- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Lymphocyte Isolation, Th17 Cell Differentiation, Activation, and Staining
Published: Vol 6, Iss 23, Dec 5, 2016 DOI: 10.21769/BioProtoc.2047 Views: 14611
Reviewed by: Ivan ZanoniAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Protocol for the Isolation and Analysis of Extracellular Vesicles From Peripheral Blood: Red Cell, Endothelial, and Platelet-Derived Extracellular Vesicles
Bhawani Yasassri Alvitigala [...] Lallindra Viranjan Gooneratne
Nov 5, 2025 1440 Views
Improved Immunohistochemistry of Mouse Eye Sections Using Davidson's Fixative and Melanin Bleaching
Anne Nathalie Longakit [...] Catherine D. Van Raamsdonk
Nov 20, 2025 1565 Views
Optimization of Adipogenic Differentiation Protocol for Murine and Human Cell Culture Models
Junwan Fan [...] Wenyan He
Jan 20, 2026 209 Views
Abstract
Background
T cells are critical to mediate host defense against bacteria, viruses and fungi as well as commensal (Kumar et al., 2016). T cells can be further subdivided into T helper (Th1), Th2 and Th17 subsets based on their ability to generate specific cytokines. Naive T cells can be differentiated into specific T cell subsets in in vitro culture in response to specific cytokine stimulation. In vitro generated Th1, Th2 and Th17 cells have helped us to understand the molecular mechanism of their differentiation and their effector functions. Here, we have described a basic protocol for Th17 cell generation.
Materials and Reagents
- 96-well tissue culture plate (CELLTREAT Scientific Products, catalog number: 229196 )
- Falcon® 70 μm cell strainers (Corning, Falcon®, catalog number: 352350 )
- 50 ml conical tube (Denville Scientific, catalog number: C1056 )
- 1 ml syringe with cap (BD, catalog number: 305217 )
- 15 ml conical tube (Denville Scientific, catalog number: C1018-P )
- 96-well round (U) bottom plate (CELLTREAT Scientific Products, catalog number: 229190 )
- C57BL\6 mice (Taconic Biosciences, catalog number: B6-F )
- Coating antibodies: anti-mouse CD28 (Affymetrix, eBioscience, catalog number: 14-0281 ), anti-mouse CD3e (Affymetrix, eBioscience, catalog number: 14-0033 )
- ELISA coating buffer (Biolegend, catalog number: 421701 )
- EasySepTM buffer (STEMCELL Technologies, catalog number: 20144 ) or PBS containing 2% fetal bovine serum (FBS) with 1 mM EDTA
- EasySepTM Mouse Naïve CD4+ T Cell Isolation Kit (STEMCELL Technologies, catalog number: 19765 )
- Staining antibodies:
- anti-mouse CD3-eFlour 450 (Affymetrix, eBioscience, catalog number: 48-0032 )
- anti-mouse CD4-Alexa Flour 700 (Affymetrix, eBioscience, catalog number: 56-0041 )
- anti-mouse IL-22-APC (Affymetrix, eBioscience, catalog number: 17-7222 )
- anti-mouse IL-17A-PE (Affymetrix, eBioscience, catalog number: 12-7177 )
- anti-mouse CD62L-FITC (Affymetrix, eBioscience, catalog number: 11-0621 )
- Phorbol 12-myristate 13-acetate (PMA) (Sigma-Aldrich, catalog number: 79346 )
- Ionomycin (Sigma-Aldrich, catalog number: I0634 )
- Fixation/Permeabilization Solution Kit with BD GolgiStopTM (BD, catalog number: 554715 )
- Cytofix/Cytoperm Plus Kit (with BD GolgiStopTM) (BD, catalog number: 554715 )
- Iscove’s modified Dulbecco’s medium (IMDM) cell culture medium + glutamax (Thermo Fisher Scientific, GibcoTM, catalog number: 31980-030 )
- HyCloneTM fetal bovine serum (U.S.), characterized FBS (GE Healthcare, catalog number: SH30071.03 )
- HyCloneTM penicillin-streptomycin 100x solution (GE Healthcare, catalog number: SV30010 )
- Recombinant porcine TGF-β1 (R&D Systems, catalog number: 101-B1 )
- Recombinant mouse IL-6 (R&D Systems, catalog number: 406-ML )
