- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
A Simple and Rapid Assay for Measuring Phytoalexin Pisatin, an Indicator of Plant Defense Response in Pea (Pisum sativum L.)
Published: Vol 7, Iss 13, Jul 5, 2017 DOI: 10.21769/BioProtoc.2362 Views: 8060
Reviewed by: Ayelign M. AdalAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Quantitative Estimation of Auxin, Siderophore, and Hydrogen Cyanide Production in Halo and Drought-Tolerant Bacterial Isolates for Cucumber Growth
Zeinab Fotoohiyan and Ali Salehi Sardoei
Oct 5, 2025 1342 Views
Reproducible Emu-Based Workflow for High-Fidelity Soil and Plant Microbiome Profiling on HPC Clusters
Henrique M. Dias [...] Christopher Graham
Jan 20, 2026 403 Views
Abstract
Phytoalexins are antimicrobial substance synthesized in plants upon pathogen infection. Pisatin (Pisum sativum phytoalexin) is the major phytoalexin in pea, while it is also a valuable indicator of plant defense response. Pisatin can be quantitated in various methods from classical organic chemistry to Mass-spectrometry analysis. Here we describe a procedure with high reproducibility and simplicity that can easily handle large numbers of treatments. The method only requires a spectrophotometer as laboratory equipment, does not require any special analytical instruments (e.g., HPLC, mass spectrometers, etc.) to measure the phytoalexin molecule quantitatively, i.e., most scientific laboratories can perform the experiment.
Keywords: PisatinBackground
Plants have host resistance and nonhost resistance depending upon the nature of plant-pathogen interactions. Host resistance is mostly controlled by R genes and less durable, whereas nonhost resistance is generally a multi-gene trait and more durable in comparison with host resistance (Gill et al., 2015; Lee et al., 2017). The pea plant has served as a model system for research on the signals that trigger the nonhost defense response when challenged by incompatible pathogens that fall outside that species’ host range (Hadwiger, 2008). An indicator of this response in peas is the induction of a secondary metabolism to the isoflavonoid, pisatin. Pisatin has strong antifungal properties but its presence is a valuable indicator of plant defense response. Pisatin can be quantitated in various ways from classical organic chemistry procedures (Schwochau and Hadwiger, 1969) to Mass-spec analysis (Seneviratne et al., 2015). However, a procedure with high reproducibility and simplicity is described herein that can easily handle large numbers of treatments. The targeted tissue is the inside layer of an immature pea pod, called endocarp. This pristine cuticle-free tissue is capable of responding rapidly to candidate microbes or elicitor compounds to generate the pea defense response. The exposed epidermal layer of cells can be monitored for light microscope-visible or stained cellular component changes. The overall changes that culminate in pisatin accumulations can be determined by immersing the pod half in 5 ml of hexane for 4 h in the dark and subsequently allowing the decanted hexane to evaporate in the air flow of a hood. The residue remaining is dissolved in 1 ml of 95% alcohol and read at OD309 using a spectrophotometer: 1 OD309 = 43.8 µg pisatin/ml in 1 cm pathlength (Cruickshank and Perrin, 1961; Perrin and Cruickshank, 1965; Teasdale et al., 1974). This reading minus the background control tissue and the characteristic UV spectrum are essentially free from other hexane soluble components of the pea tissue. This protocol was used in our recent publications (Hadwiger and Tanaka, 2014 and 2017; Tanaka and Hadwiger, 2017).
