- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
PKC-θ in vitro Kinase Activity Assay
Published: Vol 6, Iss 20, Oct 20, 2016 DOI: 10.21769/BioProtoc.1980 Views: 10319
Reviewed by: Ivan ZanoniMichael EnosMareta Ruseva

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Fluorescence Polarization-Based High-Throughput Screening Assay for Inhibitors Targeting Cathepsin L
Keyu Guo [...] Shuyi Si
Jul 20, 2025 2277 Views
Isolation and Ex Vivo Testing of CD8+ T-Cell Division and Activation Using Mouse Splenocytes
Melissa Dolan [...] John M.L. Ebos
Aug 20, 2025 3858 Views
Detecting the Activation of Endogenous Small GTPases via Fluorescent Signals Utilizing a Split mNeonGreen: Small GTPase ActIvitY ANalyzing (SAIYAN) System
Miharu Maeda and Kota Saito
Jan 5, 2026 481 Views
Abstract
Protein kinase C-θ (PKC-θ), a member of the Ca2+-independent PKC subfamily of kinases, serves as a regulator of T cell activation by mediating the T cell antigen receptor (TCR)- and coreceptor CD28-induced activation of the transcription factors NF-κB and AP-1 and, to a lesser extent, NFAT, and, subsequently, interleukin 2 (IL-2) production and T cell proliferation. In T cells, TCR and CD28 stimulation-induced activation of PKC-θ is the integrated result of diacylglycerol-mediated membrane recruitment, GLK-mediated phosphorylation at activation loop, CD28, Lck, and sumoylation-mediated central immunological synapse localization (Wang et al., 2015; Monks et al., 1997; Kong et al., 2011; Isakov and Altman, 2012; Chuang et al., 2011). Phosphatidylserine (PtdSer) and the phorbol ester Phorbol 12-myristate 13-acetate (PMA, a surrogate of diacylglycerol [DAG]) are the cofactors for the Ca2+-independent PKC subfamily that bind to PKC directly and activate it by changing its conformation (Nishizuka, 1995). A protocol to analyze the PKC-θ kinase activity in vitro is described here. Myelin basic protein is used as the substrate and its phosphorylation is detected by the incorporation of radioactive phosphate into the substrate, which is analyzed by a laser scanner.
Keywords: In vitro kinase assayBackground
Specified by their divergent regulatory domains, the PKC family can be divided into distinct subgroups: the conventional PKCs (cPKCs, comprising PKC-α, β and γ), the novel PKCs (nPKCs, including PKC-δ, ε, θ and η) and the atypical PKCs (aPKCs, PKCι and ζ). The cPKCs are activated by a combination of diacylglycerol, phospholipid and Ca2+. The nPKCs are activated by diacylglycerol and phorbol esters but do not require Ca2+. In contrast, the aPKCs do not depend on Ca2+ or diacylglycerol for activation (Rosse et al., 2010). Here we used anti-Myc or anti-PKC-θ to isolate PKC-θ from the cell lysates, then performed the radiolabeled ATP based kinase assay in the Reaction buffer containing PMA, PtdSer and EGTA (a selective chelator for Ca2+). The assay protocol described here is quick, sensitive and specific, provides a direct measurement of PKC-θ activity. This protocol could be modified to analyze the activity of other nPKC isoforms.
