- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
A Co-purification Method for Efficient Production and Src Kinase-mediated Phosphorylation of Aplysia Cortactin
Published: Vol 11, Iss 18, Sep 20, 2021 DOI: 10.21769/BioProtoc.4158 Views: 3649
Reviewed by: David PaulDeepali BhandariSeda Ekici

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
In vitro Glutamylation Inhibition of Ubiquitin Modification and Phosphoribosyl-Ubiquitin Ligation Mediated by Legionella pneumophila Effectors
Alan G. Sulpizio [...] Yuxin Mao
Nov 5, 2020 3949 Views
Proteomics Profiling of S-sulfurated Proteins in Acinetobacter baumannii
Brenna J. C. Walsh and David P. Giedroc
May 5, 2021 5153 Views
Site-specific Incorporation of Phosphoserine into Recombinant Proteins in Escherichia coli
Phillip Zhu [...] Richard B. Cooley
Nov 5, 2022 3614 Views
Abstract
Cortactin is an actin-binding protein that regulates processes like cell migration, endocytosis, and tumor cell metastasis. Although cortactin is associated with actin-cytoskeletal dynamics in non-neuronal cells and cell-free systems, the exact mechanisms underlying its fundamental roles in neuronal growth cones are not fully explored. Recent reports show that Aplysia Src2 tyrosine kinase induces phosphorylation of cortactin as a mechanism to control lamellipodia protrusion and filopodia formation in cultured Aplysia bag cell neurons (He et al., 2015; Ren et al., 2019). In order to provide in vitro evidence for Src2-mediated phosphorylation of cortactin, we developed a robust and cost-effective method for the efficient expression and purification of Aplysia cortactin and Src2 kinase that can be used for biochemical studies including phosphorylation assays. By co-purifying cortactin and Src kinase with a phosphatase (YopH) from Yersinia enterocolitica, we eliminated the problem of non-specific phosphorylation of induced proteins by bacterial kinases and also reduced costs by bypassing the need for commercial enzymatic treatments. This protocol is reproducible and can be modified to produce homogenous non-phosphorylated proteins during recombinant protein expression in Escherichia coli.
Keywords: CortactinBackground
An organism’s nervous system is highly dynamic and enables cognition, growth, breathing, physical and emotional sensation, and other daily activities. The nervous system is comprised of a vast network of specialized cells, called neurons, that work together to transmit signals between different parts of the body (Abbasi et al., 2018). Each neuron has a cell body and extensions of different lengths. Shorter extensions, called dendrites, receive and transmit signals to the neuronal cell body (Lovinger, 2008; Reference 20). Longer extensions, called axons, carry electrical impulses away from the cell body to muscles, glands, and distant neurons (Lovinger, 2008). At the cellular level, nervous system function relies on precise wiring of these neurons, breaches to which lead to neurological disorders (Goodman and Shatz, 1993; Chédotal and Richards, 2010; Engle, 2010; Kolodkin and Tessier-Lavigne, 2011; Ren et al., 2019). Specialized extensions located at the tips of dendrites and axons, called neuronal growth cones, play a key role in ensuring proper axonal growth and guidance to establish functional connectivity (Lowery and Vactor, 2009; Vitriol and Zheng, 2012). Neuronal growth cones are highly motile sensory units that can detect extracellular cues and convert them into intracellular signals (Suter and Forscher, 2000; Dent et al., 2011). Growth cones use these signals to remodel cytoskeletal proteins, including F-actin, which constitute structures like lamellipodia and filopodia that are needed for motility. Thus, growth cones control axonal growth and guidance and the formation of neuronal circuitry. Therefore, elucidating the signaling mechanisms that dictate actin organization and dynamics during growth cone motility is imperative to understand the process of nervous system development and regeneration.
Cortactin is an actin-binding protein known to regulate cytoskeletal dynamics and cell migration in non-neuronal cells. Cortactin consists of an amino terminal acidic domain (NTA), followed by an F-actin-binding domain, an alpha-helical domain, a proline-rich region (PRR), and an SH3 domain at the C-terminus (Figure 1) (Ammer and Weed, 2008; Decourt et al., 2009). It was first characterized as a substrate for Src kinase and regulates several actin-related processes, including cell migration, tumor metastasis, and response to pathogens (Schnoor et al., 2018). Cortactin is important in the assembly, branching, and stabilization of cytoskeletal structures, such as lamellipodia and filopodia (Helgeson and Nolen, 2013; He et al., 2015). While cortactin is known to undergo multiple post-translational modifications, including phosphorylation (Schnoor et al., 2018; Ren et al., 2019) and acetylation, not much is known about the exact mechanisms underlying its fundamental roles in neuronal growth cones and filopodia formation in neurons.
Using cultured Aplysia bag cell neurons as a model, recent findings demonstrated that phosphorylation of a single tyrosine 499 residue of cortactin mediated by the Src2 kinase is important for the formation of filopodia and the regulation of actin organization and dynamics in growth cones (He et al., 2015; Ren et al., 2019). We have also demonstrated with purified proteins that Aplysia Src2 phosphorylates Aplysia cortactin in vitro (Ren et al., 2019). Here, we describe in detail the robust and cost-effective assay to assess the direct phosphorylation of cortactin by Src2 kinase used in our previous study (Ren et al., 2019). Using E. coli (BL21-DE3) cells as a host, we developed a reproducible two-step protocol to produce pure cortactin and Src kinase proteins at high yields.
