3, 4 and 5.
Notes
- All procedures performed should be approved by the Institutional Animal Care and User Committee (IACUC) of the respective institutions. The animals involved in this study must be treated humanely, and in all situations in which the mice are used, pain and discomfort should be minimized.
- It is important not to mix mice from different sources as they may have different microbiome compositions that may alter the outcome of DSS induced colitis. Additionally, when using mice that are genetically modified, littermate controls are advised unless the role of maternal contribution (e.g., in-utero or vertical transfer) from a specific genotype is being studied.
- Molecular weight of DSS is highly critical (molecular weight: 35-55 kDa).
Acknowledgments
We would like to thank members of Batra lab for useful discussions. The authors on this protocols were, in parts, supported by the NCI Fred and Pamela Cancer Center Support Grant P30 CA036727.
References
- Das, S., Rachagani, S., Sheinin, Y., Smith, L. M., Gurumurthy, C. B., Roy, H. K. and Batra, S. K. (2015). Mice lacking Muc4 are more resistant to experimental colitis and colitis-associated colorectal cancer. Oncogene 35(20): 2645-2654.
- Mizoguchi, A. (2012). Animal models of inflammatory bowel disease. Prog Mol Biol Transl Sci 105: 263-320.
- Perse, M. and Cerar, A. (2012). Dextran sodium sulphate colitis mouse model: traps and tricks. J Biomed Biotechnol 2012: 718617.
- Sartor, R. B. (2006). Mechanisms of disease: pathogenesis of Crohn’s disease and ulcerative colitis. Nat Clin Pract Gastroenterol Hepatol 3(7): 390-407.
Article Information
Copyright
© 2017 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Das, S., Batra, S. K. and Rachagani, S. (2017). Mouse Model of Dextran Sodium Sulfate (DSS)-induced Colitis. Bio-protocol 7(16): e2515. DOI: 10.21769/BioProtoc.2515.
Category
Cancer Biology > Inflammation > Animal models
Cell Biology > Tissue analysis > Tissue staining
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link