Materials and Reagents
- Jurkat E6.1 cells or HEK293T cells
- Phosphate-buffered saline (PBS) (Thermo Fisher Scientific, GibcoTM, catalog number: 10010023 )
- RPMI-1640 medium (GE Healthcare, HyCloneTM, catalog number: SH30027.01 )
- Heat-inactivated fetal bovine serum (FBS) (Thermo Fisher Scientific, GibcoTM, catalog number: 10270-106 )
- Anti-PKC-θ (Santa Cruz Biotechnology, catalog number: sc-1875 )
- Anti-Myc (9E10) (Santa Cruz Biotechnology, catalog number: sc-40 )
- Protein G sepharose (GE Healthcare, catalog number: 17-0618-01 )
- Phosphatidylserine from PepTag® Non-Radioactive PKC assay kit (Promega, catalog number: V5330 )
- Tris (Sangon Biotech, catalog number: A600194 )
- Sodium chloride (NaCl) (Sigma-Aldrich, catalog number: S3014 )
- Ethylenediaminetetraacetate (EDTA) (Sangon Biotech, catalog number: A610185 )
- Nonidet P40 (Sangon Biotech, catalog number: A600385 )
- Aprotinin (EMD Millipore, catalog number: 616370 )
- Leupeptin (Calbiochem, catalog number: 108976 )
- Phenylmethanesulfonyl fluoride (PMSF) (Sigma-Aldrich, catalog number: P7626 )
- Sodium pyrophosphate tetrabasic decahydrate (NaPPi) (Sigma-Aldrich, catalog number: S6422 )
- Sodium orthovanadate (Na3VO4) (Sigma-Aldrich, catalog number: 567540 )
- HEPES (Thermo Fisher Scientific, GibcoTM, catalog number: 11344041 )
- Magnesium chloride (MgCl2) (Sigma-Aldrich, catalog number: M4880 )
- EGTA (Sigma-Aldrich, catalog number: E3889 )
- ATP (Sigma-Aldrich, catalog number: A2383 )
- [γ-32P]ATP (PerkinElmer, catalog number: NEG002Z001MC )
- Myelin basic protein (MBP) (Sigma-Aldrich, catalog number: M1891 )
- PMA (Sigma-Aldrich, catalog number: P1585 )
- Bromophenol blue (Bio-Rad Laboratories, catalog number: 1610404 )
- DL-dithiothreitol (DTT) (Sigma-Aldrich, catalog number: D9779 )
- Sodium dodecyl sulfate (SDS) (Sigma-Aldrich, catalog number: L3771 )
- Glycerol (Sangon Biotech, catalog number: A100854 )
- ATP-competitive PKC-θ inhibitor rottlerin (EMD Millipore, catalog number: 557370 )
- DMSO
- Lysis buffer (see Recipes)
- PKC-θ kinase buffer (see Recipes)
- Reaction buffer (see Recipes)
- 5x SDS-PAGE loading buffer (see Recipes)
Equipment
- Typhoon Trio+ system (GE Healthcare)
- Humidified CO2 incubator
- Laminar air flow bio-safety cabinet
- Centrifuge (Eppendorf, model: 5417R )
- Phosphor-storage screen (GE Healthcare)
- Sonicator (Sonics, model: VCX-130 )
- Thermomixer (Eppendorf)
- Exposure cassettes (GE Healthcare)
Software
- Typhoon Scanner Control software
Procedure
- Harvest 5 x 106 Jurkat E6.1 cells or 1 x 106 HEK293T cells transfected with vector, wild type (WT), kinase-dead mutant (K409R) and sumoylation deficient mutant (2KR [K325R, K506R]) of Myc-PKC-θ (indicated in Figure 2) by centrifugation (300 x g, 3 min, 25 °C).
- Wash the cells once with 1 ml ice cold PBS.
- Centrifuge the cells at 300 x g for 3 min at 25 °C.
- Add 300-500 µl lysis buffer to the cell pellets, vortex for 1-2 min.
- Place the tube on ice for 10 min.
- Centrifuge at 8,500 x g for 10 min at 4 °C.
- Transfer the clear supernatant to a new microcentrifuge tube.
- Immunoprecipitate endogenous or transfected PKC-θ by adding 2.5 µl anti-PKC-θ or anti-Myc antibodies into the clear supernatant obtained in step 7. Incubate overnight at 4 °C with rotation. Then add 30 µl protein G sepharose beads (50% [v/v] in PBS) and incubate for 2-4 h at 4 °C with rotation.
- Centrifuge for 5 min at 8,500 x g at 4 °C, carefully remove the supernatant. Wash the immunoprecipitates extensively by adding 1 ml lysis buffer and vortexing for 3-5 min.
- Repeat step 9.
- Centrifuge for 5 min at 8,500 x g at 4 °C, carefully remove the supernatant. Wash the immunoprecipitates extensively by adding 1 ml PKC-θ kinase buffer and vortexing for 3-5 min.
