- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Social Observation Task in a Linear Maze for Rats
Published: Vol 7, Iss 13, Jul 5, 2017 DOI: 10.21769/BioProtoc.2361 Views: 7307
Reviewed by: Soyun KimQing YanMarina Allerborn

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Pupillometry: A Simple and Automatic Way to Explore Implicit Cognitive Processing
Tian Yuan [...] Yi Jiang
Apr 5, 2025 1294 Views
The Mouse Social Frailty Index (mSFI): A Standardized Protocol
Charles W. Collinge [...] Alessandro Bartolomucci
Apr 20, 2025 1809 Views
Training Mice to Perform Attentional Set-Shifting Under Head Restraint
Katarina Kalajzic [...] Timothy Spellman
Sep 5, 2025 1470 Views
Abstract
Animals often learn through observing their conspecifics. However, the mechanisms of them obtaining useful knowledge during observation are beginning to be understood. This protocol describes a novel social observation task to test the ‘local enhancement theory’, which proposes that presence of social subjects in an environment facilitates one’s understanding of the environments. By combining behavior test and in vivo electrophysiological recording, we found that social observation can facilitate the observer’s spatial representation of an unexplored environment. The task protocol was published in Mou and Ji, 2016.
Keywords: HippocampusBackground
Social learning is defined as acquiring new knowledge through observing or interacting with others (Heyes and Galef, 1996; Bandura, 1997; Meltzoff et al., 2009). One form of social learning utilized by many species is the so-called ‘local enhancement’ (Heyes and Galef, 1996): an animal’s understanding of an environment is facilitated by the presence of other social subjects in the same environment. Animals achieve local enhancement possibly by heightened attention, acquiring environmental attributes such as safety or food availability, or other unspecified means (Zajonc, 1965; Heyes and Galef, 1996; Zentall, 2006). The hypothesis predicts that the presence of social subjects in an environment impacts other animals’ neural processing of information related to the environment, therefore facilitate their understanding of the environment.
It has been shown that spatial information of an environment is represented by hippocampal place cells (O’Keefe and Dostrovsky, 1971; Wilson and McNaughton, 1993; Burgess and O’Keefe, 2003) in rodents and humans. Place cells become active at specific locations of a given environment, called place fields. We asked how an observer’s place cell sequence representing an environment can be influenced by another rat navigating in the environment, even if the observer is located in a physically different environment. This protocol is designed to explore the neural basis of such local enhancement effect of social observation. Specifically, we monitored the hippocampus place cells in observer rats as they stayed in a small box while a demonstrator rat was running on a separate, nearby linear track, and then later when observer rats were running the same track themselves. Our results show that observer’s place cell sequences during track running also appeared in the box during observation, but only when a demonstrator was present on the track. Observer’s running speed, number of run laps and place cells’ specificity are significantly higher than those in control animals.
Materials and Reagents
- 3-6 month old male Long Evans rats, 450-550 g
- 4x diluted sweetened condensed milk (Eagle Brand) are used for reward
- 70% ethanol for cleaning the maze between daily training sessions
Equipment
- A 2-m long linear track made of galvanized steel (Figures 1A and 1B)
- A small 25 (length) x 25 (width) x 40 (height) cm box. Three sides of the box has opaque, high (40 cm) walls, leaving only one side open toward the track (Figure 1C)
- Milk wells located at the two ends of the linear track. Milk reward was remotely delivered by syringe and tubing from behind a curtain
- Curtain separating the experimenter and recording setup (Figure 1D)
- A 60 (length) x 60 (width) x 100 (height) cm rest box. The rest box was placed ~1 m away from the track. Animals were placed in a ceramic plate (20 cm in diameter) on top of a 30-cm tall flower pot, located at the center of the enclosed rest box for resting (Figure 1E)
- The animal’s position was tracked by red and green LED mounted over on the head. Position data were recorded by a ceiling camera at 33 Hz
- In vivo extracellular recording equipment is described earlier in Mou and Ji, 2016. Tetrode recording was made by a Digital Lynx acquisition system (Neuralynx, model: Digital Lynx Acquisition System ). Spikes from single neurons were sampled at 32 kHz and online-filtered between 600 Hz and 9 kHz. Local field potentials (LFPs) were sampled at 2 kHz and online-filtered between 0.1-1 kHz

Figure 1. Social observation apparatus. A. Schematic depiction of the recording setting; B. Linear track; C. Observation box; D. Curtain; E. Flower pot in a rest box.
Software
- Customized MATLAB script
Procedure
Note: All experimental procedures followed the guidelines by the National Institute of Health and were approved by the Institutional Animal Care and Use Committee at Baylor College of Medicine.
