- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
In vitro Flow Adhesion Assay for Analyzing Shear-resistant Adhesion of Metastatic Cancer Cells to Endothelial Cells
Published: Vol 6, Iss 4, Feb 20, 2016 DOI: 10.21769/BioProtoc.1731 Views: 14241
Reviewed by: Lee-Hwa TaiShannon RuppertAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Studying Cellular Focal Adhesion Parameters with Imaging and MATLAB Analysis
Ling-Yea Yu [...] Feng-Chiao Tsai
Nov 5, 2023 1879 Views
Isolation and Culture of Primary Pericytes from Mouse
Tamara McErlain [...] Meera Murgai
Apr 20, 2025 2986 Views
Abstract
Hematogenous metastasis is a primary cause of mortality from metastatic cancer. The shear-resistant adhesion of circulating tumor cells to the vascular endothelial cell surface under blood flow is an essential step in cell extravasation and further tissue invasion. This is similar to a process exploited by leukocytes for adhesion to inflamed blood vessels (leukocyte mimicry). The shear resistant adhesion is mediated by high affinity interactions between endothelial adhesion molecules and their counter receptor ligand expressed on circulating cells. Thus, weak interaction results in a rapid detachment of circulating cells from endothelium. Despite the critical role of vascular adhesion of cancer cells in hematogenous metastasis, our knowledge regarding this process has been limited due to the difficulty of mimicking dynamic flow conditions in vitro. In order to gain better insight into the shear-resistant adhesion of cancer cells to the endothelium, we developed a protocol for measuring the shear resistant adhesion of circulating tumor cells to endothelial cells under physiologic flow conditions by adapting a well established flow adhesion assay for inflammatory cells. This technique is useful to evaluate 1) the shear resistant adhesion competency of cancer cells and 2) the endothelial adhesion molecules necessary to support cancer cell adhesion (Kang et al., 2015).
Materials and Reagents
- Flow chamber: µ-Slide I 0.4 Luer (ibidi, catalog number: 80176 )
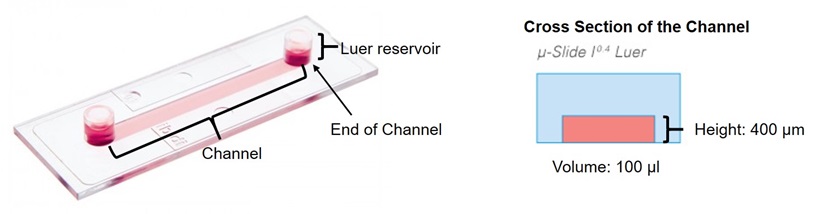
Figure 1. Flow chamber: µ-Slide I 0.4 Luer (modified the picture from www.ibidi.com) - Serial Connector for µ-Slides (ibidi, catalog number: 10830 )
- Syringe (10 ml, 30 ml)
- Silicone Tubing (0.8 mm ID) (ibidi, catalog number: 10841 )
- 50 ml tube with line connection (SARSTEDT AG & Co, catalog number: 60.596 )
- Y tube fitting (ibidi, catalog number: 10828 )
- Weight (SP Scienceware, Bel Art, catalog number: F183240000 )
- Hose clips (ibidi, catalog number: 10821 )
- Human microvascular endothelial cells (HMVEC) (Lonza, catalog number: CC2543 )
- Cancer cells
Cell line or primary cancer cells [e.g., human breast cancer cell line, MDA-MB-231 (ATCC, catalog number: CRM-HTB-26 )] - Endothelial basal medium-2 (Lonza, catalog number: CC-3156 )
- EGM SingleQuot Kit (Lonza, catalog number: CC-4133 )
- Dulbecco's Modification of Eagle's Medium (DMEM) (Thermo Fisher Scientific, Mediatech, Cellgro®, catalog number: 13-013 ) supplemented with 1% fetal bovine serum (FBS)
- Heat Inactivated Fetal Bovine Serum (FBS) (Life Technologies, catalog number: 10082-147 )
Note: Currently, it is “Thermo Fisher Scientific, GibcoTM, catalog number: 10082-147”. - Glutamax (Life Technologies, catalog number: 35050 )
Note: Currently, it is “Thermo Fisher Scientific, GibcoTM, catalog number: 35050 ”. - Antibiotic-antimycotic (Life Technologies, catalog number: 15240 )
Note: Currently, it is “Thermo Fisher Scientific, GibcoTM, catalog number: 15240 ”. - Collagen I, Rat Tail (Life Technologies, catalog number: A1048301 )
Note: Currently, it is “Thermo Fisher Scientific, GibcoTM, catalog number: A1048301”. - Fibronectin from human plasma (Sigma-Aldrich, catalog number: F0895 )
- Cell culture medium for HMVEC (see Recipes)
- Cell culture medium for Cancer cells (see Recipes)
Equipment
- Glass beaker (1,000 ml)
- Stir bar
- 37 °C, 5% CO2 cell culture incubator (NuAire, model: In-VitroCell ES NU-5800 )
- Cell culture centrifuge (Eppendorf, model: 5702 )
- Inverted Microscope with camera (Nikon, model: Eclipse TS100 )
- Camera (McCrone, model: MicroPublisher 3.3 RTV )
- KDS LegatoTM 200 syringe pump (KD Scientific)

