- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
An Assay to Determine NAD(P)H: Quinone Oxidoreductase Activity in Cell Extracts from Candida glabrata
(*contributed equally to this work) Published: Vol 11, Iss 21, Nov 5, 2021 DOI: 10.21769/BioProtoc.4210 Views: 3086
Reviewed by: Alexandros AlexandratosSascha BrunkeYingnan Hou

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Measurement of Energy-dependent Rhodamine 6G Efflux in Yeast Species
Yvetta Gbelska [...] Alexandra Konecna
Aug 5, 2017 8985 Views
Isolation of Mitochondria from Ustilago maydis Protoplasts
Juan Pablo Pardo [...] Lucero Romero-Aguilar
Jan 5, 2022 3603 Views
Abstract
Flavodoxin-like proteins (Fld-LPs) are an important constituent of the oxidative stress defense system in several organisms and highly conserved from bacteria to humans. These proteins possess NAD(P)H:quinone oxidoreductase activity and convert quinones to hydroquinones through two-electron reduction, using NAD(P)H and quinone as electron donor and acceptor, respectively. Purified yeast and bacterial Fld-LPs exhibit NAD(P)H:quinone oxidoreductase activity in vitro. Here, we describe a protocol to measure oxidoreductase activity of Fld-LPs that are present in extracts of whole cells. We have recently shown that the assembly and activity of a Fld-LP, CgPst2, is regulated by an aspartyl protease-mediated cleavage of its C-terminus in the pathogenic yeast Candida glabrata. Mutant yeast where the CgPST2 gene was deleted lacked cellular NAD(P)H:quinone oxidoreductase activity and displayed elevated susceptibility to menadione stress. The protocol described herein is based on the measurement of NADH oxidation (conversion of NADH to NAD+) by endogenous Fld-LPs in the presence of quinone menadione. This assay can be performed with whole cell lysates prepared by the mechanical lysis of C. glabrata cells and does not require expression and purification of Fld-LPs from a heterogeneous system, thereby allowing researchers to study the effect of different posttranslational modifications and varied structural states of Fld-LPs on their enzymatic activities. Since many FLP-LPs are known to exist in dimeric and tetrameric states possessing differential activities, our efficient and easy-to-use assay can reliably detect and validate their quinone reductase activities. Although we have used menadione with CgPst2 enzyme in our study, the protocol can easily be modified to examine the presence of Fld-LPs with specificity for other quinones. As this assay does not require many expensive chemicals, it can readily be scaled up and adapted for other medically important fungi and potentially be a useful tool to characterize fungal oxidative stress response systems and screen inhibitors specific for fungal Fld-LPs, thereby contributing to our understanding of fungal pathogenesis mechanisms.
Keywords: FlavodoxinBackground
Invasive fungal infections are a common cause of morbidity, mortality, and high healthcare costs in hospitals worldwide (Bongomin et al., 2017). Candida species are opportunistic human fungal pathogens, with Candida glabrata being the second to fourth most prevalent etiological agent of Candida bloodstream infections (Chakrabarti et al., 2015; Bongomin et al., 2017; Lamoth et al., 2018; Pfaller et al., 2019). Cells of the innate immune system play an important role in the control of Candida infections through an armamentarium of reactive oxygen and nitrogen species generation, antifungal peptide production, and facilitating fungal antigen presentation to activate the adaptive immune system (Netea et al., 2015; Heung, 2020). C. glabrata encounters reactive oxygen species (ROS)-induced stress upon internalization by host macrophages (Seider et al., 2011; Rai et al., 2012). However, it probably possesses a robust oxidative stress defense system that facilitates its survival and replication in macrophages (Kaur et al., 2007; Seider et al., 2011; Rai et al., 2012). Of note, macrophages are thought to act as Trojan horses for C. glabrata, providing it with a safe niche in the human host (Brunke and Hube, 2013).
Efficient detoxification of ROS is necessary to survive an oxidative environment. C. glabrata shows high levels of resistance towards oxidative stress caused by hydrogen peroxide in vitro (Cuéllar-Cruz et al., 2008). The sole catalase CgCta1 and two superoxide dismutases, CgSod1 and CgSod2, contribute to the effective oxidative stress response system in C. glabrata (Cuéllar-Cruz et al., 2008; Briones-Martin-Del-Campo et al., 2015). Another arm of the oxidative stress defense system is represented by proteins containing flavodoxin-like fold that are implicated in quinone detoxification (Sancho, 2006; Carey et al., 2007; Lodeyro et al., 2012). The flavodoxin-fold is characterized by a 5-stranded parallel β-sheet surrounded by α-helices on both sides, and present in proteins that are involved in electron transfer between varied redox compounds (Sancho, 2006; Carey et al., 2007; Lodeyro et al., 2012; Farías-Rico et al., 2014; Houwman and van Mierlo, 2017). Many proteins with flavodoxin-like fold possess NAD(P)H:quinone oxidoreductase activity and detoxify quinones to hydroquinones by preventing the formation of unstable and highly reactive semiquinone species, and use FMN (flavin mononucleotide) or FAD (flavin adenine dinucleotide) as cofactors (Patridge and Ferry, 2006; Sancho, 2006; Carey et al., 2007; Farías-Rico et al., 2014; Koch et al., 2017). This enzymatic reaction follows a ping-pong bi-bi mechanism, occurs in two half-reactions, reductive and oxidative, and involves two direct hydride transfer steps (Deller, 2008). The reductive half-reaction involves electron donation by the reduced form of nicotinamide adenine dinucleotide, NADH, to cofactors FMN or FAD, with NADH getting oxidized to NAD+. In the oxidative half-reaction, the reduced cofactors, FMN(H2) or FAD(H2), transfer the electrons to quinone substrates, which leads to the reduction of quinones to hydroquinones (Deller, 2008).