- Repeat step 11.
- Centrifuge for 5 min at 8,500 x g at 4 °C, carefully remove the supernatant and resuspend the pellet in 25 μl of the reaction buffer.
- Incubate samples in the Thermomixer with gentle shaking at 300 rpm for 30 min at 30 °C. Stop the reaction by adding 6.25 µl 5x SDS-PAGE loading buffer.
- Heat samples at 95 °C for 10 min and subject the samples to 15% SDS-PAGE, then cover the gel with plastic wrap to minimize the chance of radioactive contamination.
- Open the exposure cassette, place the wrapped gel on the internal surface of the cassette, and then place the phosphor-storage screen on top the wrapped gel with the phosphor (white) side facing down onto the gel. Close and lock the cassette, place it for 30 min to 6 h at room temperature and then take the phosphor-storage screen out of the cassette avoiding direct light. Scan the screen with the Typhoon Trio+ system.
- (Optional) A pilot experiment to verify the specificity of this kinase assay is recommended. In detail, HEK293T cells were transfected with wild type Myc-PKC-θ, which has proven to be an active kinase as T538, S676, and S695 are constitutively phosphorylated on recombinant PKC-θ isolated from HEK293T expression systems (Wang et al., 2012). 24 h after transfection, Myc-PKC-θ was immunoprecipitated and subjected to the kinase assay as described previously. When doing the kinase assay, the ATP-competitive PKC-θ inhibitor or DMSO was added into the reaction buffer which served as the negative and the positive control individually.
