- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Extraction of Total Proteins from Rice Plant
Published: Vol 4, Iss 21, Nov 5, 2014 DOI: 10.21769/BioProtoc.1277 Views: 44972
Reviewed by: Feng LiCindy AstAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Streamlining Protein Fractional Synthesis Rates Using SP3 Beads and Stable Isotope Mass Spectrometry: A Case Study on the Plant Ribosome
Dione Gentry-Torfer [...] Federico Martinez-Seidel
May 5, 2024 2855 Views

An Activity-Based Proteomics with Two-Dimensional Polyacrylamide Gel Electrophoresis (2D-PAGE) for Identifying Target Proteases in Arabidopsis Apoplastic Fluid
Sayaka Matsui and Yoshikatsu Matsubayashi
Mar 5, 2025 1975 Views
Advancing 2-DE Techniques: High-Efficiency Protein Extraction From Lupine Roots
Sebastian Burchardt [...] Emilia Wilmowicz
Oct 5, 2025 1763 Views
Abstract
This protocol provides an efficient method for preparation of high-quality proteins from rice leaves and grains. The method involves phenol extraction to separate proteins from the non-protein components such as polysaccharides, lipids and phenolic compounds that are commonly enriched in plant tissues. Following isolation, proteins are precipitated with ammonium acetate/methanol and then solubilized for proteomic analysis. As the protocol is simple, universal, and most importantly compatible with silver staining, it has been applied to our routine protein extraction from rice and many other plant tissues and it even works fine in animal tissues for the requirement of electrophoretic separation.
Keywords: RiceMaterials and Reagents
- Tainung 67
Note: TNG67, an elite japonica type rice variety, has been a leading variety in Taiwan for more than 30 years since it been released in 1978. - SA0420
Note: An aroma mutant, exhibits an agreeable taro-like flavor, was selected from the TNG67 mutation pool developed via sodium azide mutagenesis. - Liquid nitrogen
- Acetone (Merck KGaA, catalog number: 1.00012.4000 )
- NH4OAc (Merck KGaA, catalog number: 1.01116.1000 )
- DTT (Merck KGaA, catalog number: 1.11474.0001 )
- CHAPS (GE Healthcare, catalog number: 17-1314-01 )
- HCl (Merck KGaA, catalog number: 1.09063.1000 )
- 2-mercaptoethanol (Sigma-Aldrich, catalog number: M6250 )
- 2-propanol (Merck KGaA, catalog number: 1.09634.2500 )
- Methanol (Merck KGaA, catalog number: 1.06007.4000 )
- NaCl (Merck KGaA, catalog number: 7710CN )
- NaOH (Merck KGaA, catalog number: 1.06469.1000 )
- KCl (Merck KGaA, catalog number: 1.04936.1000 )
- PMSF (Merck KGaA, catalog number: 52332 )
- Phenol (saturated, pH 6.6/7.9) (AMRESCO, catalog number: 0945 )
- PVPP (Merck KGaA, catalog number: 1.07302.0100 )
- Seasand (Merck KGaA, catalog number: 107711 )
- Sucrose (GERBU Biotechnik GmbH, catalog number: 1366 )
- Triton X-100 (GERBU Biotechnik GmbH, catalog number: 2000 )
- Tris-base (AMRESCO, catalog number: 0497 )
- Urea (AMRESCO, catalog number: 0378 )
- Miracloth (Calbiochem®, catalog number: 475855-1R )
- Extraction buffer (see Recipes)
- Precipitation buffer (see Recipes)
- 0.5 M EDTA (see Recipes)
- Lysis buffer (see Recipes)
- 40 mM PMSF (see Recipes)
Equipment
- Mortar (Φ 9 cm) and pestle (attached with Φ 12 cm mortar) (Figure 1)

Figure 1. Mortar and pestle - Pipetman (Gilson, model: p5000 , p1000 , p200 , p100 , p20 and p10 )
- High-Speed Refrigerated Centrifuges and No.46 Rotor (Hitachi, model: himac CR22F )
- 50 ml centrifuge tube (Thermo Fisher Scientific, Nalgene®, model: 3119-0050 )
- -20 °C refrigerator (Firstek Scientific, model: F-25 )
- SpeedVac system (Thermo Fisher Scientific, model: SPD speed Vac and UVS400 universal vacuum system )
- Ultrasonic machine (ULTRASONIC, model: LC130H )
- Chemical fume hood (CHUNG FU, model: HAP-090 )
Procedure
- Preparation of protein samples from rice leaves
- Cut 0.5 g fresh or -80 °C frozen leaf samples into small pieces, add little sea sand (Figure 2A) and freeze the tissue by pouring liquid nitrogen on the cut leaves in the mortar (Video 1).
