- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Measuring the Arsenic Content and Speciation in Different Rice Tissues
Published: Vol 5, Iss 8, Apr 20, 2015 DOI: 10.21769/BioProtoc.1445 Views: 10268
Reviewed by: Arsalan DaudiMohan TCZhaohui Liu

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Targeting Ultrastructural Events at the Graft Interface of Arabidopsis thaliana by A Correlative Light Electron Microscopy Approach
Clément Chambaud [...] Lysiane Brocard
Jan 20, 2023 2973 Views
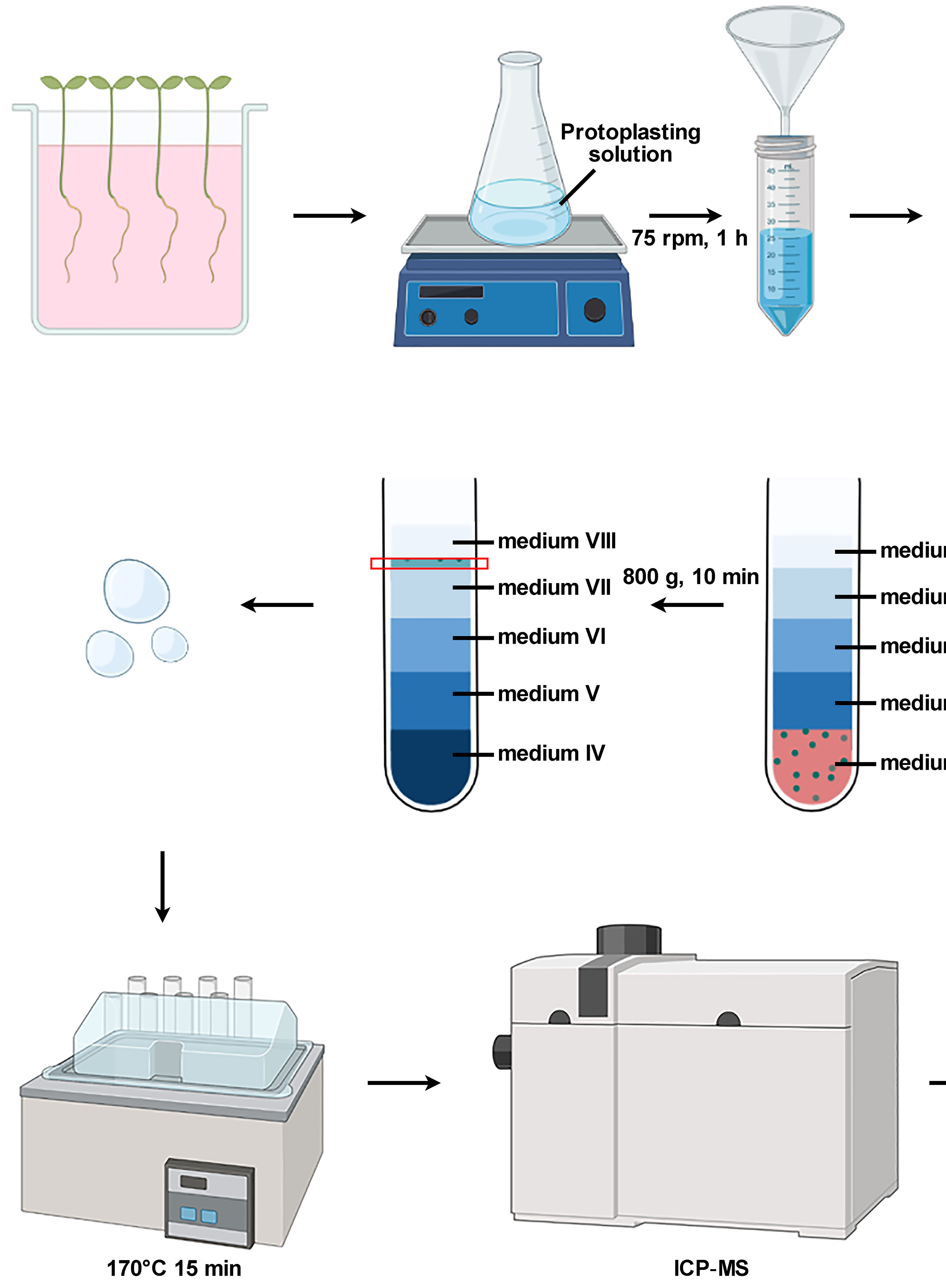
Isolation of Intact Vacuoles from Arabidopsis Root Protoplasts and Elemental Analysis
Chuanfeng Ju [...] Zhenqian Zhang
Mar 5, 2023 2035 Views
High-Performance Liquid Chromatography Quantification of Glyphosate, Aminomethylphosphonic Acid, and Ascorbate in Culture Medium and Microalgal Cells
Juan Manuel Ostera [...] Gabriela Malanga
Apr 5, 2025 1171 Views
Abstract
Arsenic (As) plays an important role in rice production as vast soils used for rice cultivation contain As. To understand how rice plants deal with inorganic As (III) and As (V) and organic As in their tissue, it is important to obtain specific information on how much and what species of As are present in which tissue of the rice plant. The protocol presented here allows to analyse the As contents and As speciation in roots, shoots, and husks of rice plants, and thus permits direct comparison of the As contents of these rice plant tissues.