Data analysis
- The kinase assay system works specifically if there is a strong signal for the DMSO added sample while no signal for the PKC-θ inhibitor added sample in the pilot experiment.
- Analyze signals with Typhoon Scanner Control software as followed:
- Double-click the Scanner Control shortcut icon on the desktop.
- After the current instrument state is ‘Ready’, select the acquisition mode as ‘Storage Phosphor’ (Figure 1).
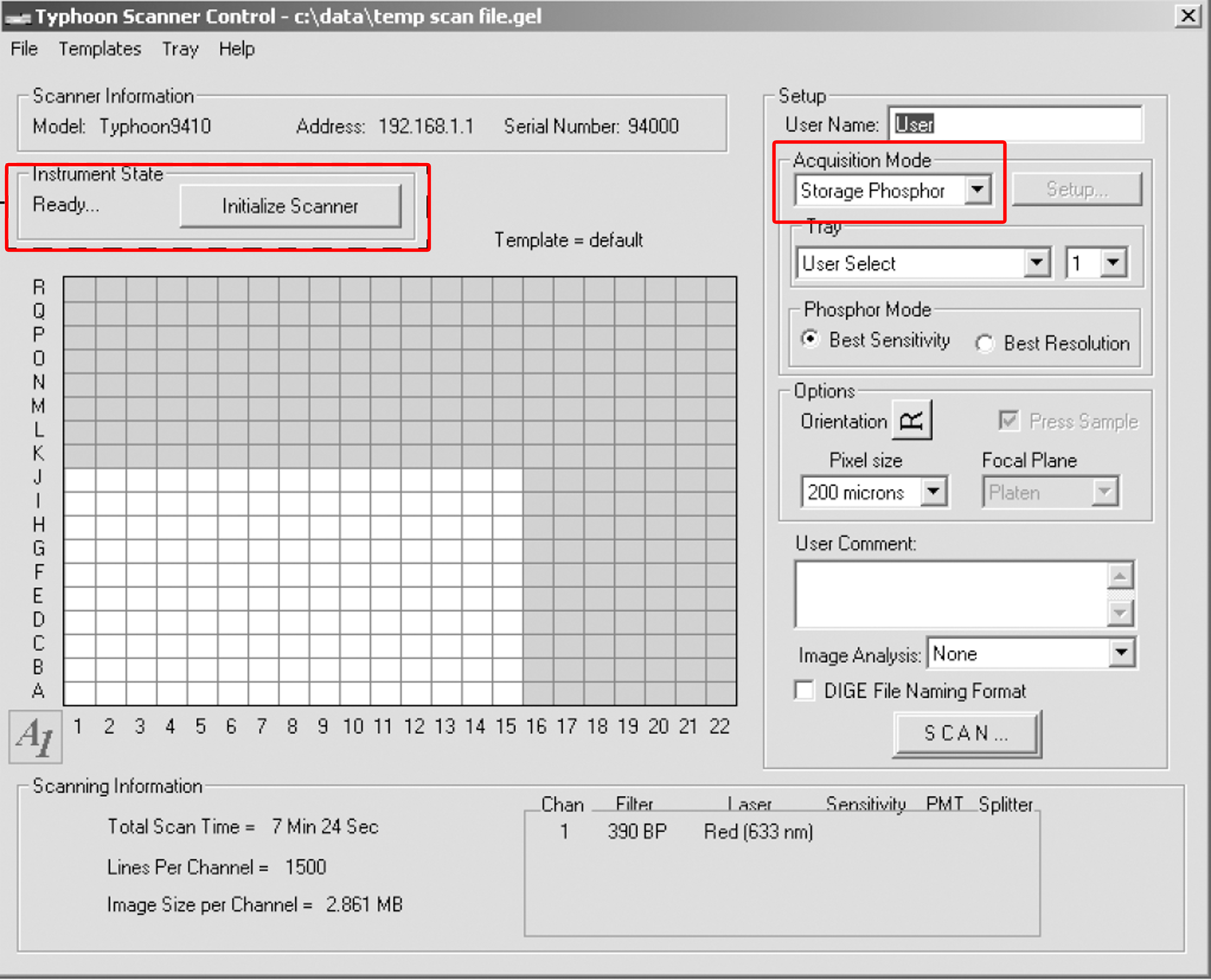
Figure 1. The scanner control window - Place the storage phosphor screen in the Typhoon instrument. Remove the storage phosphor screen from the exposure cassette. Keep the screen face down. Position the screen on the glass platen so that the A1 corner of the screen aligns with the A1 corner of the glass platen. Close the sample lid.
- Select the grid area and pixel size, click ‘SCAN’ to start scanning.
- Save the data and evaluate the results. A representative data set for the samples from transfected HEK293T cells was shown in Figure 2.
Representative data
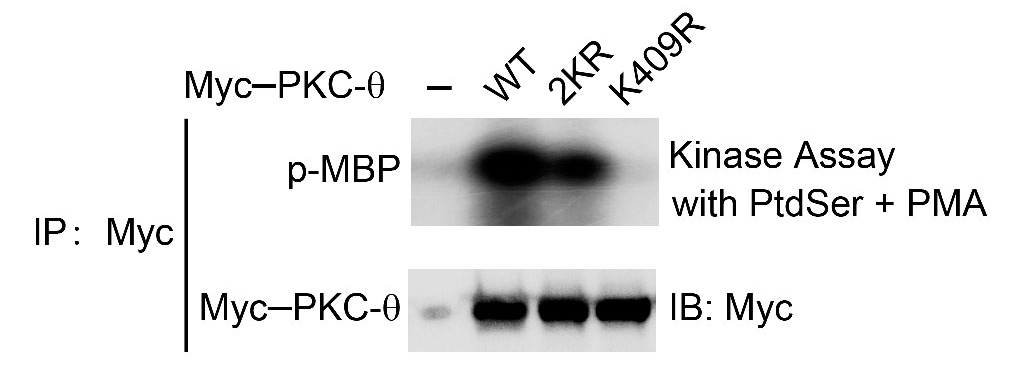
Figure 2. In vitro kinase assay of Myc-tagged PKC-θ. Myc-PKC-θ-WT, -2KR (K325R, K506R) or -K409R were immunoprecipitated (IP) from lysates of HEK293T cells transfected with empty vector or the indicated Myc-tagged PKC-θ plasmids, MBP was used as a substrate in the presence of PtdSer/PMA cofactors (upper panel). Aliquots of the IPs were immunoblotted with anti-Myc to confirm similar expression levels of PKC-θ (bottom panel).