- Recombinant mouse IL-23 (R&D Systems, catalog number: 1887-ML )
- Anti-mouse IFNγ (R&D Systems, catalog number: MAB485 )
- Anti-mouse IL-4 (R&D Systems, catalog number: MAB404 )
- Phosphate buffer saline (PBS) (Boston BioProduct, catalog number: BM220S )
- Bovine serum albumin (BSA) (Sigma-Aldrich, catalog number: A3059 )
- Sodium azide (Sigma-Aldrich, catalog number: S2002 )
- Complete IMDM medium (see Recipes)
- 2x Th17 differentiation condition medium (see Recipes)
- FACS buffer (see Recipes)
Equipment
- Tissue culture incubator (NuAire, model: LabGard Class II type A2 )
- Centrifuge (Thermo Fisher Scientific, Thermo ScientificTM, model: Sorvall Legend XFR )
- BD LSRII Flow cytometer - BD
- Scientific Industries Vortex Genie2 (Stellar Scientific, catalog number: SI-236 )
- EasyEightTM EasySepTM magnet (STEMCELL Technologies, catalog number: 18103 )
Software
- FACS Diva or Flow Jo software
Procedure
- Lymphocyte isolation
- Coat desired number of wells in a 96-well plate with 50 μl of anti-mouse CD3 (2.5 μg/ml) and anti-mouse CD28 (2.0 μg/ml) for overnight at 4 °C. We used 1x ELISA coating buffer for antibodies dilution and coating. We washed plate once with 1x PBS before adding the cells.
- Next day, euthanize a C57BL\6 mice and harvest spleen.
- Place 70 micron cell strainer on 50 ml conical tube.
Place spleen on the cell strainer and homogenize/disrupt using 1 ml syringe cap. Syringe cap is placed on top of the spleen and rotate handle with hand to crush/homogenize the tissue. Add 1 ml EasySep buffer in cell strainer to facilitate homogenization. Collect flow through (contains cells) and add additional 9 ml EasySep buffer directly into the centrifuge tube. Centrifuge 300 x g for 10 min at 4 °C. Discard supernatant and re-suspend cells pellet with 1 ml EasySep buffer. Count cells (RBC lysis step is not required) and add 1 ml EasySep buffer/1 x 108 cells. Transfer cells into a 15 ml centrifuge tube.
Follow EasySep mouse CD4+CD62L+ naïve T cells isolation protocol.
Steps for naïve CD4+CD62L+ T cell isolation as per the EasySepTM kit
Notes:- The total number of cells in a naïve mouse spleen is (~1 x 108) without RBC lyses. We usually get 5-7 million naïve CD4 T cells after the purification steps.
- Make sure the purity of isolated CD4+CD62L+ T cells is above 95% after enrichment (see Figure 1). Take small aliquots of purified cells (5,000-10,000 cells) and stain with anti-CD4-Alexa Flour 700 (1:100) and anti-CD62L-FITC (1:100) in ice cold FACS buffer for 30 min at 4 °C. After washing with FACS buffer check purity using flow cytometer by gating for CD4+CD62L+ T cells.
- The total number of cells in a naïve mouse spleen is (~1 x 108) without RBC lyses. We usually get 5-7 million naïve CD4 T cells after the purification steps.
- Count the enriched cells and plate them as follows:
For a 96-well-plate, add 2 x 105 cells/well in 100 μl complete IMDM medium (see Recipes).
- Coat desired number of wells in a 96-well plate with 50 μl of anti-mouse CD3 (2.5 μg/ml) and anti-mouse CD28 (2.0 μg/ml) for overnight at 4 °C. We used 1x ELISA coating buffer for antibodies dilution and coating. We washed plate once with 1x PBS before adding the cells.
- Th17 cell differentiation
- Prepare 2x Th17 differentiation condition medium (see Recipes)
- Add 100 μl 2x Th17 differentiation condition medium to each well.
- Culture for 3 additional days at 37 °C and 5% CO2 in a tissue culture incubator. You will see several T cell clusters on day 2 post differentiation and medium will turn slight yellowish. If medium turned very yellowish then add additional 50 μl complete IMDM medium (see Recipes).