Src kinases are phosphoryl transferases that transfer the γ-phosphate of ATP to tyrosine residues on specific substrate proteins. A common method used to detect kinase activity is a radioactive ATP kinase assay (Karra et al., 2017). This kinase assay protocol tracks the transfer of the radio-isotope 32P from ATP [γ-32P] to the substrate. Incorporation of this radiolabeled phosphate into the kinase substrate is then measured using autoradiography. Despite the sensitivity of this assay using optimized protocols with human Src and cortactin (Tehrani et al., 2007), we were unable to detect phosphorylation of Aplysia cortactin by Src kinase in vitro. Because bacteria also possess kinases, we reasoned and found to be true that our bacterially expressed and purified Src and cortactin proteins were non-specifically phosphorylated by bacterial kinases (Shrestha et al., 2012) during the purification process, therefore preventing Src2-mediated phosphorylation of cortactin. We circumvented these issues by co-purifying Aplysia Src2 and cortactin in the presence of a phosphatase called YopH (encoded by Yersinia enterocolytica) (Zhang et al., 1992), followed by sequential affinity and size exclusion chromatography, which yielded pure, unphosphosphorylated proteins that could be used in an in vitro kinase assay. By incorporating YopH into the purification scheme, we removed residual phosphate groups from the proteins that may have been introduced during the heterologous expression step. Here, we describe the co-purification process and present data obtained using this method, revealing the first direct biochemical evidence that Aplysia Src2 phosphorylates Aplysia cortactin and that Y505 (Cort-FYF) is preferentially modified in vitro, whereas Y499 is the essential tyrosine that is phosphorylated by Src2 in neuronal growth cones (see Figure 6). This procedure could be more generally adopted to remove non-specific phosphorylation of other proteins expressed in a prokaryotic host.
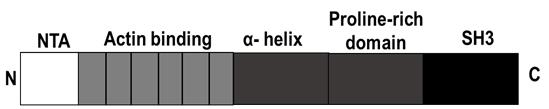
Figure 1. Schematic representation of the functional domain structure of Cortactin (human). N = N terminus, C = C-terminus, NTA = amino-terminal acidic region, SH3 = Src homology 3 domain. Schematics were created with Biorender.com.
Materials and Reagents
pSMT3 plasmid (Sanyal et al., 2015)
Ampicillin (AMRESCO, catalog number: 0339-100G)
Kanamycin (Fisher Bioreagents, catalog number: BP906-5)
Isopropyl p-Dthiogalactoside (IPTG) (Fisher Bioreagents, catalog number: BP1755-10)
Liquid Broth (LB) (Fisher Bioreagents, catalog number: BP1426500 )
Glycerol (Fisher Reagents, catalog number: BP229-4)
Phenylmethylsulfonyl fluoride (PMSF) (Sigma-Aldrich, catalog number: p7626-25g)
Amicon ultra-4 centrifugal filter unit (Sigma-Aldrich, catalog number: UFC 805024)
Liquid nitrogen
Escherichia coli (E. coli) BL21 (DE3) (Strategene, catalog number: 200131)
10% Mini-PROTEAN® TGXTM Precast Protein Gels (Bio-Rad Laboratories, catalog number: 456-1033)
Adenosine -5′-triphosphate [γ-32P] (Perkin-Elmer)
Coomassie Stain (AMRESCO, catalog number: 0472-25G)
Dithiothreitol (DTT) (Alfa Aesar, catalog number: A15797)
Sodium chloride (NaCl) (Fisher Scientific, catalog number: S271-10)
β-Mercaptoethanol (Sigma-Aldrich, catalog number: M3148-250ML)
Tris (Thermo Fisher Scientific, catalog number: BP152-5)
Imidazole (Sigma-Aldrich, catalog number: I2399-500G)
Eppendorf Microcentrifuge (Fisherbrand, catalog number: Q5-408-129)
Nitrocellulose membrane (Thermo Fisher Scientific, catalog number: 88018)
Monoclonal anti-polyhistidine antibody (Sigma-Aldrich, catalog number: H1029-5ML)
Triton X-100 (Sigma-Aldrich, catalog number: T9284-500ML)
Agarose (AMRESCO, catalog number: N605-500G)
Bacto agar (Beckmon, Dickson and Company, catalog number: 214010)
Magnesium chloride hexahydrate (Alfa Aesar, catalog number: 12288)
Manganese chloride tetrahydrate (Sigma-Aldrich, catalog number: M3634-500G)
Sodium orthovanadate (MP Biomedicals, catalog number: 159664)
[γ-32P] radioactive ATP (Perkin-Elmer, catalog number: BLU502A100UC)
Bovine serum albumin (Fisher Bioreagents, catalog number: BP1605-100)
Tween 20 ( Bio-Rad Laboratories, catalog number: 1706531)
IRDye® 680LT Goat anti-Mouse IgG Secondary Antibody (P/N: 926-68020)
Lambda protein phosphatase (New England Biolabs, catalog number: P0753S)
Recombinant proteins
WT Aplysia cortactin and single tyrosine mutants were subcloned as full-length N-terminal His6-SUMO fusion into pSMT3 expression vector using restriction enzyme cloning (Ren et al., 2019).