We have recently identified and characterized four flavodoxin-like proteins (Fld-LPs) in C. glabrata: CgPst2, CgRfs1, CgPst3, and CgYcp4, and shown that CgPst2 is uniquely required to survive menadione and benzoquinone stress (Battu et al., 2021). Since the bacterially purified CgPst2 showed no NAD(P)H:quinone oxidoreductase activity (Battu et al., 2021), we developed a protocol to measure NAD(P)H:quinone oxidoreductase activity in whole cell lysates of wild-type and CgPST2 gene-deleted strains to establish the role of CgPst2 in quinone detoxification. The step-by-step procedure followed to carry out the assay is represented in Figure 1. Our protocol is based on the conserved ping-pong mechanism of flavodoxin-like proteins, wherein we monitored the oxidation of NADH to NAD+ at 340 nm as a measure of quinone conversion to quinol/hydroquinone. Since the reduced form of NADH absorbs at 340 nm, while the oxidized form NAD+ has no absorbance at 340 nm, NADH oxidation was recorded as a decrease in the absorbance at 340 nm. This reaction mechanism is schematically illustrated in Figure 2.
Using this assay, we were able to show that menadione treatment leads to an increase in cellular NAD(P)H:quinone oxidoreductase activity, which could be partially attributed to elevated CgPst2 levels and enhanced aspartyl protease CgYps1-mediated cleavage of CgPst2, stimulating homo-tetramerization of CgPst2 (Battu et al., 2021). Notably, purified Fld-LPs WrbA and Pst2 exhibited NAD(P)H:quinone oxidoreductase activity in Escherichia coli and Saccharomyces cerevisiae, respectively (Patridge and Ferry, 2006; Koch et al., 2017). However, using the protocol developed, we could show NAD(P)H:quinone oxidoreductase activity in whole cell extracts of C. glabrata leading to NADH oxidation in the presence of menadione in vitro (Battu et al., 2021). Importantly, this activity was absent in strains deleted for the CgPST2 gene, and the activity was regained upon expression of CgPST2 gene in mutant strains (Battu et al., 2021), thereby unequivocally correlating the cellular NAD(P)H:quinone oxidoreductase activity with the presence of CgPst2 in cells.
The advantages of our protocol are many-fold. First, it is an efficient way to measure NAD(P)H:quinone oxidoreductase activity in cellular extracts and does not require cloning, expression, and purification of Fld-LPs in a heterologous expression system. Second, it neither requires expensive chemicals nor sophisticated equipment. Third, our assay is easy-to-use and can readily be applied to study different quinone detoxification activities under varied stress and host-mimicking conditions, as well as in mutants lacking various genes of the oxidative stress response machinery and yeast cells recovered from host immune cells, including macrophages. It can also be adapted to screen inhibitors that are specific for fungal Fld-LPs. Fourth, unlike activity measurement in purified enzymes, no additional supply of FMN/FAD is required as cofactor in our assay. Finally, since this protocol estimates NAD(P)H:quinone oxidoreductase activity in cellular extracts, the loss of enzymatic activity often observed during protein purification is avoided. Thus, this assay can provide key insights into the activity of Fld-LPs present in their native states, including Fld-LPs containing different posttranslational modifications and/or oligomeric Fld-LPs.
