- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Cytokinin Analysis: Sample Preparation and Quantification
Published: Vol 4, Iss 13, Jul 5, 2014 DOI: 10.21769/BioProtoc.1167 Views: 16128
Reviewed by: Tie LiuAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Seed Coat Permeability Test: Tetrazolium Penetration Assay
Sollapura J. Vishwanath [...] Owen Rowland
Jul 5, 2014 14655 Views

Extraction and Quantification of Plant Hormones and RNA from Pea Axillary Buds
Da Cao [...] Christine A. Beveridge
Oct 5, 2022 2958 Views
Targeting Ultrastructural Events at the Graft Interface of Arabidopsis thaliana by A Correlative Light Electron Microscopy Approach
Clément Chambaud [...] Lysiane Brocard
Jan 20, 2023 2973 Views
Abstract
Cytokinins are a group of phytohormones discovered about half a decade ago by Miller et al. (1955) and Skoog et al. (1965). Since then they were found to participate in many plant physiological processes, including the regulation of the source/sink transitions, plant growth and organ development, responses to environmental conditions such as light, nutrient and water availability and biotic interactions with mutualists, pathogens and herbivores (Werner and Schmülling, 2009; Giron et al., 2013). To aid the quantification of cytokinins for analyzing their changes after environmental stress conditions, we developed this cytokinin extraction and analysis method. This protocol is based on the cytokinin extraction with an acidic methanol-water solution and purification with a mixed-mode solid phase extraction procedure described by Dobrev and Kamı́nek (2002) and the modifications of Kojima et al. (2009). The protocol was successfully used to verify cytokinin overproduction in transgenic Nicotiana attenuata plants expressing the cytokinin biosynthesis gene Tumor morphology root (Tmr) from Agrobacterium tumefaciens under the control of the chemical inducible expression system pOp6/LhGR in the glasshouse and under field conditions (Schäfer et al., 2013) to study the role of cytokinins in plant-herbivore interactions.
Materials and Reagents
- Plant tissue
- MeOH
- 1 N HCOOH
- 0.35 N NH4OH
- 0.35 N NH4OH in 60% MeOH
- 0.1% (v/v) acetic acid
- 0.05% (v/v) HCOOH (for mass spectrometry) in Milli-Q H2O
- Acetonitrile (gradient grade)
- [2H5] tZ (Olchemim, catalog number: 030 0301 )
- [2H5] tZR (Olchemim, catalog number: 030 0311 )
- [2H5] tZROG (Olchemim, catalog number: 030 5131 )
- [2H5] tZ7G (Olchemim, catalog number: 030 5111 )
- Extraction buffer (see Recipes)
- Extraction buffer + deuterated standards (see Recipes)
Equipment
- 96-well BioTubes (1.1 ml individual tubes) (Arctic White LLC, catalog number: AWTS-X22100 )
- Steel balls (ASKUBAL, catalog number: 3 MM-G100-1.4034 )
- Caps for 96-well BioTubes (strips of 8 plug caps) (Arctic White LLC, catalog number: AWSM-T100-30 )
- Pipet
- ArctiSeal 96 Round Well Sealing Mats for 96-well BioTubes (Arctic White LLC, catalog number: AWSM-2002RB )
- Nunc 96-well Deep Well Plates (Thermo Fisher Scientific, catalog number: 278752 )
- Nunc 96-Well Cap Mats (Thermo Fisher Scientific, catalog number: 276002 )
- 96-well PCR plates (Kaneka Corporation, Eurogentec, catalog number: RT-PL96-MQ )
- Sealing film (OMNILAB-LABORZENTRUM, Schubert & Weiss, catalog number: 5420203 )
- Machery Nagel Multi 96 HR-X (96 x 25 mg) (MACHEREY-NAGEL, catalog number: 738530.025M )
- Machery Nagel Multi 96 HR-XC (96 x 25 mg) (MACHEREY-NAGEL, catalog number: 738540.025M )
- Chromabond Multi 96 vacuum manifold (MACHEREY-NAGEL, catalog number: 738630.M )
- Evaporator system (Glas-Col, catalog number: 099A EV9624S )
- Geno/Grinder 2000 (SPEX SamplePrep)
- Eppendorf Centrifuge 5804 R equipped with a Swing-bucket rotor A-2-DWP (Eppendorf)
- Ultrasonic bath Bransonic Models 1200 (BRANDSONTM)
- Agilent 1200 HPLC system (Agilent)
- Zorbax Eclipse XDB-C18 column (50 x 4.6 mm, 1.8 µm) (Agilent)
- API 5000 tandem mass spectrometer (Applied Biosystems®) equipped with a Turbospray ion source
Procedure
- Preparation of plant material
- Collect plant tissue and immediately freeze it in liquid nitrogen. Stored at -80 °C.