Materials and Reagents
- Spatula–smooth narrow tip and smooth glass rod
- Plastic Petri dishes (60 x 15 mm) (Corning, catalog number: 351007 )
- Plastic container with wet Kimwipes inside for humidity
- Paper towel or Kimwipe
- Immature pea pods (1.5-2.0 cm in length) grown in sand and clay pots at 65-70 F under greenhouse conditions and freshly harvested (use within 3 h of applying a treatment). Remove calyx and retain briefly in sterile water (Figure 1). Endocarp will be used for the assay (see Note 1 in detail)
- Glass vials 30 ml
- Candidate elicitor solutions best dissolved in deionized water (For exceptions see Procedure 1)
- DMSO
- Hexane
- 95% ethanol
Equipment
- Adjustable pipettes (P-200 and P-1000 and corresponding tips)
- Flask 500 ml with 5 ml dispenser top or 5 ml pipet for dispensing hexane
- Glass beakers, 30 ml
- Room temperature dark cabinet space for pathogen or elicitor treatments (as described in step 3b)
- UV spectrometer (Shimadzu, model: UV160 )
- 1 cm Pathlength quartz cuvettes (Sigma-Aldrich, catalog number: C5178 )
Note: This product has been discontinued.
Procedure
- Preparation of elicitors
The selection of elicitors is by design open to innovation. Follow the directions of manufactures for solubility procedures. Water soluble compounds dissolved at near neutral pH are preferred. When solubility depends on ethanol, DMSO etc., there must be suitable control applications with only the respective solvent. Incompatible pathogen can be a positive control for an inducer of nonhost resistance. See Note 2 in detail.
- Preparation of pod halves
- Harvest pods and remove calyx as shown in Figure 1. Hold these pods in a sterile deionized water reservoir to keep the tissue moist.
- Select uniform sized and conditioned pea pods from their water reservoir.
- Separate the pod halves with a smooth spatula avoiding wounding as much as possible.
- Fresh weight of pod halves is determined.
- Lay endocarp (inner) surface layer up in a Petri dish (Figure 1).
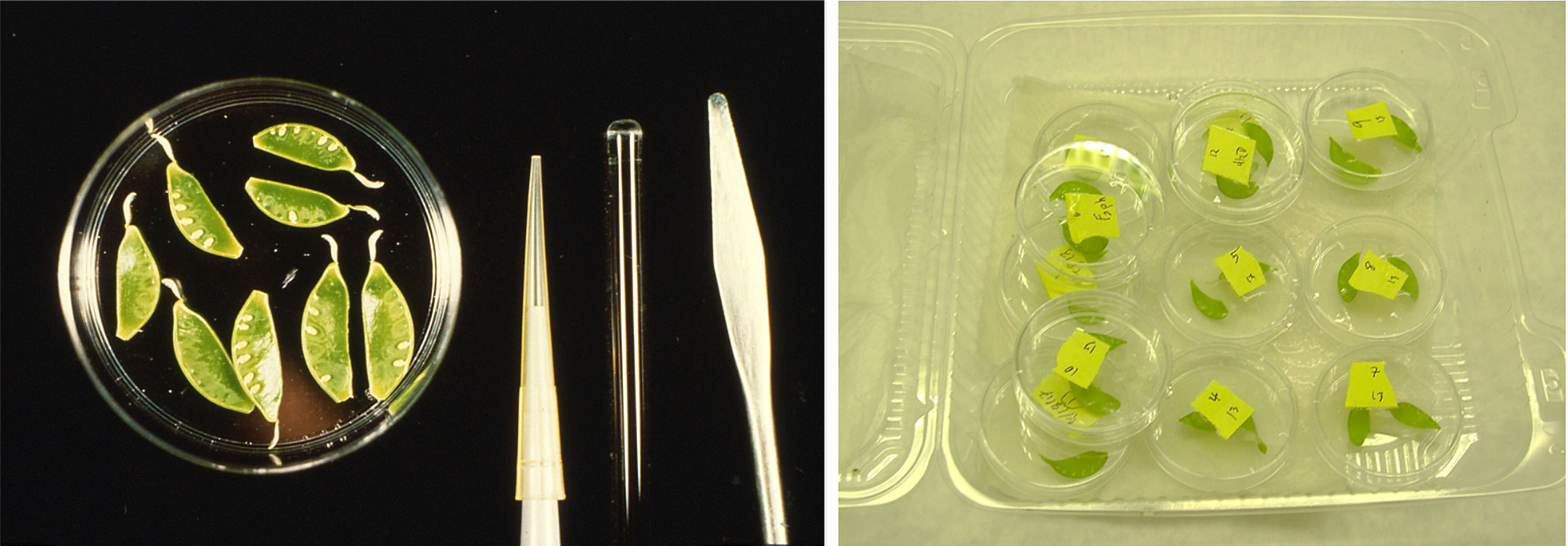
Figure 1. Pea endocarp. The inside layer (endocarp) of an immature pea pod is cuticle-free tissue capable of responding rapidly to microbes or elicitor compounds. Right picture shows endocarp tissues treated in an elicitor solution in Petri dishes. Incubation is performed in a plastic container with wet paper towels to maintain humidity.