Note: Cortactin mutant proteins are indicated as YFF, FYF, and FFY where tyrosine was mutated to phenylalanine to test which of the three putative tyrosine residue (Y499, Y505, and Y509), respectively, in Aplysia cortactin is the preferred phosphorylation sites in vitro versus what was previously observed in vivo in neuronal growth cones. FFF is the triple tyrosine phosphorylation-defective mutant.
Src2 kinase (Src2) and the enzymatically inactive dominant negative Src2 (DNSrc2) cloned into pET-Duet with an N-terminal His6-tag (Ren et al., 2019).
GST-tagged, truncated YopH phosphatase missing 184 residues at the N-terminal and with an Arg in place of Ser at position 235 from Yersinia enterocolitica cloned in the plasmid pT7-7 (YopH) (Zhang et al.,1992); provided by Jack Dixon, University of California, San Diego.
Note: All plasmids are available upon request from the corresponding author.
Protein sample buffer for SDS-PAGE (made in house; see Recipes)
Liquid LB media
Solid LB plate
Stock preparation (see Recipes)
1 M IPTG
1,000× kanamycin stock
1,000× ampicillin stock
Buffer Preparation (see Recipes)
Lysis Buffer
Wash Buffer
Elution Buffer
SEC Buffer
Kinase Buffer
Sample loading buffer
10× Transfer Buffer
1× Tris-Buffered Saline, 0.1% Tween® 20 Detergent (TBST)
5% BSA
1× Transfer Buffer
Equipment
Note: Alternate yet equivalent equipment can be used.
4-L Erlenmeyer flask (Avantor VWR, catalog number: 10545-846)
UV/VIS spectrophotometer (Beckman Coulter, model: DU 350)
Refrigerated benchtop centrifuge (Beckman Coulter, model: ALLEGRA X-14R)
ÄKTA Purifier chromatography system (GE Healthcare, model: ÄKTA Pure)
High-speed centrifuge (Beckman Coulter, model: Avanti J-E)
Rotor capable of spinning 250 ml bottles (Nalgene, catalog number: 3120-1000)
Shaker incubator (New Brunswick, model: Innova®43) or an equivalent incubator that can be set at 37°C
Ice-water bath (VWR International, model: 1225PC)
Pipetman (Rainin)
Hiload 16/600 Superdex 200 PG column (GE Healthcare, catalog number: 28-9893-35)
PCR machine (Thermo Fisher Scientific, model: Mastercycler)
DNA agarose gel imaging equipment (UV transilluminator)
Sonicator (Branson Sonic Power Co., model: cell disruptor 350)
Odyssey imaging system (LI-COR, Biosciences, Lincoln, NE)
SDS-PAGE apparatus with power supply (Bio-Rad Laboratories)
Microwave
Bunsen burner
Typhoon phosphorimager (FLA 9500, GE Healthcare)
ImageQuant TL software (GE Healthcare)
Procedure
Transformation of plasmids into competent E. coli cells
Obtain wild-type (WT) or mutant plasmids and thaw freshly prepared aliquots of competent BL21(DE3) cells while on ice.
Add 2 µl (75-100 ng) of plasmid DNA to 50-100 µl of chemically competent cells.
Mix thoroughly by flicking each tube and then placing the tubes on ice for 10 min.
Transfer the tubes to a water bath set at 42°C for 45-60 s to ensure the uptake of plasmid DNA by E. coli cells.
Following heat shock, put the mixture on ice for 5 min and then add 800 µl of room temperature LB media.
Note: No antibiotic should be added to the media.
Incubate the tubes at 37°C while shaking for 1 h to allow the bacteria to express the antibiotic resistance proteins.
After 1 h, spread 50-100 µl of transformed cells onto solid LB media supplemented with antibiotics.
Allow colonies to grow by incubating the plates at 37°C overnight (Figure 2a).
Protein Expression and Purification
Transform kinase (Src2 or DNSrc2; pET-Duet), yopH (pT7-7) along with His6-SUMO fused WT cortactin or mutants (pSMT3) into E. coli BL21-DE3 competent cells as described above.
Note: Each pET-Duet, His6-SUMO fusion, and GST-fusion constructs must be transformed separately into the competent cells.
Pick a single colony of E. coli BL21 cells transformed with the expression plasmid and inoculate 10 ml of LB media containing 50 µg/ml kanamycin (for pSMT3) or 100 µg/ml ampicillin (for pT7-7 or pET-Duet) (Figure 2a). Incubate the bacterial culture overnight at 37°C while shaking.
The next day, transfer the 10 ml starter culture to 1 liter of LB media containing 50 µg/ml of kanamycin (pSMT3) or 100 µg/ml of ampicillin (pT7-7 or pET-Duet).
Note: 1 ml of overnight culture can be used to make 25% glycerol stocks and used in subsequent protein expression to save time and resources.
