- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Cortical Laminar Recording of Multi-unit Response to Distal Forelimb Electrical Stimulation in Rats
Published: Vol 11, Iss 22, Nov 20, 2021 DOI: 10.21769/BioProtoc.4153 Views: 3361
Reviewed by: Oneil Girish BhalalaZijian ZhangAndrew L. EagleAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Conditioned Lick Suppression: Assessing Contextual, Cued, and Context-cue Compound Fear Responses Independently of Locomotor Activity in Mice
Youcef Bouchekioua [...] Yu Ohmura
Dec 5, 2022 1666 Views
In situ Microinflammation Detection Using Gold Nanoclusters and a Tissue-clearing Method
Fayrouz Naim [...] Masaaki Murakami
Apr 5, 2023 2705 Views
A One-Step Mouse Model of Parkinson’s Disease Combining rAAV-α-Synuclein and Preformed Fibrils of α-Synuclein
Santhosh Kumar Subramanya [...] Poonam Thakur
Dec 5, 2025 1747 Views
Abstract
Severe traumatic brain injury (sTBI) survivors experience permanent functional disabilities due to significant volume loss and the brain’s poor capacity to regenerate. Chondroitin sulfate glycosaminoglycans (CS-GAGs) are key regulators of growth factor signaling and neural stem cell homeostasis in the brain. In this protocol, we describe how to perform recordings to quantify the neuroprotective and regenerative effect of implanted engineered CS-GAG hydrogel (eCS) on brain tissue. This experiment was performed in rats under three conditions: healthy without injury (Sham), controlled cortical impact (CCI) injury on the rostral forelimb area (RFA), and CCI-RFA with eCS implants. This protocol describes the procedure used to perform the craniotomy, the positioning of the cortical recording electrode, the positioning of the stimulation electrode (contralateral paw), and the recording procedure. In addition, a description of the exact electrical setup is provided. This protocol details the recordings in the brain of injured animals while preserving most of the uninjured tissue intact, with additional considerations for intralesional and laminar recordings of multi-unit response.
Graphic abstract:
Sensorimotor response to paw stimulation using cortical laminar recordings.
Background
Traumatic brain injury (TBI) is a common and increasingly prevalent problem that affects approximately 69 million people globally, without an effective treatment to date (Dewan et al., 2018). Given the failure of secondary neuroprotective strategies, such as decompressive craniotomy, or tight blood pressure regulation, in ameliorating poor functional outcomes, increased attention has turned to re-establishing damaged neuronal circuitry in the brain through biomaterial implants, with or without concurrent cell transplantation (Tan et al., 2020). Apart from providing the ability to inject any one of several natural or synthetic biomaterials into an injury lesion, it is unclear what functional effect these implants have on restoring native neuronal circuits and on higher-order cognitive and motor outcomes. In our manuscript (Latchoumane et al., 2021, DOI: 10.1126/sciadv.abe0207), we implanted rats with engineered chondroitin-sulfate glycosaminoglycans (eCS) as a potential treatment for the loss of tissue and consequent loss of motor function following TBI. To assess the physiological recovery promoted by eCS implants, we recorded the laminar cortical activity in response to electrical stimulation of the paw in anesthetized rats. Previous works in the field have used ex vivo planar multielectrode arrays on 300 µm brain slices to evaluate field excitatory postsynaptic potentials (fEPSPs) post-biomaterial implant (Yang et al., 2015; Hao et al., 2017). Other labs have performed steady-state evoked potentials (SSEP) recordings with chronically implanted electrodes in the mouse brain to measure brain responses post-implant (Fernández-García et al., 2016). We present a simple protocol that can be carried out in a single procedure, using a multichannel system recording and stimulation setup for the evaluation of the sensorimotor integration in animals implanted with eCS following TBI. Our protocol demonstrates the feasibility and reproducibility of recording perilesionnally to assess biomaterial integration, the impact of eCS on surrounding tissue, and the extent of live neuronal proliferation within and around the implant.
