- EN - English
- CN - 中文
Generating Loss-of-function iPSC Lines with Combined CRISPR Indel Formation and Reprogramming from Human Fibroblasts
人成纤维细胞通过CRISPR 插入缺失的形成联合重编程生成功能丧失iPSC细胞系
发布: 2018年04月05日第8卷第7期 DOI: 10.21769/BioProtoc.2794 浏览次数: 12407
评审: David CisnerosSara E HowdenGanesh Swaminathan

相关实验方案
利用EpiCRISPR系统通过靶向DNA甲基化诱导Alpha TC1-6细胞产生胰岛素
Marija B. Đorđević [...] Melita S. Vidaković
2025年10月20日 1291 阅读
Abstract
For both disease and basic science research, loss-of-function (LOF) mutations are vitally important. Herein, we provide a simple stream-lined protocol for generating LOF iPSC lines that circumvents the technical challenges of traditional gene-editing and cloning of established iPSC lines by combining the introduction of the CRISPR vector concurrently with episomal reprogramming plasmids into fibroblasts. Our experiments have produced nearly even numbers of all 3 genotypes in autosomal genes. In addition, we provide a detailed approach for maintaining and genotyping 96-well plates of iPSC clones.
Keywords: Induced pluripotent stem cells (诱导的多能干细胞)Background
CRISPR/Cas9 technology has allowed easy and specific targeting of a particular genomic location for gene-editing. Combining this technology with the disease modeling and regenerative medicine potential of induced pluripotent stem cells (iPSCs) will continue to have unprecedented impacts on biomedical research. However, adapting the CRISPR/Cas9 system to iPSCs has presented several challenges. The traditional approach for gene-editing in cell lines is to transfect the cells with a plasmid expressing the Cas9 protein and guide RNA (gRNA) after which single clones are generated and screened for the desired genetic alteration. Unfortunately, iPSCs are not amenable to single cell cloning. Several media supplements and cloning approaches have been developed to overcome this difficulty but are still fraught with either expensive equipment (low oxygen incubators), difficult technological steps (survival of FACS sorted single iPSCs), or labor intensive protocols (sub-cloning) (Forsyth et al., 2006; Miyaoka et al., 2014). Moreover, single cell passaging has been linked to increased genomic abnormalities in iPSCs (Bai et al., 2015). Fluorescent or antibiotic resistance genetic markers have been used to overcome issues with clonality and the overall low efficiency of gene-editing in these cells, but require homologous recombination of a large cassette via a targeting plasmid designed with long homology arms (400-800 bp) (Hendel et al., 2014). Designing these plasmids takes a great deal of time.
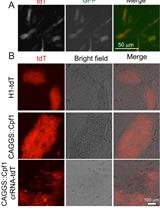
To overcome many of these obstacles, we utilized a combined approach of simultaneous reprogramming and CRISPR/Cas9 mutagenesis to generate both heterozygous and homozygous loss-of-function (LOF) iPSC lines. This combined approach was first presented by Howden and colleagues for homologous recombination gene-editing (Howden et al., 2015 and 2016), but we have expanded upon and further defined their original results for indel formation in a recent publication (Tidball et al., 2017). This procedure takes advantage of the cloning step inherent to iPSC reprogramming as well as the greater ease of transfection in fibroblast culture (Figure 1). The ability to efficiently generate a large number of clones with a similar proportion of all three genotypes (wild-type/wild-type, wild-type/indel, and indel/indel) will allow rapid development of LOF iPSC lines for disease modeling and basic research. 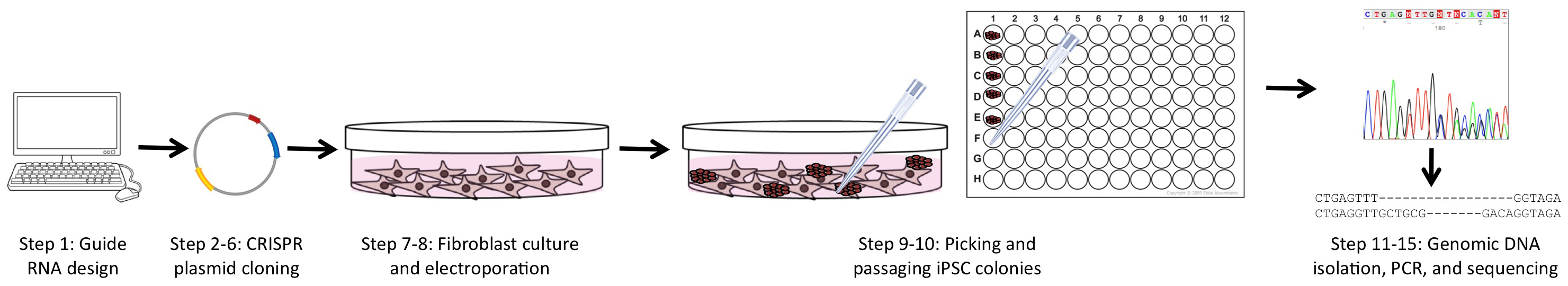
Figure 1. Overview of the experimental workflow. The protocols in this article are broken into 4 major sections, including: guide RNA and PCR primer design (Step 1), CRISPR plasmid generation and purification (Steps 2-6), fibroblast culture and electroporation with CRISPR and reprogramming plasmids (Steps 7-8), and isolating genomic DNA followed by PCR of the targeted region and sequencing (Steps 11-15).