Allow the large cultures to grow while shaking at 200 rpm at 37°C to an optical density (OD600nm) between 0.6 and 0.9 (3-4 h), and then induce expression with IPTG to a final concentration of 0.4 mM (Figure 2b).
Grow induced cultures for an additional 4 h at 37°C while shaking.
To eliminate the phosphorylation of induced proteins by bacterial kinase, combine YopH phosphatase cultures with cultures overexpressing cortactin or kinase (Src2 or DNSrc2) in a 1:1 ratio and 1:2 ratio, respectively (Figure 2b).
Harvest the cells by centrifugation at 7500-8,000 × g at 4°C for 5-10 min.
Flash freeze and store the pellets at -80°C until needed.
Lysis and co-purification of pre-phosphorylated proteins with YopH phosphatase
Resuspend the bacterial pellets containing prephosphrylated proteins and YopH in 50 ml lysis buffer (see Recipes) supplemented with 5 mM PMSF protease inhibitor.
Lyse cells by sonicating on ice for 4 min with a 30% duty cycle (30 s with continuous setting and 1 min rest).
Transfer the lysate to a 50 ml centrifuge bottle. Isolate the soluble fraction by spinning at 27,000 × g for 35-40 min at 4°C (Figure 2c).
Load the clarified lysate onto an equilibrated gravity flow column filled with 4 ml of cobalt resin. To accomplish binding of the his6-tagged proteins to the cobalt resin, shake gently at 4°C for 1 h.
Wash the resin twice with 15-20 ml of wash buffer and then elute the protein of interest using elution buffer (see Recipes) stored at 4°C.
Assess the purity of protein via 10% SDS-PAGE.
To ensure cortactin purity, further subject the eluted cortain fraction to size exclusion chromatography (SEC) in SEC buffer stored at 4°C. Transfer the eluted protein onto a Hiload 16/600 Superdex 200 PG column using an ÄKTA pure system at a flow rate of 1 ml/min (Figure 4a and 4b).
Note: Wavelength at 280 nm can be used to monitor the protein elution profile and chromatograms recorded using UNICORN software.
Pool the peak fractions based on the SEC chromatogram and concentrate using an Amicon Ultra-4 concentrator (60 kDa cut-off) in a refrigerated benchtop centrifuge at 4°C.
Determine the total protein concentration spectrophotometrically at 280 nm and/ or by Bradford assay of the fractions and measuring the absorbance at 595 nm. The extinction coefficient of Aplysia cortactin is 55,810 M-1 cm-1, and its predicted molecular weight is 100 kDa.
Confirm the purity by 10% SDS-PAGE and western blotting (Figure 3a). Flash-freeze the proteins in aliquots and store at -80°C for further downstream experiments.

