- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Whole Spleen Flow Cytometry Assay
Published: Vol 3, Iss 15, Aug 5, 2013 DOI: 10.21769/BioProtoc.834 Views: 37093
Reviewed by: Fanglian He

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Detection of Autophagy in Human Peripheral Blood Mononuclear Cells Using Guava® Autophagy and Flow Cytometry
Melanie Scherer [...] Jörg Bergemann
Sep 20, 2025 1444 Views
Protocol for the Isolation and Analysis of Extracellular Vesicles From Peripheral Blood: Red Cell, Endothelial, and Platelet-Derived Extracellular Vesicles
Bhawani Yasassri Alvitigala [...] Lallindra Viranjan Gooneratne
Nov 5, 2025 1508 Views
Dual Phospho-CyTOF Workflows for Comparative JAK/STAT Signaling Analysis in Human Cryopreserved PBMCs and Whole Blood
Ilyssa E. Ramos [...] James M. Cherry
Nov 20, 2025 2393 Views
Abstract
In the Whole Spleen Flow Cytometry Assay, we used splenocytes directly ex vivo for stimulation with a variety of TLR ligands. The splenocytes were stimulated for a total of 4 hours, then stained for intracellular cytokines. We then examined cytokine production via flow cytometry. This allowed us to compare the responses of minimally manipulated primary macrophages/monocytes and conventional dendritic cells.
Materials and Reagents
- PerCP/Cy5.5 anti-mouse CD11b (Biolegend, catalog number: 101228 )
- PE/Cy7 anti-mouse CD11c (BD Bioscience, catalog number: 558079 )
- APC/Cy7 anti-mouse CD19 (BD Bioscience, catalog number: 557655 )
- APC anti-mouse CD3e (Biolegend, catalog number: 100312 )
- FITC anti-mouse TNFa (Caltag, catalog number: RM9011 )
- PE anti-mouse IL-12 (BD Bioscience, catalog number: 554479 )
- aCD16/32 FcBlock (BD Bioscience, catalog number: 553142 )
- FITC anti-mouse CD8 (BD Bioscience, catalog number: 553031 )
- PE anti-mouse CD8 (BD Bioscience, catalog number: 553033 )
- PerCP/Cy5.5 anti-mouse CD4 (Biolegend, catalog number: 100539 )
- PE/Cy7 anti-mouse CD8 (Biolegend, catalog number: 100722 )
- APC anti-mouse CD8 (BD Bioscience, catalog number: 553035 )
- Ultrapure O111:B4 EC LPS (Life Technologies, InvitrogenTM, catalog number: tlrl-pelps )
- Synthetic Lipid IVa (Peptides International, catalog number: CLP-24006-S )
- ODN1826 (Coley Pharmaceuticals)
- Frosted glass slides, nonsterile (Fisherbrand, catalog number: 12-550-343 )
- Cytofix/Cytoperm Solution (BD Biosciences, catalog number: 554722 )
- 10x Perm Wash Buffer (BD Biosciences, catalog number: 554723 )
- 10% Paraformaldehyde (Electron Microscopy Sciences, catalog number: 15712-S )
- RPMI 1640 Medium (with glutamax, HEPES, Phenol red) (Life Technologies, catalog number: 72400-120 )
- Fetal bovine serum (FBS) (Heat Inactivated) (Hyclone, catalog number: SH30070.03HI )
- Pen/Strep (Life Technologies, catalog number: 15140-122 )
- Beta-ME (Sigma-Aldrich, catalog number: M-7522 )
- NaCl
- KCl
- KH2PO4
- Na2HPO4
- NaOH
- NH4Cl
- NaHCO3
- 10x Phosphate buffered saline (PBS) (see Recipes)
- 1x PBSA/azide (PBSA/az) (see Recipes)
- RBC Lysis Buffer (see Recipes)
- Culture Medium (see Recipes)
- Brefeldin A (Sigma-Aldrich, catalog number: B6542 ) (see Recipes)
Equipment
- 15 ml conical tubes (BD Biosciences, Falcon®, catalog number: 352196 )
- 50 ml conical tubes (BD Biosciences, Falcon®, catalog number: 352070 )
- FACS titertube microtubes (Bio-Rad Laboratories, catalog number: 223-9391 )
- 70 μM filters (BD Biosciences, Falcon®, catalog number: 352350 )
- Tissue culture (TC) dish (100 x 20 mm) (Corning Incorporated, catalog number: 430167 )
- 96-well round bottom plates (Corning Incorporated, catalog number: 3799 )
- BD FACS Canto I (6 color analyzer) flow cytometer: Equipped with 2 lasers; blue laser 488 nm four color detection and red laser 633 nm 2 color detection. Uses FACSDiva software
- Centrifuge (Beckman Coulter, model: Allegra X-15R )
Software
- FACSDiva software
- BD FACS Canto I software
Procedure
- Splenocyte Isolation
- Extract whole spleen from mouse and place in a 15 ml polypropylene tube on ice (no liquid).
