- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Cell Wall-bound p-Coumaric and Ferulic Acid Analysis
Published: Vol 6, Iss 19, Oct 5, 2016 DOI: 10.21769/BioProtoc.1955 Views: 8107
Reviewed by: Marisa RosaAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Isolation of Intact Vacuoles from Arabidopsis Root Protoplasts and Elemental Analysis
Chuanfeng Ju [...] Zhenqian Zhang
Mar 5, 2023 2035 Views

Closed Systems to Study Plant–Filamentous Fungi Associations: Emphasis on Microscopic Analyses
Vasiliki Skiada and Kalliope K. Papadopoulou
Feb 20, 2025 2853 Views
High-Performance Liquid Chromatography Quantification of Glyphosate, Aminomethylphosphonic Acid, and Ascorbate in Culture Medium and Microalgal Cells
Juan Manuel Ostera [...] Gabriela Malanga
Apr 5, 2025 1171 Views
Abstract
Hydroxycinnamic acids, such as p-coumaric acid and ferulic acid, are a major class of compounds derived from the phenylpropanoid pathway. These compounds are widely conserved in plants and primarily accumulate in the secondary cell wall. They serve as important structural components that contribute to the overall strength and rigidity of plant cell walls and are also potent antioxidants valued for nutritional consumption. This protocol describes a method for analyzing hydroxycinnamic acids that are released after incubation under alkaline conditions.
Keywords: Cell wallMaterials and Reagents
- 50 ml conical tubes (MIDSCI, catalog number: C50B )
- 2 ml microfuge tubes (MIDSCI, catalog number: AVX-T-20-C )
- Arabidopsis inflorescence tissue
- Liquid nitrogen
- Ethanol, pure (Sigma-Aldrich, catalog number: E7023 )
- Acetone (Sigma-Aldrich, catalog number: 320110 )
- 1 N NaOH (Sigma-Aldrich, catalog number: S8045 )
- 3,4,5-trimethoxycinnamic acid (Sigma-Aldrich, catalog number: T70408 )
- HCl (concentrated hydrochloric acid) (36-38%) (Sigma-Aldrich, catalog number: H1758 )
- Ethyl acetate (Sigma-Aldrich, catalog number: 270989 )
- Acetonitrile (Sigma-Aldrich, catalog number: 271004 )
- 0.1% formic acid (Sigma-Aldrich, catalog number: 94318 )
- p-coumaric acid (Sigma-Aldrich, catalog number: C9008 )
- Ferulic acid (Sigma-Aldrich, catalog number: 128708 )
- 50% methanol (Sigma-Aldrich, catalog number: 34860 )
Equipment
- Mortar and pestle
- Plastic spoon (appropriate for handling small volumes)
- Shaking incubator, 37 °C
- Centrifuge with a rotor that can accommodate 50 ml conical tubes and can reach 10,000 x g
- Microcentrifuge
- Vortex
- Drying oven, 70 °C
- Water bath, 65 °C
- Chemical scoop (appropriate for handling small volumes)
- Micropipette and tips (Mettler-Toledo, catalog number: L-STARTXLS+ )
- Repeating pipettor with adaptor syringe for dispensing 200 µl (Mettler-Toledo, model: AR-E1 )
- Speed Vac
- Analytical balance
- HPLC system with UV detector and autosampler (Shimadzu prominence modular HPLC: system controller CBM-20A, solvent delivery unit LC-20A, auto-sampler SIL-20A, column oven CTO-20A, UV-VIS detector SPD-20A)
- HPLC glass vials (VWR, catalog number: 89220-128 )
- Shim-pack XR-ODS column (column dimensions 3.0 x 75 mm, bead size 2.2 µm) (Shimadzu, model: Shim-pack XR-ODS )
Procedure
- Triplicate samples from three independent biological populations should first be washed to remove soluble metabolites. About 1 g of fresh Arabidopsis inflorescence tissue, stripped of leaves and seeds, was used for the analysis described in Anderson et al. (2015); however, this method can be used to analyze all plant tissues. Briefly, this process includes:
- Grind the tissue to powder form in liquid nitrogen using a mortar and pestle.
- Transfer the ground tissue to 50 ml conical tubes using a plastic spoon, a larger volume container may be needed depending on the amount of tissue used. The transfer of tissue becomes more cumbersome if the tissue is allowed to thaw. Immediate transfer of tissue is recommended.