Materials and Reagents
Suture 4-0 Ethicon Absorbable plus antibacterial (Vicryl, catalog number: 109162)
Self tapping screws (18-8 Stainless Steel Slotted Flat Head Screws, M0.8 × 0.2 mm Thread, 2 mm Long; McMastercarr, catalog number: 91430A143)
Sterile cotton swabs, Cotton Tipped Applicators 6"/Sterile 100/box (Dynarex)
Stimulation needles, sterile stainless steel needle 24 gauge (BD, Microlance, catalog number: 1730738)
Animals: Sprague-Dawley Rats, male, age (7-10 weeks) (Charles River, catalog number: 400)
32 channel linear probe (Neuronexus, A1x32-6mm-50-177-CM32, 15 µm thickness, Gen4, lot# P994)
Ketamine 100 mg/ml (Coventrus, catalog number: 056347361-4)
Xylazine cocktail, 100mg/ml (Sigma-Aldrich, catalog number: 7361-61-7)
Isofluorane (Coventrus, catalog number: 029405 )
Buprenorphine, 0.03 mg/ml (Coventrus, catalog number: 059122)
Marcaine, 0.5% (Coventrus, catalog number: 054893)
Povidone-Iodine, 10% topical solution (CVS, catalog number: 59779-085)
Etch-Gel, phosphoric acid 40% (DMG, catalog number: 61901)
Gel Foam® (Pfizer, catalog number: 115631)
SeaKem® agarose (Lonza, catalog number: 50004)
Triple antibiotics, 0.9 g Pouch (25 ct. Box) (Safetec, catalog number: 53205)
Ketamine/Xylazine Cocktail (see Recipes)
Ketamine/Xylazine Cocktail:
For 1 ml solution: 0.9 ml of Ketamine (100 mg/ml) + 0.1 ml of Xylazine (100 mg/ml)
KX rat cocktail 0.1 ml/100g rat wt. IP (Ketamine: 90 mg/kg, Xylazine: 9.0 mg/kg)
Equipment
Multichannel acquisition system (MCS, Wireless Recording, model: W2100)
Multichannel recording and stimulation headstage (MCS, headstage, model: HS32-EXT0.5mA)
Dell PC i7, RAM:8Go, SSD: 500 GoPremium Silicone
Kopf Stereotaxic frame for rodent with manipulating arm
Electronic breadboard (Half-size breadboard; 63; adafruit.com)
Electronic cables (Covered Male-Male Jumper Wires, 200 mm × 40; Adafruit.com, catalog number: 4482)
MCS coaxial TTL cable (C-BNC-Lemo1m; Multichannel system accessories)
Electric Shaver (Philips, Norelco oneblade QP2520/90)
Electric Drill and trephine bur (Micromotor shiyang, H102S)
CCI tip, 3 mm diameter (Custom made)
CCI impactor machine (UGA workshop custom made)
Software
Acquisition Software: MCS experimenter (Multichannel Systems, https://www.multichannelsystems.com/)
System configuration: MCS IPconfig (Multichannel Systems, https://www.multichannelsystems.com/)
Data analysis: Matlab + MCS toolbox (Multichannel Systems, https://www.multichannelsystems.com/)
Matlab R2019b (Mathworks Inc., mathworks.com)
Procedure
System setup
The system setup is described in detail in Figure 1.
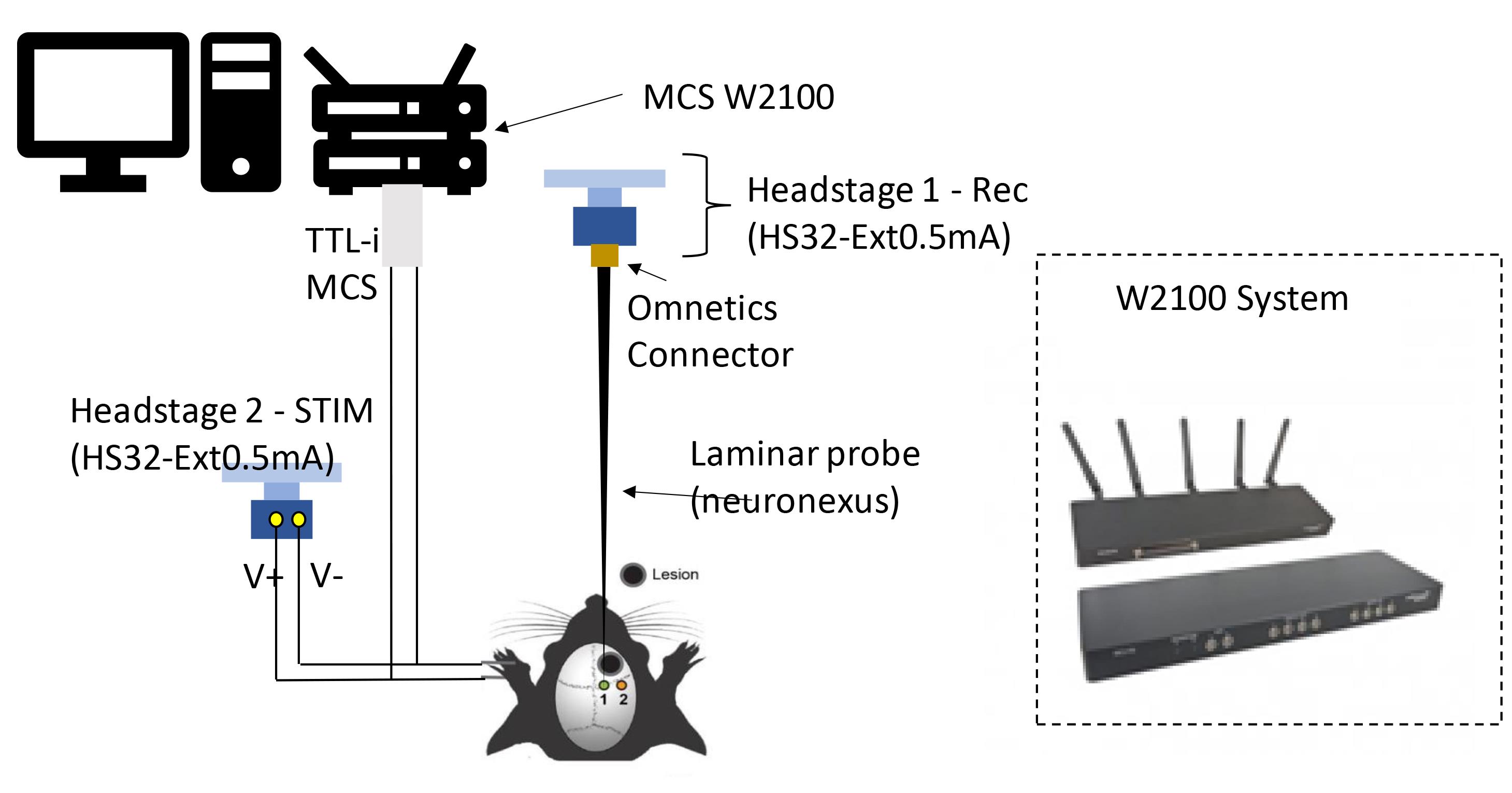