Figure 2. Experimental outline of recombinant protein expression and co-purification process for Cortactin. A flow chart of the overall experimental steps described in this procedure. Pre-phosphorylated proteins (Cortactin or Src) are co-purified in the presence of YopH phosphatase followed by sequential affinity and Size Exclusion Chromatography (SEC).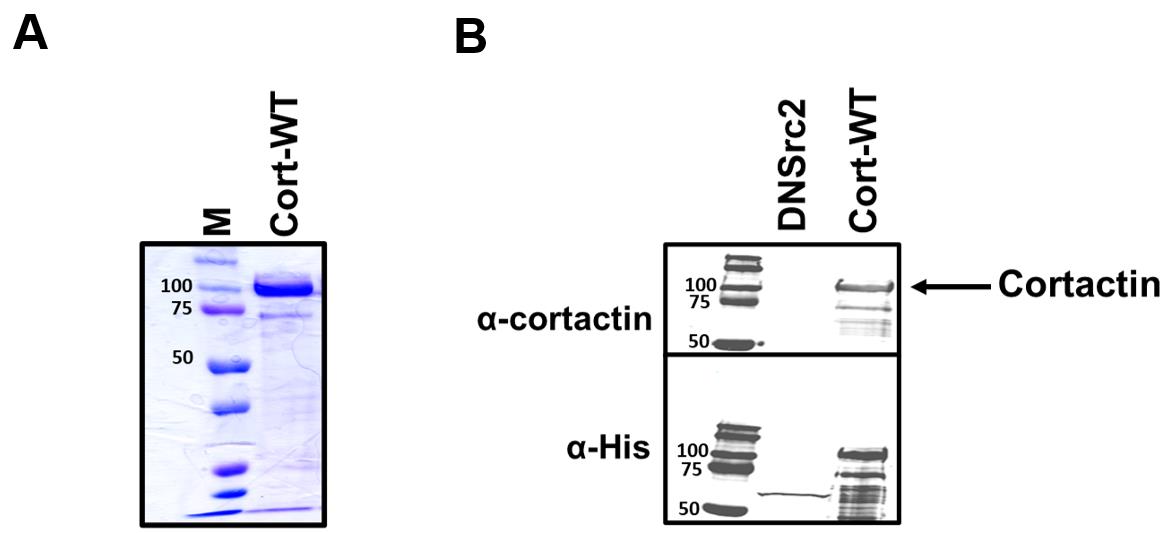
Figure 3. Recombinant expression of wild-type Aplysia cortactin and Src kinase in E. coli. (A) SDS-PAGE gel of affinity-purified WT cortactin co-purified with YopH phosphatase. The sample shows cortactin migrating as a predominant band corresponding to cortactin’s molecular weight of 95 kDa and fainter bands of lower molecular weight contaminants prompting the need for further purification by SEC. (B) Immunostaining of samples with 4F11 monoclonal cortactin antibody and monoclonal anti-His antibody confirmed the heterologous expression of Aplysia His6-tagged cortactin. His6-tagged dominant negative Src2 kinase (DNSrc2) was used as a control for both conditions.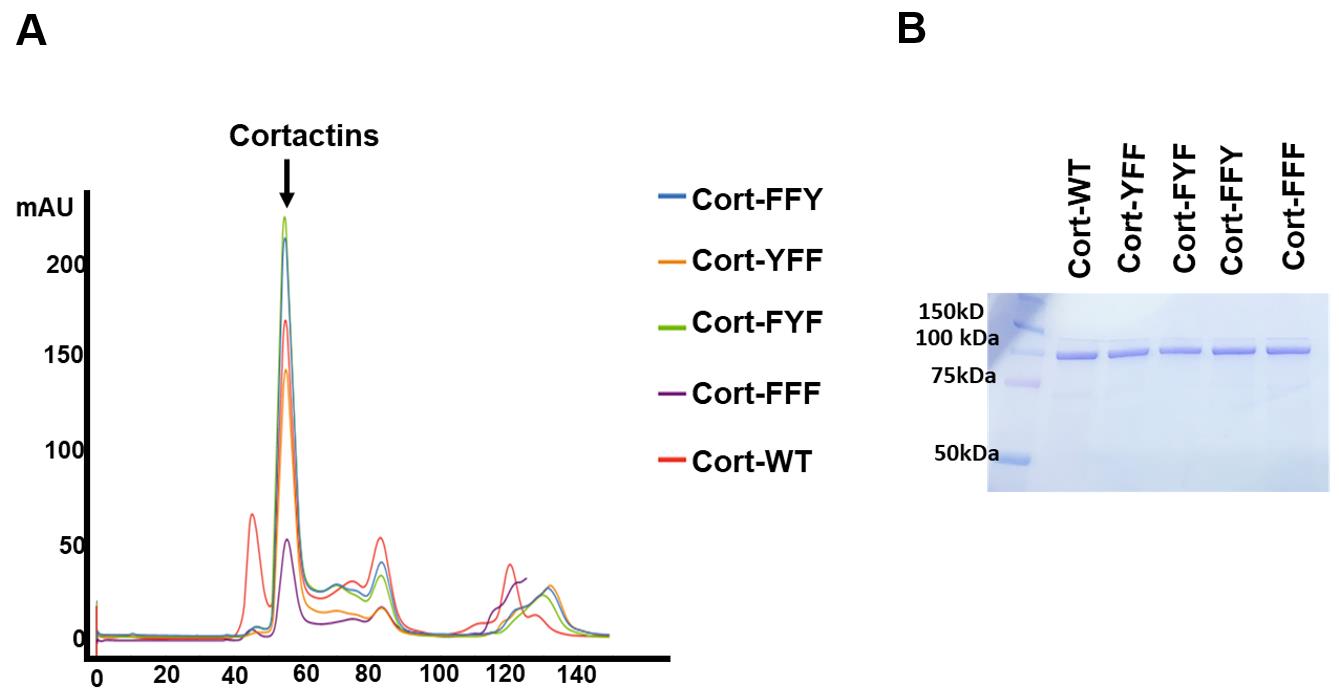
Figure 4. Purification profile of wild-type cortactin and mutants following Size Exclusion Chromatography (SEC). (A) Representative elution chromatogram of bacterially expressed and affinity-purified, recombinant WT and tyrosine phosphorylation mutants of cortactin (YFF, FYF, FFY, and FFF) (see Materials and Reagents section for details about mutants). The single peak at 55 ml elution volume corresponds to cortactin. Smaller peaks at 85 and 130 ml represent low molecular weight contaminants. WT cortactin also showed a second small peak at 45 ml, which may represent potentially oligomeric forms. (B) Coomassie-stained gel of WT and mutant cortactin proteins collected in the 55-ml elution volumes of the SEC peak in A. This figure is adapted from Figure 5A and 5B in our previous publication (Ren et al., 2019).
Verifying cortactin expression using a specific antibody
Obtain purified WT cortactin or mutant (pSMT3) and separate by SDS-PAGE on a 10% gel and then transfer to a nitrocellulose membrane.
Block the membrane with 5% bovine serum albumin dissolved in TBST (see Recipes) for 30 min at room temperature.
Probe the blots with the primary antibodies anti-cortactin (4F11) (Ren et al., 2019) or anti-His for 2 hours at room temperature.
After washing with TBST, incubate the membrane with goat anti-mouse 680 antibody for 30 min at room temperature.
Following extensive washing of the membrane with TBST, detect the signal using Odyssey imaging system (Figure 3B).
