- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Inoculation of Rice with Different Pathogens: Sheath Blight (Rhizoctonia solani), Damping off Disease (Pythium graminicola) and Barley Powdery Mildew (Blumeria graminis f. sp. hordei)
Published: Vol 6, Iss 24, Dec 20, 2016 DOI: 10.21769/BioProtoc.2070 Views: 14308
Reviewed by: Marisa RosaAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Quantification of the Composition Dynamics of a Maize Root-associated Simplified Bacterial Community and Evaluation of Its Biological Control Effect
Ben Niu and Roberto Kolter
Jun 20, 2018 10488 Views

Tracking Root Interactions System (TRIS) Experiment and Quality Control
Hassan Massalha [...] Asaph Aharoni
Apr 20, 2019 7336 Views

A Quick Method for Screening Biocontrol Efficacy of Bacterial Isolates against Bacterial Wilt Pathogen Ralstonia solanacearum in Tomato
Heena Agarwal [...] Niraj Agarwala
Nov 20, 2020 5547 Views
Abstract
To prevent yield losses in plant cultivation due to plant pathogens, it is an important task to find new disease resistance mechanisms. Recently, Weidenbach et al. (2016) reported about the capacity of the rice gene OsJAC1 to enhance resistance in rice and barley against a broad spectrum of different pathogens. Here, we describe the respective protocols used by Weidenbach and colleagues for inoculation of rice with the basidiomycete Rhizoctonia solani, the oomycete Pythium graminicola and the ascomycete Blumeria graminis f. sp. hordei (Bgh).
Keywords: RiceBackground
Following the observation that transcripts of the rice gene OsJAC1 accumulated after pathogen attack or treatment with chemical resistance inducers, transgenic rice plants with constitutive expression or knockout of this gene were investigated in response to inoculation with fungal pathogens. To cover a broad pathogen spectrum, economically important representatives of ascomycete fungi (Magnaporthe oryzae, Blumeria graminis f. sp. hordei), basidiomycete fungi (Rhizoctonia solani) and oomycetes (Pythium graminicola) were chosen. Using protocols for standardized and even inoculation, an enhanced disease resistance phenotype was established for the transgenic plants constitutively expressing OsJAC1 while the respective knockout plants showed enhanced susceptibility (Weidenbach et al., 2016). A detailed bio-protocol for M. oryzae inoculation on rice is already available (Akagi et al., 2015), therefore we focus here on the inoculation protocols for R. solani, P. graminicola and Bgh.
As causal agent of rice sheath blight R. solani is one of the two most important rice diseases (Lee and Rush, 1983). The fungus overwinters as sclerotia or mycelium in the soil and infects rice sheaths by cuticular penetration or through stomata resulting in lesions, necrosis and leaf death (Ou, 1985). Of different methods available for R. solani inoculation, in the present study a time- and space-saving detached leaf assay is described, that was slightly modified from a protocol provided by Monika Höfte (Ghent University, personal communication).
P. graminicola is a causal agent of seedling damping-off and root rot resulting in stunting and yield loss (Hendrix and Campbell, 1973). In this study P. graminicola was inoculated on rice roots growing on agar plates using a protocol adapted from Van Buyten and Höfte (2013).
Fungi of the B. graminis species invade epidermal cells of their host plants with specialized feeding structures called haustoria. All other parts of the fungal mycelium are developed on the leaf surface thereby causing typical powdery mildew disease symptoms. The disease is of permanent importance in cereal agriculture (Dean et al., 2012). Rice plants do not have any powdery mildew pathogens. However, rice can be inoculated with Bgh which allows the investigation of nonhost resistance mechanisms (e.g., Abbruscato et al., 2012; Weidenbach et al., 2016). For an evenly distributed inoculation density, rice leaves have to be fixed and inoculated in a settling tower, as described for barley (Weidenbach et al., 2014).