- Grind the tissue to a fine powder, using a mortar and pestle (Figure 2B, Video 1).
- Add 5 ml extraction buffer and immediately grind the sample until foaming (Figure 2C, Video 1).
- Add 0.5 g PVPP and grind completely. Then add 5 ml extraction buffer and grind the sample until foaming (Video 1).
- Add 250 μl 40 mM PMSF and 5 ml extraction buffer, and then grind the mixture until foaming (Video 1).
- Transfer the homogenate into a sterilized 50 ml centrifuge tube (Video 1).
- The remaining plant material sticking on mortar and pestle is rescued by carefully rinsing mortar and pestle with 5 ml extraction buffer, and poured into the tube and mixed well with the homogenate (Video 1).
- Centrifuge 20 min at 15,000 rpm (in No. 46 Rotor), 4 °C.
- Filter the supernatant through 2 layers of Miracloth into another fresh tube (Figure 2F).
- Extract the filtrate with an equal volume of buffer saturated phenol (about 16 ml). Mix well by inversion. Centrifuge 20 min at 15,000 rpm, 4 °C.
- Transfer the top layer (phenol phase) into another fresh tube (Figure 2G and 2H).

Figure 2. Preparation of protein sample. A-E. Leaves were ground and extracted by extraction buffer. F-H. Protein sample was purified by phenol extraction. - Extract the phenol phase with an equal volume of cold extraction buffer twice (1st 14 ml and 2nd 12 ml, respectively). Mix well by inversion. Centrifuge 20 min at 15,000 rpm, 4 °C. Recover the top layer (phenol phase).
- Add 3x volume of cooled precipitation buffer to the recovered phenol phase and mix well by inversion.
- Incubate at -20 °C for 4 h (or even overnight) to precipitate the proteins. Then centrifuge 20 min at 15,000 rpm, 4 °C and discard the supernatant.
- Wash the pellet with 1.8 ml precipitation buffer twice. Recover the pellet by centrifugation at 8,000 rpm for 5 min.
- Wash the pellet with 1 ml of cold 100% acetone containing with 0.7 μl 2-mercaptoethanol (14.3 M). Recover the pellet by centrifugation at 8,000 rpm for 5 min.