Keywords: O. sativaMaterials and Reagents
- Rice tissue (roots, shoots, husks) (Muehe et al., 2014)
- Liquid nitrogen
- 65% HNO3 (Merck KGaA, catalog number: 100456 )
- Double-distilled or MilliQ water
- >30% H2O2 (Merck KGaA, catalog number: 107298 )
- 85% phosphoric acid (Merck KGaA, catalogue number: 100563 )
- 0.05 mol/L as stock solution as arsenite (Merck KGaA, catalog number: 106277 ), a 1,000 mg/L as stock solution as arsenate (Merck KGaA, catalog number: 119773 ) diluted to the appropriate concentrations
- NH4NO3 (Merck KGaA, catalog number: 102596 )
- Eluent A for LC-HPLC-ICP-MS (see Recipes)
- Eluent B for LC-HPLC-ICP-MS (see Recipes)
Equipment
- Mortar (diameter of at least 10 cm) and pestle (approximately 15 cm long)
- Drying oven (adjustable to 60 °C)
- WhatmanTM #1 filter papers (GE Healthcare, catalog number: 1440-125 )
- Duran® test tubes, straight rim (13 x 100 mm) (Duran Group, catalog number: 26 131 12 )
- Polyethylene flanged plug caps for tubes, natural (Globe Scientific, catalog number: 118240 )
- Polytetrafluoroethylene (PTFE)-coated spatula (Fisher Scientific, catalog number: 10081074 ) to minimize metal contamination of the plant material from the spatula
- Volumetric flask with glass stopper (10 ml)
- Tube rack
- Water bath
- Glass funnel
- Inductively coupled plasma atomic emission spectrometry ICP-AES (CIROS)
- Inductively coupled plasma mass spectroscopy ICP-MS (Agilent Technologies, model: 7700 )
- Autosampler vials (8 mm, brown glass, WICOM, vials are free of As) and screw caps with Teflon septum
- Liquid chromatography (Agilent Technologies, model: 1200 ) equipped with a Shodex RSpak NN-614 column (150 mm x 6 mm; Shodex) coupled to inductively coupled plasma mass spectroscopy HPLC-ICP-MS (Agilent Technologies, model: 7700 )
Procedure
- Material preparation
Glassware (tubes, volumetric flasks), pestle, and mortar are soaked in 1 M HCl (wear gloves and safety goggles) for 24 h and washed in distilled water three times and dried before use. This is done to remove metal contaminants sorbed to the equipment.
- Harvesting of plant material
- Rice plant material is separated into different tissues (roots, shoots, husks, and seeds) using clean scissors and tweezers and wearing gloves.
- Plant material is dipped in distilled water for washing.
- (Approximately 1-5 g of fresh weight) tissue is ground in liquid nitrogen using a mortar and pestle (3 times, for 30 sec each, see Figure 1) (ground material can also be stored at -80 °C for molecular biology analysis) (Mueller et al., 2013).
- An approximately 1 g fresh weight aliquot of the liquid-nitrogen-ground plant material is filled into an opened falcon tube and immediately dried at 60 °C in an oven for a minimum of three days.

Figure 1. Liquid N2-ground rice shoots
- Rice plant material is separated into different tissues (roots, shoots, husks, and seeds) using clean scissors and tweezers and wearing gloves.
- Total arsenic quantification in rice material (wear gloves and goggles)
- 20-70 mg of the ground and dried material is weighed into Duran tubes (ensure that plant material is not aggregated).
- 2 ml of 65% HNO3 is added to the plant material; followed by mixing the acid and plant material by gentle shaking/swirling of the tube.
- Duran tubes are closed with plastic lids.
- Rice material is digested over night at room temperature.
- Tubes are placed in a rack, which is placed in a water bath.
- The material is boiled for 2 h at 100 °C.
- 2 ml of ≥30% H2O2 is added to the tubes.
- The rice material is boiled until brown gasses stop evolving.
- Samples are cooled to room temperature by incubating on the lab bench for an hour.
- Material is fully digested if no more tissue pieces are present in the extract.
- (If plant material is not fully digested (visible by small bits of see-through tissue), the extract is filtered through Whatman #1 filter papers).
- The extract is filled into a volumetric flask.
