- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Density Gradient Centrifugation for Enrichment and Identification of GFP-tagged Chitosomal Microvesicles of Filamentous Fungi
(*contributed equally to this work) Published: Vol 5, Iss 19, Oct 5, 2015 DOI: 10.21769/BioProtoc.1611 Views: 16046
Reviewed by: Fanglian HeAksiniya AsenovaSadri Znaidi

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
An Assay to Determine NAD(P)H: Quinone Oxidoreductase Activity in Cell Extracts from Candida glabrata
Anamika Battu [...] Rupinder Kaur
Nov 5, 2021 3054 Views
Isolation of Mitochondria from Ustilago maydis Protoplasts
Juan Pablo Pardo [...] Lucero Romero-Aguilar
Jan 5, 2022 3578 Views
Use of the Fluorescent Dye Thioflavin T to Track Amyloid Structures in the Pathogenic Yeast Candida albicans
Thierry Mourer [...] Sophie Bachellier-Bassi
Feb 5, 2024 2339 Views
Abstract
Density gradient centrifugation has been utilized to characterize the subcellular distribution of physiologically relevant enzymes in yeasts and filamentous fungi (Leal-Morales et al., 1988; Martínez et al., 1989; Kamada et al., 1991). This approach is now potentiated by protein tagging and live imaging techniques, which make possible to relate a single protein with, for example, a discrete population of intracellular vesicles and their in vivo dynamics (Verdín et al., 2009; Fajardo-Somera et al., 2013; Sánchez-León et al., 2015). Here, we describe the density gradient centrifugation and fractionation analysis of cell-free homogenates of a Neurospora crassa (N. crassa) strain that expresses CHS-6 chitin synthase fused to the green fluorescent protein (Riquelme et al., 2007).
Keywords: Neurospora crassaMaterials and Reagents
- Tygon R-3603 tubing, I.D. 1/16 inch (Saint-Gobain, catalog number: AAC00002 )
- Grade 1 Qualitative Filter Papers (GE Healthcare, catalog number: 1001-090 )
- Neurospora crassa conidia
- Sucrose (Sigma-Aldrich, catalog number: S0389 )
- Sodium phosphate monobasic (NaH2PO4.H2O) (Thermo Fisher Scientific, Fisher Scientific, catalog number: S369-500 )
- Sodium phosphate dibasic anhydrous (Na2HPO4) (Sigma-Aldrich, catalog number: S-7907 )
- Protease Inhibitor Cocktail, Complete ULTRA tablets EDTA-free (Roche Diagnostics, catalog number: 04693132001 )
Note: Currently, it is “Sigma-Aldrich, catalog number: 04693132001 ”. - Glass beads (500 μm) (Bio Spec Product, catalog number: 11079105 )
- 10% (w/v) sucrose in 33 mM (final) phosphate buffer (pH 8.2) (steam sterilize and store at 4 °C until use)
- 65% (w/v) sucrose in 33 mM (final) phosphate buffer (pH 8.2) (steam sterilize and store at 4 °C until use)
- D-Biotin (Faga Labs, catalog number: CAS-58-85-5 )
- Vogel’s complete medium (see Recipes)
- 50x salt solution (see Recipes)
- Trace element solution (see Recipes)
- 100 mM phosphate buffer (pH 8.2) (see Recipes)
- Laemmli buffer (see Recipes)
Equipment
- Braun MSK 50 ml shaking bottles (LABEQUIP LTD, catalog number: 8541302 )
- VWR® Standard Hot Plate Stirrers (VWR International, catalog number: 12365-382 )
- Quick-seal centrifuge tube (Beckman Coulter, catalog number: 344326 )
- Shaker incubator (Lab-LineX, model: Orbit Environ Shaker )
- Vacuum filtration system
- Braun MSK cell homogenizer (LABEQUIP LTD, catalog number: 953030 )
- Bright field microscope (Olympus, model: Vanox-S )
- Beckman L8-70M Ultracentrifuge (pre-cooled at 4 °C) (Beckman Coulter)
- Type 70Ti rotor (pre-cooled at 4 °C) (Beckman Coulter)
- GM-40 Linear Gradient maker (C. B. S. Scientific)
- Peristaltic (Multi-staltic) pump (Buchler Lab, catalog number: N/A )
- Tube sealer (Beckman Coulter, catalog number: 342420 )
- Density gradient fractionator (ISCO, 185)
Note: This model is not available anymore. Brandel BR-186 Gradient Fractionator with Syringe Pump can be used instead. - CIGNETTM Fraction collector (ISCO)
Note: This item is also discontinued, but Bio-Rad 2110 Fraction Collector can be used instead. - 6505 W UV-Vis Spectrophotometer (Bibby-scientific, Jenway)
- Refractometer (ZEISS, catalog number: 12230 )
Procedure
- Homogenate (lysate) preparation
- Inoculate 400 ml Vogel’s Complete Medium (Vogel, 1956) with Neurospora crassa conidia (1 x 106 conidia/ml) in a 1 L flask. Incubate for 14-20 h at 30 °C, 200 rpm.