- Put 5 ml RBC lysis buffer into 15 ml tube with spleen.
- Pour out spleen with RBC lysis buffer into a 10 cm tissue culture dish.
- Smash spleen thoroughly between frosted glass slides by placing the spleen on the rough side of the frosted part of the slide (wetted with RBC lysis buffer) and grind it between the two slides until spleen is dissociated (Video 1). Video 1. Homogenization of spleen between two glass slides
- Pipette up the RBC lysis containing splenocytes back into the 15 ml tube.
- Add 10 ml of culture medium to the tube, invert 2-3 times.
- Spin at 1,300 rpm at 4 °C for 5 min.
- Discard the supernatant and resuspend the pellet in 3 ml medium.
- Filter through a 70 μM filter over a 50 ml conical tube.
- Count 1:100 dilution of cells.
- Dilute single-cell suspensions to 1 x 107 cells/ml in RPMI medium (for Flow Assays, counting and calculating of cells is not necessary).
- Place splenocytes on ice until ready to plate into 96-well plate.
- Extract whole spleen from mouse and place in a 15 ml polypropylene tube on ice (no liquid).
- Stimulation of splenocytes
- Prepare a stock of RPMI medium with Brefeldin A added to it at 10 μg/ml.
- Vortex and spin down all LPS stocks, then sonicate each for 10 min. After sonication, vortex and spin down again.
- Each stimulus (EC LPS, PA, YP, Lipid IVa) is prepared to twice (2x) its desired final concentration using the Brefeldin A containing medium.
- Plate 100 μl of each stimulus into the appropriate wells of the 96-well plate, include unstimulated wells as a control.
- Plate 100 μl of the splenocytes into the appropriate wells (plate an extra set of splenocytes to be used as single stain controls).
- Incubate the plate for 4 h at 37 °C.
- Prepare a stock of RPMI medium with Brefeldin A added to it at 10 μg/ml.
- Immunocytochemistry (ICC) staining
- After 4 h, treat wells with 20 μl of 20 mM EDTA.
- Mix and incubate at 37 °C for 10 min.
- Mix again after incubation and then spin at 1,300 rpm for 5 min.
- Aspirate or flick medium off.
- Block with 100 μl of diluted FcBlock (1:100 FcBlock in PBSA/az) to all wells.
- Incubate on ice for 15 min.
- Add 100 μl of PBSA/az, then spin at 1,300 rpm for 5 min.
- Aspirate or flick medium off.
- Resuspend/stain with 100 μl of Antibody mix: CD11b PerCP-Cy5.5, CD11c PE-Cy7, CD19 APC-Cy7, and CD3 APC (1:100 Ab in PBSA/az) to the stimulated splenocyte samples.
- Add 100 μl of PBSA/az to the single stain controls, then add 1 μl of each of these single stain Ab: CD8 FITC, CD8 PE, CD4 PerCP-Cy5.5, CD8 PE-Cy7, CD19 APC-Cy7, and CD8 APC.
- Incubate on ice in dark (or wrap in foil) for 20 min.
- Add 100 μl PBSA/az, then spin at 1,300 rpm for 5 min.
- Aspirate or flick medium off.
- Resuspend with 100 μl of BD cytofix/cytoperm solution to all wells.
- Incubate on ice in dark for 20 min.
- Add 100 μl of 1x BD Perm wash (dilute 10x Perm wash in dH2O) to all wells.
- Spin at 1,300 rpm for 5 min, then aspirate or flick medium off.
- Resuspend/wash once with 200 μl of 1x BD Perm wash by pipetting up and down a few times to all wells.
- Spin at 1,300 rpm for 5 min, then aspirate or flick medium off.