Figure 2. KDS LegatoTM 200 syringe pump (modified picture from http://www.kdscientific.com/products/pumps/Legato200.asp) - Ibidi Heating System [Ibidi Temperature Controller ©ibidimodel (ibidi, model: Version 3.3)]

Figure 3. Ibidi Heating System (modified picture from http://ibidi.com/xtproducts/en/Instruments-Accessories/Heating-Incubation-Systems/ibidi-Heating-System-Universal-Fit) - Magnetic hot stir (Thermo Fisher Scientific, catalog number: 200-48SH)
- Thermometer (Thermo Fisher Scientific, catalog number: 13-201-695 )
- Level (Stanley Black & Decker, catalog number: 43-511)
Software
- Q-capture pro 7.0 (Qimaging)
Procedure
- Grow HMVEC monolayer in flow chamber (µ-Slide I 0.4 Luer)
- Dilute collagen (250 µg/ml) and fibronectin (75 µg/ml) with sterilized PBS. Fill the flow chamber with the collagen and fibronectin mixture solution and incubate for 1-3 h in a culture hood at RT (e.g., 100 µl of collagen and fibronectin mixture solution for µ-Slide I 0.4 Luer).
- To wash the flow chamber, add sterilized water into one channel end of flow chamber and take out the same volume from the other side channel end. Use 5 times the volume of flow chamber (e.g.,500 µl for µ-Slide I 0.4 Luer).
- Fill the chamber with 100 µl of fresh culture medium.
- Infuse HMVEC cells (1 x 105 of HMVEC cells in 100 μl HMVEC cells culture medium) into the collagen/fibronectin-coated flow chamber and remove pre-filled culture medium from the opposite side channel end. Do not remove culture medium from step 3 before infusing HMVEC cells to avoid the formation of air bubbles. Fill both Luer reservoirs of flow chamber (Figure 1). Incubate culture until HMVEC cells have grown to 100% confluence, approximately 2-3 days. It is critical to use 100% confluent endothelial monolayer to avoid cancer cells to be stuck in between the endothelial cells. To prevent drying of medium and keep sterile conditions, put caps (provided with flow chamber from manufacturer) on both Luer reservoirs. To change the culture medium inside the flow chamber, first, remove the old medium from both Luer reservoirs not inside the channel. Then, add fresh culture medium into one channel end of the flow chamber and remove the same volume of old medium from the opposite side channel end. Refill both Luer reservoirs of flow chamber (Figure 2).
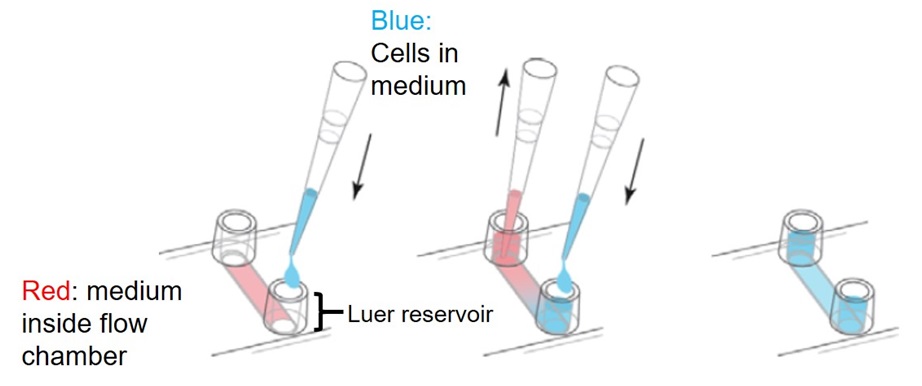
Figure 4. Cell seeding in flow chamber-Modified from Ibid Application Note 3 (http://ibidi.com/fileadmin/support/application_notes/AN03_Growing_cells.pdf)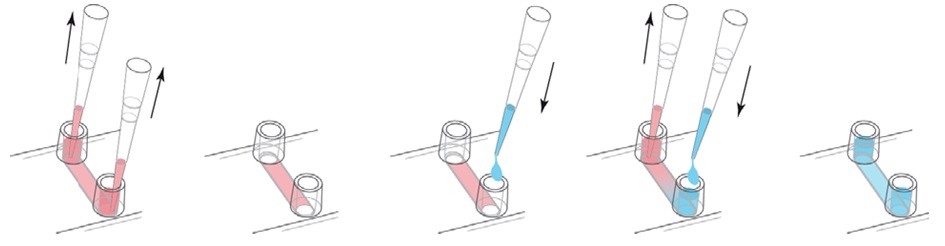
Figure 5. Change medium in flow chamber. (http://ibidi.com/fileadmin/support/application_notes/AN03_Growing_cells.pdf)
- Dilute collagen (250 µg/ml) and fibronectin (75 µg/ml) with sterilized PBS. Fill the flow chamber with the collagen and fibronectin mixture solution and incubate for 1-3 h in a culture hood at RT (e.g., 100 µl of collagen and fibronectin mixture solution for µ-Slide I 0.4 Luer).
- Assemble the flow chamber with pump and perform the flow adhesion assay
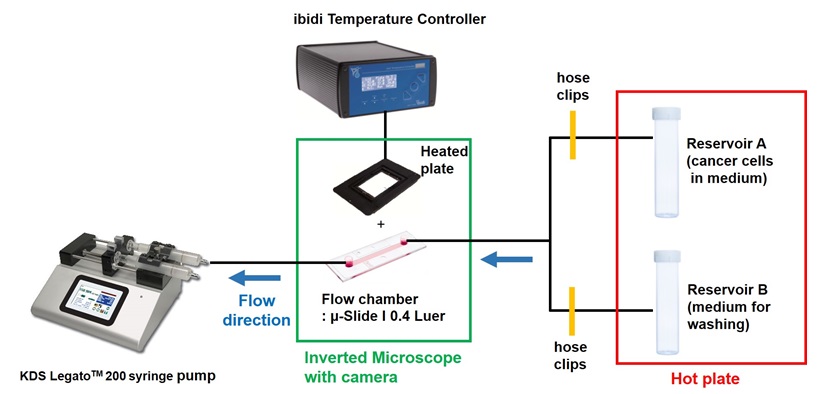
Figure 6. The diagram of overall setting for flow adhesion assay- Assemble reservoirs and turn on the hot plate to warm the water to 37 °C.