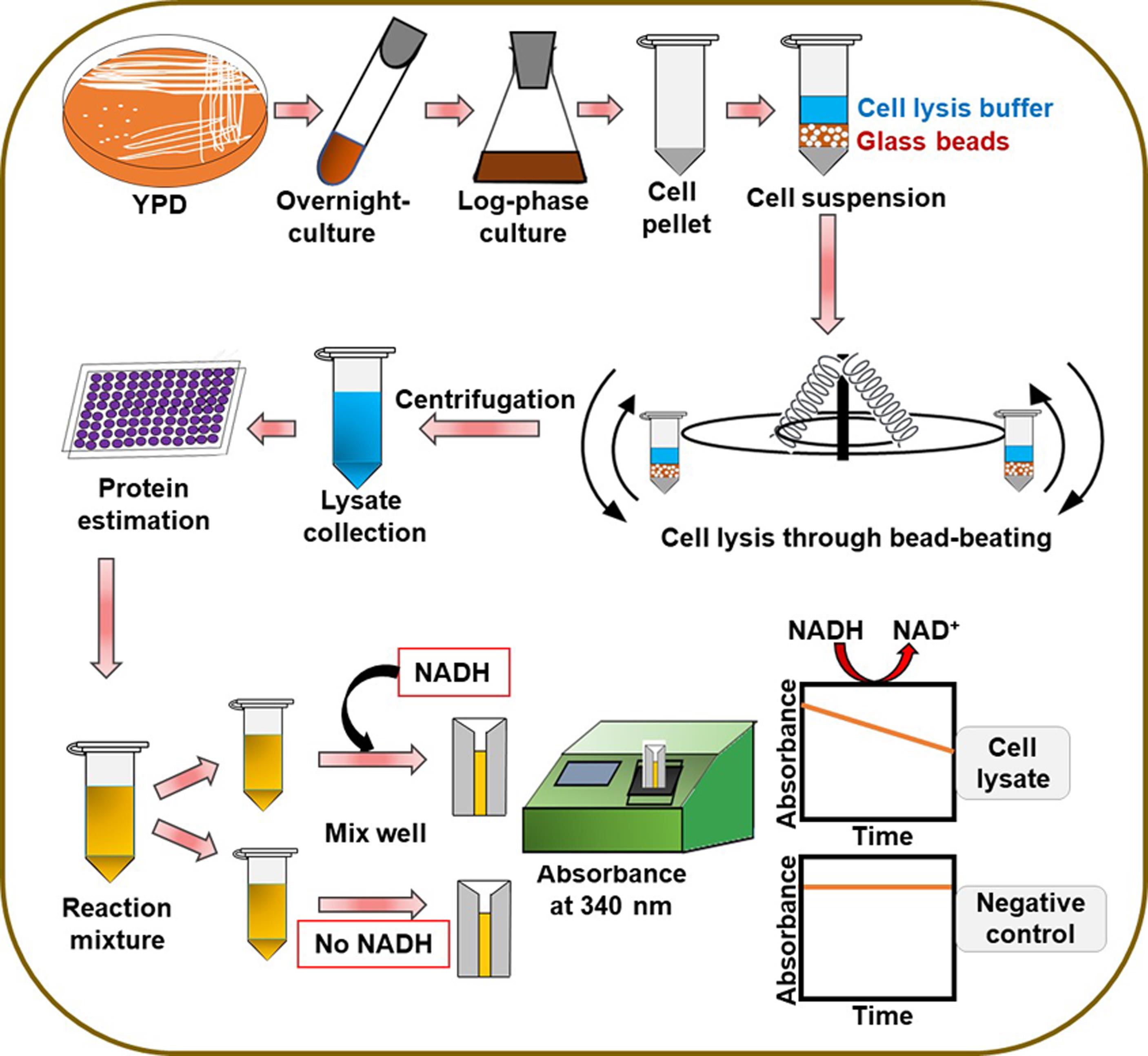
Figure 1. Schematic representation of the steps in the protocol
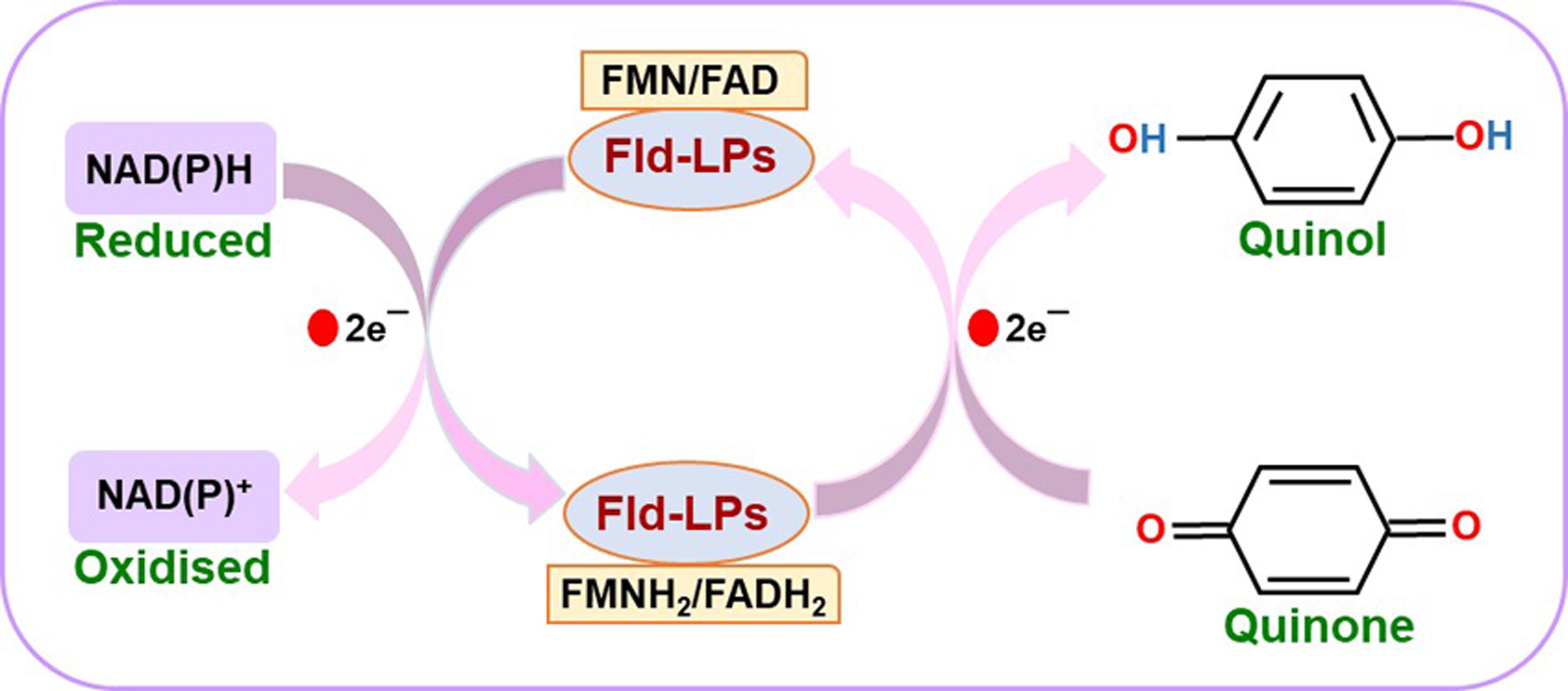
Figure 2. Schematic illustration of the reaction mechanism of flavodoxin-like proteins (Fld-LPs)
Materials and Reagents
40 ml glass test tube
Cotton wads
1.5 ml centrifuge tubes (Tarsons, catalog number: 500010)
15 ml Falcon tubes (Tarsons, catalog number: 546021)
0.5 mm diameter glass beads (BioSpec Products, catalog number: 11079105)
96-well plates (Biofil, catalog number: 011096)
Menadione (Sigma-Aldrich, catalog number: M5625)
Ethanol (PHARMCO-AAPER, catalog number: 111WORLDUV200)
200 mM phenylmethylsulphonyl fluoride (PMSF, serine proteases inhibitor) (Amresco, catalog number: 0754-5G)
100 mM sodium orthovanadate (NaOVa, Tyr and alkaline phosphatase inhibitor) (Sigma-Aldrich, catalog number: S6508)
200 mM sodium fluoride (NaF, Ser/Thr and acid phosphatase inhibitor) (Sigma-Aldrich, catalog number: 450022)
100× protease inhibitor cocktail (Sigma-Aldrich, catalog number: P8215)
PierceTM BCA protein assay kit (Thermo Scientific, catalog number: 23227)
Nicotinamide adenine dinucleotide (NADH; reduced form of NAD), disodium salt, grade 2 approx. 98% (500 µM) (Roche, catalog number: 10128023001)
NAD(P)H: FMN oxidoreductase from Photobacterium fischeri (Roche, catalog number: 10476480001)
Culture media: Yeast extract (1%)-Peptone (2%)-Dextrose (2%) YPD (Becton, Dickinson and Company (BD) Diagnostic Systems, catalog number: 242810) (see Recipes)
Phosphate Buffer Saline (PBS) (see Recipes)
NaCl (Affymetrix, catalog number: 21618)
KCl (G-Biosciences, catalog number: RC1167)
Na2HPO4 (Fisher Scientific, catalog number: 27785)
KH2PO4 (Fisher Scientific, catalog number: 13405)
Cell lysis buffer (see Recipes)
Tris base (MP Biosciences, catalog number: 808229)