In vitro kinase assay to assess cortactin phosphorylation by Src2
Note: This procedure uses radioactive material, which should be handled with care. Consult the radiation guidelines of your institution to make sure you are authorized to work with radioactivity. Wear the proper personal protection equipment (PPE), keeping in mind exposure can cause deleterious effects.
Prepare the phosphorylation reaction by adding 2 µg of SEC co-purified His6-SUMO-tagged WT or mutant cortactin with 0.2 µg Src2 or DNSrc2. In addition, perform reactions with WT Src2 and DNSrc2 alone to assess autophosphorylation as a control (see Figure 5).
Incubate the proteins in kinase reaction buffer while on ice (20 µl aliquots).
Initiate the kinase reaction by adding 2.5 uCi [γ-32P] ATP. Incubate for 20 min at 30°C.
Quench the reaction by adding 5 µl of 4x SDS PAGE loading buffer.
Separate the samples on a 10% polyacrylamide gel, and image the sample by auto-radiograpy or by using the Typhoon phosphor-imager (Figures 5 and 6).
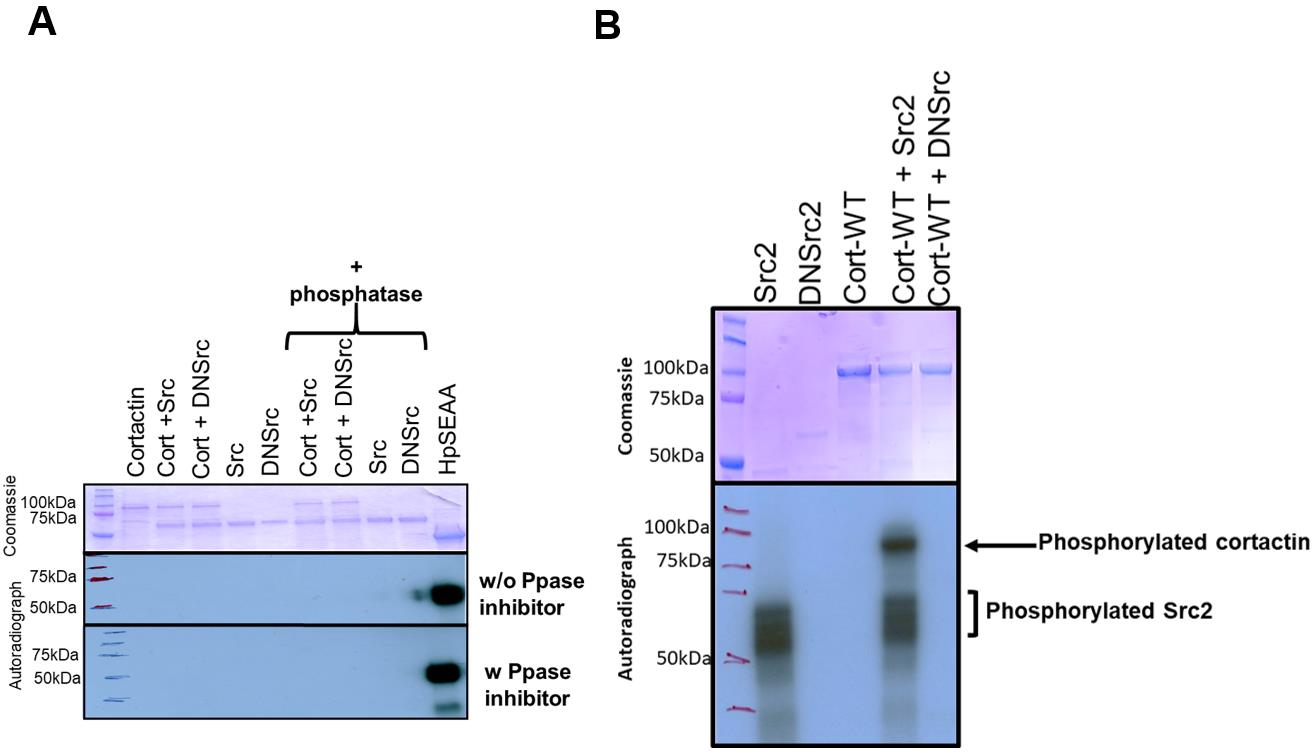
Figure 5. Treatment with YopH phosphatase allowed the functional characterization of cortactin as a target for Src2. (A) Non-specific phosphorylation inhibited the ability of Src2 to phosphorylate cortactin in vitro. Representative autoradiograph and corresponding Coomassie stained gel of an in vitro kinase reaction showing no phosphorylation signal when the WT cortactin is incubated with Src2 at 30°C. The reaction was also examined in the presence and absence of phosphotyrosyl phosphatase inhibitor, Na3VO4, while being treated with a commercially available phosphatase; the results showed no phospho-specific signal. A known bacterial kinase (hpSEAA) was used as a positive control; no inhibitor or phosphatase was used here. (B) Autoradiograph and corresponding Coomassie stained gel of a representative in vitro kinase reaction showing that co-purification of YopH eliminated residual ATP from the protein, thus showing that Aplysia cortactin is a direct substrate of Src2 kinase in vitro. Dominant negative Src2 (DNSrc2) failed to phosphorylate cortactin.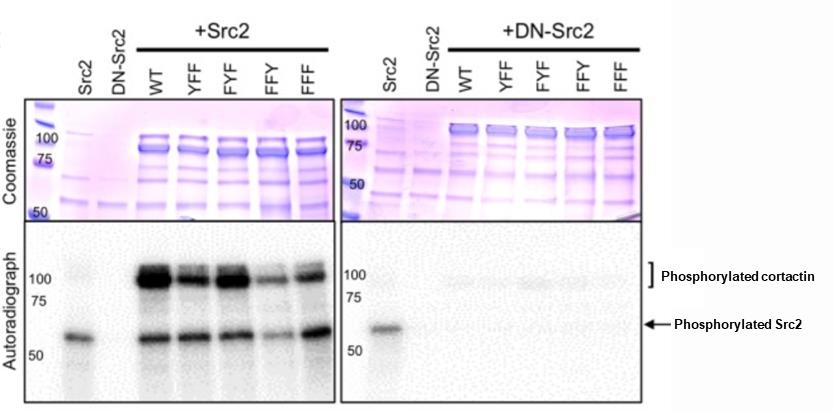
Figure 6. Co-purifying Aplysia proteins with YopH allowed the visualization of differences in phosphorylation pattern of cortactin by Src2 in vitro. Autoradiograph and corresponding Coomassie-stained gel of a representative in vitro kinase reaction showing the phosphorylation of WT or cortactin mutants by active Src2 or enzymatically dead dominant negative DNSrc2. Bands corresponding to phosphorylated Src2 (~65 kDa) and phosphorylated cortactin (95-110 kDa doublet) are indicated (Ren et al., 2019); see the Materials and Reagents section for detailed information about each construct. In contrast to living neurons, where phosphorylation of Y499 appears to have the most significant phenotypic effects, in vitro kinase data suggest that Y505 is preferentially phosphorylated. This figure is adapted from Figure 5C in our previous publication (Ren et al., 2019).