- 3-6 month-old male Long Evans rats, weighted between 450 g to 550 g, are housed 2-3 per cage and handled daily for ~7 days.
- After the acclimation period, demonstrator rats are food restricted to 85-90% of their baseline body weight before experiment. In the meantime, they are trained to run the linear track back and forth for milk reward for at least one week. Training session takes place once a day and lasted about 20 min. An adult rat can achieve satisfactory behavioral performance (continuous run back and forth without prolonged pause) within 2-3 days.
- While demonstrator rats are being trained, non-trained naïve rats are implanted with hyperdrive that contains 15 independently movable tetrodes and one reference electrode. Tetrodes target the right dorsal hippocampal CA1 region (coordinates: anteroposterior -3.8 mm, mediolateral 2.4 mm relative to bregma). During surgery, all tetrodes are placed right above the surface of exposed brain tissue without touching it. Then tetrodes are advanced individually to reach dorsal CA1 area. Since the dorsal CA1 area is not flat, the final depths vary among individual tetrodes, but approximately 2 mm below the dura. The reference tetrode is placed in white matter above dorsal CA1 pyramidal cell layer, approximately 1.7 mm below the dura.
- After having fully recovered from surgery (typically within 3 days), the implanted rats are food deprived to 85-90% of their baseline weight.
- During the following 3-4 weeks after implantation, tetrodes are slowly advanced to the CA1 pyramidal layer until sharp-wave ripples signal were observed (Figure 2). The reference tetrode is placed in the white matter above the CA1. Recordings are not conducted within 24 h after tetrodes movement.
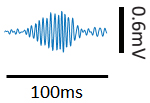
Figure 2. A representative LFP sharp-wave ripple - Pre-recording training phase: Each rat is placed in the observation box for 2 or 3 days, 15-30 min each day. For a group of implanted rats, there is a well-trained demonstrator running on the track in this pre-recording phase. For the other group of implanted rats, the track is left empty without a demonstrator.
- Recording procedure: The recording starts and lasts for 6-12 consecutive days after pre-recording training. The recording procedure is depicted in Figure 3. A typical recording took ~2 h. Each recording consists of three sessions.
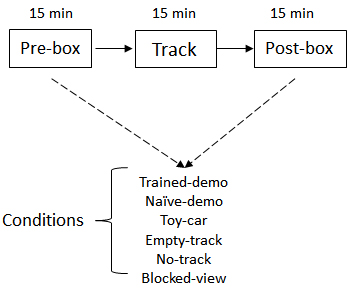
Figure 3. Schematic of daily recording procedure. The Pre- and Post-box sessions were configured with various conditions.
For the group of rats that have watched a well-trained demonstrator in the pre-recording training phase, on the first recording day:- The implanted rats first stay in the observation box while a well-trained demonstrator is running the linear track for 15 min (Pre-box session).
- Then the implanted rat runs the linear track for the first time (Track session).
- Then the implanted rat stays in the observation box again while the well-trained demonstrator is running the track (Post-box session).
Note: The recorded rats have never been exposed to the track before the first recording day.
In each of the following days, the Pre-box and Post-box session is set up in various ways as the following, while the Track session remains the same. Each condition is recorded for 1-3 days.- Empty-track: removing the demonstrator from the track.
- No-track: removing both the track and demonstrator.
- Naïve-demo: replacing the demonstrator with a naïve demonstrator that have never been exposed to the track.
- Toy-car: replacing the demonstrator with a toy car remotely controlled by the experimenter behind the curtain. The car is maneuvered to move at a speed comparable to a rat’s. The toy car stops when it reaches the end of the track then reverses its direction.
- Blocked-view: implanted rats stay in the box but with the view blocked while a well-trained demonstrator is running on the track. In this condition, the observation box is rotated 180° such that the opening side now is facing a nearby wall of the room 20 cm away. The implanted rat in the box can not see either the track or the demonstrator, but has access to the auditory and olfactory information associated with the demonstrator.
For the other group of rats that have seen only the empty track in the pre-recording training, the Pre-box and Post-box sessions on the first day are under Empty-track condition. In the following days, the Pre-box and Post-box sessions are replaced by Trained-demo and other conditions as described above.
- Empty-track: removing the demonstrator from the track.
- The implanted rats first stay in the observation box while a well-trained demonstrator is running the linear track for 15 min (Pre-box session).
