- EN - English
- CN - 中文
Improving CRISPR Gene Editing Efficiency by Proximal dCas9 Targeting
通过近端dCas9靶向提高CRISPR基因编辑效率
发布: 2017年08月05日第7卷第15期 DOI: 10.21769/BioProtoc.2432 浏览次数: 10757
评审: Jihyun KimVera Karolina SchoftAnonymous reviewer(s)

相关实验方案
在哺乳动物细胞中通过具有 3' 突出端的长 dsDNA 介导的 CRISPR 敲入 (LOCK) 实现高效的大 DNA 片段敲入
Wenjie Han [...] Jianqiang Bao
2023年10月20日 2786 阅读
Abstract
Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR) and CRISPR-associated (Cas) systems function as an adaptive immune system in bacteria and archaea for defense against invading viruses and plasmids (Barrangou and Marraffini, 2014). The effector nucleases from some class 2 CRISPR-Cas systems have been repurposed for heterologous targeting in eukaryotic cells (Jinek et al., 2012; Cong et al., 2013; Mali et al., 2013; Zetsche et al., 2015). However, the genomic environments of eukaryotes are distinctively different from that of prokaryotes in which CRISPR-Cas systems have evolved. Mammalian heterochromatin was found to be a barrier to target DNA access by Streptococcus pyogenes Cas9 (SpCas9), and nucleosomes, the basic units of the chromatin, were also found to impede target DNA access and cleavage by SpCas9 in vitro (Knight et al., 2015; Hinz et al., 2015; Horlbeck et al., 2016; Isaac et al., 2016). Moreover, many CRISPR-Cas systems characterized to date often exhibit inactivity in mammalian cells and are thus precluded from gene editing applications even though they are active in bacteria or on purified DNA substrates. Thus, there is a need to devise a means to alleviate chromatin inhibition to increase gene editing efficiency, especially on difficult-to-access genomic sites, and to enable use of otherwise inactive CRISPR-Cas nucleases for gene editing need. Here we describe a proxy-CRISPR protocol for restoring nuclease activity of various class 2 CRISPR-Cas nucleases on otherwise inaccessible genomic sites in human cells via proximal targeting of a catalytically dead Cas9 (Chen et al., 2017). This protocol is exemplified here by using Campylobacter jejuni Cas9 (CjCas9) as nuclease and catalytically dead SpCas9 (SpdCas9) as proximal DNA binding protein to enable CjCas9 to cleave the target for gene editing using single stranded DNA oligo templates.
Keywords: CRISPR-Cas nuclease (CRISPR-Cas核酸酶)Background
By creating targeted chromosomal DNA double strand breaks (DSBs) or single strand breaks (nicks) or serving as a DNA binding module for other DNA modification effectors, programmable endonucleases have become an important tool for genome modification in eukaryotic cells (Gaj et al., 2013). In response to targeted DNA breaks, host cells can invoke various repair pathways to mend the damages to maintain the genome integrity. Insertions and/or deletions derived from NHEJ repair errors can be capitalized for gene knockout and homologous recombination can be exploited for introducing pre-determined changes on gene of interest by providing a DNA donor. In addition to these more traditional gene editing applications, catalytically inactive forms of programmable endonucleases are increasingly used as DNA binding modules for other DNA modification effectors, such as cytidine deamination enzymes (Komor et al., 2016). However, no matter which forms of programmable nucleases are utilized, target site binding is the prerequisite step and local chromatin structure can determine whether or not or how efficiently a programmable nuclease can bind the target site (Knight et al., 2015; Hinz et al., 2015; Horlbeck et al., 2016; Isaac et al., 2016, Chen et al., 2017). We hypothesize that binding at proximal locations by a programmable DNA binding protein could change the local chromatin structure and render an otherwise inaccessible target site accessible for binding.
Previous generations of programmable nucleases, such as meganucleases, zinc finger nucleases (ZFNs), and transcription activator-like effector nucleases (TALENs), solely rely on protein structure to recognize target sites, and thus re-targeting of these nucleases requires rather laborious protein structural change. In contrast, class 2 CRISPR-Cas effector nucleases use protein structure to recognize a protospacer adjacent motif (PAM) and employ CRISPR RNA (crRNA) to bind the target site adjacent to the PAM. Because PAM is typically a short DNA sequence, such as 5’-NGG-3’ for SpCas9, and thus occurs frequently in a genome, re-targeting of CRISPR-Cas nucleases is a simple process of changing the crRNA sequence by molecular cloning or chemical synthesis. This targeting modality makes CRISPR-Cas systems very suitable for use as nucleases or as DNA binding proteins. This protocol combines these two utilities together to expand CRISPR gene editing capability. The CRISPR-Cas system used as nuclease must be orthogonal to the CRISPR-Cas system used as DNA binding protein to avoid binding site sharing. In general, different subtypes of class 2 CRISPR-Cas systems (e.g., type II-A, type II-B, type II-C, and type V) are orthogonal to one another. Within each subtype, some systems are highly divergent and could be also orthogonal to one another, but they need to be experimentally verified. Currently, it is highly recommended to use SpdCas9 as proximal DNA binding protein, for SpCas9 is the most robust system in mammalian cells to date, although it can also be inactive at certain genomic sites. However, it is anticipated that more robust Cas9 systems will be developed for use as DNA binding proteins.