- Harvest pods and remove calyx as shown in Figure 1. Hold these pods in a sterile deionized water reservoir to keep the tissue moist.
- Application of elicitors
- Apply 25 µl of elicitor candidate solution and lightly distribute over the entire surface with a glass rod. For the control, apply the same solvent used for dissolving the elicitor.
- Treated pods are retained in a plastic container with wet paper towels (Figure 1) to maintain humidity and then incubate in the dark or moderate light for up to 24 h.
- Apply 25 µl of elicitor candidate solution and lightly distribute over the entire surface with a glass rod. For the control, apply the same solvent used for dissolving the elicitor.
- Extraction and measurement of pisatin
- Pods are transferred to 30 ml glass vials using forceps and immersed in 5 ml of hexane for 4 h in the dark. Typically, 400 mg fresh weight per 5 ml of hexane.
- The hexane is decanted off into 30 ml beakers and the hexane evaporated in the air stream of a hood in low light because pisatin is not stable in bright light (typically light strength in the lab is not incandescent).
- One milliliter of 95% ethanol is added to the residue and read at 309 nm in a cuvette using spectrophotometer.
- To insure purity, a UV spectrum is measured in the range of 220-320 nm to verify the characteristic pisatin spectrum (Figure 2). See Note 3 in detail.
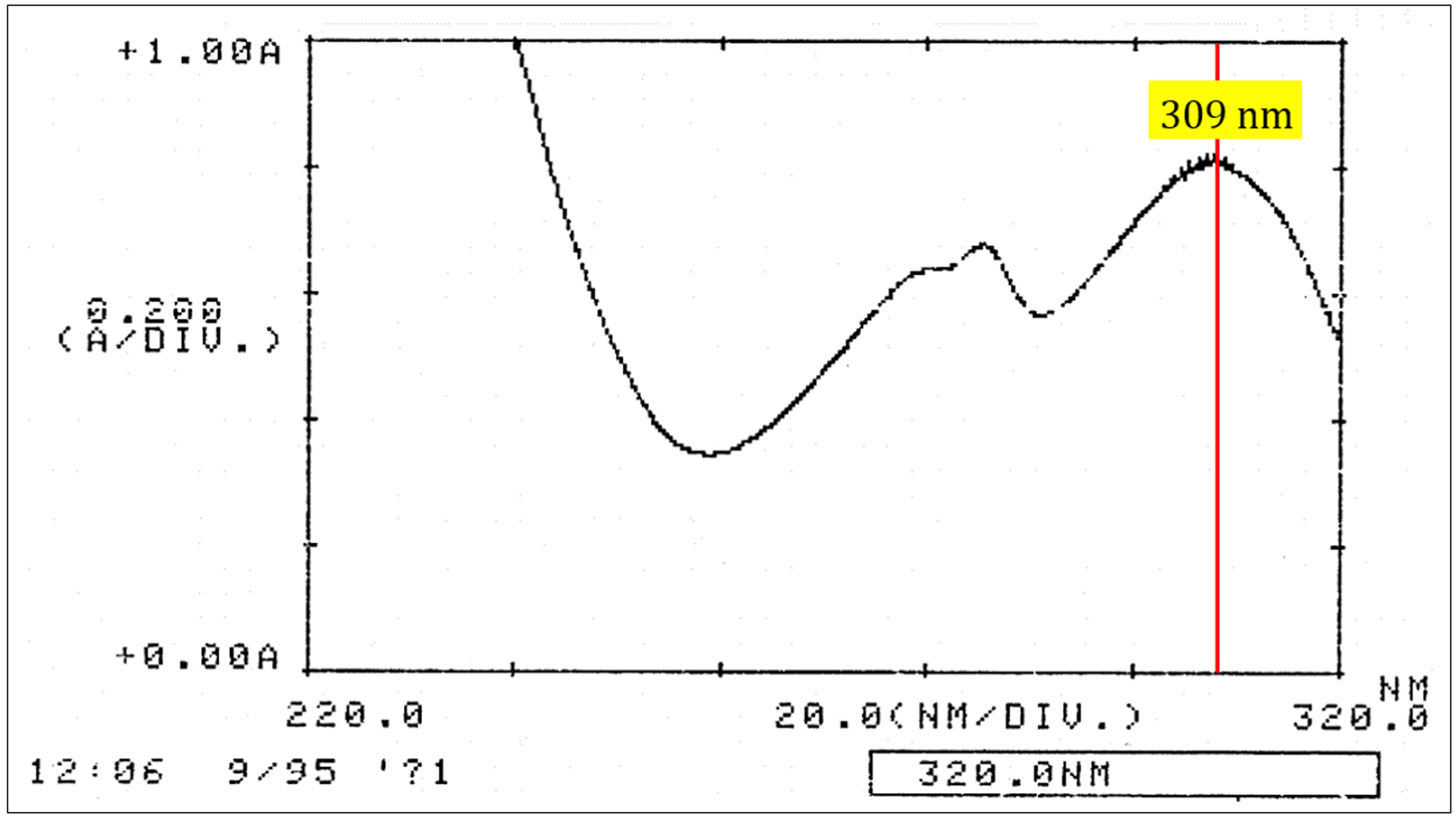
Figure 2. Pisatin spectrum. UV spectrum in the range of 220-320 nm was run to verify the purity of pisatin.
- Pods are transferred to 30 ml glass vials using forceps and immersed in 5 ml of hexane for 4 h in the dark. Typically, 400 mg fresh weight per 5 ml of hexane.
