- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Gene Dosage Experiments in Enterobacteriaceae Using Arabinose-regulated Promoters
Published: Vol 7, Iss 14, Jul 20, 2017 DOI: 10.21769/BioProtoc.2396 Views: 7912
Reviewed by: Yoko EguchiAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Development and Quantitation of Pseudomonas aeruginosa Biofilms after in vitro Cultivation in Flow-reactors
Yingdan Zhang [...] Haihua Liang
Aug 20, 2021 3681 Views
Quantification of RuBisCO Expression and Photosynthetic Oxygen Evolution in Cyanobacteria
Mateusz Kędzior and Betul Kacar
Oct 20, 2021 3335 Views
A Practical CRISPR-Based Method for Rapid Genome Editing in Caulobacter crescentus
Xuezhou Yuan [...] Jingxian Sun
Nov 5, 2025 1812 Views
Abstract
This protocol is used to assay the effect of protein over-expression on fitness of E. coli. It is based on a plasmid expression of a protein of interest from an arabinose-regulated pBAD promoter followed by the measurement of the intracellular protein abundance by Western blot along with the measurement of growth parameters of E. coli cell expressing this protein.
Keywords: Gene-dosage toxicityBackground
Gene dosage experiments are crucial for understanding the effects of protein over-expression on fitness and determining the optimal levels of protein abundance. Several genes are toxic even when expressed at very low levels. It is therefore important to express the protein from a tightly regulated promoter to minimize leaky expression. Here we have elucidated conditions for expression of proteins under the well-characterized arabinose induced pBAD promoter, and designed protocols to measure the intracellular abundance and fitness of E. coli cells harboring the overexpression plasmid.
Materials and Reagents
- Sterile tips
- 1.5 ml microfuge tubes (Corning, Axygen®, catalog number: MCT-175-C )
- 14 ml polypropylene tubes for bacterial culture (Corning, Falcon®, catalog number: 352059 )
- 50 ml tubes for bacterial culture (Corning, Falcon®, catalog number: 352070 )
- Sterile needle for inoculation (Thermo Fisher Scientific, Thermo ScientificTM, catalog number: 253988 )
- Honeycomb plates (Bioscreen, catalog number: 95025BIO )
- Gene of interest cloned in pBAD/MCS-vector (https://www.embl.de/pepcore/pepcore_services/strains_vectors/vectors/bacterial_expression_vectors/popup_bacterial_expression_vectors/ obtained from European Molecular Biology Laboratory EMBL) using appropriate restriction sites in the multiple cloning site
- E. coli BW27783 cells (CGSC, catalog number: 12119 )
- LB/Agar plates
- Ampicillin (Sigma-Aldrich, catalog number: A0166 )
- Sodium phosphate buffer, pH 7.4
- Tris-HCl pH 8.0
- Pierce BCA protein assay kit (Thermo Fisher Scientific, Thermo ScientificTM, catalog number: 23227 )
- 4x protein gel loading dye (Thermo Fisher Scientific, InvitrogenTM, catalog number: NP0007 )
- Pre-cast 12% Bis-Tris SDS polyacrylamide gel (Bio-Rad Laboratories, catalog number: 3450123 )
- Trans-blot Turbo midi nitrocellulose transfer packs (Bio-Rad Laboratories, catalog number: 1704158 )
- Antibody raised against the protein of interest
- Western-breeze Chromogenic Immunodetection kit (Thermo Fisher Scientific, InvitrogenTM, catalog number: WB7105 for anti-rabbit)
- M9 salts (BD, DifcoTM, catalog number: 248510 )
- Magnesium sulfate heptahydrate (MgSO4·7H2O) (Sigma-Aldrich, catalog number: 230391 )
- Thiamine (Sigma-Aldrich, catalog number: T1270 )
- Casamino acid (AMRESCO, catalog number: J851 )
- Glucose (Sigma-Aldrich, catalog number: G7021 )
- L(+) arabinose (EMD Millipore, catalog number: 178680 )
- 10x Bugbuster reagent (Novagen, catalog number: 70921-3 )
- Benzonase nuclease (EMD Millipore, catalog number: 70664 )
- Supplemented M9 medium (see Recipes)
- Lysis buffer (see Recipes)
- L(+) arabinose solution (Concentrations used) (see Recipes)
Equipment
- Pipettes
- Incubator
- Heated Orbital shaker
- Centrifuge (Eppendorf, models: 5810 R and 5417 R )
- Spectrophotometer (BioTek Instruments, model: PowerWave HT )
- Rotator-mixer
- Trans-Blot Turbo transfer system (Bio-Rad Laboratories, catalog number: 1704155 )
- Bioscreen C (Growth Curves USA)
Software
- ImageJ software
Procedure
- For abundance measurement
- Transform pBAD plasmid containing the gene of interest in to electro-competent BW27783 cells (see Notes, point #1), and spread on LB/Agar plates containing 100 µg/ml ampicillin. Incubate plates overnight at 37 °C.