- Homogenize plant tissue under liquid nitrogen.
- Aliquot 100 mg plant tissue per sample in liquid nitrogen-precooled 96-well BioTubes (with two steel balls per tube, closed with caps).
Note: Make small holes in the caps to prevent tubes from exploding. - Keep at least for 30 min at -20 °C (for longer storage at -80 °C) before starting with the extraction to evaporate liquefied gases from the tubes.
- Extraction
Note: In-between the steps, while buffer addition and sample collection samples are kept on ice.- Add 800 µl precooled (-20 °C) extraction buffer + deuterated standards to each tube, and cover with a precooled sealing mat.
- Shake in Geno/Grinder for 60 s (1,150 strokes *min-1).
- Incubate over night at -20 °C.
- Shake in the Geno/Grinder for 60 s (1,150 strokes *min-1).
- Centrifuge (20 min, 1,913 x g, 4 °C).
- Collect 600 µl supernatant in 96-well BioTubes and keep it at -20 °C.
- Add 600 µl extraction buffer to the pellet.
- Shake in the Geno/Grinder for 60 s (1,150 strokes *min-1).
- Incubate for 30 min at -20 °C.
- Centrifuge (20 min, 1,913 x g, 4 °C).
- Collect 600 µl supernatant and combine it with the supernatant from the first extraction steps. Cover samples with a sealing mat.
- Centrifuge supernatants (20 min, 1,913 x g, 4 °C) and continue with the sample purification.
- Purification
Note: If not further mentioned the following steps are performed at room temperature.
HR-X- Condition the HR-X column with 0.6 ml MeOH. Discard flow-through.
Note: Use the Vacuum manifold to suck the samples and buffers through the column. Same accounts for the following steps. - Condition the column with 0.6 ml extraction buffer. Discard flow-through.
- Load the samples to the column (collect flow-through) and subsequent wash with 0.2 ml extraction buffer (collect flow-through). Collection of both flow-through can be done in a Nunc 96-well Deep Well Plate.
- Evaporate the samples at 45 °C under a constant nitrogen stream to remove MeOH from the samples, utilizing the evaporator system (see Equipment).
Note: MeOH evaporates fastest and after evaporation of the MeOH, approximately 350 µl liquid should be left, which can be tested with a pipet. Samples only have to be evaporated until this point. - Add 850 µl 1 N HCOOH per sample.
- Cover the plate with a sealing mat and shake in the Geno/Grinder for 30 s (1,000 strokes *min-1).
- Centrifuge (20 min, 1,913 x g, 4 °C).
- Condition the HR-XC column with 0.6 ml MeOH. Discard flow-through.
- Condition the column with 0.6 ml 1 M HCOOH. Discard flow-through.
- Load the samples to the column. Discard flow-through.
- Wash column with 1 ml 1 M HCOOH. Discard flow-through.
- Wash column with 1 ml MeOH. Discard flow-through.
- Wash column with 1 ml 0.35 N NH4OH. Discard flow-through.
- Elute cytokinin-nucleobases, -ribosides and -glucosides from the column with 0.35 N NH4OH in 60% MeOH. Collect flow-through in 96-well BioTubes.
- Completely evaporate the samples at 45 °C under a constant nitrogen stream utilizing the evaporator system.
- Reconstitute samples in 50 µl 0.1% (v/v) acetic acid.
- Cover the plate with a sealing mat and shake in the Geno/Grinder for 60 s (1,000 strokes *min-1).
- Sonicate the samples for 10 min in an Ultrasonic bath.
Note: Remove the bottom plate of the 96-well rack and place the whole rack in the Ultrasonic bath. Do not use 96-well racks without bottom plate for shaking and centrifugation! - Centrifuge (20 min, 1,913 x g, 4 °C).