Recipes
Preparation of media for protein expression
Liquid LB media
Dissolve 20 g of LB broth into 1 L ultrapure water. Autoclave and allow the mixture to cool and then add the desired antibiotics.
Solid LB plate
Dissolve 15 g bacto-Agar (Becton Dickinson and Company) into 1 L LB broth. Once media is autoclave and cooled, add antibiotics and pour mixture onto petri dishes. Store at 4°C for future use.
Stock preparation
1 M IPTG (IsoPropyl-1-Thio-β-D-Galactopyranoside)
Weigh out 2.38 g of IPTG and dissolve in 10 ml of sterile water. Store at -20°C
1,000× kanamycin stock (50 mg/ml stock)
Weigh out 0.5 g kanamycin and dissolve it in 10 ml of sterile water. Store at -20°C
1,000× ampicillin stock (100 mg/ml stock)
Weigh out 1 g ampicillin and dissolve it in 10 ml of sterile water. Store at -20°C
Buffer Preparation
Lysis Buffer
50 mM Tris pH 8.0
300 mM NaCl
20 mM Imidazole
5 mM β-mercaptoethanol (BME)
0.5% Triton X-100
10% glycerol, store at 4°C
Wash buffer
50 mM Tris pH 8.0
300 mM NaCl
20 mM Imidazole
5 mM BME, store at 4°C
Elution buffer
100 mM Tris pH 8.0
150 mM NaCl
300 mM Imidazole
10% glycerol, store at 4°C
SEC Buffer
100 mM Tris pH 8.0
150 mM NaCl
Kinase Buffer
50 mM Tris pH 7.5
5 mM MgCl2
5 mM MnCl2
1 mM DTT
0.25 mM sodium orthovanadate (optional)
Sample loading buffer
14.3 M BME
1 M Tris-HCl pH 6.8
1.0 g SDS
0.1% brompophenol blue
4 ml 100% glycerol
Bring to 10ml with H2O
TBS Buffer
24.2 g Tris base
80 g NaCl
Dissolve in 1 L diH2O
10× Transfer Buffer
30.3 g Tris base
144 g glycine
Add 1 L H2O
1× Tris-Buffered Saline, 0.1% Tween® 20 Detergent (TBST)
100 ml 10× TBS
1 ml Tween 20
Add 900 ml H2O
5% BSA
Dissolve 2.5 g BSA in 50 ml 1× TBST buffer
1× Transfer Buffer
200 ml methanol
100 ml 10× transfer buffer
Add 700 ml H2O
Acknowledgments
S.M. was supported by a grant from NIH (R01-GM10092). S.L.B. was supported by National Institute Of Allergy And Infectious Diseases of the National Institutes of Health under Award Number T32AI148103 and a Bird Stair Graduate Student Fellowship from the Department of Biochemistry, Purdue University. D.M.S. was supported by a grant from the National Science Foundation (1146944-IOS) and grants from the Purdue Research Foundation.
Competing interests
The authors declare that no competing interests exist.
References
- Abbasi, N. A., Lafci, D. and Akan, O. B. (2018). Controlled Information Transfer Through An In Vivo Nervous System. Sci Rep 8(1): 2298.