- Next day, pick a single colony from the plate and inoculate into 2 ml of supplemented M9 medium (see Recipes and Notes, point #2) containing 100 µg/ml of ampicillin. Grow overnight with shaking (250 rpm) at 37 °C in 14 ml polypropylene tubes.
- Next day, dilute 500 µl of the overnight culture 1/100 in to 50 ml of fresh M9 medium containing 100 µg/ml of ampicillin and varying concentrations of arabinose (0-0.05%) (see Recipes), and grow for 4 h with shaking (250 rpm) at 37 °C.
- After 4 h, measure OD600 of the cultures, and spin down in a table-top centrifuge at 3,000 x g for 15 min. Aspirate as much culture supernatant as possible. Store the cell pellets at -20 °C.
- Prepare the lysis buffer, which is the buffer of choice (50 mM sodium phosphate buffer, pH 7.4, 10 mM Tris-HCl pH 8.0, etc.) supplemented with the detergent Bugbuster and Benzonase nuclease (see Recipes below). Based on the measured OD600 of the cultures, re-suspend the pellets in the prepared lysis buffer such that the final OD600 is 2.0 (see Notes, point #3). Allow the lysis to proceed for 20 min at room temperature on a rotator-mixer.
- Following lysis, spin down the cell debris for 30 min at 7500 x g in a centrifuge that has been pre-chilled at 4 °C.
- Separate the supernatant and quantify the total protein in each sample using the BCA assay kit using the manufacturer’s instructions.
- Load 20 µl of the supernatants on a 12% Bis-Tris SDS polyacrylamide gel, after required dilution of the samples (see Notes, point #4).
- Resolve the samples at a voltage gradient of 10 V/cm of gel.
- Once the dye front has reached the base, the gel is ready to be transferred to a membrane for blotting. Use pre-assembled nitrocellulose membrane sandwiches (see Notes, point #5).
- Following transfer, wash the membrane with water two times to remove transfer buffer components and weakly bound proteins. From this step onwards, use Invitrogen’s Western-breeze Chromogenic Immunodetection kit to develop the membrane.
- Transform pBAD plasmid containing the gene of interest in to electro-competent BW27783 cells (see Notes, point #1), and spread on LB/Agar plates containing 100 µg/ml ampicillin. Incubate plates overnight at 37 °C.
- For growth rate measurement
- From step A2 above, dilute the overnight culture to a final OD600 of 0.01 (see Notes, point #3) into fresh M9 medium containing 100 µg/ml of ampicillin and varying concentrations of arabinose (see Recipes). Aliquot 150 µl of this into three wells of the honeycomb Bioscreen plate (see Notes, point #6). This serves as replicates for a single colony. To obtain standard error of biological replicates, inoculate 3 independent colonies, and subject each of them to varying arabinose concentration.
- Aliquot 150 µl M9 medium into three independent wells. This serves as the background for OD measurements.
- Measure OD600 values at 15 min intervals over a period of 12 h in Bioscreen C system at 37 °C with shaking (see Notes, point #6).
- From step A2 above, dilute the overnight culture to a final OD600 of 0.01 (see Notes, point #3) into fresh M9 medium containing 100 µg/ml of ampicillin and varying concentrations of arabinose (see Recipes). Aliquot 150 µl of this into three wells of the honeycomb Bioscreen plate (see Notes, point #6). This serves as replicates for a single colony. To obtain standard error of biological replicates, inoculate 3 independent colonies, and subject each of them to varying arabinose concentration.
Data analysis
- Use ImageJ software to evaluate the band intensities from the Western blot (For a complete tutorial, please visit ‘https://imagej.nih.gov/ij/docs/guide/146-30.html#toc-Subsection-30.13’).
- Normalize the intensity by the total protein concentration obtained by BCA assay, and also scale up the values by the dilution factor. If the protein of interest is an endogenous E. coli protein, then calculate the fold-overexpression of the protein of interest based on the intensity obtained for untransformed BW27783 cells (set to 1). For a foreign protein, the expression level obtained with 0% arabinose should be set to 1.
- Fit the growth curves obtained from Bioscreen C as OD vs. time (t) using the following 4-parameter Gompertz equation to obtain growth rate parameters (Adkar et al., 2017).
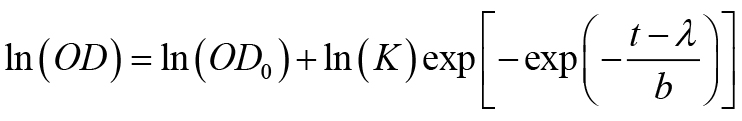
Where, K is the fold-increase over initial population at saturation, b is the shape factor and defined as b = ln(K)/(μ·exp(1)) where μ is the maximum growth rate, and the lag time λ is the time taken to achieve the maximum growth rate. - Make a plot of gene-dosage effect using measured growth rates and intracellular protein abundance (Figure 1) (see Notes, point #7).