Figure 1. Setup for the recording of cortical activity with simultaneous electrical stimulation of the paw in the anesthetized rat. We used two battery-powered HS32-Ext0.5 mA headstages synchronized through the W2100 from multichannel systems (MCS). This system allowed the use of separate grounds for recording and stimulation, limiting the eStim-induced artefacts (i.e., electrical artifacts). The HS32 headstages can electrically stimulate in a bipolar setup while providing simultaneous recording through a 32-channel omnetics connector. The recording headstage was connected to a 32-lead linear silicone probe (Neuronexus). To obtain the exact time of stimulation, the stimulation headstage was connected to the paw of the animal and returned the voltage to the MSC. This system allowed a precisely synchronized stimulation and recording of events with neural response. We recorded from 2 regions (sequentially) in order to assess the laminar response of the S1 region and S1/M1 regions in the rat following paw stimulation.Controlled Cortical Injury (CCI) Surgical Procedure and eCS gel implant
Prior to CCI injury, anesthetize each animal with 5% isoflurane (switched to maintenance 2-3% as soon as the incision was performed – Step B4).
Inject buprenorphine (0.3 mg/ml, 0.05 ml/300g, Henry Schein) subcutaneously,
Place animals with their scalp shaved and sanitized (70% Ethanol and 3% povidone-iodine) on a stereotaxic frame attached to a temperature-controlled heating pad (37°C).
Perform a sagittal incision and clean the periosteum using Etch-gel (phosphoric acid etching, Henry Schein).
Perform a craniotomy using a 5-mm-diameter trephine bur fitted to an electric drill.
Fit a 3-mm CCI tip onto the pneumatic piston, position it in contact with the surface of the dura (fully extended position), and then retract it to adjust for an impact depth of 2 mm.
Launch the 3-mm CCI tip to create an impact. A severe CCI injury is induced by programming the piston speed to 2.25 m/s with a dwell time of 250 ms, resulting in an initial 3-mm-diameter injury with a depth of 2 mm.
Apply absorbable gelatin (Gel Foam®, Pfizer pharmaceutical) to the injury site, using sterile cotton swabs to remove excess blood.
Remove the gelatin and completely cover the injury site with a layer of 1% sterile agarose (SeaKem®, Lonza).
Suture skin flaps were together to close the wound. Apply triple antibiotic cream on the sutured skin.
Optional: For the eCS group, the eCS gel was implanted 48 h post-CCI using a pre-photocrosslinked gel delivered through a 32 gauge Hamilton syringe (speed: 20 μl/10min) in the center of the CCI lesion (1 mm depth).