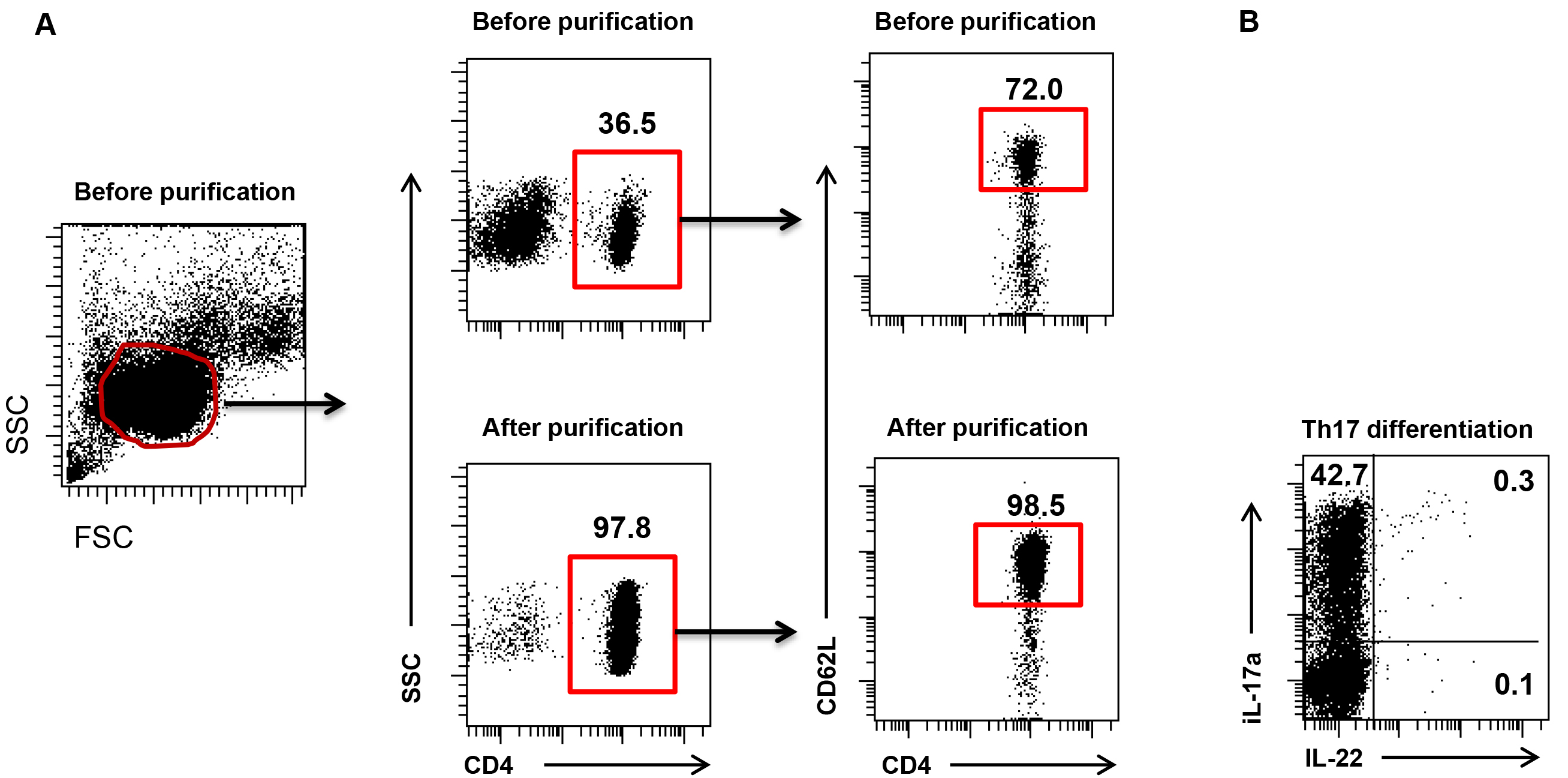
Figure 1. Naïve CD4 T cells purification and Th17 differentiation. A. Flow cytometry dot plot gate shows spleen lymphocytes (left), percentage of CD4+ cells (middle) and frequency of purified naïve CD4+CD62L+ T cells. B. Data show IL-17 producing Th17 cells on day 3 post differentiation.
- Prepare 2x Th17 differentiation condition medium (see Recipes)
- Activation and staining
- After day 3 post differentiation, transfer cells into a U-bottom 96-well plate using a multi-channel pipette (gently pipetting up and down to resuspend the cells) and centrifuge at 300 x g for 5 min at room temperature.
Discard supernatant by flicking plate in a quick single motion. To flick off the supernatant, move the plate upward and bring it straight down. Gently touch plate with the tissue paper to remove drops. Vortex plates using a benchtop Vortex Genie2 or any mini vortex mixer. Follow these steps for washing and decanting the supernatant. - Add 200 μl of complete IMDM medium containing 50 ng/ml PMA, 750 ng/ml ionomycin and GolgiStop (1:1,000) to the cells and incubate for 4 h at 37 °C and 5% CO2 in a tissue culture incubator.