- Wash the ground tissue using 40 ml 80% ethanol, allowing the mixture to incubate at 65 °C for 1 h with thorough mixing by vortex every 15 min. After 1 h, centrifuge the mixture for 10 min at 10,000 x g. Decant the supernatant being careful to not disturb the pelleted tissue.
- Repeat the washing, in 1 h intervals, 4 to 6 times until the solvent is clear (please see Figure 1).
Note: Other plant tissues, such as leaves or roots, may require less or more washes. - Wash once using 20 ml acetone. Do not incubate at 65 °C. Mix well by vortex and then centrifuge directly for 10 min at 10,000 x g. Decant the supernatant being careful to not disturb the pelleted tissue.
Place in a drying oven (70 °C) until dried. For the sample described, sufficient drying occurred overnight. More tissue will increase the drying time. Proper drying is important for storage and handling. This washing step has previously been published in Meyer et al. (1998). - Add 20 mg of the dried, washed cell wall tissue using a chemical scoop into 2 ml microfuge tubes.
- Add 600 µl of 1 N NaOH, containing a final concentration of 50 µM 3,4,5-trimethoxycinnamic acid as an internal standard, and incubate for 24 h at 37 °C with constant agitation.
- Acidify the solution by using 200 µl concentrated HCl using a repeating pipette. The final solution should be acidic for the proper extraction of hydroxicinnamic acids into ethyl acetate.
- Mix the samples well by vortex and centrifuged for 20 min at 14,000 x g.
- Carefully transfer 500 µl of the supernatant to a new tube and add 1 ml ethyl acetate.
- Mix samples well by vortex and centrifuge for 5 min at 14,000 x g.
- Transfer 900 µl of the organic phase into a new tube and dry to completion using a speed Vac.
- Redissolve the dried extracts in 50% MeOH and analyze by HPLC. Extracts were separated on a Shim-pack XR-ODS column using a gradient of increasing acetonitrile from 2.0% to 25% for 29.5 min in 0.1% formic acid at a flow rate of 0.9 ml min-1 (Figure 2).
- Quantify p-coumaric and ferulic acid content in samples. Authentic standards (purchased p-coumaric acid and ferulic acid in their purified form) were run separately using the same method for compound identification by retention time. Standard curves of these compounds of known concentrations were used to generate a linear formula that compares peak area to concentration. This formula was then used to quantify the concentration of the compounds in the sample. Concentrations for the standard curve should be selected so that they are similar to the amount of compound present in the sample. Peak area for these compounds should be adjusted to the peak area of the internal standard to account for transfer and pipetting error.