- Fill samples in 96-well PCR plates and cover with the sealing film.
- Centrifuge (20 min, 1,913 x g, 4 °C) and continue with Ultra-performance LC coupled MS/MS.
- Condition the HR-X column with 0.6 ml MeOH. Discard flow-through.
- Ultra-performance LC coupled MS/MS
Inject 2 µl per sample in an Agilent 1200 HPLC system equipped with a Zorbax Eclipse XDB-C18 column. The mobile phase comprises of 0.05% (v/v) HCOOH in Milli-Q H2O as solvent A and acetonitrile as solvent B with following settings:
Flow rate: 1.1 ml min–1
Column temperature: 25 °C
Solvent gradient for chromatographic separation: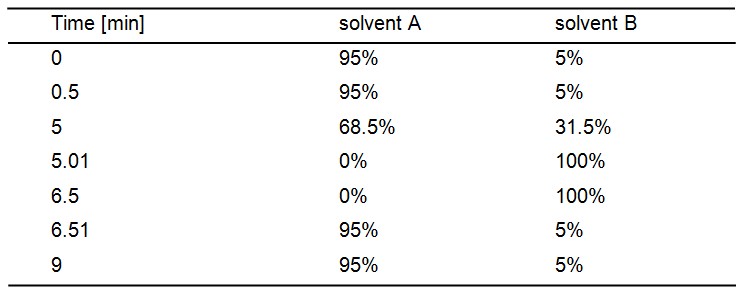
Analysis is done with an API 5000 tandem mass spectrometer equipped with a Turbospray ion source. Multiple-reaction-monitoring mode is used with following settings for quantification:
Ionization mode: positive
Ion spray voltage: 5,500 eV
Turbo gas temperature: 700 °C
Nebulizing gas: 70 psi
Curtain gas: 25 psi
Heating gas: 60 psi