Materials and Reagents
- Materials
- 1.5 ml Eppendorf tubes (VWR, catalog number: 89000-028 )
- 10 µl, 200 µl, and 1 ml pipette tips (USA Scientific, catalog numbers: 1111-3700 , 1111-0700 , and 1111-2721 )
- 14 ml tubes for bacterial culture (Corning, Falcon®, catalog number: 352059 )
- Conical flasks (Corning, PYREX®, catalog number: 4980-500 )
- 10 cm tissue culture dish (Corning, Falcon®, catalog number: 353003 )
- 6-well plate (Corning, Costar®, catalog number: 3516 )
- 5 ml, 10 ml, and 25 ml serological pipettes (Aikali Scientific, ASI, catalog numbers: SP205 , SP210 , and SP225 )
- 96-well plate (Corning, Falcon®, catalog number: 353072 )
- Sterile 50 ml disposable pipette basins (Fisher Scientific, catalog number: 13-681-502 )
- PCR tube strips (USA Scientific, catalog number: 1402-4700 )
- 15 ml centrifuge tubes (Fisher Scientific, catalog number: 05-539-12 )
- Nalgene General Long-Term Storage Cryogenic Tubes (Thermo Fisher Scientific, Thermo ScientificTM, catalog number: 5000-1012 )
- 1.5 ml Eppendorf tubes (VWR, catalog number: 89000-028 )
- Cells
- Human foreskin fibroblasts (MTI-GlobalStem, catalog number: GSC-3002 )
- Human foreskin fibroblasts (MTI-GlobalStem, catalog number: GSC-3002 )
- Reagents
- CRISPR plasmid pX330-U6-Chmeric_BB-CBh-hSpCas9 (Addgene, catalog number: 42230 )
- Reprogramming plasmids pCXLE-hOCT3/4-p53shRNA, pCXLE-hUL, and pCXLE-hSK (Addgene, catalog numbers: 27077 , 27080 , 27078 )
- Subcloning Efficiency DH5α competent cells (Thermo Fisher Scientific, InvitrogenTM, catalog number: 18265017 )
- BbsI restriction enzyme, supplied with NEBuffer 2.1 (New England Biolabs, catalog number: R3539 )
- QIAGEN PCR purification kit (QIAGEN, catalog number: 28104 )
- Custom oligonucleotides (Thermo Fisher Scientific)
- T4 DNA ligase kit (Thermo Fisher Scientific, InvitrogenTM, catalog number: 15224017 )
- SOC medium (Thermo Fisher Scientific, InvitrogenTM, catalog number: 15544034 )
- LB broth base (Thermo Fisher Scientific, InvitrogenTM, catalog number: 12780052 )
- LB agar (Thermo Fisher Scientific, InvitrogenTM, catalog number: 22700025 )
- Ampicillin sodium salt (Sigma-Aldrich, catalog number: A9518-25G )
- QIAGEN Miniprep Kit (QIAGEN, catalog number: 27104 )
- Glycerol (Sigma-Aldrich, catalog number: G5516-100ML )
- LKO.1 5’ sequencing primer (5’-GACTATCATATGCTTACCGT-3’)
- Endo-Free QIAGEN Maxi Prep Kit (QIAGEN, catalog number: 12362 )
- Phosphate buffered saline (PBS), pH 7.4 (Thermo Fisher Scientific, GibcoTM, catalog number: 10010023 )
- 0.25% trypsin solution (Thermo Fisher Scientific, GibcoTM, catalog number: 25200056 )
- Growth factor depleted Matrigel (Corning, catalog number: 356230 )
- Neon Transfection System 100 µl Kit (Thermo Fisher Scientific, InvitrogenTM, catalog number: MPK10025 )
- TeSR-E7 (STEMCELL Technologies, catalog number: 05910 )
- mTeSR1 (STEMCELL Technologies, catalog number: 85850 )
- ROCK inhibitor Y-27632 (Tocris Bioscience, catalog number: 1254 )
- Accutase (Innovative Cell Technologies, catalog number: AT104 )
- ZR-96 Quick-gDNA Kit (Zymo Research, catalog number: D3010 )
- GoTaq green master mix (Promega, catalog number: M7122 )
- Molecular biology grade water (Thermo Fisher Scientific, InvitrogenTM, catalog number: 10977023 )