Electrode placement surgical procedure (8 weeks post-CCI)
See Structure Denomination for brain region names.
Structure Denomination:
M1: primary motor cortex
RFA: rostral forelimb area, part of the M1 region for hand control
CFA: caudal forelimb area, part of the M1 region for hand and leg control
S1: primary sensory cortex
AP/ML/DV: stereotaxic positioning relative to the bregma point (reference); AP: anterior-posterior, ML: middle-lateral, DV: dorsal-ventral
From 8 to 10 weeks post-CCI, anesthetize rats that received RFA-targeted lesions using a ketamine/xylazine cocktail (100 mg/Kg) and place them on a stereotaxic frame.
Following sagittal scalp incision, perform a craniotomy (Figure 2) caudally to the injury (AP: 0 mm, ML: 2 to 4mm, relatively to bregma). Using a dental drill with trephine bur (2 mm diameter), draw a square shape within the skull, from the half width of the injury site down to the bregma (AP: 0 mm), with a width of 3-4 mm, and covering the entire lateral side (from the midline to the temporal cranial muscles). Then, lift and carefully remove the bone flap with fine forceps.
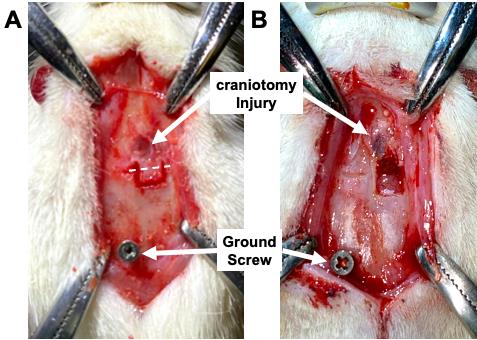
Figure 2. Position and description of the craniotomy post-lesion.Perform a durotomy to allow for the insertion of a 32-channel silicone probe (Neuronexus). Using the tip of a 1 ml insulin needle bent at 90° penetrate the dura tangentially to the brain to avoid any damage to the grey matter. Move the needle across the length of the craniotomy to tear the dura, and flip its pieces outside of the craniotomy region.
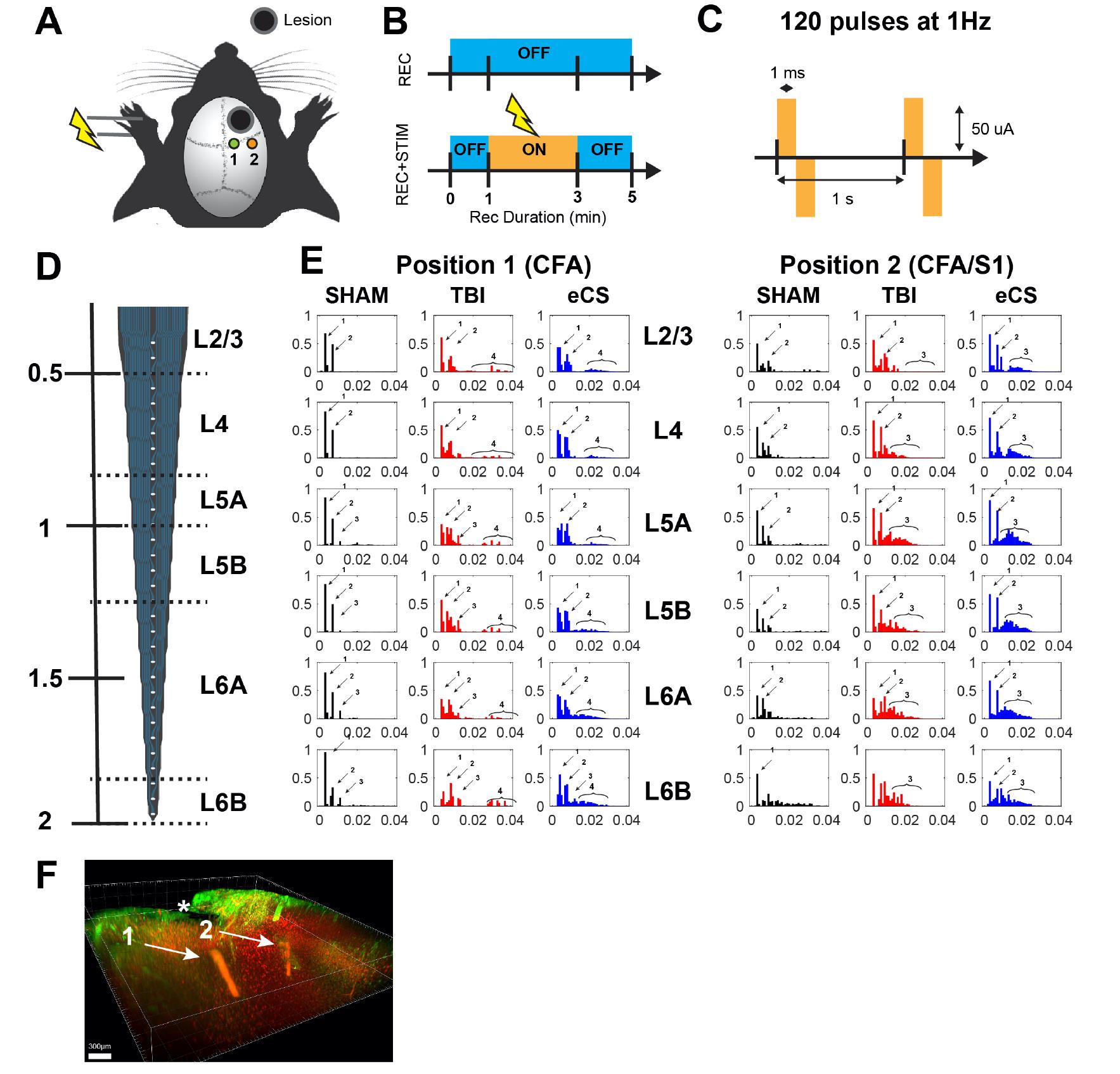
Insert the silicone probe (Figure 3, Video 1) in the motor area adjacent to the RFA (position 1: CFA, AP: 0 mm, ML: 2.5 mm) or in the sensorimotor area (position 2: S1, AP: 0 mm, ML: 3.5 mm) at a depth of 2 mm from the surface of the brain. Important: since the CCI was performed on the RFA, the CFA region should remain intact. If necrosis is observed in the CFA region, chances are that the recording will yield a low number of healthy unit spiking. Necrosis in the CFA/S1 region can be identified by the presence of liquified or brownish tissue with poor consistency, missing tissue volume, or dark tissue with a mix of coagulated and non-coagulated blood.