Collision gas: 6 psi
Precursor-to-product ion transitions used for cytokinin analysis: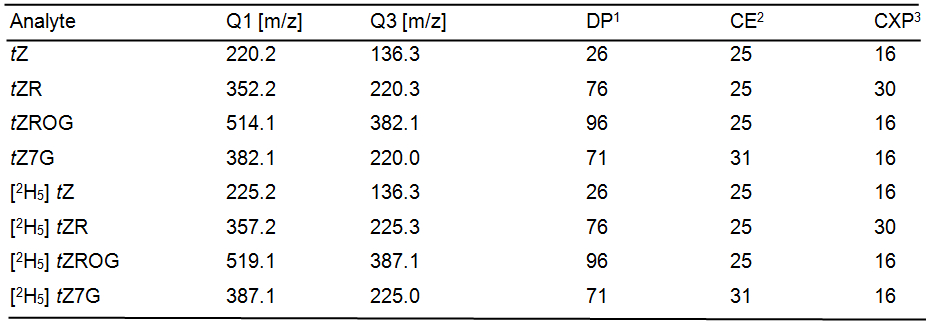
tZ, trans-zeatin, tZR, trans-zeatin riboside, tZROG, trans-zeatin riboside O-glucoside, tZ7G, trans-zeatin N7-glucoside
1Declustering potential, 2Collision energy, 3Collision cell exit potential
Representative data
- Chromatograms
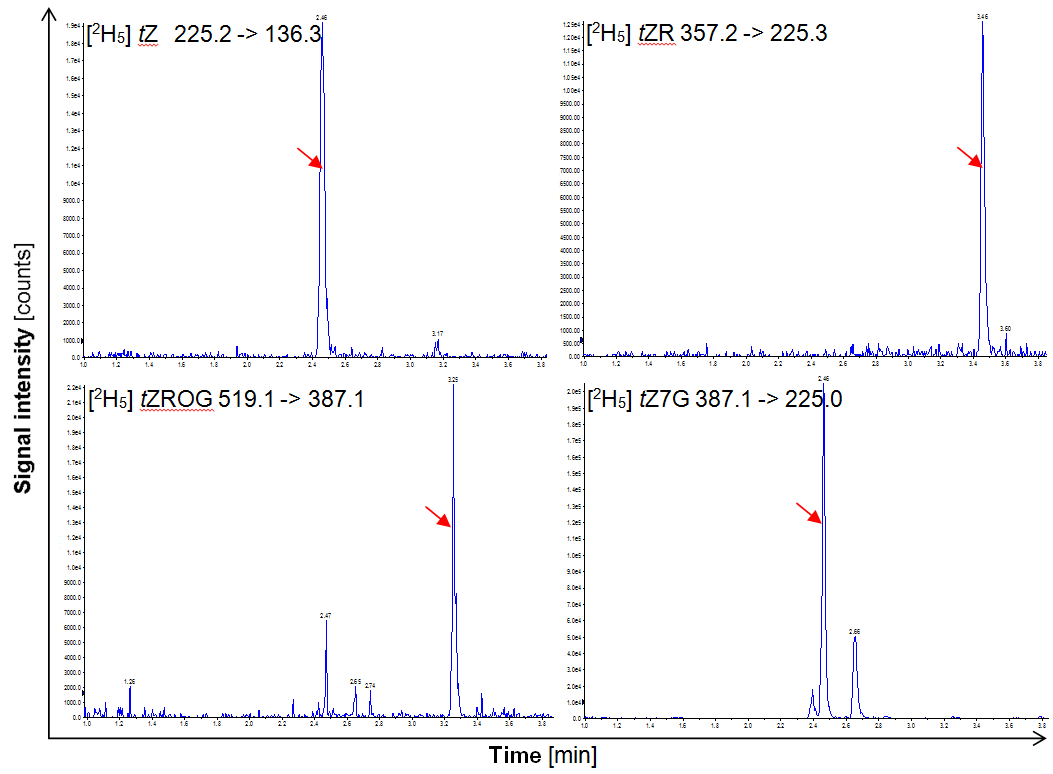
Figure 1. Separation of deuterated internal standards in the ultra-performance LC coupled MS/MS analysis. The red arrows indicate the respective peaks.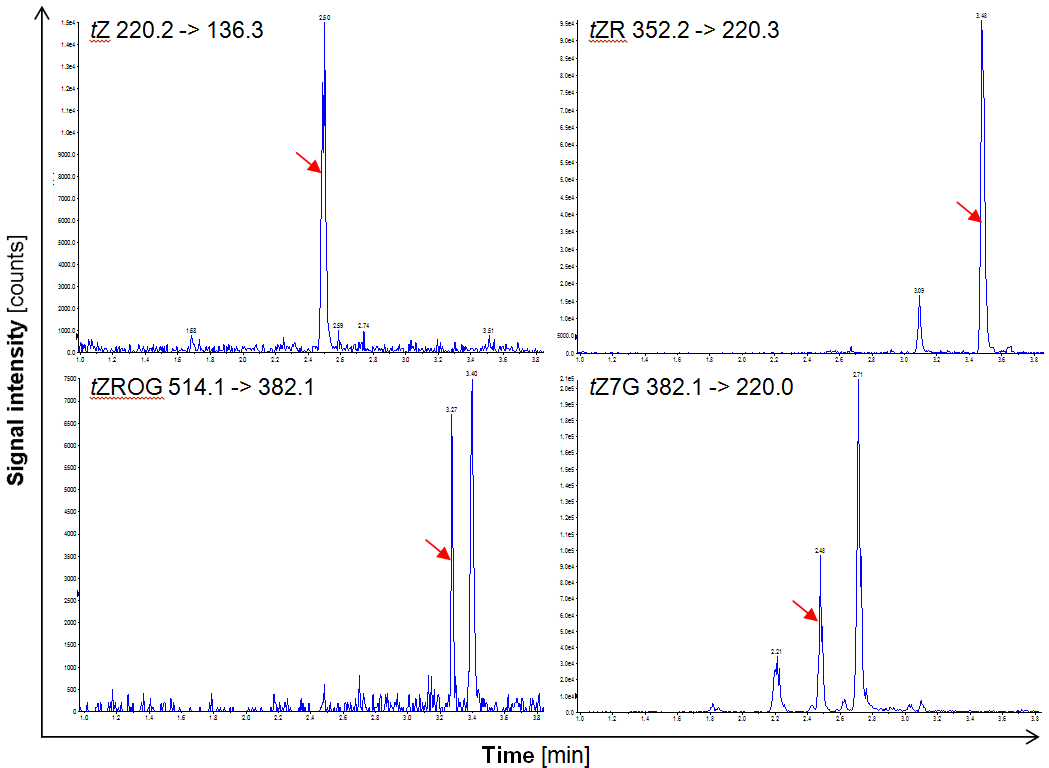
Figure 2. Separation of cytokinins from the plant tissue in the ultra-performance LC coupled MS/MS analysis. The red arrows indicate the respective peaks. - Calculation
Exemplary tZ quantification in 0.1 g leaf tissue, supplemented with 1 ng deuterated tZ as internal standard. The obtained peak areas were 23,700 counts and 38,400 counts for tZ and [2H5] tZ, respectively.