Data analysis
- We tracked animals’ positions using the red and green LED mounted on their heads. Position data were recorded by a ceiling camera and analyzed off-line using customized MATLAB script. The observer rats’ behavior in the Track session was quantified by mean running speed and number of running laps per trajectory (one running direction, Figure 4A). Our data show that both parameters in the observer rats after watching the well-trained demonstrator were significantly greater than those watching the empty track during pre-recording training phase.
- All extracellular recording data, including spike timestamps, LFPs, were digitized and analyzed off-line using customized MATLAB routines (MATLAB routines are available upon reasonable request). After watching well-trained demonstrator, observer rats’ place cell firing appeared to be less dispersed than those watching Empty-track. Place cells’ firing sparsity measured by spatial information was significantly greater in the former group (Figure 4B). Taken together, our data suggest an improvement in understanding of a novel environment measured by track-running performance and place field development. Detailed analysis can be found in Mou and Ji, 2016.
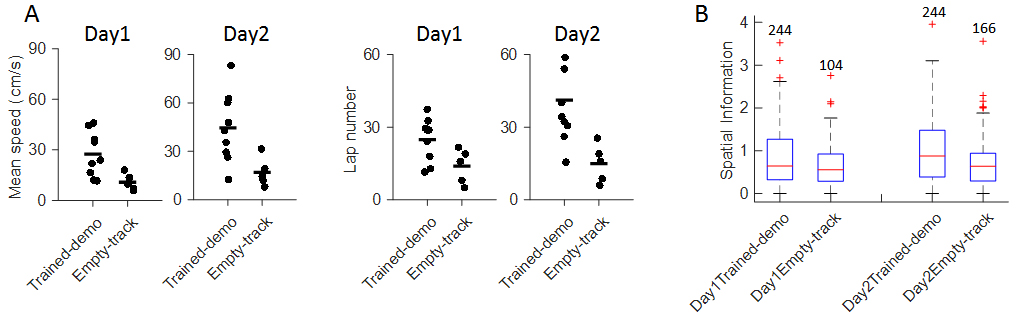
Figure 4. Experience in the box improved behavioral performance and novel place field development on the track. A. Mean running speed and number of laps per trajectory in Track sessions of first two days (Day1, Day2) for the Trained-demo (N = 9) and Empty-track (N = 5) rats; B. Spatial information for all active cells under each condition (Trained-demo, Empty-track) on each day (Day1, Day2). Number on top of each bar: the number of cells. There was a significant main effect between the conditions (F(1,791) = 21, p = 0, Two-way ANOVA), but not between days (F(1,791) = 1.4, p = 0.25). Reproduced from Mou and Ji, 2016 with permission.
Acknowledgments
The authors would like to thank the entire Ji lab for help on constructing and configuring the apparatus, and for suggestions on preparation of the manuscript. This work was supported by grants NIMH R01MH106552, Simons Foundation 273886 to D.J.
References
- Bandura, A. (1997). Social learning theory. General Learning Press.
- Burgess, N. and O’Keefe, J. (2003). Neural representations in human spatial memory. Trends Cogn Sci 7(12): 517-519.
- Heyes, C. M. and Galef Jr, B. G. (1996). Social learning in animals: the root of culture. Academic Press.
- Meltzoff, A. N., Kuhl, P. K., Movellan, J. and Sejnowski, T. J. (2009). Foundations for a new science of learning. Science 325(5938): 284-288.
- Mou, X and Ji, D. (2016). Social observation enhances cross-environment activation of hippocampal place cell patterns. eLife 5: e18022.
- O’Keefe, J. and Dostrovsky, J. (1971). The hippocampus as a spatial map. Preliminary evidence from unit activity in the freely-moving rat. Brain Res 34(1):171-175.
- Wilson, M. A. and McNaughton, B. L. (1993). Dynamics of the hippocampal ensemble code for space. Science 261(5124): 1055-1058.
- Zajonc, R. B. (1965). Social facilitation. Science 149(3681): 269-274.
- Zentall, T. R. (2006). Imitation: definitions, evidence, and mechanisms. Anim Cogn 9(4): 335-353.
Article Information
Copyright
Mou and Ji. This article is distributed under the terms of the Creative Commons Attribution License (CC BY 4.0).
How to cite
Readers should cite both the Bio-protocol article and the original research article where this protocol was used:
- Mou, X. and Ji, D. (2017). Social Observation Task in a Linear Maze for Rats. Bio-protocol 7(13): e2361. DOI: 10.21769/BioProtoc.2361.
- Mou, X and Ji, D. (2016). Social observation enhances cross-environment activation of hippocampal place cell patterns. eLife 5: e18022.
Category
Neuroscience > Behavioral neuroscience > Cognition
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link