EDTA (Sigma-Aldrich, catalog number: E6635)
Dextrose (Becton, Dickinson and Company (BD) Biosciences, catalog number: 215530)
Quinone reductase activity assay buffer (see Recipes)
Tris base (MP Biosciences, catalog number: 808229)
NaCl (Affymetrix, catalog number: 21618)
HPLC grade water (Merck Life Science Pvt. Limited, catalog number: 61765010001730)
Equipment
30°C shaker incubator (New Brunswick Innova, model: 43R)
Spectrophotometer/Cell density meter (Biowave-WFA, model: C08000)
Fine weighing balance (Mettler Toledo, model: MS205DU)
FastPrep 24 homogenizer or Bead beater (MP Biomedicals, model: 19083976)
Cooling table top centrifuge (Eppendorf, model: 5424R)
Quartz cuvette, 10 mm light path (Hellma Analytics, catalog number: 104-10-40)
SpectraMax multimode microplate reader (Molecular Devices, model: M5)
Needle and Spatula (Local stores)
Software
Excel 2018 (Microsoft)
SoftMax Pro Software 7.0
GraphPad Prism 7
Procedure
C. glabrata culturing
Inoculate one single colony from the C. glabrata-streaked YPD agar plate in a 40 ml glass test tube with 5 ml of YPD medium.
Place the tubes in a plastic beaker, and put cotton wads around the tubes so that these are tightly arranged in the beaker.
Place the beaker in an incubator shaker, and grow the cultures with agitation at 200 rpm (rotation per minute) at 30°C overnight (14 to 16 h).
The next morning, measure the optical density of the cultures at 600 nm (OD600) using the spectrophotometer, by diluting 20 times in sterile PBS.
To obtain logarithmic (log)-phase cultures, inoculate the fresh YPD medium (10 ml, in 100 ml flask) with overnight-grown cultures at an OD600 of 0.1 and incubate with 200 rpm agitation at 30°C for 4-5 h, until the OD600 reaches 0.4-0.6. The OD600 may be measured every half an hour after 4 h of incubation.