Figure 7. Assemble reservoirs (step B1) - Set the syringe pump next to the inverted microscope and turn ibidi heating system on at 37 °C.
- Fill reservoirs A and B with tissue culture medium and wash the line by plunging out with DMEM containing 1% FBS, ensuring there are no air bubbles. Close both tubings with hose clips.
- Prepare single cell suspension of cancer cells (MDA-MB-231 cells: 1 x 105 cell/ml in 10 ml of DMEM containing 1% FBS) and add to reservoir A with magnetic stir bar. (Concentration of cells may vary).

Figure 8. Addition of cells into Reservoir A, corresponding with step B4 - Place the flow chamber with HMVEC monolayer onto the ibidi heating system.

Figure 9. The process of line washing with culture medium via line connected to Reservoir B - Release the hose clip for reservoir B and drip culture media from this reservoir. Attach the serial connector end of tubing to Luer connector (IN). Avoid letting any air bubbles enter the channel.
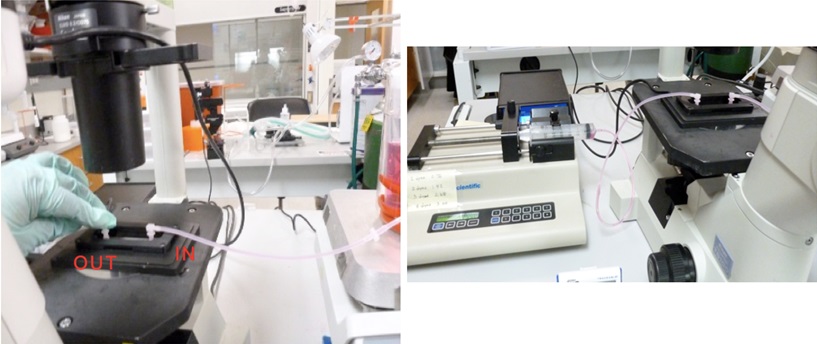
Figure 10. Connecting the line to the syringe pump (step B6) - Once the channel is filled with culture media, attach the serial connector from the syringe pump to Luer connector (OUT).
- Set the pump for syringe withdrawal at a constant flow rate. The flow rate will vary depending on shear stress calculations for each type of flow chamber. (e.g., for shear stress of 1 dyn/cm2 using µ-Slide I 0.4 Luer, the flow rate setting is 0.76 ml/min. For more information about flow rate and shear stress in different types of flow chambers, check Application Note 11 in http://ibidi.com/fileadmin/support/application_notes/AN11_Shear_stress.pdf). Different shear stresses should be tested. (*See Notes following procedure)