Materials and Reagents
- Pipette tips (Thermo Fisher Scientific, Thermo ScientificTM, catalog numbers: 2140 , 2149 , 2065E , 2069 , and 2079E )
- 75 cm2 U-shaped canted neck cell culture flask with vent cap (Corning, catalog number: 430641U )
- Microcentrifuge tubes, 1.5 ml (Sigma-Aldrich, catalog number: T6649 )
- Tissue culture plate, 6-well (Corning, Costar®, catalog number: 3516 )
- Serological pipettes
5 ml (Corning, Costar®, catalog number: 4051 )
10 ml (Corning, Costar®, catalog number: 4488 )
25 ml (Corning, Costar®, catalog number: 4489 )
- Cuvettes
- Coverslip
- 50-ml conical centrifuge tube (Corning, catalog number: 430828 )
- Human K562 cells (ATCC, catalog number: CCL-243 )
- Catalytically inactive Streptococcus pyogenes Cas9 (SpdCas9) plasmid DNA (from MilliporeSigma; see Figure 1A)
- SpCas9 sgRNA plasmid constructs with the guide sequences 5’-CCAAGGGTGAGGCCGGGAAG-3’ and 5’-CATCTCCCCCATGTACACCT-3’ for binding two human POR target sites (from MilliporeSigma; see Figure 1B)
- Campylobacter jejuni Cas9 (CjCas9) plasmid DNA (from MilliporeSigma; see Figure 1C)
- CjCas9 sgRNA plasmid construct with the guide sequence 5’-TTCGCCAGTACGAGCTTGTG-3’ for binding a human POR target site (from MilliporeSigma; see Figure 1D)
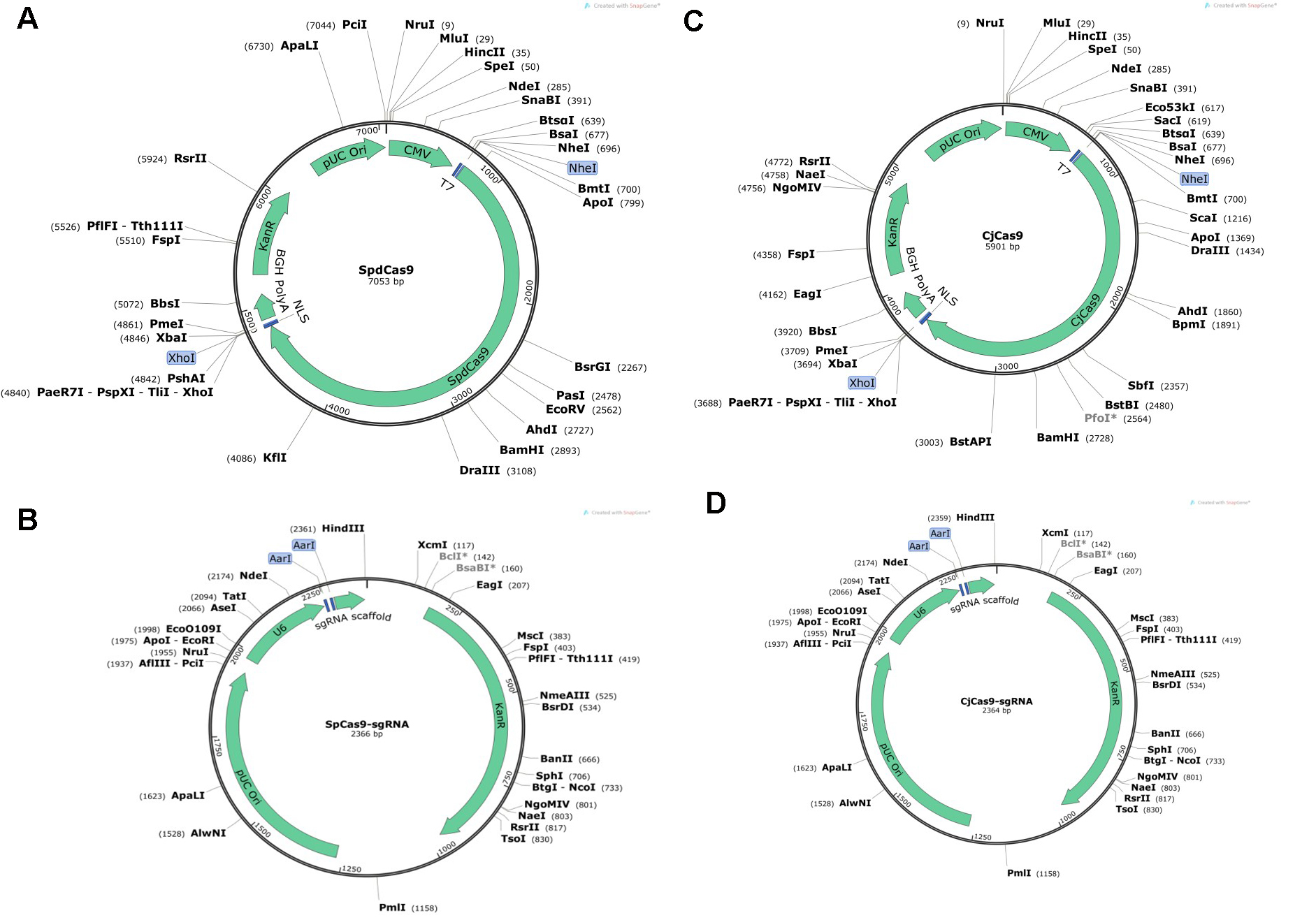
Figure 1. Vector maps
- Single stranded DNA oligo donor for introducing a diagnostic EcoRI site at the CjCas9 cleavage site in POR. The oligo sequence is: 5’-CACCCTTGGTCTCCCCTTTCCAGCATTCGCCAGTACGAGCGAATTCTTGTGGTCCACACCGACATAGATGCGGCCAAGGTGTACATGG-3’ (Underlined: EcoRI site)
Note: Synthesize the oligo at 0.2 µmol scale and purify by PAGE. Re-suspend the oligo in 10 mM Tris buffer (pH 7.6) at 200 µM.
- PCR primers for amplification of the targeted POR genomic region: forward 5’-CTCCCCTGCTTCTTGTCGTAT-3’, reverse 5’-ACAGGTCGTGGACACTCACA-3’
- Alcohol
- Hank’s balanced salt solution (Sigma-Aldrich, catalog number: H6648 )