Materials and Reagents
- Square Petri dishes, (120 x 120) x 17 with vents (Greiner Bio One, catalog number: 688102 )
- Round Petri dishes, 94 x 16 with vents (Greiner Bio One, catalog number: 633180 )
- Filter paper, e.g., Rotilabo-round filters, type 113A, 125 mm (Carl Roth, Germany)
- 50 ml tubes (Cellstar tubes) (Greiner Bio One, Germany)
- Surgical 3M Micropore tape (3M Deutschland, Germany)
- Aluminum foil
- Cover glass
- Removable adhesive labels, e.g., multipurpose labels (Avery Zweckform, Germany)
- Rice (Oryza sativa L. japonica), transgenic plants and respective wild type
Note: Cultivar Nipponbare used in this study was kindly provided by the Center de cooperation internationale en recherche agronomique pour le developpement (CIRAD, Montpellier, France). - Barley (Hordeum vulgare) susceptible to Bgh.
Note: Cultivar Ingrid used in this study was kindly provided by Paul Schulze Lefert (Max-Planck Institute for Plant Breeding Research, Cologne, Germany). - Rhizoctonia solani.
Note: The isolate NL84 used in this study was kindly provided by Monica Höfte (Ghent University, Gent, Belgium). - Pythium graminicola.
Note: The isolate 132 used in this study was kindly provided by Monica Höfte (Ghent University, Gent, Belgium). - Blumeria graminis f. sp. hordei.
Note: In this study race K1 (Hinze et al., 1991) was used, which was kindly provided by Paul Schulze-Lefert (Max Planck Institute for Plant Breeding Research, Cologne, Germany). - Potato extract glucose agar (PDA) (Carl Roth, Germany)
- Distilled water
- Gamborg B5 medium including vitamins (Duchefa Biochemie, catalog number: G0210.0025 )
- Agar-Agar, Kobe I (Carl Roth, Germany)
- Sodium hypochlorite (or commercially available bleach water, ‘Eau de Javel’)
Equipment
- Scissor, scalpel
- Plant growth chamber for rice cultivation (settings: 15 h light/9 h dark period, 24 °C, 75-80% humidity)
- Plant growth cabinet for constant Bgh propagation on barley (settings:16 h light/8 h dark period, 18 °C, 65% humidity)
- Incubator for maintenance of fungal cultures (22 °C, constant darkness)
- Horizontal shaker
- Thoma cell counting chamber (Marienfeld, Germany)
- Light microscope (ca. 150x magnification)
- Trays for Bgh inoculation (min. 30 x 15 cm2)
Note: Area should at least correspond to the magnitude of two-week old rice plants. - Spore settling tower (e.g., a plastic tent of ca. 1 m height with an inoculation opening at the top, see example in Figure 3C)
Procedure
- Inoculation protocol for Rhizoctonia solani

Figure 1. Images of R. solani inoculation. A. Typical R. solani culture on PDA seven days after sub-cultivation. B. Four-week old rice plants with fully expanded fourth leaves. C. Four-week old rice plant with fourth leaf fully expanded (leaf numbers indicated). D. Rice leaves placed on filter paper and inoculated with agar blocks overgrown with R. solani. E. Example of necrotic lesions developing on an inoculated leaf.- Cultivate R. solani on potato extract glucose agar (PDA) at 22 °C in the dark. For maintenance, transfer agar blocks overgrown with fungal mycelium to fresh PDA plates every two weeks. For inoculation, use a R. solani culture seven days after sub-cultivation (Figure 1A).
- Grow rice plants for approx. 4 weeks until full expansion of the fourth leaf (Figures 1B and 1C).
- Prepare square Petri dishes (12 x 12 cm) lined with filter paper. Moisten the filter paper with distilled water.
- Cut off the upper 10 cm of the fourth leaves of different rice plants and immediately place them on the moist filter paper.
- Cut small agar blocks (ca. 0.5 x 0.5 cm) overgrown with fungal mycelium from a 7-day old R. solani culture. For inoculation, place one agar block in the middle of each rice leaf (Figure 1D).