ConcentrationCompound = Peak areaCompound/PeakareaInternal Standard/mExtracted tissue * mInternal Standard
ConcentrationtZ = Peak areatZ/Peak area[2H5] tZ/mLeaf tissue * m[2H5] tZ
ConcentrationtZ = 23,700 counts/38,400 counts/0.1 g * 1 ng = 6.2 ng tZ per g leaf tissue
Recipes
- Extraction buffer
750 ml MeOH
200 ml ddH2O
50 ml HCOOH - Extraction buffer + deuterated standards
Prepare Extraction buffer spiked with 1 ng [2H5] tZ, 0.1 ng [2H5] tZR, 4 ng [2H5] tZROG and 2 ng [2H5] tZ7G per 800 µl extraction buffer.
Note: Deuterated cytokinins are used as internal standards for quantification.
Acknowledgments
The presented work was adopted from Schäfer et al. (2013) and is based on the procedure described by Dobrev and Kamı́nek (2002) and the modifications of Kojima et al. (2009). We thank Radomira Vanková for valuable help with the method development. MS, MR and IB are funded by the Max-Planck-Society and SM is funded by Advanced Grant no. 293926 of the European Research Council to IB. Additional financial support was given by the Human Frontier Science Program (RGP0002/2012) and the Global Research Lab program (2012055546) from the National Research Foundation of Korea.
References
- Dobrev, P. I. and Kaminek, M. (2002). Fast and efficient separation of cytokinins from auxin and abscisic acid and their purification using mixed-mode solid-phase extraction. J Chromatogr A 950(1-2): 21-29.
- Giron, D., Frago, E., Glevarec, G., Pieterse, C. M. and Dicke, M. (2013). Cytokinins as key regulators in plant–microbe–insect interactions: connecting plant growth and defence. Funct Ecol 27(3): 599-609.
- Kojima, M., Kamada-Nobusada, T., Komatsu, H., Takei, K., Kuroha, T., Mizutani, M., Ashikari, M., Ueguchi-Tanaka, M., Matsuoka, M., Suzuki, K. and Sakakibara, H. (2009). Highly sensitive and high-throughput analysis of plant hormones using MS-probe modification and liquid chromatography-tandem mass spectrometry: an application for hormone profiling in Oryza sativa. Plant Cell Physiol 50(7): 1201-1214.
- Miller, C. O., Skoog, F., Von Saltza, M. H. and Strong, F. (1955). Kinetin, a cell division factor from deoxyribonucleic acid1. J American Chem Soci 77(5): 1392-1392.
- Schäfer, M., Brütting, C., Gase, K., Reichelt, M., Baldwin, I. and Meldau, S. (2013). ‘Real time’genetic manipulation: a new tool for ecological field studies. Plant J 6(3): 506-518.
- Skoog, F., Strong, F. M. and Miller, C. O. (1965). Cytokinins. Science 148(3669): 532-533.
- Werner, T. and Schmulling, T. (2009). Cytokinin action in plant development. Curr Opin Plant Biol 12(5): 527-538.
Article Information
Copyright
© 2014 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Schäfer, M., Reichelt, M., Baldwin, I. T. and Meldau, S. (2014). Cytokinin Analysis: Sample Preparation and Quantification. Bio-protocol 4(13): e1167. DOI: 10.21769/BioProtoc.1167.
Category
Plant Science > Plant biochemistry > Plant hormone > Membrane protein detection
Plant Science > Plant physiology > Tissue analysis
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link