- Ammer, A. G. and Weed, S. A. (2008). Cortactin branches out: roles in regulating protrusive actin dynamics. Cell Motil Cytoskeleton 65(9): 687-707.
- Chédotal, A. and Richards, L. J. (2010). Wiring the brain: the biology of neuronal guidance. Cold Spring Harb Perspect Biol 2(6): a001917.
- Decourt, B., Munnamalai, V., Lee, A. C., Sanchez, L. and Suter, D. M. (2009). Cortactin colocalizes with filopodial actin and accumulates at IgCAM adhesion sites in Aplysia growth cones. J Neurosci Res 87(5): 1057-1068.
- Dent, E. W., Gupton, S. L. and Gertler, F. B. (2011). The growth cone cytoskeleton in axon outgrowth and guidance. Cold Spring Harb Perspect Biol 3(3).
- Engle, E. C. (2010). Human genetic disorders of axon guidance. Cold Spring Harb Perspect Biol 2(3): a001784.
- Goodman, C. S. and Shatz, C. J. (1993). Developmental mechanisms that generate precise patterns of neuronal connectivity. Cell 72 Suppl: 77-98.
- He, Y., Ren, Y., Wu, B., Decourt, B., Lee, A. C., Taylor, A. and Suter, D. M. (2015). Src and cortactin promote lamellipodia protrusion and filopodia formation and stability in growth cones. Mol Biol Cell 26(18): 3229-3244.
- Helgeson, L. A. and Nolen, B. J. (2013). Mechanism of synergistic activation of Arp2/3 complex by cortactin and N-WASP. Elife 2: e00884.
- Karra, A. S., Stippec, S. and Cobb, M. H. (2017). Assaying Protein Kinase Activity with Radiolabeled ATP. J Vis Exp(123): 55504.
- Kolodkin, A. L. and Tessier-Lavigne, M. (2011). Mechanisms and molecules of neuronal wiring: a primer. Cold Spring Harb Perspect Biol 3(6): a001727.
- Lovinger, D. M. (2008). Communication networks in the brain: neurons, receptors, neurotransmitters, and alcohol. Alcohol Res Health 31(3): 196-214.
- Lowery, L. A. and Van Vactor, D. (2009). The trip of the tip: understanding the growth cone machinery. Nat Rev Mol Cell Biol 10(5): 332-343.
- Ren, Y., He, Y., Brown, S., Zbornik, E., Mlodzianoski, M. J., Ma, D., Huang, F., Mattoo, S. and Suter, D. M. (2019). A single tyrosine phosphorylation site in cortactin is important for filopodia formation in neuronal growth cones. Mol Biol Cell 30(15): 1817-1833.
- Sanyal, A., Chen, A. J., Nakayasu, E. S., Lazar, C. S., Zbornik, E. A., Worby, C. A., Koller, A., Mattoo, S. (2015). A novel link between Fic (filamentation induced by cAMP)-mediated adenylylation/AMPylation and the unfolded protein response. J Biol Chem 290(13):8482-99.
- Schnoor, M., Stradal, T. E. and Rottner, K. (2018). Cortactin: Cell Functions of A Multifaceted Actin-Binding Protein. Trends Cell Biol 28(2): 79-98.
- Shrestha, A., Hamilton, G., O'Neill, E., Knapp, S. and Elkins, J. M. (2012). Analysis of conditions affecting auto-phosphorylation of human kinases during expression in bacteria. Protein Expr Purif 81(1): 136-143.
- Suter, D. M. and Forscher, P. (2000). Substrate-cytoskeletal coupling as a mechanism for the regulation of growth cone motility and guidance. J Neurobiol 44(2): 97-113.
- Tehrani, S., Tomasevic, N., Weed, S., Sakowicz, R. and Cooper, J. A. (2007). Src phosphorylation of cortactin enhances actin assembly. Proc Natl Acad Sci U S A 104(29): 11933-11938.
- The principles of nerve cell communication. (1997). Alcohol Health Res World 21(2): 107-108. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6826821/.
- Vitriol, E. A. and Zheng, J. Q. (2012). Growth cone travel in space and time: the cellular ensemble of cytoskeleton, adhesion, and membrane. Neuron 73(6): 1068-1081.
- Zhang, Z. Y., Clemens, J. C., Schubert, H. L., Stuckey, J. A., Fischer, M. W., Hume, D. M., Saper, M. A. and Dixon, J. E. (1992). Expression, purification, and physicochemical characterization of a recombinant Yersinia protein tyrosine phosphatase. J Biol Chem 267(33): 23759-23766.
Article Information
Copyright
© 2021 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Brown, S. L., Ren, Y., Suter, D. M. and Mattoo, S. (2021). A Co-purification Method for Efficient Production and Src Kinase-mediated Phosphorylation of Aplysia Cortactin. Bio-protocol 11(18): e4158. DOI: 10.21769/BioProtoc.4158.
Category
Neuroscience > Cellular mechanisms > Protein isolation
Biochemistry > Protein > Posttranslational modification
Biological Sciences > Biological techniques
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link