Figure 3. Poisitioning and setup of the recording electrode following craniotomy.
Video 1. General setup for the terminal recording of cortical activity following paw stimulation (somatosensory evoked response). (This video was made at the University of Georgia (UGA) according to guidelines from the IACUC of UGA under protocol # A2020-06-002.)Use the probe reference (top ground electrode) as the main reference, and a screw positioned above the left cerebellum (posterior, contralateral hemisphere) as ground. The probe reference is described in the main manufacturer website: https://www.neuronexus.com/files/technicalsupportdocuments/Chronic-Wiring-Configurations.pdf (see chronic probe reference description for internal reference).
Paw electrode placement procedure
Use a bipolar configuration for the paw stimulation. Use close positioning of the sink and source electrode to produce a short distance electrical stimulation, which results in a very localized electrical field excitation of the tissue. The bipolar configuration is the opposite of a monopolar configuration, where the sink and source are placed far apart, with the stimulation area becoming larger and less specific as a consequence.
Connect the anode and cathode of the stimulator to stainless steel needles.
Insert the two electrode tips at 2 mm from each other in the ventral section of the forelimb (palm of the paw). Then, place the paw and the entire forearm on the sterile pad.
Recording Procedure
Digitize and record neural data at 20 kHz (unit gain) using a multichannel system (MCS) W2100 acquisition module and a wireless headstage (HS32-EXT-0.5 mA, 16 bit; Figure 4A).
For spike analysis, filter the broadband electrophysiological through a real-time bandpass (300-5,000 Hz) and use a baseline pre-calculated threshold as trigger to save spike waveforms and spike event timestamps (threshold: 5 standard deviations). Do not sort for single-unit data; rather use multiunit spikes as a measure of population activity (Figure 4B, Video 2).
Perform all recordings for 5 min after stabilization for 10 min.
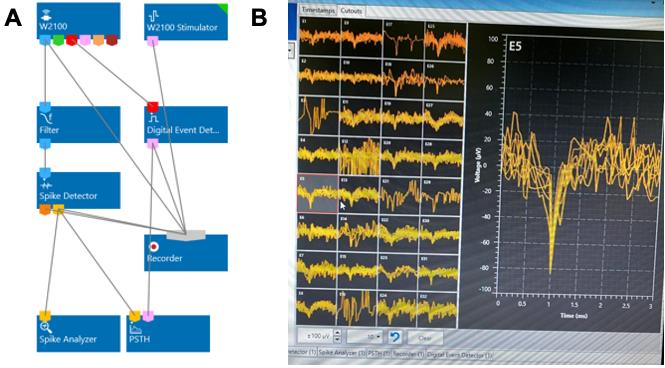
Figure 4. Recording setup using multichannel system software.