- Dry the pellet by SpeedVac for about 2-3 min (less than 5 min) and resuspend in an appropriate volume of lysis buffer.
- The protein samples can be quantified by Bradford method (Bradford, 1976) and stored at -20 °C.
- The obtained protein samples can be used for proteomic analysis (Lin et al., 2014; Liu et al., 2012) or Western blot (Lin and Jeang, 2005) (Figure 3).
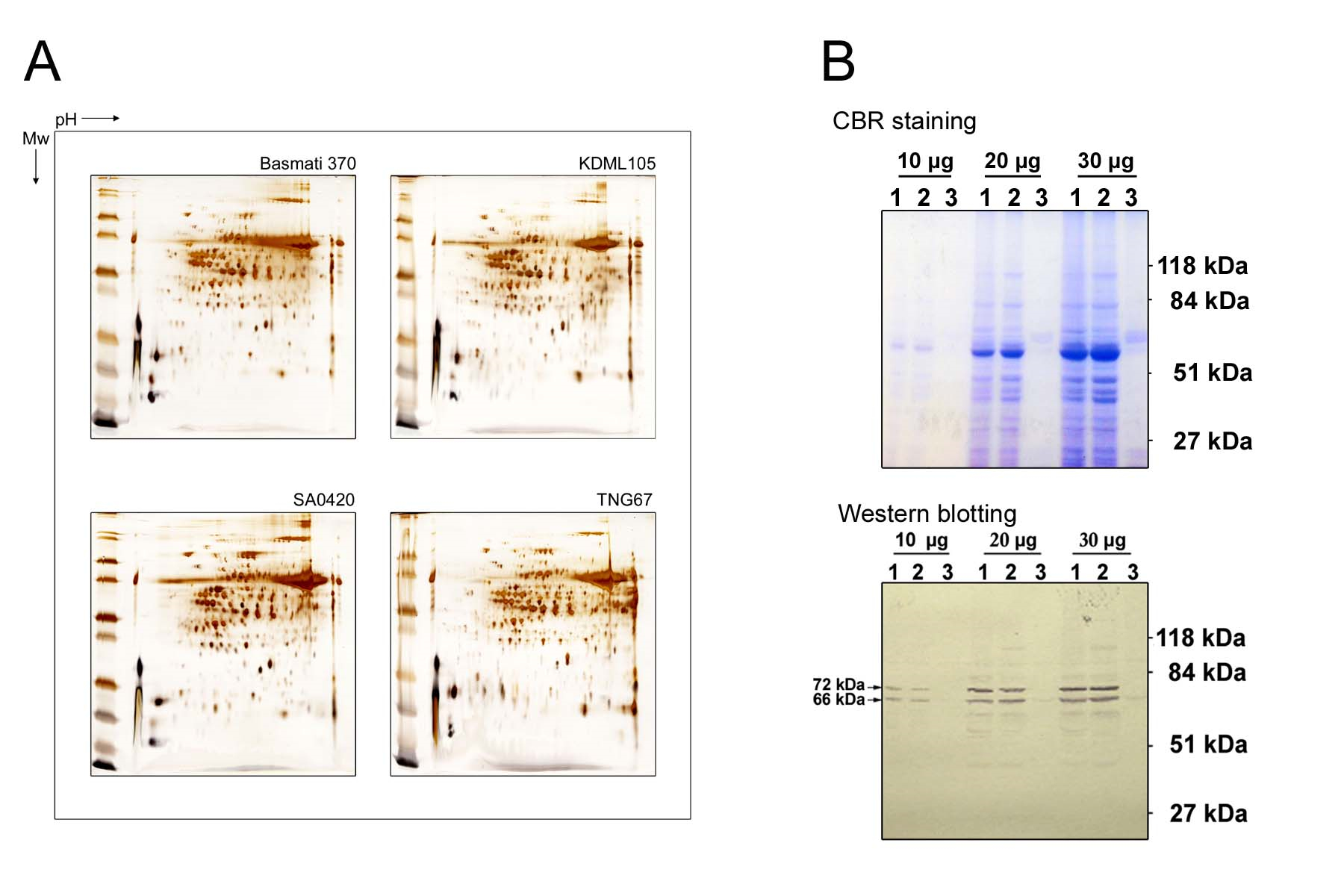
Figure 3. Proteomic analysis and Western blot analysis. A. Proteomic analysis of leaf proteins from various rice varieties. B. Proteomic analysis of taro leaf proteins and western blotting probed with anti-SSSI (Lin and Jeang, 2005).
- Cut 0.5 g fresh or -80 °C frozen leaf samples into small pieces, add little sea sand (Figure 2A) and freeze the tissue by pouring liquid nitrogen on the cut leaves in the mortar (Video 1).
- Preparation of the protein samples from rice grains
- Freeze 0.5 g dehulled grains (rice grains without glumes) by liquid nitrogen.
- Place the frozen tissue in a mortar containing little seasand and liquid nitrogen. Grind the tissue to a powder with the mortar and pestle.
- Add 10 ml extraction buffer, contains with 140 μl 40 mM PMSF. Grind sample for 2 min or to a homogenate and then transfer into a sterilized 50 ml centrifuge tube.
- Centrifuge 20 min at 15,000 rpm (in No. 46 Rotor), 4 °C. Transfer the supernatant into another fresh tube.
- Add 3x volume of cold 0.1 M NH4OAc/Methanol/10 mM 2-mercaptoethanol to the recovered phenol phase and mix well by inversion.
- Incubate at -20 °C for 4 h (or overnight) to precipitate the proteins. Then centrifuge 20 min at 15,000 rpm, 4 °C and discard the supernatant.
- Wash the pellet with 1.8 ml 0.1 M NH4OAc/ Methanol/ 10 mM 2-mercaptoethanol twice. Recover the pellet by centrifugation at 8,000 rpm for 5 min.
- Wash the pellet with 1 ml of cold 100% acetone containing with 0.7 μl 2-mercaptoethanol (14.3 M). Recover the pellet by centrifugation at 8,000 rpm for 5 min.
- Dry the pellet by the SpeedVac system for about 2-3 min (less than 5 min) and resuspend in an appropriate volume of lysis buffer.