Figure 11. Leveling the heights of chamber and reservoir - To avoid gravity-based infusion, use a level to ensure the bottom of reservoir A and the flow are at the same height.

Figure 12. Start of flow adhesion assay - Run the syringe pump and check fluid flow inside the channel under microscope. Ensure there are no bubbles in the route or channel.
- Open the hose clip for reservoir A and close the hose clip for reservoir B. Infuse cancer cells (MDA-MB-231 cells: 1 x 105 cell/ml in 10 ml of DMEM containing 1% FBS) for 5 min.
- Close reservoir A and open B to wash the line with culture medium.
- Count the cancer cells adherent to HUMVEC under the inverted microscope or take photos (at least 5 fields) using Q-capture pro 7.0 and count the adherent cancer cells. Red arrow in Figure 13 shows MDA-MB-231 cells adhering to human endothelial cells at 1 dyn/cm2 for 5 min at 37 °C.
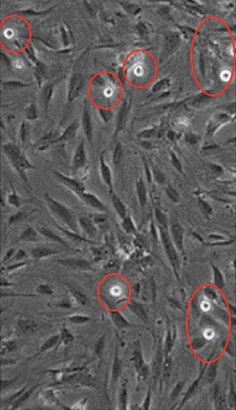
Figure 13. Image of cancer cell adhesion to monolayer of endothelial cells. Red arrow indicates cancer cells adhering to endothelial cells.
- Assemble reservoirs and turn on the hot plate to warm the water to 37 °C.
Notes
- The adhesion of each cell line is different. Experimental conditions may need to be optimized based on the cells of interest.
- It is worthwhile to test different flow rates. We usually use 1-5 dyn/cm2.
- It is important that endothelial cells are fully confluent with no empty space in between cells.
Recipes
- Cell culture medium for HMVEC
Endothelial basal medium-2 supplemented with 2% FBS
EGM SingleQuot Kit containing:
Epidermal growth factor (EGF) 10 ng/ml
Hydrocortisone 1 μg/ml
Gentamicin 30 μg/ml
Amphotericin B (GA-1000) 15 ng/ml - Cell culture medium for cancer cells (e.g., MDA-MB-231)
Dulbecco's modification of Eagle's medium (DMEM) supplemented with 10% fetal bovine serum (FBS)
1% glutamax
1% antibiotic-antimycotic
Acknowledgments
This work was supported by the Department of Defense (W81XWH-11-1-0238 to T. T), the National Institutes of Health (1R01CA160271-01A1 to T. T), the American Cancer Society (IRG-08-060-04), and the Pennsylvania Breast Cancer Coalition (T. T). Images were shared by ibidi USA, Inc., Madison WI (ibidi.com).
References
- Kang, S. A., Hasan, N., Mann, A. P., Zheng, W., Zhao, L., Morris, L., Zhu, W., Zhao, Y. D., Suh, K. S., Dooley, W. C., Volk, D., Gorenstein, D. G., Cristofanilli, M., Rui, H. and Tanaka, T. (2015). Blocking the adhesion cascade at the premetastatic niche for prevention of breast cancer metastasis. Mol Ther 23(6): 1044-1054.
Article Information
Copyright
© 2016 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Kang, S., Bajana, S. and Tanaka, T. (2016). In vitro Flow Adhesion Assay for Analyzing Shear-resistant Adhesion of Metastatic Cancer Cells to Endothelial Cells. Bio-protocol 6(4): e1731. DOI: 10.21769/BioProtoc.1731.
Category
Cancer Biology > Invasion & metastasis > Cell biology assays > Cell adhesion
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link