Video 2. Example of recording during ketamine anesthesia with laminar electrode.
Stimulation Protocols
Perform all paw stimulations using a single pulse (biphasic, on phase 1 ms) with a 1 s inter-pulse interval (1 Hz). Deliver the stimulation for a total of 120 pulses (2 min). Record continuously for 5 min and set the stimulation to start after 1 min of baseline and stop 2 min before the end of the recording, to obtain a return to baseline period.
Choose a stimulation intensity of 50 µA, as this induced a visible S1 response without the presence of artifacts due to electrical stimulation. Use two separate, battery-powered headstages to perform the stimulation on the paw and the recording of the electrode, allowing for independent grounding and low cross-electrical artifact formation.
Co-register each stimulation triggering to the recording system to match the real delivery of stimulation, rather than the estimated onset of the stimulation.
Data analysis
Analyze all data using custom-made Matlab® codes (Mathworks Inc.) and the provided Multichannel systems matlab toolbox. Upon reasonable request, the script used for analysis can be shared via email.
Record data from a 32-channel linear electrode, and group recorded channels according to the laminar organization as described in Figure S11D (abe0207 manuscript) orthe graphical abstract. Use the same channel grouping for all recordings, considering that the depth (2mm from the dura) and ground position (electrode ref/ground placed in layer 1) were placed similarly (Layer 1: none; Layer 2/3: ch1-3; Layer 4: ch4-10; Layer 5A: ch11-14; Layer 5B: ch15-19; Layer 6A: ch20-29; Layer6B: ch30-32). Data analysis included:
Stim-triggered histograms: use bin histograms to estimate the number of spikes elicited following the stimulation (Figure 5E; Figure S11E in Latchoumane et al., 2021).
From the average multiunit waveform (Figure 6A), estimate the spike wave width and peak-to-trough values to obtain the distribution of cell type (i.e., regular spiking units showing wider widths and lower amplitudes; fast spiking units showing shorter widths and larger amplitude).
Using the 5 min recording time separated into pre (1 min), STIM (2 min), and post (last 3 min), estimate the mean firing rate across channels in each layer. Compute the z-score using the mean and standard deviation of the firing rate during baseline (either a preceding recording without STIM or the pre-STIM period). This estimation can be obtained for each group (Sham, TBI, eCS) and each location (CFA and CFA/S1) (Figure 6B).