Representative data

Figure 1. A representative sampling of the extract from washed tissues. A progression showing the reduction of color present in the solvent during the washing process. The tube on the left shows what the solvent might look like after the first washing step. The tube in the middle shows what the solvent might look like after the third washing step and the tube on the right shows what it might look like after five washes where it is ready for the final wash with acetone.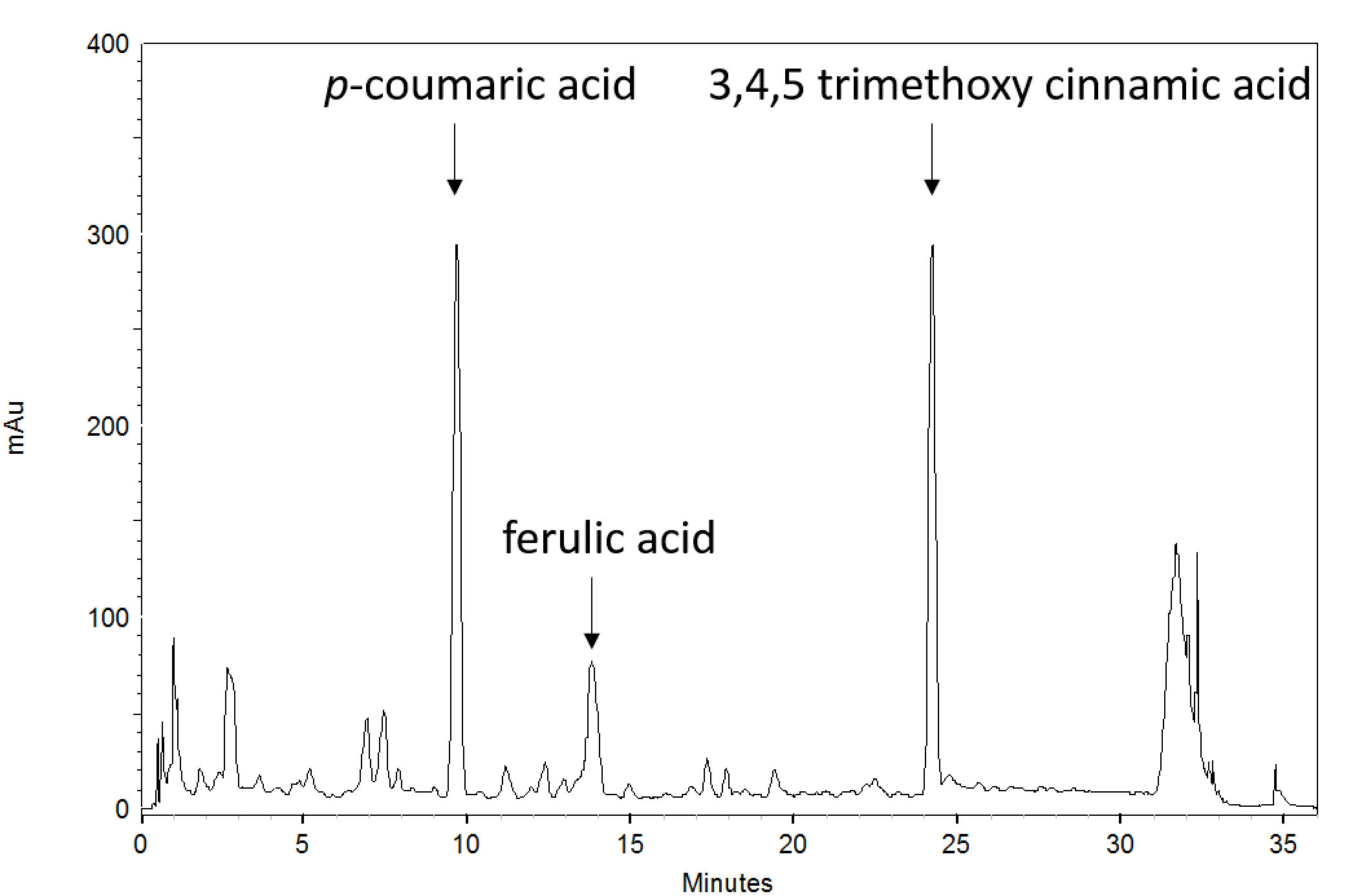
Figure 2. A representative chromatogram from an Arabidopsis sample at 330 nm. Peaks corresponding to p-coumaric acid, ferulic acid, and 3,4,5-trimethoxycinnamic acid have been identified on this chromatogram; however, authentic standards should always be run separately for peak identification.
Acknowledgments
This protocol was originally modified from Meyer et al. (1998) and was published in Anderson et al. (2015).
References
- Anderson, N. A., Bonawitz, N. D., Nyffeler, K. and Chapple, C. (2015). Loss of FERULATE 5-HYDROXYLASE leads to mediator-dependent inhibition of soluble phenylpropanoid biosynthesis in Arabidopsis. Plant Physiol 169(3): 1557-1567.
- Meyer, K., Shirley, A. M., Cusumano, J. C., Bell-Lelong, D. A. and Chapple, C. (1998). Lignin monomer composition is determined by the expression of a cytochrome P450-dependent monooxygenase in Arabidopsis. Proc Natl Acad Sci U S A 95(12): 6619-6623.
Article Information
Copyright
© 2016 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Readers should cite both the Bio-protocol article and the original research article where this protocol was used:
- Anderson, N. (2016). Cell Wall-bound p-Coumaric and Ferulic Acid Analysis. Bio-protocol 6(19): e1955. DOI: 10.21769/BioProtoc.1955.
- Anderson, N. A., Bonawitz, N. D., Nyffeler, K. and Chapple, C. (2015). Loss of FERULATE 5-HYDROXYLASE leads to mediator-dependent inhibition of soluble phenylpropanoid biosynthesis in Arabidopsis. Plant Physiol 169(3): 1557-1567.
Category
Biochemistry > Other compound > Acid
Plant Science > Plant biochemistry > Other compound
Plant Science > Plant cell biology > Tissue analysis
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link