- Nucleofector instrument (Lonza, catalog number: AAB-1001 )
- Amaxa Cell Line Nucleofector Kit V (Lonza, catalog number: VCA-1003 )
- GenElute Mammalian Genomic DNA Miniprep Kit (Sigma-Aldrich, catalog number: NA2010 )
- JumpStart Taq ReadyMix (Sigma-Aldrich, catalog number: P2893 )
- GenElute PCR Clean-Up Kit (Sigma-Aldrich, catalog number: NA1020 )
- EcoRI restriction enzyme (New England Biolabs, catalog number: R0101S )
- 10% Mini-PROTEAN TGX Precast Protein Gels, 15-well, 15 µl (Bio-Rad Laboratories, catalog number: 4561036 )
- Tris buffered saline (Sigma-Aldrich, catalog number: T5912 )
- Gel loading buffer (Sigma-Aldrich, catalog number: G2526 )
- DirectLoad Wide Range DNA Marker (Sigma-Aldrich, catalog number: D7058 )
- Iscove’s modified Dulbecco’s medium (Sigma-Aldrich, catalog number: I3390 )
- Fetal bovine serum (FBS) (Sigma-Aldrich, catalog number: F2442 )
- L-glutamine, 200 mM (Sigma-Aldrich, catalog number: G7513 )
- K562 culture medium (see Recipes)
Equipment
- Water bath (Polyscience)
- Laminar flow hood for sterile tissue culture, biosafety level 2 approved (Thermo Fisher Scientific)
- Microbiological incubator with 37 °C, atmospheric CO2 (Thermo Fisher Scientific)
- Pipettes (Gilson)
- Bench-top centrifuge (Eppendorf, model: 5417 C )
- Mini-PROTEAN Tetra Vertical Electrophoresis Cell (Bio-Rad Laboratories, catalog number: 1658004 )
- PCR thermocycler (Bio-Rad Laboratories, catalog number: 1861096 )
Software
- ImageJ (https://imagej.nih.gov/ij/)
Procedure
文章信息
版权信息
© 2017 The Authors; exclusive licensee Bio-protocol LLC.
如何引用
Chen, F., Ding, X., Feng, Y., Seebeck, T., Jiang, Y. and Davis, G. D. (2017). Improving CRISPR Gene Editing Efficiency by Proximal dCas9 Targeting. Bio-protocol 7(15): e2432. DOI: 10.21769/BioProtoc.2432.
分类
分子生物学 > DNA > DNA 修饰
您对这篇实验方法有问题吗?
在此处发布您的问题,我们将邀请本文作者来回答。同时,我们会将您的问题发布到Bio-protocol Exchange,以便寻求社区成员的帮助。
Share
Bluesky
X
Copy link


.jpg)