- The protein samples can be quantified by Bradford method (Bradford, 1976) and stored at -20 °C.
- The obtained protein samples can be used for proteomic analysis or Western blot.
- Freeze 0.5 g dehulled grains (rice grains without glumes) by liquid nitrogen.
Representative data
- Video 1. Protein extraction
Notes
- In this protocol, the critical steps which affect the yield and intactness of protein samples are as follows:
- The lyophilized sample is not recommended, because some dehydrated proteins might be difficult to dissolve in the extraction buffer.
- The complete homogenization can be achieved by grinding the samples in extraction buffer (see Video 1).
- Do not dry the pellet too long in the step B9. It takes about 2-3 min (less than 5 min). The over-dried pellet will be very difficult to resuspend and dissolve completely in the lysis buffer.
- Resuspension of protein sample can be achieved by placing the Eppendorf tube containing the protein solution into an ice cold ultrasonication bath (Figure 4).

Figure 4. The dissolution of protein with ultrasonic vibration. Left panel is the ultrasonic machine. Right panel shows the protein sample is resuspending in the cold bath with ultrasonication.
- The lyophilized sample is not recommended, because some dehydrated proteins might be difficult to dissolve in the extraction buffer.
- Phenol waste has to be stored separately in a glass bottle that can be properly sealed. Phenol waste has to be discarded according to the chemical waste regulations of respective institution.
Recipes
- Extraction buffer
23.96 g Sucrose
6.05 g Tris-base
10 ml 0.5 M EDTA (pH 8.0)
0.75 g KCl
250 μl HCl
2 ml 2-mercaptoethanol
Add dH2O to 100 ml
Note: This buffer should be prepared freshly and pre-cooled at 4 °C. - Precipitation buffer
1.54 g NH4OAc
140 μl 2-mercaptoethanol (14.3 M)
Add methanol to 200 ml and stored at -20 °C - 0.5 M EDTA (pH 8.0)
186.1 g EDTA
800 ml dH2O
Adjust pH to 8.0 with NaOH (about 20 g NaOH)
Add dH2O to 1,000 ml, autoclave and store at room temperature - Lysis buffer
57 g Urea
4 ml Triton X-100
4 g CHAPS
484.4 mg Tris-base
231.4 mg DTT
Add dH2O to 100 ml, dispense in 1ml/ tube and stored at -20 °C - 40 mM PMSF
6.96 mg PMSF
Add 2-propanol to 1 ml and stored at -20 °C
Acknowledgments
This protocol was developed by modifying the phenol-methanol method (Hurkman and Tanaka, 1986). The authors acknowledge the financial support from the Council of Agriculture (91-3112-P-055-001-Y) and the National Science Council (NSC 95-2317-B-005 -01, NSC 96-2317-B-005-01) of Taiwan to C. S. Wang. We also thank Miss Yu-Hisn Yeh at the Biotechnology Division, Taiwan Agricultural Research Institute, for helping with the preparation of pictures and videos.
References
- Bradford, M. M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72: 248-254.
- Hurkman, W. J. and Tanaka, C. K. (1986). Solubilization of plant membrane proteins for analysis by two-dimensional gel electrophoresis. Plant Physiol 81(3): 802-806.
- Lin, D. G. and Jeang, C. L. (2005). Cloning, expression, and characterization of soluble starch synthase I cDNA from taro (Colocasia esculenta Var. esculenta). J Agric Food Chem 53(20): 7985-7990.
- Lin, D. G., Chou, S. Y., Wang, A. Z., Wang, Y. W., Kuo, S. M., Lai, C. C., Chen, L. J. and Wang, C. S. (2014). A proteomic study of rice cultivar TNG67 and its high aroma mutant SA0420. Plant Sci 214: 20-28.
- Liu, C. W., Hsu, Y. K., Cheng, Y. H., Yen, H. C., Wu, Y. P., Wang, C. S. and Lai, C. C. (2012). Proteomic analysis of salt-responsive ubiquitin-related proteins in rice roots. Rapid Commun Mass Spectrom 26(15): 1649-1660.
Article Information
Copyright
© 2014 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Lin, D. and Wang, C. (2014). Extraction of Total Proteins from Rice Plant. Bio-protocol 4(21): e1277. DOI: 10.21769/BioProtoc.1277.
Category
Plant Science > Plant biochemistry > Protein > Isolation and purification
Biochemistry > Protein > Isolation and purification
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link