- Harvest mycelium by vacuum filtration through Whatman filter paper number 1. Wash twice with 200 ml cold sterile distilled H2O to eliminate remnant culture media and then twice with 50 ml cold 33 mM phosphate buffer (pH 8.2) to equilibrate mycelium for the next step.
- Mix 10 g wet mycelium and 10 ml 5% (w/v) sucrose in 33 mM phosphate buffer (pH 8.2), supplemented with a protease inhibitor cocktail (1 tablet/50 ml) into a Braun bottle and add 20 g of glass beads (500 μm; 2 g/g mycelium). Keep all solutions, tubes and bottles at 4 °C to prevent protein degradation.
- Homogenize in a Braun homogenizer 4 x 30 sec in presence of a CO2 stream directed to the Braun bottle so that a temperature increase during homogenization is prevented. The CO2 line must be firmly attached to the metallic arm of the Braun homogenizer so that the CO2 stream can be directed to the bottle. After homogenization, verify the broken cells by observing the sample in a bright field microscope.
- Transfer homogenate into a 50 ml tube (on ice) and centrifuge at 1,000 x g (Rav), 10 min, 4 °C.
- Save the supernatant at 4 °C using a glass pipette to avoid the lipid layer formed on top. This will be the homogenate or lysate, which would contain mitochondria, microvesicles, ribosomes, and soluble proteins. Discard pellet containing whole cells, cell walls, and nuclei.
- Inoculate 400 ml Vogel’s Complete Medium (Vogel, 1956) with Neurospora crassa conidia (1 x 106 conidia/ml) in a 1 L flask. Incubate for 14-20 h at 30 °C, 200 rpm.
- Density gradient centrifugation
- Construct a 10-65% sucrose linear gradient (10% and 65% sucrose stock solutions must be prepared in final 33 mM phosphate buffer, pH 8.2) in a quick-seal centrifuge tube using a linear gradient maker and a peristaltic pump set up to obtain a flow rate of 3.5 ml/min (Figure 1).
- Gently layer the N. crassa homogenate (supernatant saved in step A6) on top of the 10-65% sucrose gradient by using a Pasteur pipette via single drops close to the surface of the sucrose gradient. Grease the centrifuge inlet to seal and cap the centrifuge tube. Centrifuge at 184,000 x g (Rav), for 4 h and 4 °C in a pre-cooled Beckman rotor 70Ti.
- Fractionate the gradient from the top with an ISCO fractionation system using 70% (w/v) sucrose as chase at a flow rate of 6 ml/min. Make sure no air bubbles form in the tube system that conducts the chase sucrose to the gradient or else they will break it. Collect 2 ml fractions (3 fractions/min) into ice-cold tubes and keep at 4 °C.
- Characterize each fraction by measuring absorbance at 280 nm, refraction index (to infer the density) and, ideally, an enzyme activity associated with intracellular particles or vesicles (Figure 2A). Refractometers output the refractive index, which can be converted to density (g/ml) using this table: http://homepages.gac.edu/~cellab/chpts/chpt3/table3-2.html.
- Mix 50 to 100 μl of each fraction with Laemmli buffer (2x), boil for 5 min and store at -20 °C until analysis by Western blot (Figure 2B).