- Incubate the Petri dishes with the inoculated leaves in the plant chamber. Avoid desiccation of the leaves by daily moistening the filter paper with distilled water (Note 1).
- Observe the development of necrotic lesions (Figure 1E) within four days after inoculation.
- Cultivate R. solani on potato extract glucose agar (PDA) at 22 °C in the dark. For maintenance, transfer agar blocks overgrown with fungal mycelium to fresh PDA plates every two weeks. For inoculation, use a R. solani culture seven days after sub-cultivation (Figure 1A).
- Inoculation protocol for Pythium graminicola
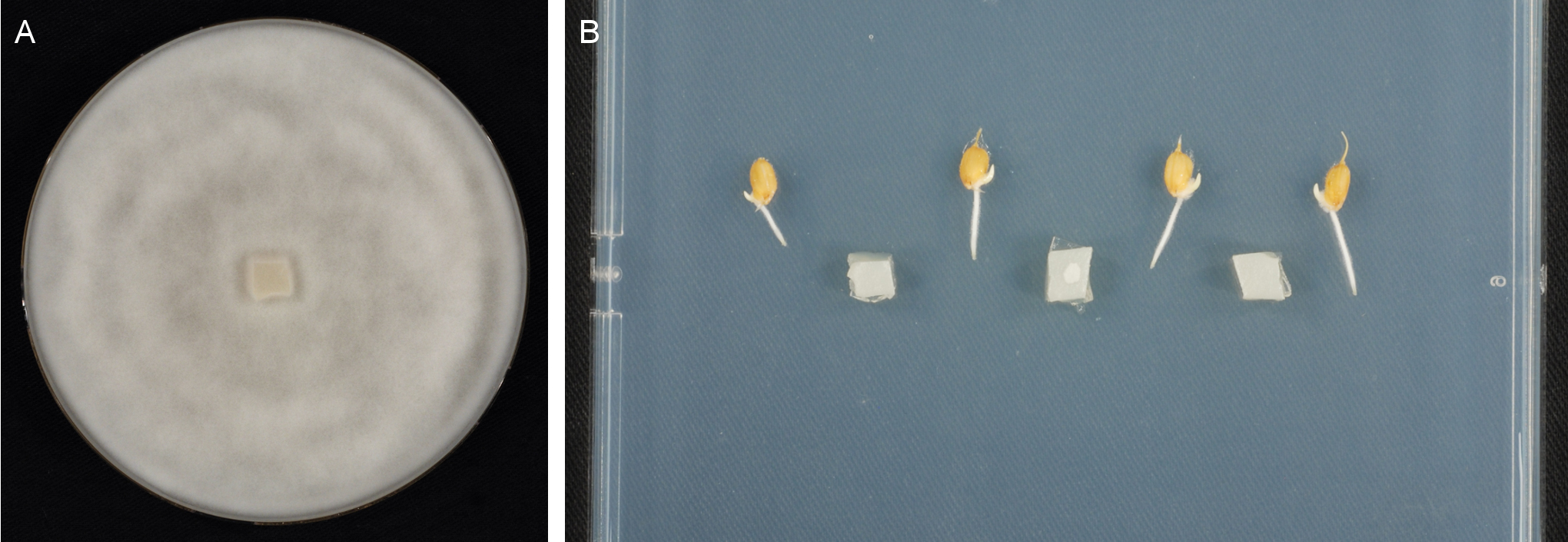
Figure 2. Images of P. graminicola inoculation. A. Typical P. graminicola culture on PDA seven days after sub-cultivation. B. Rice seedlings on Gamborg B5 medium inoculated with agar blocks overgrown with P. graminicola.- Cultivate P. graminicola on potato extract glucose agar (PDA) at 22 °C in the dark. For maintenance, transfer agar blocks (ca. 0.5 x 0.5 cm) overgrown with fungal mycelium to fresh PDA plates every two weeks. For inoculation, use P. graminicola cultures seven days after sub-cultivation (Figure 2A).