Results
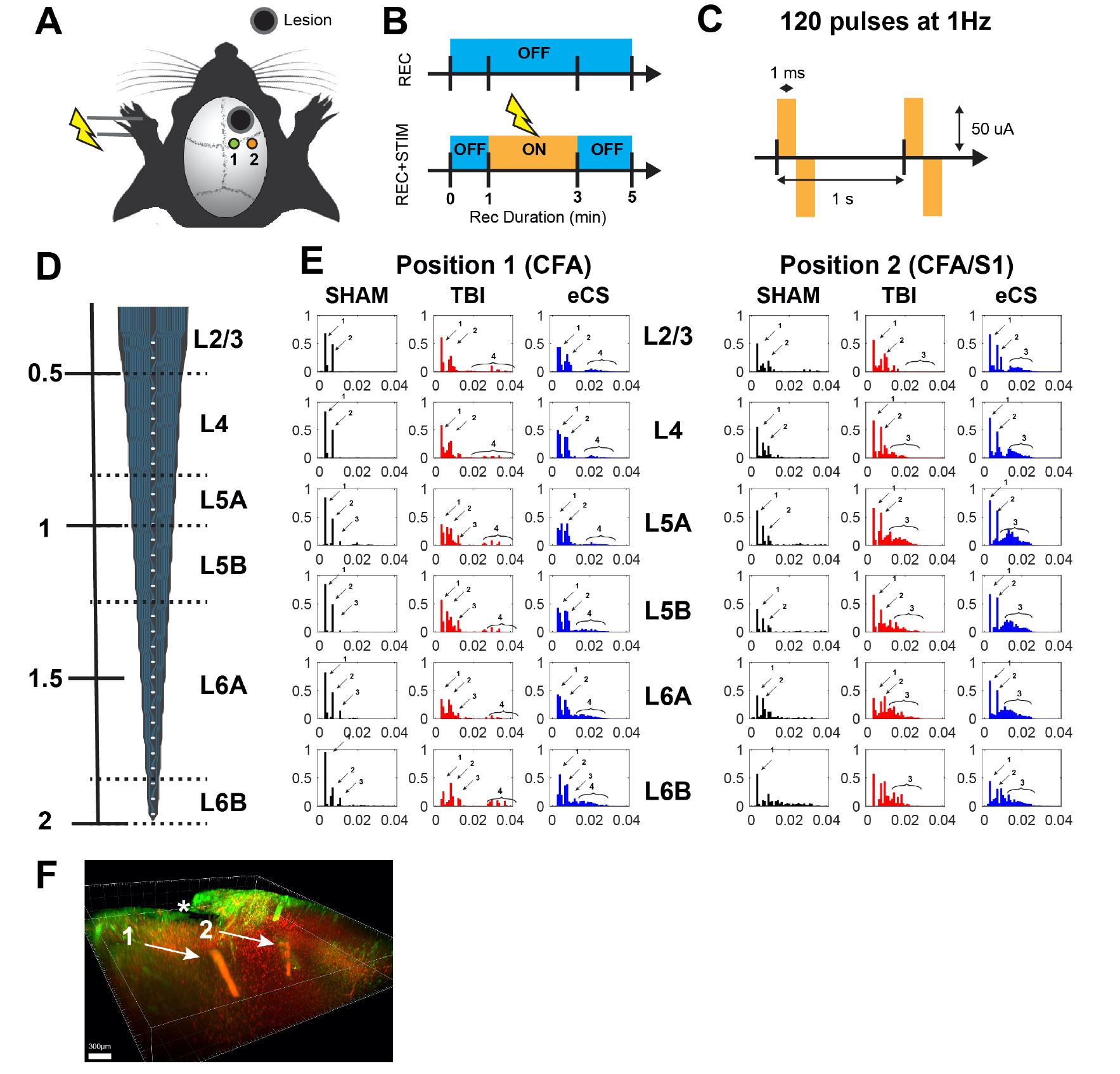
Figure 5. eCS matrix implants promote sensorimotor connectivity 10 weeks post-sTBI. A. Experimental schedule for recording session in ketamine anesthetized rats. To avoid disruption to the injury or eCS implant, use two positions for recording perilesionally for each rat. Position 1: CFA; Position 2: CFA/S1. B. Perform the recording for each position only after the probe insertion has been stabilized (top), followed by a stimulation session (2 min) with pre and post stimulation baseline recording of 1 and 2 min, respectively. Deliver 120 stimulation pulses for each position and each rat. C. The stimulation protocol used a bipolar pulse of 1 ms width (phase 1) at 1 Hz with an amplitude fixed at 50 µA for all rats. The stimulation amplitude was determined to minimize stimulation artifacts on recording while still eliciting sensorimotor response. Note: Perform stimulations and recordings using two separate wireless headstages (Multichannel Systems, W2100 HS32-Ext0.5mA) to guarantee ground and stimulation isolation from recording electrodes. Record each rat without (5 min) and with stimulation (5 min, out of which 2 min where stimulated) for the two positions CSA and CSA/S1. D. Use a 32 channel linear silicone electrode (iridium-iridium oxide, recording sites: 50 µm spacing) with a total span of approximately 1.6 mm. Perform implantation up to a depth of 2 mm from the surface of the pia. The layer position was based on the depth of the electrode and previously characterized layer distribution in the sensorimotor cortex. E. For the two recording positions, a treatment- and layer-dependent stimulation-locked response was observed. Following stimulation, a multimodal distribution typically revealed two major sharp peaks of neuronal activity (mono/di-synaptic; arrow indicating peaks 1 and 2) and later a response that revealed multi-synaptic activation of the area post-paw stimulation (arrow indicating peaks 3 and 4). Arrows indicate detected peaks of activity response, numbered in order of delay from stimulation start. F. Representative localization of the electrode positioning in a TBI rat brain, post-recording/stimulation. The arrow indicates the position of recording for position 1 and 2 shown in A. * indicates the position of the lesion. Scale bar: 300 µm.