- Resuspend/stain with 50 μl of Antibody mix: TNF FITC and IL-12/IL-23p40 PE (1:100 Ab in 1x perm wash) to the stimulated splenocyte samples (add 50 μl of 1x perm wash to the single stain controls).
- Incubate on ice in dark for 30 min.
- Add 150 μl 1x perm wash to all wells, then spin at 1,300 rpm for 5 min.
- Aspirate or flick medium off.
- Resuspend/wash once with 200 μl 1x perm wash by pipetting up and down a few times to all wells.
- Spin at 1,300 rpm for 5 min, then aspirate or flick medium off.
- Resuspend in final volume of 200 μl of 1% Paraformaldehyde solution (dilute 10% Paraformaldehyde in 1x PBS).
- Store at 4 °C in dark (or wrapped in foil) until ready for Flow Cytometry (recommended to store no longer than 24 h) – transfer samples from 96-well plate into titer tubes immediately prior to FACS.
- After 4 h, treat wells with 20 μl of 20 mM EDTA.
- Flow Cytometry
- On the BD FACS Canto I software, select for a 'New Experiment' to set up.
- Select for Area, Height, Width for FSC and SSC; select for Log and Area for the colors FITC, PE, PerCP/Cy5.5, PE/Cy7, APC/Cy7, and APC (under Inspector box).
- Create compensation controls and adjust the gating for unstained splenocytes (adjust FSC and SSC voltages as necessary).
- Adjust each color of single stains in the voltage panel so that the positive peak is at the 104 mark.
- For single stains, use anti-CD4 for PerCP/Cy5.5, anti-CD19 for APC/Cy7, and anti-CD8 for FITC, PE, PE/Cy7, and APC, see Figure 1 for example.
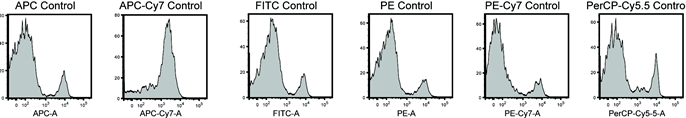
Figure 1. Example of single stain controls. Splenocytes were stained with the single antibodies listed under IV-5 and voltages adjusted to give depicted histograms. - Record the desired voltages after any adjustments for each of the single stains and unstain, then calculate compensation controls.
- Events are now ready to be recorded – set up to collect 500,000 – 1,000,000 events.
- On the BD FACS Canto I software, select for a 'New Experiment' to set up.
Recipes
- 10x Phosphate buffered saline (PBS) (for 1 L)
75 g NaCl
2 g KCl
2 g KH2PO4
11.5 g Na2HPO4
1 ml 10 N NaOH
pH to 7.4 and autoclave - 1x PBSA/azide (for 1 L)
Dilute 10x PBS to 1x PBS using ddH2O
10 g BSA
18 ml of 5% NaN3 (final concentration = 0.09% sodium azide)
Filter sterilize - RBC Lysis buffer
0.15 M NH4Cl
1 mM NaHCO3
0.1 mM EDTA dissolved in sterile irrigation water
pH to 7.2-7.4 with 1 M HCl
Filter sterilize - Culture medium
RPMI 1640 Medium (with glutamax, HEPES, Phenol red)
10% FBS
5 ml Pen/Strep
5 ml 5 mM beta-ME - Brefeldin A
Received as 25 mg of dry powder
Dissolve in ethanol to 2 mg/ml and aliquot into dark eppendorf tubes (light sensitive)
Store at -20 °C
References
- Hajjar, A. M., Ernst, R. K., Fortuno, E. S., 3rd, Brasfield, A. S., Yam, C. S., Newlon, L. A., Kollmann, T. R., Miller, S. I. and Wilson, C. B. (2012). Humanized TLR4/MD-2 mice reveal LPS recognition differentially impacts susceptibility to Yersinia pestis and Salmonella enterica. PLoS Pathog 8(10): e1002963.
Article Information
Copyright
© 2013 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Yam, C. S. and Hajjar, A. M. (2013). Whole Spleen Flow Cytometry Assay. Bio-protocol 3(15): e834. DOI: 10.21769/BioProtoc.834.
Category
Immunology > Immune cell function > Cytokine
Cell Biology > Cell-based analysis > Flow cytometry
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link