Cell lysis
Transfer 10 ml of log-phase culture (0.4-0.6 OD600) to a 15 ml conical centrifuge/Falcon tube.
Centrifuge tubes at 4,700 × g for 5 min at 4°C.
Discard the supernatants and add 10 ml of PBS to the cell pellets.
Centrifuge tubes at 4,700 × g for 5 min at 4°C and discard the supernatants. Add 1 ml of PBS and suspend cell pellets in PBS by gentle pipetting.
Transfer cell suspensions to 1.5 ml microcentrifuge tubes and centrifuge tubes at 2,400 × g for 5 min at 4°C in a refrigerated microcentrifuge.
Remove the supernatant completely using a 1 ml micropipette tip, without disturbing the pellet, add 200 μl cell lysis buffer to each tube, and suspend the cell pellet by gentle pipetting.
Add 0.5 mm glass beads to each tube up to a volume equal to that of the cell suspension, and lyse cells using the FastPrep 24 homogenizer at the maximum speed setting of 6.5 M/s for five cycles of 60 s each, with 3-5 min ice incubations in between cycles. Of note, based on the fungal species, this cell lysis step may require standardization for the optimal number of cycles and bead-beating speed settings.
Take the tubes out from the FastPrep homogenizer and puncture them at the bottom using a needle. Place the cell homogenate-containing punctured tube inside a new 1.5 ml microcentrifuge tube whose lid stays open. Arrange the tubes in a centrifuge with the open lids facing inwards and towards the center of the rotor, and centrifuge at 2,400 × g for 5 min at 4°C. The supernatant will be transferred to the lower tube, while the glass beads will stay in the upper tube. Alternatively, centrifuge the cell homogenate-containing tubes at 21,000 × g for 10 min at 4°C to remove the beads, and transfer the supernatant to a new tube.
Centrifuge tubes again at 21,000 × g for 10-15 min at 4°C to remove cell debris and non-lysed cells, and collect the clear supernatants containing cellular proteins (approximately 150 μl) in new microcentrifuge tubes.
Measure protein concentration in cell lysates using the BCA protein assay kit, following the manufacturer’s instructions. For this, add 11.5 μl sterile water and 1 μl cell lysate per assay. The reaction mixture volume in each well of a 96-well plate will be 12.5 μl. For standard plot generation, add 1 μl to 5 μl of BSA (Bovine serum albumin; 2 mg/ml concentration provided with the kit) in five separate wells of the 96-well plate. Make up the volume of each well to 12.5 μl with sterile water. Keep one well containing 12.5 μl of sterile water as blank, to which no protein sample is added. Add 100 μl of BCA solution (Solution A and Solution B in 50:1 ratio) to each well. Cover the plate with aluminum foil and incubate it at 37°C for 30 min. Measure sample absorbance at 562 nm in a multimode microplate reader. Calculate the protein concentration in cell lysate samples, which usually ranges from 5 to 10 μg/μl, using the BSA standard curve.
Setting up of reaction mixture
Start preparing 1 ml total volume of the reaction mixture by placing a 1.5 ml centrifuge tube on ice. It will contain 500 µg protein, 500 µM menadione, and 500 µM NADH.
Add the cell lysate corresponding to 500 µg of protein and 10 μl of 50 mM menadione stock solution (freshly prepared by dissolving 8.609 mg powder in 1 ml absolute ethanol in a 1.5 ml microcentrifuge tube) to the reaction mixture. Of note, menadione is required for this assay, as no enzymatic activity was observed in the absence of exogenous menadione. However, menadione may be replaced with other quinones to check the specificity of different quinone reductase enzymes present in cell lysates. Similarly, since the levels and activities of Fld-LPs may change under various stress conditions, it is important to standardize the assay with 2-3 different concentrations of cell lysates and measure the absorbance values in the linear range.
Make up the reaction volume to 950 µl with the assay buffer.
For each cell lysate, prepare one blank control that contains all reagents.
Set up a positive control by adding 20 μl of NAD(P)H: FMN oxidoreductase (1 Unit; Roche) instead of the cell lysate.
Prepare 10 mM NADH stock solution by dissolving 6.6343 mg of powder in 1 ml HPLC grade water, before proceeding to the absorbance measurement step.
Absorbance measurement
Set up the Spectramax M5 plate reader to measure absorbance at 340 nm over a period of 60 s at 10 s intervals.
Perform all the following steps in the dark.
Add 50 µl of 10 mM NADH solution to the reaction mixture, mix well, transfer to a quartz cuvette (1-cm-path-length), and read the absorbance at 340 nm over a period of 60 s at 10 s intervals. Since NADH addition will start the quinone detoxification reaction, this step needs to be performed as quickly as possible. Therefore, it is important to start reactions individually just before the absorbance measurement.
As a negative control, measure the absorbance of the blank sample (without adding NADH) at 340 nm over a period of 60 s at 10 s intervals.
Repeat the above-mentioned steps for all cell lysate and positive control samples.
Data analysis
For each sample, a graph displaying absorbance values as a function of time will appear on the SpectraMax M5 multimode microplate system, which is generated using the SoftMax Pro Software 7.0. Save the raw file as pdf and export the raw values to an excel file. This can be done by a right click on the graph and choosing the copy cuvette data option, followed by pasting data in the excel sheet.