- The Duran tubes are washed with a few ml of distilled water, which is then added to the extract in the volumetric flask.
- The extract is filled up to 10 ml with distilled water and stored cool and dark until analysis.
- Total As is determined by ICP-AES (concentrations > 0.1 mg/L) or for lower concentrations by ICP-MS (Limit of detection: 0.2 µg/L).
- 20-70 mg of the ground and dried material is weighed into Duran tubes (ensure that plant material is not aggregated).
- Arsenic speciation in rice material
- 20-50 mg of fresh plant material (roots, shoots, and husks) are cut into 1 cm long pieces and are weighed into Falcon tubes immediately after sampling.
- 10 ml 10 mM phosphoric acid is added to the plant material (Daus et al., 2008).
- Rice material is extracted for 2 h by shaking at 150 rpm and room temperature.
- The material is filtered through Whatman #1 filter paper.
- To prevent changes in As speciation, samples are immediately filled into the autosampler vials. If unavoidable, samples can be stored in the dark at low temperature (6 °C) until measurement.
- The redox species of arsenate and arsenite are analysed with HPLC–ICP–MS using brown glass autosampler vials.
- Additionally, the dry weight of the plant material is estimated by weighing an aliquot before and after drying at 60 °C.
- The amount of each As species in the fresh plant material is calculated per dry plant material and can be correlated to the total As content of the material.
- Please refer to Muehe et al. (2014) for representative amounts of As species calculated using this protocol.
- 20-50 mg of fresh plant material (roots, shoots, and husks) are cut into 1 cm long pieces and are weighed into Falcon tubes immediately after sampling.
Notes
- Seeds can also be sampled, but should be ground in a grinder beforehand.
- Grinding of the sample material in liquid nitrogen assures the destruction of the material and thorough mixing of the entire sample material, and thus, unbiased analysis when using small sample aliquots for further procedures.
- Storing the liquid N2-ground plant material at -80 °C allows for molecular analysis of the material as well. For example, in Muehe et al. (2014) we extracted RNA from these samples and performed qPCR on the cDNA product of the extracted RNA.
- The use of the phosphoric acid as extractant yields an almost complete recovery of the total As present in the material. The As species in these extracts are stable up to 4 weeks if they are stored cool and dark (Daus et al., 2006).
- More detailed information on ICP-AES and HPLC-ICP-MS can be obtained in Daus et al. (2002), Daus et al. (2006), Daus et al. (2008) and Noelte (2003).
Recipes
- Eluent A for LC-HPLC-ICP-MS
5 mM HNO3
- Eluent B for LC-HPLC-ICP-MS
5 mM HNO3 + 50 mM NH4NO3 [for As (III) and As (V) only 7 min isocratic 94% A]
Flow: 1 ml/min, injection volume: 50 µl, LOD = 0.2 µg/L
Acknowledgments
This work was funded by the scholarship program of the German Federal Environmental Foundation to EMM and the Deutsche Forschungsgemeinschaft (DFG) to AK (KA 1736/14-1). The procedure was previously employed in Muehe et al. (2014).
References
- Daus, B., Mattusch, J., Wennrich, R. and Weiss, H. (2002). Investigation on stability and preservation of arsenic species in iron rich water samples. Talanta 58(1): 57-65.
- Daus, B., Mattusch, J., Wennrich, R. and Weiss, H. (2008). Analytical investigations of phenyl arsenicals in groundwater. Talanta 75(2): 376-379.
- Daus, B., Weiss, H., Mattusch, J. and Wennrich, R. (2006). Preservation of arsenic species in water samples using phosphoric acid--limitations and long-term stability. Talanta 69(2): 430-434.
- Muehe, E. M., Eisele, J. F., Daus, B., Kappler, A., Harter, K. and Chaban, C. (2014). Are rice (Oryza sativa L.) phosphate transporters regulated similarly by phosphate and arsenate? A comprehensive study. Plant Mol Biol 85(3): 301-316.
- Muller, K., Daus, B., Mattusch, J., Vetterlein, D., Merbach, I. and Wennrich, R. (2013). Impact of arsenic on uptake and bio-accumulation of antimony by arsenic hyperaccumulator Pteris vittata. Environ Pollut 174: 128-133.
- Noelte, D. (2003). ICP Emission Spectrometry, Wiley-VCH, 1st Edition.
Article Information
Copyright
© 2015 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Muehe, E. M., Kappler, A., Chaban, C. and Daus, B. (2015). Measuring the Arsenic Content and Speciation in Different Rice Tissues. Bio-protocol 5(8): e1445. DOI: 10.21769/BioProtoc.1445.
Category
Plant Science > Plant biochemistry > Other compound
Plant Science > Plant physiology > Tissue analysis
Plant Science > Plant physiology > Ion analysis
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link