Notes
Phosphatidylserine easily aggregates to form micelles. To achieve maximal PKC-θ activation, sonicate the phosphatidylserine solution with a probe sonicator for 20-30 sec or until warm to disrupt the micelles.
Recipes
- Lysis buffer
20 mM Tris-HCl (pH 7.5)
150 mM NaCl
5 mM EDTA
1% Nonidet P40
10 μg/ml aprotinin
10 μg/ml leupeptin
1 mM PMSF
5 mM NaPPi
1 mM Na3VO4 - PKC-θ kinase buffer
20 mM HEPES
10 mM MgCl2
0.1 mM EGTA - Reaction buffer
5 μCi [γ-32P]ATP, 20 μM ATP
1 μg of myelin basic protein (MBP)
10 μM PMA cofactors
200 μg/ml phosphatidylserine (PtdSer) (presence or absence) - 5x SDS-PAGE loading buffer
0.25% bromophenol blue
0.5 M DTT
50% glycerol
10% SDS
0.25 M Tris-HCl (pH 6.8)
Acknowledgments
Supported by the National Natural Science Foundation of China (31170846) and the Ministry of Science and Technology of China (2013CB835300). This protocol was modified from the in vitro kinase assay (Bi, 2001).
References
- Bi, K., Tanaka, Y., Coudronniere, N., Sugie, K., Hong, S., van Stipdonk, M. J. and Altman, A. (2001). Antigen-induced translocation of PKC-θ to membrane rafts is required for T cell activation. Nat Immunol 2(6): 556-563.
- Chuang, H. C., Lan, J. L., Chen, D. Y., Yang, C. Y., Chen, Y. M., Li, J. P., Huang, C. Y., Liu, P. E., Wang, X. and Tan, T. H. (2011). The kinase GLK controls autoimmunity and NF-κB signaling by activating the kinase PKC-θ in T cells. Nat Immunol 12(11): 1113-1118.
- Isakov, N. and Altman, A. (2012). PKC-theta-mediated signal delivery from the TCR/CD28 surface receptors. Front Immunol 3: 273.
- Kong, K. F., Yokosuka, T., Canonigo-Balancio, A. J., Isakov, N., Saito, T. and Altman, A. (2011). A motif in the V3 domain of the kinase PKC-θ determines its localization in the immunological synapse and functions in T cells via association with CD28. Nat Immunol 12(11): 1105-1112.
- Monks, C. R., Kupfer, H., Tamir, I., Barlow, A. and Kupfer, A. (1997). Selective modulation of protein kinase C-θ during T-cell activation. Nature 385(6611): 83-86.
- Nishizuka, Y. (1995). Protein kinase C and lipid signaling for sustained cellular responses. FASEB J 9(7): 484-496.
- Rosse, C., Linch, M., Kermorgant, S., Cameron, A. J., Boeckeler, K. and Parker, P. J. (2010). PKC and the control of localized signal dynamics. Nat Rev Mol Cell Biol 11(2): 103-112.
- Xie, J. J., Liang, J. Q., Diao, L. H., Altman, A. and Li, Y. (2013). TNFR-associated factor 6 regulates TCR signaling via interaction with and modification of LAT adapter. J Immunol 190(8): 4027-4036.
- Wang, X., Chuang, H. C., Li, J. P. and Tan, T. H. (2012). Regulation of PKC-θ function by phosphorylation in T cell receptor signaling. Front Immunol 3: 197.
- Wang, X. D., Gong, Y., Chen, Z. L., Gong, B. N., Xie, J. J., Zhong, C. Q., Wang, Q. L., Diao, L. H., Xu, A., Han, J., Altman, A. and Li, Y. (2015). TCR-induced sumoylation of the kinase PKC-θ controls T cell synapse organization and T cell activation. Nat Immunol 16(11): 1195-1203.
Article Information
Copyright
© 2016 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Wang, X. and Li, Y. (2016). PKC-θ in vitro Kinase Activity Assay. Bio-protocol 6(20): e1980. DOI: 10.21769/BioProtoc.1980.
Category
Immunology > Immune cell function > Antigen-specific response
Biochemistry > Protein > Activity
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link