Arrange the data in a new excel sheet by placing sample absorbance values in an ascending order by time of measurement.
Considering the absorbance of the substrate NADH as 100 at 0 s time point, calculate NADH oxidation in each sample by dividing the absorbance at each time point by 0 s absorbance and multiplying the number by 100. The number obtained reflects the NADH (substrate) amount remaining in the reaction mixture at that particular time point.
Plot the amount of substrate remaining versus the time point (in seconds) at which the absorbance was measured, using GraphPad Prism 7 or any other software. A representative graph is shown in Figure 3. Perform the experiment in biological triplicates.
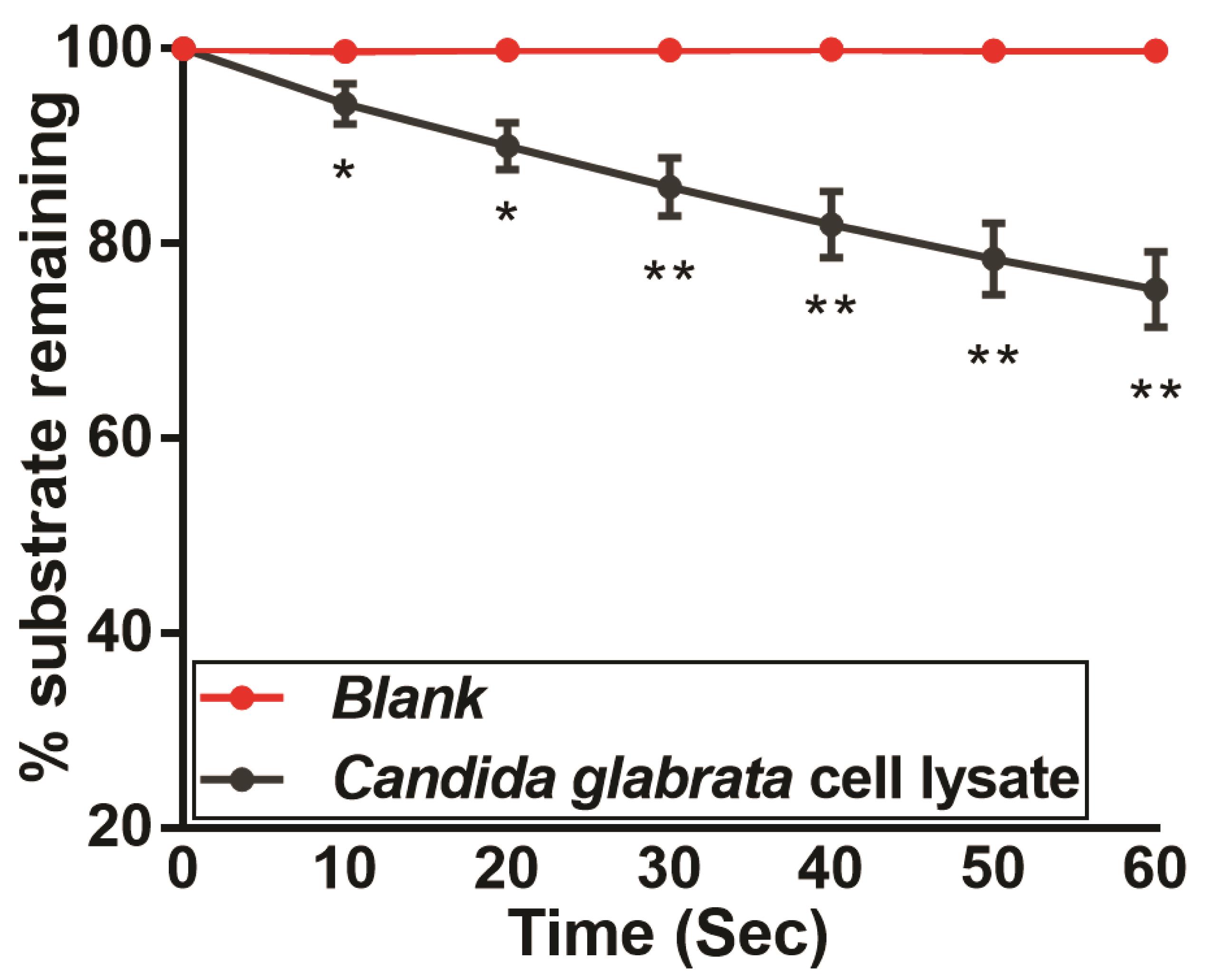
Figure 3. NADH:quinone oxidoreductase activity measurement in C. glabrata cell lysates, as measured using menadione and NADH. The C. glabrata wild-type strain BG2 was grown in the YPD medium to log-phase, followed by cell collection and lysis using glass beads. Cell lysate (500 μg) was incubated with menadione (500 µM) and NADH (500 µM), and absorbance was recorded at 340 nm over a period of 60 s. Absorbance of the substrate NADH was taken as 100 at 0 h time point, and NADH oxidation was calculated using the formula: [(absorbance at each time point/0 h absorbance) × 100]. Data represent mean ± SEM (n = 3). Asterisks indicate statistically significant differences in activity between the wild-type cell lysate and the blank sample, which contained all reagents except NADH. *, P < 0.0332; **, P < 0.0021; Grouped multiple t-test.