Figure 1. Preparation and fractionation of sucrose density gradients. A. To prepare the density gradients, sucrose stock solutions are independently poured into each column of the gradient maker (keep locked the Teflon valve that connects one column to the other). A magnetic stir bar is placed in the 10% sucrose solution column, from which a silicon tube (purged of any bubble) exits towards the peristaltic pump and, via a capillary tube attached to the tip of the silicon one, is introduced into the quick-seal centrifuge tube down to the bottom. After turning the peristaltic pump on, the Teflon valve is opened to allow the sucrose solutions mixing. Then, increasing concentration sucrose flows to the quick-seal tube. Lower concentration sucrose arrives first to the tube, which is pushed up by higher concentration sucrose (the capillary tip is always touching the quick-seal tube bottom). Immediately before the transfer of total volume of the sucrose gradient to the quick-seal tube, turn off the peristaltic pump and take out the capillary tube very gently to avoid breaking the gradient. The remaining volume of the quick-seal tube is filled up with the mycelial lysate. B. After centrifugation and isopycnic separation of mycelial homogenates, the gradient is carefully fractionated using a 70% sucrose solution as chase. This solution is injected into the quick-seal tube from the bottom so that the gradient is integrally pushed up toward the exit tubing and the fraction collector. Before fractionation, it is critical to purge any bubble from the syringe that contains the chase solution and the tubing that conducts it to the bottom of the quick-seal tube; otherwise, the gradient could be broken. Collected fractions are subsequently characterized (enzyme activity, refractive index, absorbance, etc.) and stored at 4 °C.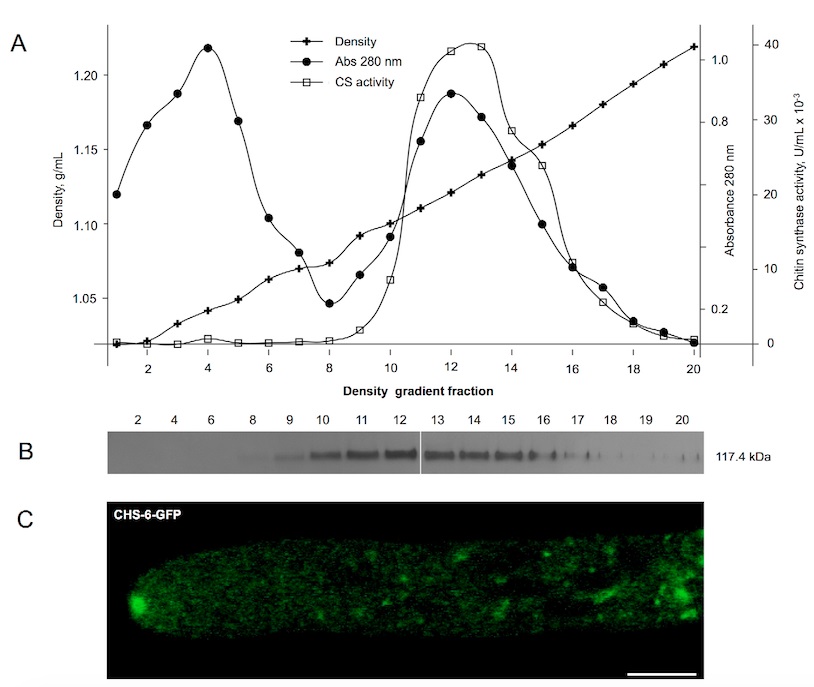
Figure 2. Density gradient centrifugation of homogenates of N. crassa expressing CHS-6 chitin synthase co-translationally fused to GFP (Riquelme et al., 2007). A. Density (inferred from the refractive index), total protein (Abs 280 nm) and chitin synthase activity were estimated for each fraction of the density gradient collected. Chitin synthase activity sedimented at 1.13 g/ml, the buoyant density of chitosomes. B. Western blot analysis demonstrated the presence of CHS-6-GFP around fraction 13 (1.13 g/ml). C. Live imaging of a N. crassa hyphae by confocal microscopy showed that CHS-6-GFP localizes at the tip mainly at the core of the Spitzenkörper (an apical body that directs the growth of the hypha), where microvesicles (chitosomes) accumulate, as confirmed by transmission electron microscopy (Riquelme et al., 2002). Scale bar, 5 μm
- Construct a 10-65% sucrose linear gradient (10% and 65% sucrose stock solutions must be prepared in final 33 mM phosphate buffer, pH 8.2) in a quick-seal centrifuge tube using a linear gradient maker and a peristaltic pump set up to obtain a flow rate of 3.5 ml/min (Figure 1).
Recipes
- Vogel’s complete medium
Dilute 50x salt solution 50 fold with distilled water
Add:
Sucrose 15 g/L
0.5% yeast extract
0.5% N-Z case (casamino acids)
Steam sterilize and store at room temperature - 50x salt solution
Bring up to 100 ml with ddH2ONa3C6H5O7.5.5 H2O Sodium citrate 420 mM KH2PO4 Potassium phosphate monobasic anhydrous 1.8 M NH4NO3 Ammonium nitrate anhydrous 1.2 M MgSO4.7H2O Magnesium sulfate heptahydrate 40.5 mM CaCl2.2H2O Calcium chloride dihydrate 34 mM Trace element solution* 500 μl Biotin solution (0.1 mg/ml, in ethanol) 250 μl
Add 2 ml of chloroform as preservative
Alternatively, steam sterilize and store at 4 °C - Trace element solution
Note: Concentrations in brackets are final concentrations.
