Materials and Reagents
- Mice of interest
- 1x DPBS (Lonza, catalog number: 17-512F )
- ACK lysing buffer (Lonza, catalog number: 10-548E )
- Dulbecco’s Modified Eagle’s Medium with high glucose and without L-glutamine (DMEM) (Lonza, catalog number: 12-614F )
- Fetal Calf Serum (FCS) (Thermo Scientific, catalog number: SH30070.03 )
- Murine M-CSF (Peprotech, catalog number: 315-02 )
- 100x Penicillin/Streptomycin (Invitrogen, catalog number: 10378-016 )
- Complete DMEM medium (see Recipes)
Equipment
- 40 μm nylon strainer (BD Falcon, catalog number: 352340 )
- Needle (Gauge #25)
- Centrifuge
Procedure
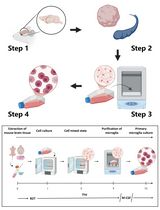
- Mouse bone marrow (BM) cells are harvested from femurs by syringe and needle (Gauge #25) with 5 ml 1x DPBS. (Cut two ends of femurs and rinse out the cells by 1x DPBS)
- Centrifuge the cell suspension at 200 x g at room temperature (RT) for 5 min.
- Remove the supernatant and suspend the cell pellet with 2 ml ACK lysing buffer for 1 min to deplete red blood cells.
- The cell suspension is directly filtered through a 40 μm nylon strainer (observe the cell lysate under the microscope to determine whether red blood cells are depleted completely. If there are still too many red blood cells, add more ACK lysing buffer until there are very few red blood cells).
- Wash the strainer with 2 ml 1x DPBS and centrifuge the filtered cell suspension at 200 x g at RT for 5 min.
- The cell pellet is washed once again with 1x DPBS and re-suspended in 15 ml complete DMEM medium with 20 ng/ml murine M-CSF in a 100 mm Petri dish (one femur cells per dish) in an incubator (37 °C, 5% CO2).
- After 3 days, half of the medium is replaced with fresh complete DMEM medium.
- On Day 4, the cells containing more than 80% CD11b+/F4/80+ macrophages are ready for further characterization and functional experiments.
Recipes
- Complete DMEM medium (505 ml)
100 ml FCS
5 ml 100x Penicillin/Streptomycin
400 ml DMEM
Acknowledgments
This protocol has been adapted from Liu et al. (2013).
References
- Liu, Y., Chen, K., Wang, C., Gong, W., Yoshimura, T., Liu, M. and Wang, J. M. (2013). Cell surface receptor FPR2 promotes antitumor host defense by limiting M2 polarization of macrophages. Cancer Res 73(2): 550-560.
Article Information
Copyright
© 2013 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Liu, Y., Chen, K., Wang, C., Gong, W., Yoshimura, T., Wang, J. M. and Liu, M. (2013). Mouse Macrophage Differentiation by Induction with Macrophage Colony-Stimulating Factor . Bio-protocol 3(17): e893. DOI: 10.21769/BioProtoc.893.

 Download Citation in RIS Format
Download Citation in RIS Format
Category
Immunology > Immune cell isolation > Maintenance and differentiation
Cell Biology > Cell isolation and culture > Cell isolation