- Wash twice with 200 μl ice cold FACS buffer (300 x g, 5 min, 4 °C). Discard supernatant by flicking plate in a quick single motion.
- Re-suspend cells in 100 μl ice cold FACS buffer containing anti-mouse CD3-eFluor 450 (1:100), anti-mouse CD4-Alexa Fluor 700 (1:100) and incubate for 30 min at 4 °C (refrigerator).
- Wash cells with FACS buffer (300 G, 2 min, 4 °C).
- Fix cells with 100 μl BD Cytofix/Cytoperm for 20 min at 4 °C. Discard supernatant by flicking plate in a quick single motion.
- Wash twice with 200 μl 1x BD Perm/WashTM buffer (300 x g, 2 min, 4 °C). Discard supernatant by flicking plate in a quick single motion.
- Add 100 μl 1x BD Perm/WashTM buffer containing anti-IL-17A-PE (1:100) and anti-IL-22-APC (1:100) antibodies and incubate for 45 min/overnight at 4 °C.
Instead of surface staining steps, all antibodies including surface (anti-mouse CD3-eFluor 450, anti-mouse CD4-Alexa Fluor 700) and intracellular (anti-mouse IL-17A-PE and anti-mouse IL-22-APC) can be used together in a single step after fixation and Perm/Wash. We have stained cells with intracellular cytokines along with surface antibodies against CD3 and CD4. - Wash cells twice with 200 μl 1x BD Perm/WashTM buffer (300 x g, 5 min, 4 °C). Re-suspend pellet in 200 μl FACS buffer.
- Confirm the lineage of Th17 cell by determining the levels of intracellular IL-17A and IL-22 by FACS. A good Th17 differentiation will result into 20-40% IL-17A producing CD4 T cells.
- After day 3 post differentiation, transfer cells into a U-bottom 96-well plate using a multi-channel pipette (gently pipetting up and down to resuspend the cells) and centrifuge at 300 x g for 5 min at room temperature.
Data analysis
FACS Diva or Flow Jo software can be used to analyze data. Plot a linear FSC versus SSC dot plot and create a gate (P1) to select all cells. Using P1 population plot a linear CD3 versus CD4 dot plot. Majority of differentiated cells will be positive for both CD3 and CD4. CD3+CD4+ double positive population (gate P2) will be analyzed for intracellular IL-17 and IL-22 staining.
Recipes
- Complete IMDM medium (Pre-warmed at 37 °C before use)
IMDM medium + glutamax
10% FBS
1% penicillin/streptomycin solution - 2x Th17 differentiation condition medium
Complete IMDM medium (Pre-warmed at 37 °C) containing:
10 ng/ml TGF-β1
80 ng/ml recombinant IL-6
20 ng/ml recombinant IL-23
10 μg/ml anti-IFNγ
10 μg/ml anti-IL-4 - FACS buffer
PBS with
0.5 % bovine serum albumin (BSA)
0.05% sodium azide
Acknowledgments
The authors would like to acknowledge support from the following PHS grants: P50HL084932, 5R01HL061271, and R37HL079142 to J.K.K. P.K is supported from Children’s Hospital of Pittsburgh Research Advisory Committee Grant from Children’s Hospital of Pittsburgh of the UPMC Health System.
References
- Kumar, P., Monin, L., Castillo, P., Elsegeiny, W., Horne, W., Eddens, T., Vikram, A., Good, M., Schoenborn, A. A., Bibby, K., Montelaro, R. C., Metzger, D. W., Gulati, A. S. and Kolls, J. K. (2016). Intestinal interleukin-17 receptor signaling mediates reciprocal control of the gut microbiota and autoimmune inflammation. Immunity 44(3): 659-671.
Article Information
Copyright
© 2016 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Kumar, P. and Kolls, J. K. (2016). Lymphocyte Isolation, Th17 Cell Differentiation, Activation, and Staining. Bio-protocol 6(23): e2047. DOI: 10.21769/BioProtoc.2047.
Category
Cell Biology > Cell isolation and culture > Cell differentiation
Cell Biology > Cell staining > Whole cell
Cell Biology > Cell-based analysis > Flow cytometry
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link