Data analysis
After subtracting the OD309 value of non-treated control, the numbers are converted based on the equation: 1.0 OD309 unit = 43.8 µg/ml pisatin in 1 cm pathlength (see Note 4 in detail). Data should be shown with pisatin quantity (µg) per fresh weight of tissues (g). Data obtained should be analyzed using ANOVA followed by Student’s t-test. Difference with P < 0.05 is considered significant.
Notes
- The pea endocarp tissue has some potential to condition or partially take up small percentages of insoluble materials in suspension, such as the cell wall fragments released by fungal spores, detected in electron microscope view (Hadwiger et al., 1981).
- Effect of elicitors can be compared with that of an incompatible fungal pathogen, e.g., Fusarium solani f.sp. phaseoli (Fsph), which is a pathogen for bean (not for pea). An example result was shown in Figure 3, in which the data show that no detectable pisatin above background in water-treated tissue, and accumulation of pisatin induced by a fungal wall carbohydrate (i.e., chitosan) and by an authentic inducer of nonhost resistance in pea, a fungal pathogen of bean (i.e., Fsph).
- Pisatin has been purified from the final step with both thin layer chromatography and mass spectrometry (Teasdale et al., 1974; Seneviratne et al., 2015). The assay is accurate because pisatin only absorbs at 309 nm.
- Pisatin in ethanol has a characteristic UV absorption spectrum with two peaks at 286 nm and 309 nm (Figure 2). When pisatin is the only light-absorbing compound in the solution, the ratio OD309 to OD286 is 1.47 (Cruickshank and Perrin, 1961).