Bring up to 100 ml with ddH2OCitric Acid.H2O 5.0 g (238 mM) ZnSO4.7H2O 5.0 g (174 mM) Fe(NH4)2(SO4)2.6H2O 1.0 g (25.5 mM) CuSO4.H2O 0.25 g (14 mM) MnSO4.H2O 0.05 g (2.3 mM) H3BO3 anhydrous 0.05 g (8 mM) Na2MoO4.2H2O 0.05 g (2.3 mM)
Add 1 ml of chloroform for storage at room temperature - 100 mM phosphate buffer (pH 8.2)
NaH2PO4.H2O 1.23 g Na2HPO4 12.92 g H2O up to 1 L - Laemmli bufferStored at -20 °C
0.5 M Tris (pH 6.8) 1.25 ml 10% SDS 2.0 ml Glycerol 2.5 ml ddH2O 9.5 ml Bromophenol blue 1 mg
Add 25 μl β-mercaptoethanol to 475 μl Laemmli buffer before use
Dilute samples 1:2 in Laemmli buffer and boil for 4 min
Acknowledgments
This work was supported by Mexican National Council for Science and Technology (CONACYT) grants CB-222375, CB2008-105600-Q, and U45818-Q.
References
- Fajardo-Somera, R. A., Bowman, B. and Riquelme, M. (2013). The plasma membrane proton pump PMA-1 is incorporated into distal parts of the hyphae independently of the Spitzenkörper in Neurospora crassa. Eukaryot Cell 12(8): 1097-1105.
- Kamada, T., Bracker, C. E., Lippman, E. and Bartnicki-García, S. (1991). Unexpected destruction of chitosomal chitin synthetase by an endogenous protease during sucrose density gradient purification. J Cell Sci 99 (Pt 3): 565-570.
- Leal-Morales, C. A., Bracker, C. E. and Bartnicki-García, S. (1988). Localization of chitin synthetase in cell-free homogenates of Saccharomyces cerevisiae: chitosomes and plasma membrane. Proc Natl Acad Sci U S A 85(22): 8516-8520.
- Martínez, J. P., Gimenez, G., Bracker, C. E. and Bartnicki-García, S. (1989). Sedimentation properties of chitosomal chitin synthetase from the wild-type strain and the 'slime' variant of Neurospora crassa. Biochim Biophys Acta 990(1): 45-52.
- Riquelme, M., Roberson, R. W., McDaniel, D. P. and Bartnicki-Garcia, S. (2002). The effects of ropy-1 mutation on cytoplasmic organization and intracellular motility in mature hyphae of Neurospora crassa. Fungal Genet Biol 37(2): 171-179.
- Riquelme, M., Bartnicki-García, S., González-Prieto, J. M., Sánchez-León, E., Verdín-Ramos, J. A., Beltrán-Aguilar, A. and Freitag, M. (2007). Spitzenkörper localization and intracellular traffic of green fluorescent protein-labeled CHS-3 and CHS-6 chitin synthases in living hyphae of Neurospora crassa. Eukaryot Cell 6(10): 1853-1864.
- Sánchez-León, E., Bowman, B., Seidel, C., Fischer, R., Novick, P. and Riquelme, M. (2015). The Rab GTPase YPT-1 associates with Golgi cisternae and Spitzenkörper microvesicles in Neurospora crassa. Mol Microbiol 95(3): 472-490.
- Verdín, J., Bartnicki-García, S. and Riquelme, M. (2009). Functional stratification of the Spitzenkörper of Neurospora crassa. Mol Microbiol 74(5): 1044-1053.
- Vogel, H. J. (1956). A convenient growth medium for Neurospora (medium N). Microbiol Genet Bull 13:42-43.
Article Information
Copyright
© 2015 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Verdín, J., Sánchez-León, E., Fajardo-Somera, R., Morales, C. A. L., Bartnicki-García, S. and Riquelme, M. (2015). Density Gradient Centrifugation for Enrichment and Identification of GFP-tagged Chitosomal Microvesicles of Filamentous Fungi. Bio-protocol 5(19): e1611. DOI: 10.21769/BioProtoc.1611.
Category
Microbiology > Microbial biochemistry > Protein
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link