Notes
NADH and menadione solutions are to be freshly prepared just before use.
The HPLC-grade water was used to prepare reagents and buffer to avoid variations associated with water quality.
pH of the cell lysis buffer should be strictly maintained, as enzymatic reactions are standardized for a buffer of pH 7.4-7.5 only.
The NADH solution should be wrapped with aluminum foil to minimize exposure to light and stored at 4°C during the enzyme assay.
In case the assay is being performed with flavodoxin-like proteins purified from E. coli, add FMN (100 μM) or FAD (100 μM) as cofactors to the purified enzyme and incubate for 1 h at 4°C, followed by the addition of menadione and other reagents to the reaction mixture.
The NAD(P)H:quinone oxidoreductase activity can also be measured in cells treated with different quinones. Using this protocol, we have measured NAD(P)H:quinone oxidoreductase activity in log-phase menadione (90 µM)-treated C. glabrata cultures (Battu et al., 2021). Menadione treatment was given by adding 20 μl of menadione stock solution (50 mM) to 10 ml log-phase culture and incubating cultures with 200 rpm agitation for 90 min at 30°C. One culture aliquot was kept as untreated control, to which menadione was not added. Cultures were processed using the steps described above in the protocol [Menadione treatment can be replaced by exposure to any other stressor or growth of a mutant strain].
Recipes
Yeast extract-Peptone-Dextrose (YPD) medium (1 L)
YPD medium can either be prepared using BD DifcoTM YPD Broth (# 242810) or adding the following ingredients to sterile water, followed by autoclaving.
10 g Yeast extract
20 g Peptone
20 g Dextrose
Phosphate Buffer Saline (PBS) (1 L)
137 mM NaCl
2.7 mM KCl
10 mM Na2HPO4
1.8 mM KH2PO4
Dissolve 80 g of NaCl, 2 g of KCl, 14.4 g of Na2HPO4, and 2.4 g of KH2PO4 in water to prepare a 10× solution.
Adjust the final pH to 7.4 with HCl. Dilute 10× solution to 1× solution with distilled water.
Cell lysis buffer (100 ml)
50 mM Tris-HCl (pH 7.5) (50 ml of 1 M stock solution)
2 mM EDTA (133.3 µl of 1.5 M stock solution)
2% dextrose (4 ml of 50% stock solution)
10 mM Sodium fluoride (NaF)*
1 mM Phenylmethylsulphonyl fluoride (PMSF)*
1 mM Sodium orthovanadate (NaOV)*
1× Protease inhibitor cocktail*
*Inhibitor stock solutions can be prepared in advance and stored in small aliquots at -20°C. Thaw an aliquot and add to the lysis buffer just before use.
Quinone reductase activity assay buffer (10 ml)
10 mM Tris-HCl (pH 7.4) (100 µl of 1 M stock solution)
150 mM NaCl (300 µl of 5 M stock solution)
Make up the volume to 10 ml with HPLC grade water.
Acknowledgments
This work was fully supported by the DBT/Wellcome Trust India Alliance Senior Fellowship to RK (IA/S/15/1/501831; www.indiaalliance.org/). Research in the Kaur laboratory is also supported by grants from the Department of Biotechnology (BT/HRD/NBA/37/01/2014; http://www.dbtindia.gov.in/) and Science and Engineering Research Board, Department of Science and Technology (EMR/2016/005375; http://www.serb.gov.in/home.php), Government of India. AB is the recipient of a Research Fellowship of the Department of Biotechnology, New Delhi, India (http://dbtindia.gov.in/).
The protocol is derived from the activity assays previously described for E. coli.-purified proteins (Herrou et al., 2016). This protocol for NAD(P)H:quinone oxidoreductase activity in C. glabrata cell extracts is described in our recent publication (Battu et al., 2021).
Competing interests
The authors declare that they have no conflict of interests.
References
- Battu, A., Purushotham, R., Dey, P., Vamshi, S. S. and Kaur, R. (2021). An aspartyl protease-mediated cleavage regulates structure and function of a flavodoxin-like protein and aids oxidative stress survival. PLoS Pathog 17(2): e1009355.
- Bongomin, F. and Gago, S. (2017). Global and Multi-National Prevalence of Fungal Diseases-Estimate Precision. J Fungi 3(4): 57.
- Briones-Martin-Del-Campo, M., Orta-Zavalza, E., Cañas-Villamar, I., Gutiérrez-Escobedo, G., Juárez-Cepeda, J., Robledo-Márquez, K., Arroyo-Helguera, O., Castaño, I. and De Las Peñas, A. (2015). The superoxide dismutases of Candida glabrata protect against oxidative damage and are required for lysine biosynthesis, DNA integrity and chronological life survival. Microbiology(Reading) (2): 300-310.
- Brunke, S. and Hube, B. (2013). Two unlike cousins: Candida albicans and C. glabrata infection strategies. Cell Microbiol 15(5): 701-708.