- Prepare square Petri dishes (12 x 12 cm) with 1.5 % agar in Gamborg B5 medium.
- Fill rice seeds in a 50 ml tube and add 4 % sodium hypochlorite for surface-sterilization, shake for 10 min. Subsequently, wash three times with sterile distilled water.
- Place 3-6 seeds in a line on one Gamborg B5 agar-plate.
- Seal the plates with 3M micropore tape and protect root growth from light by covering with aluminum foil.
- Incubate the plates upright in a plant growth chamber (conditions see above) until roots have grown to a length of ca. 1 cm (after approx. 5 days).
- For inoculation, cut small agar blocks (ca. 0.5 x 0.5 cm) from the marginal zone of a 7 days old P. graminicola culture. Place one agar block between the roots of adjacent plants (Figure 2B). Incubate the plates in a growth chamber as before.
- Monitor the disease development for 1 to 2 weeks after inoculation.
- Cultivate P. graminicola on potato extract glucose agar (PDA) at 22 °C in the dark. For maintenance, transfer agar blocks (ca. 0.5 x 0.5 cm) overgrown with fungal mycelium to fresh PDA plates every two weeks. For inoculation, use P. graminicola cultures seven days after sub-cultivation (Figure 2A).
- Inoculation protocol for Blumeria graminis f. sp. hordei (Bgh)

Figure 3. Images of Bgh inoculation. A. Two-week old rice plants with second leaves fixed on a tray. B. Barley plants heavily infected with Bgh (with white powdery mildew pustules on primary leaves). C. Example for a settling tower that was used for Bgh inoculation (plants were fixed on a tray which was placed at the bottom of the tower). At the top an opening is visible through which infected barley plants can be introduced into the tower.- As the fungus is an obligate biotroph, maintainance has to be done on barley plants in a growth cabinet separated from the test plants. Weekly transfer of Bgh onto fresh, one-week old plants is done by shaking off conidiospores from heavily infected barley plants (Figure 3B).
- Grow rice plants in soil for approx. 2 weeks until full expansion of the second leaf. Use squared plant pots in order to facilitate a stable fixation on the inoculation trays (see step C4).
- One day before inoculation, remove old Bgh conidiospores from barley plants by shaking (Note 2).
- For the inoculation of rice, place the plant pots laterally on a tray and fix the second leaf of each plant, adaxial surface up, on the tray by placing 2-3 mm stripes of removable adhesive labels at the tip and the base of each leaf (Note 3, Figure 3A).
- Place a Thoma cell counting chamber (without cover glass) beside or between the fixed leaves to monitor the inoculation density.
- Cover the trays with a settling tower (Figure 3C).
- For inoculation, gently transfer leaves or whole plant pots of Bgh-infected barley plants to the opening of the settling tower and release freshly emerged conidiospores into the tower by shaking. Subsequently, wave with a small tray in the air space of the settling tower to cause a turbulent flow and evenly dispense the conidia.
- Let the conidiospores settle for 30-60 min.
- Apply a cover glass to the Thoma counting cell and determine the spore density by microscopy. Therefore count the conidiospores in the large central square of each of the two chambers (1 mm2 in a standard Thoma chamber) and take the average. Under our laboratory conditions, 1-5 spores mm-2 may result in distinct, macroscopically observable mildew pustules, while approx. 15-20 spores mm-2 are recommended for microscopic evaluation. If spore density is too low, return to step C5.
- Gently remove the adhesive labels from the rice leaves, raise the pots and transfer them to the plant growth chamber.
- On host plants, first powdery mildew pustules (as shown in Figure 3B) can be observed within the first week after inoculation. Since rice is a nonhost plant for Bgh, evaluation of the infection process has to be done by microscopy.
- As the fungus is an obligate biotroph, maintainance has to be done on barley plants in a growth cabinet separated from the test plants. Weekly transfer of Bgh onto fresh, one-week old plants is done by shaking off conidiospores from heavily infected barley plants (Figure 3B).