Figure 1. Representative gene-dosage toxicity curves for E. coli Dihydrofolate Reductase (DHFR) expressed from pBAD-plasmid (Bhattacharyya et al., 2016). A. Plot of relative growth rate as a function of arabinose concentration; B. Plot of relative growth rate as a function of intracellular abundance of DHFR protein.
Notes
- The transporter araE in E. coli helps in uptake of arabinose from the medium. However, as the expression of araE is all or none in WT E. coli cells (e.g., MG1655), induction with arabinose results in a heterogeneous population. BW27783 that is used in this protocol is a strain of E. coli MG1655 that has been engineered to constitutively express araE, resulting in a uniform and homogeneous uptake of arabinose (Khlebnikov et al., 2001).
- The gene-dosage experiments can be done in any medium of choice (LB, M9, etc.). However over-expression of the protein of interest may show different phenotypes in different media conditions. For example as discussed in (Bhattacharyya et al., 2016), over-expression of E. coli Dihydrofolate Reductase (DHFR) was found to be toxic in supplemented M9 medium, but not in LB medium. We therefore suggest choosing the growth medium which allows the study of the phenotype of interest.
- OD600 is defined for 1 cm path-length.
- The typical amount of total protein from cell lysate loaded on to the gel was 5 μg. However, for higher levels of induction, dilution will be necessary. We therefore suggest doing a pilot experiment to find the necessary dilution.
- Instead of pre-assembled nitrocellulose membrane sandwiches, one can prepare their own using pre-cut nitrocellulose membranes, blotting papers, and tris-glycine-methanol transfer buffer to perform this step successfully.
- Bioscreen C is an absorbance based microplate reader that is used to measure growth curves of microorganisms (http://www.growthcurvesusa.com/description.html). As opposed to a conventional microplate reader, which can measure 96 wells at a time, honeycomb plates used in Bioscreen C have 100 wells, and two plates can be used at a time. The design of the honey comb plate ensures uniform temperature across the wells without any significant condensation/evaporation, thereby greatly reducing errors among replicates. However, it should be mentioned that in absence of Bioscreen C instrument, any conventional microplate reader or spectrophotometer with cuvette can also be used to perform this step successfully.
- If it is desirable to find out the absolute amount of protein required to achieve a particular growth inhibition, it can be done by purifying the protein of interest, estimating its concentration and then using a known amount of the purified protein as a standard/reference to estimate the amount of protein from the crude lysate at each given induction level.
Recipes
- Supplemented M9 medium (1 L)
11.28 g of M9 salts (Difco) (identical to the classical M9 salts recipe in Molecular Cloning by Maniatis)
1 ml 1 M MgSO4
5 µl 100 mM thiamine
10 ml 10% casamino acid
10 ml 20% glucose
Make up the volume to 1,000 ml, sterilize by filtration - Lysis buffer (10 ml)
1 ml 10x Bugbuster reagent
10 µl Benzonase nuclease
Make up the volume to 10 ml using buffer of choice - L(+) arabinose solution (concentrations used)
Start with 0.05%, and then do 4-fold serial dilution up to 1.22 x 10-5%, in addition to 0% arabinose
Acknowledgments
This work was funded by NIH RO1 GM111955.
References
- Adkar, B. V., Manhart, M., Bhattacharyya, S., Tian, J., Musharbash, M. and Shakhnovich, E. I. (2017). Optimization of lag phase shapes the evolution of a bacterial enzyme. Nature Ecology & Evolution 1: 0149.
- Bhattacharyya, S., Bershtein, S., Yan, J., Argun, T., Gilson, A. I., Trauger, S. A. and Shakhnovich, E. I. (2016). Transient protein-protein interactions perturb E. coli metabolome and cause gene dosage toxicity. Elife 5.
- Khlebnikov, A., Datsenko, K. A., Skaug, T., Wanner, B. L. and Keasling, J. D. (2001). Homogeneous expression of the P(BAD) promoter in Escherichia coli by constitutive expression of the low-affinity high-capacity AraE transporter. Microbiology 147(Pt 12): 3241-3247.
Article Information
Copyright
Bhattacharyya et al. This article is distributed under the terms of the Creative Commons Attribution License (CC BY 4.0).
How to cite
Readers should cite both the Bio-protocol article and the original research article where this protocol was used:
- Bhattacharyya, S., Bershtein, S. and Shakhnovich, E. I. (2017). Gene Dosage Experiments in Enterobacteriaceae Using Arabinose-regulated Promoters. Bio-protocol 7(14): e2396. DOI: 10.21769/BioProtoc.2396.
- Bhattacharyya, S., Bershtein, S., Yan, J., Argun, T., Gilson, A. I., Trauger, S. A. and Shakhnovich, E. I. (2016). Transient protein-protein interactions perturb E. coli metabolome and cause gene dosage toxicity. Elife 5.
Category
Microbiology > Microbial genetics > Gene expression
Molecular Biology > DNA > Gene expression
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link