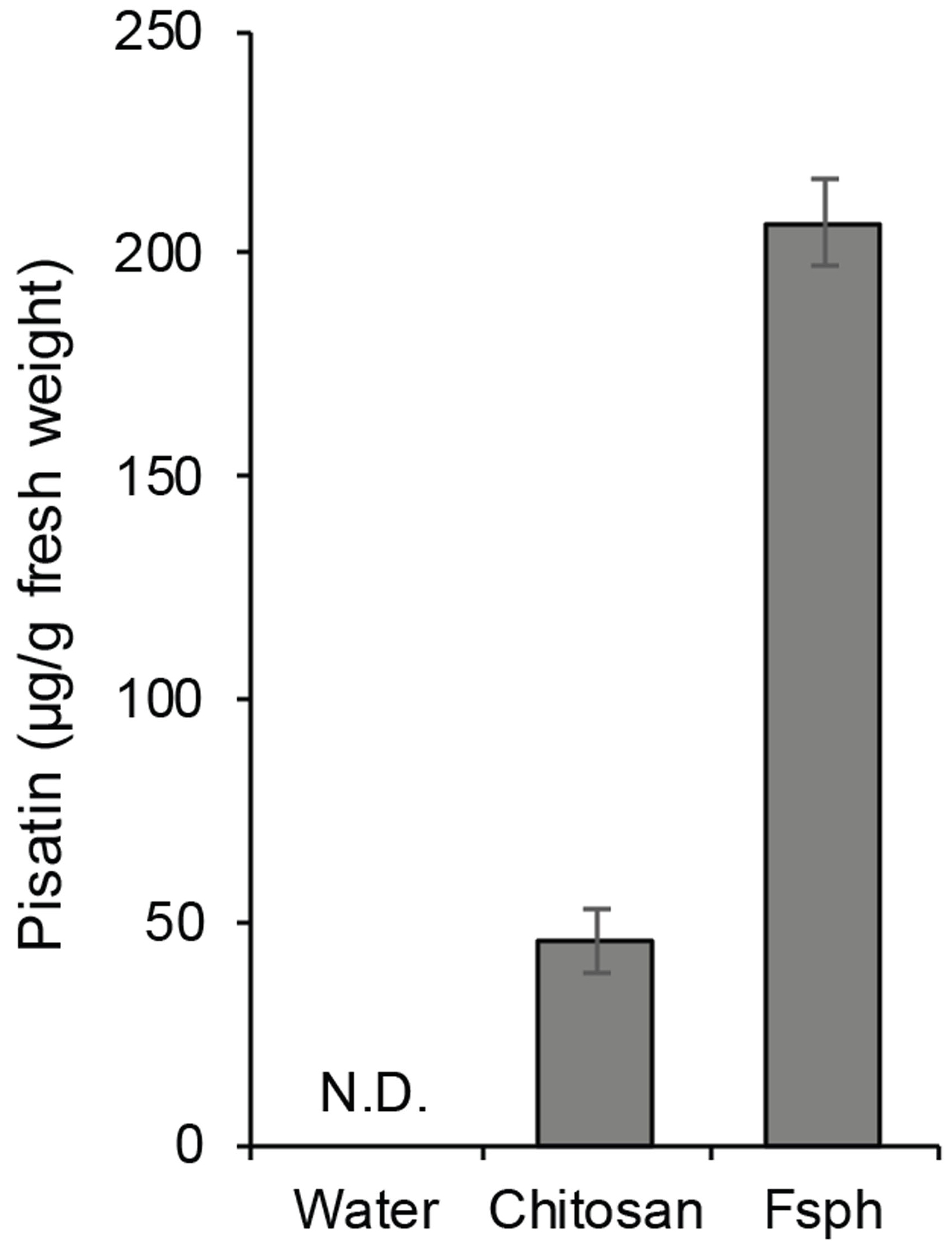
Figure 3. An example result of pisatin measurement. Pisatin was extracted and measured from endocarps (n = 3) after 24 h incubation with water (control), 1 mg/ml chitosan (elicitor), or 4 x 106 spores of Fsph (Fusarium solani f.sp. phaseoli). Data was modified from our previous publication (Hartney et al., 2006). There were statistically significant differences between group means as determined by one-way ANOVA (P < 0.05).