- Carey, J., Brynda, J., Wolfova, J., Grandori, R., Gustavsson, T., Ettrich, R. and Smatanova, I. K. (2007). WrbA bridges bacterial flavodoxins and eukaryotic NAD(P)H:quinone oxidoreductases. Protein Sci 16(10): 2301-2305.
- Chakrabarti, A., Sood, P., Rudramurthy, S. M., Chen, S., Kaur, H., Capoor, M., Chhina, D., Rao, R., Eshwara, V. K. and Xess, I. (2015). Incidence, characteristics and outcome of ICU-acquired candidemia in India. Intensive Care Med 41(2): 285-295.
- Cuéllar-Cruz, M., Briones-Martin-del-Campo, M., Cañas-Villamar, I., Montalvo-Arredondo, J., Riego-Ruiz, L., Castaño, I. and De Las Peñas, A. (2008). High resistance to oxidative stress in the fungal pathogen Candida glabrata is mediated by a single catalase, Cta1p, and is controlled by the transcription factors Yap1p, Skn7p, Msn2p, and Msn4p. Eukaryot Cell 7(5): 814-825.
- Deller, S., Macheroux, P. and Sollner, S. (2008). Flavin-dependent quinone reductases. Cell Mol Life Sci 65(1): 141-60.
- Farías-Rico, J. A., Schmidt, S. and Höcker, B. (2014). Evolutionary relationship of two ancient protein superfolds. Nat Chem Biol 10(9): 710-715.
- Herrou, J., Czyż, D. M., Willett, J. W., Kim, H. S., Chhor, G., Babnigg, G., Kim, Y. and Crosson, S. (2016). WrpA Is an Atypical Flavodoxin Family Protein under Regulatory Control of the Brucella abortus General Stress Response System. J Bacteriol 198(8): 1281-1293.
- Heung, L. J., (2020). Monocytes and the Host Response to Fungal Pathogens. Front Cell Infect Microbiol 1034.
- Houwman, J. A. and van Mierlo, C. P. M. (2017). Folding of proteins with a flavodoxin-like architecture. FEBS J 284(19): 3145-3167.
- Kaur, R., Ma, B. and Cormack, B. P. (2007). A family of glycosylphosphatidylinositol-linked aspartyl proteases is required for virulence of Candida glabrata. Proc Natl Acad Sci U S A 104(18): 7628-7633.
- Koch, K., Hromic, A., Sorokina, M., Strandback, E., Reisinger, M., Gruber, K. and Macheroux, P. (2017). Structure, biochemical and kinetic properties of recombinant Pst2p from Saccharomyces cerevisiae, a FMN-dependent NAD(P)H:quinone oxidoreductase. Biochim Biophys Acta Proteins Proteom 1865(8): 1046-1056.
- Lamoth, F., Lockhart, S. R., Berkow, E. L. and Calandra, T. (2018). Changes in the epidemiological landscape of invasive candidiasis. J Antimicrob Chemother (1): i4-i13.
- Lodeyro, A. F., Ceccoli, R. D., Pierella Karlusich, J. J. and Carrillo, N. (2012). The importance of flavodoxin for environmental stress tolerance in photosynthetic microorganisms and transgenic plants. Mechanism, evolution and biotechnological potential. FEBS Lett 586(18): 2917-2924.
- Netea, M. G., Joosten, L. A., van der Meer, J. W., Kullberg, B. J. and van de Veerdonk, F. L. (2015). Immune defence against Candida fungal infections. Nat Rev Immunol 15(10): 630-642.
- Patridge, E. V. and Ferry, J. G. (2006). WrbA from Escherichia coli and Archaeoglobus fulgidus is an NAD(P)H:quinone oxidoreductase. J Bacteriol 188(10): 3498-3506.
- Pfaller, M. A., Diekema, D. J., Turnidge, J. D., Castanheira, M. and Jones, R. N. (2019). Twenty Years of the SENTRY Antifungal Surveillance Program: Results for Candida Species From. Open Forum Infect Dis (1): S79-S94.
- Rai, M. N., Balusu, S., Gorityala, N., Dandu, L. and Kaur, R. (2012). Functional genomic analysis of Candida glabrata-macrophage interaction: role of chromatin remodeling in virulence. PLoS Pathog 8(8): e1002863.
- Sancho, J., (2006). Flavodoxins: sequence, folding, binding, function and beyond. Cell Mol Life Sci 63(7-8): 855-864.
- Seider, K., Brunke, S., Schild, L., Jablonowski, N., Wilson, D., Majer, O., Barz, D., Haas, A., Kuchler, K., Schaller, M. and Hube, B. (2011). The facultative intracellular pathogen Candida glabrata subverts macrophage cytokine production and phagolysosome maturation. J Immunol 187(6): 3072-3086.
Article Information
Copyright
© 2021 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Battu, A., Purushotham, R. and Kaur, R. (2021). An Assay to Determine NAD(P)H: Quinone Oxidoreductase Activity in Cell Extracts from Candida glabrata. Bio-protocol 11(21): e4210. DOI: 10.21769/BioProtoc.4210.
Category
Microbiology > Microbial biochemistry > Protein > Activity
Microbiology > Microbial physiology
Biochemistry > Protein > Activity
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link