Data analysis
Analyses of the inoculated plants can be performed by macroscopic or microscopic evaluation (as described in Weidenbach et al., 2016).
Notes
- Carefully avoid ‘flooding’ of the leaves and fungal inoculum.
- This step insures fresh inoculum, since the old conidiospores, which differ in virulence, are depleted.
- By fixing the position of the leaves on the trays, an even inoculation becomes possible.
Acknowledgments
We thank Monika Höfte for providing R. solani and P. graminicola isolates and for her kind advise in respective inoculation methods. All the protocols presented here were used and described in Weidenbach et al. (2016), whose first author was funded in the framework of the BMBF funding activity ‘Plant Biotechnology for the future, PLANT 2030’ within the project ‘BarleyFortress’.
References
- Abbruscato, P., Nepusz, T., Mizzi, L., Del Corvo, M., Morandini, P., Fumasoni, I., Michel, C., Paccanaro, A., Guiderdoni, E., Schaffrath, U., Morel, J. B., Piffanelli, P. and Faivre-Rampant, O. (2012). OsWRKY22, a monocot WRKY gene, plays a role in the resistance response to blast. Mol Plant Pathol 13(8): 828-841.
- Akagi, A., Jiang, C. and Takatsuji, H. (2015). Magnaporthe oryzae inoculation of rice seedlings by spraying with a spore suspension. Bio-protocol 5(11): e1486.
- Dean, R., Van Kan, J. A., Pretorius, Z. A., Hammond-Kosack, K. E., Di Pietro, A., Spanu, P. D., Rudd, J. J., Dickman, M., Kahmann, R., Ellis, J. and Foster, G. D. (2012). The Top 10 fungal pathogens in molecular plant pathology. Mol Plant Pathol 13(4): 414-430.
- Hendrix, F. F. and Campbell, W. A. (1973). Pythiums as plant pathogens. Annu Rev Phytopathol 11: 77-98.
- Hinze, K., Thompson, R. D., Ritter, E., Salamini, F. and Schulze-Lefert, P. (1991). Restriction fragment length polymorphism-mediated targeting of the ml-o resistance locus in barley (Hordeum vulgare). Proc Natl Acad Sci U S A 88(9): 3691-3695.
- Lee, F. N., and Rush, M. C. (1983). Rice sheath blight: A major rice disease. Plant Dis 67(7): 829-833.
- Ou, S. H. (1985). Rice diseases. Second edition. Commonwealth Mycological Institute.
- Van Buyten, E. and Höfte, M. (2013). Pythium species from rice roots differ in virulence, host colonization and nutritional profile. BMC Plant Biol 13: 203.
- Weidenbach, D., Esch, L., Möller, C., Hensel, G., Kumlehn, J., Höfle, C., Hückelhoven, R. and Schaffrath, U. (2016). Polarized defense against fungal pathogens is mediated by the jacalin-related lectin domain of modular Poaceae-specific proteins. Mol Plant 9: 514-527.
- Weidenbach, D., Jansen, M., Franke, R. B., Hensel, G., Weissgerber, W., Ulferts, S., Jansen, I., Schreiber, L., Korzun, V., Pontzen, R., Kumlehn, J., Pillen, K. and Schaffrath, U. (2014). Evolutionary conserved function of barley and Arabidopsis 3-ketoacyl-CoA synthases in providing wax signals for germination of powdery mildew fungi. Plant Physiol 166(3): 1621-1633.
Article Information
Copyright
© 2016 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Delventhal, R., Loehrer, M., Weidenbach, D. and Schaffrath, U. (2016). Inoculation of Rice with Different Pathogens: Sheath Blight (Rhizoctonia solani), Damping off Disease (Pythium graminicola) and Barley Powdery Mildew (Blumeria graminis f. sp. hordei). Bio-protocol 6(24): e2070. DOI: 10.21769/BioProtoc.2070.
Category
Plant Science > Plant immunity > Disease bioassay
Microbiology > Microbe-host interactions > In vivo model > Plant
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link