Acknowledgments
This work was partly supported by Biologically-Intensive Agriculture and Organic Farming (BIOAg) grant from the Center for Sustaining Agriculture and Natural Resources (CSANR) at Washington State University. PPNS No. 0740, Department of Plant Pathology, College of Agricultural, Human, and Natural Resource Sciences, Agricultural Research Center, Hatch Project No. WNP01844, Washington State University, Pullman, 99164-6430, USA.
References
- Cruickshank, I. A. M. and Perrin D. R. (1961). Studies on phytoalexins III. The isolation, assay, and general properties of a phytoalexin from Pisum sativum L. Aust J Biol Sci 14: 336-348.
- Gill, U. S., Lee, S. and Mysore, K. S. (2015). Host versus nonhost resistance: distinct wars with similar arsenals. Phytopathology 105: 580-87.
- Hadwiger, L. A. (2008). Pea-Fusarium solani interactions contributions of a system toward understanding disease resistance. Phytopathology 98: 372-379.
- Hadwiger, L. A., Beckman, J. M. and Adams, M. J. (1981). Localization of Fungal components in the Pea-Fusarium interaction detected immunochemically with anti-chitosan and anti-fungal cell wall antisera. Plant Physiol 67: 170-175.
- Hadwiger, L. A. and Tanaka, K. (2014). EDTA a novel inducer of pisatin, a phytoalexin indicator of the non-host resistance in peas. Molecules 20: 24-34.
- Hadwiger, L. A. and Tanaka, K (2017). Non-host resistance: DNA damage is associated with SA signaling for induction of PR genes and contributes to the growth suppression of a pea pathogen on pea endocarp tissue. Front Plant Sci 8: 446.
- Hartney, S., Carson, J. and Hadwiger, L. A. (2006). The use of chemical genomics to detect functional systems affecting the non-host disease resistance of pea to Fusarium solani f.sp. phaseori. Plant Sci 172: 45-56.
- Lee, H-A., Lee, H-Y., Seo, E., Lee, J., Kim, S-B., Oh, S., Choi, E., Choi, e., Lee, s. E. and Choi, D. (2017). Current understandings of plant nonhost resistance. Mol Plant-Microbe Interact 30: 5–15
- Perrin, D. R. and Cruickshank, I. A. M. (1965). Studies on phytoalexins VII. Chemical stimulation of pisatin formation in Pisum sativum L. Aust J Biol Sci 18: 803-816.
- Schwochau, M. E. and Hadwiger, L. A. (1969). Regulation of gene expression by actinomycin D and other compounds which change the conformation of DNA. Arch Biochem Biophys 134: 34-41.
- Seneviratne, H. K., Dalisay, D. S., Kim, K. W., Moinuddin, S. G., Yang, H., Hartshorn, C. M., Davin, L. B. and Lewis, N. G. (2015). Non-host disease resistance response in pea (Pisum sativum) pods: Biochemical function of DRR206 and phytoalexin pathway localization. Phytochemistry 113: 140-148.
- Tanaka, K. and Hadwiger, L. A. (2017). Nonhost resistance: reactive oxygen species (ROS) signal causes DNA damage prior to the induction of PR genes and disease resistance in pea tissue. Physiol Mol Plant Pathol 98: 18-24.
- Teasdale, J., Daniels, G., Davis, W. C., Eddy, R., Hadwiger, L. A. (1974). Physiological and cytological similarities between disease resistance and cellular incompatibility responses. Plant Physiol 54: 690-695.
Article Information
Copyright
© 2017 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Hadwiger, L. A. and Tanaka, K. (2017). A Simple and Rapid Assay for Measuring Phytoalexin Pisatin, an Indicator of Plant Defense Response in Pea (Pisum sativum L.). Bio-protocol 7(13): e2362. DOI: 10.21769/BioProtoc.2362.
Category
Plant Science > Plant immunity > Host-microbe interactions
Cell Biology > Cell metabolism > Other compound
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link