- ZR-96 DNA Sequencing Clean-up Kit (Zymo Research, catalog number: D4052 )
- Gel 6x Loading Dye, Purple (New England Biolabs, catalog number: B7024S )
- GeneMate LE Quick Dissolve Agarose (BioExpress, GeneMate, catalog number: E-3119-500 )
- GelRed Nucleic Acid Gel Stain 10,000x in DMSO (Biotium, catalog number: 41002 )
- Accugene 10 TBE Buffer (Lonza, catalog number: 50843 )
- Quant-IT PicoGreen dsDNA Assay Kit (Thermo Fisher Scientific, InvitrogenTM, catalog number: P7589 )
- mFreSR (STEMCELL Technologies, catalog number: 05855 )
- Liquid nitrogen
- Dulbecco modified Eagle’s medium (high glucose) (Thermo Fisher Scientific, GibcoTM, catalog number: 11965118 )
- Fetal bovine serum (FBS) (Thermo Fisher Scientific, GibcoTM, catalog number: 10437010 )
- Penicillin-streptomycin solution (10,000 U/ml) (Thermo Fisher Scientific, GibcoTM, catalog number: 15140122 )
- MEM Non-essential amino acids solution 100x (Thermo Fisher Scientific, GibcoTM, catalog number: 11140050 )
- Fibroblast growth medium (see Recipes)
- CRISPR plasmid pX330-U6-Chmeric_BB-CBh-hSpCas9 (Addgene, catalog number: 42230 )
Equipment
- Portable Pipet-aid (Drummond, model: DP-101, catalog number: 4-000-101 )
- Laminar flow hood (Thermo Fisher Scientific, Thermo ScientificTM, model: 1300 Series Class II Type A2 , catalog number: 1335)
- Neon electroporator (Thermo Fisher Scientific, InvitrogenTM, catalog number: MPK5000 )
- Heraguard ECO Clean Bench (Thermo Fisher Scientific, Thermo ScientificTM, model: HeraguardTM ECO, catalog number: 51029701 )
- NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific, Thermo ScientificTM, model: NanoDropTM 2000 )
- iCycler Thermal cycler (Bio-Rad Laboratories, model: iCycler Thermal cycler )
- Water bath (Thermo Electron Corporation, catalog number: 51221073 )
- Refrigerator (Frigidaire, model: FFTR2021QW1 )
- Tissue culture incubator set at 37 °C, 5% CO2 (Thermo Fisher Scientific, Thermo ScientificTM, model: HeracellTM 150i , catalog number: 51026283)
- Thermo Scientific Sorvall St8 120v cell culture centrifuge with plate adapters (Thermo Fisher Scientific, Thermo ScientificTM, model: SorvallTM St 8 , 120v)
- Bright-Line Hemocytometer (Hausser Scientific, catalog number: 3110 )
- HandEvac Handheld Aspirator with 8-channel adapter (Argos Technologies, catalog number EV500 )
- Olympus CKX41 inverted cell culture microscope (Olympus, model: CKX41 )
- Multichannel pipettes (Thermo Fisher Scientific, Thermo ScientificTM, catalog numbers: 4662000 and 4662010 )
- Sub-cell GT Electrophoresis cell (Bio-Rad Laboratories, model: Sub-Cell® GT Cell )
- Gel Imager
- Beckman Coulter DTX 880 Multimode Detector Microplate Reader (Beckman Coulter, model: DTX 880 )
- CoolCell LX Freezing Container (Biocision, catalog number: BCS-405 )
Procedure
文章信息
版权信息
© 2018 The Authors; exclusive licensee Bio-protocol LLC.
如何引用
Tidball, A. M., Swaminathan, P., Dang, L. T. and Parent, J. M. (2018). Generating Loss-of-function iPSC Lines with Combined CRISPR Indel Formation and Reprogramming from Human Fibroblasts. Bio-protocol 8(7): e2794. DOI: 10.21769/BioProtoc.2794.
分类
干细胞 > 多能干细胞 > 细胞诱导
细胞生物学 > 细胞工程 > CRISPR-cas9
分子生物学 > DNA > 诱/突变
您对这篇实验方法有问题吗?
在此处发布您的问题,我们将邀请本文作者来回答。同时,我们会将您的问题发布到Bio-protocol Exchange,以便寻求社区成员的帮助。
Share
Bluesky
X
Copy link