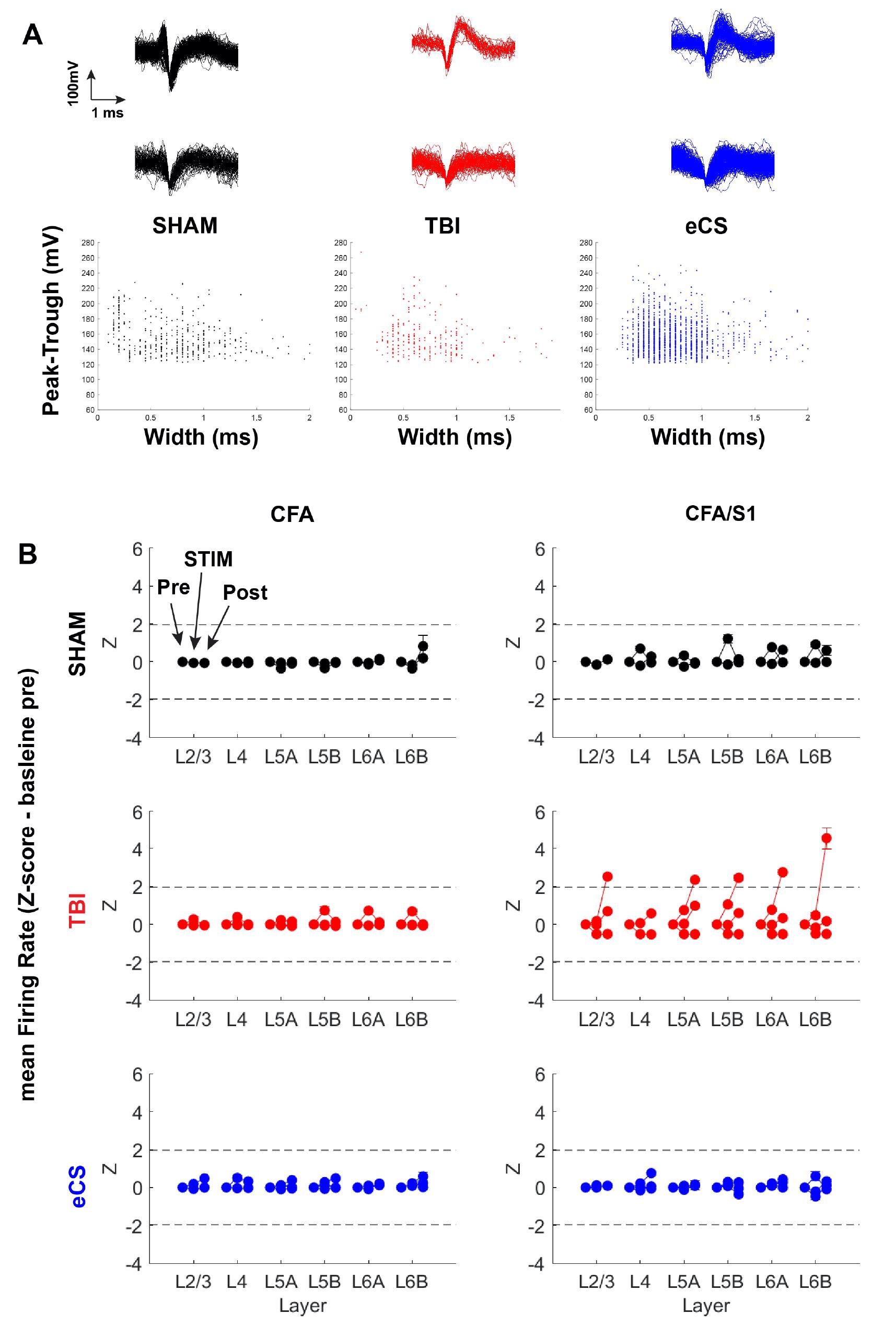
Figure 6. Characterization of multi-unit activity in all treatment groups using linear probe recording under ketamine anesthesia. A. Extract a multi-unit spike wave form during the resting period for each treatment group and for all recording sites. The top panel shows two representative waveforms for each treatment group, and the bottom panel shows the width vs. peak-trough length scatter plot for all detected multi-units. B. Derive a Z-score from the average population firing frequency normalized to the pre-stimulation period (1 min) for each treatment and the two recording positions CFA and CFA/S1/CFA. Note: the TBI group showed maladaptive sustained firing post stimulation for two rats out of three. Graphs show mean ± S.E.M.
Notes
Important consideration for reproducibility:
Grounding issue: consider the use of battery-based separate recording and stimulation systems to reduce the chance of recording noise, electrical stimulation-induced artifacts, and other electrical interactions that might affect the recording quality.
If using multi-shank silicone probes (not described in this protocol): Using probes that can span across the space and would be inserted once for the entire recording could be advantageous for this kind of recording. It would help reduce the total recording time, as well as provide simultaneous response for all layers and neighboring cortical regions involved in sensorimotor processing.
Positioning of the stimulation probes: As for every silicone probe recording, use a slow speed during the insertion of the electrode to avoid sheering damages.
Anesthetic depth: Perform recordings within the first 40-60 min of the ketamine-induced anesthesia to obtain the most stable data. Following a ketamine boost, the level of anesthesia might get more variable, leading to lower reproducibility. The recordings provided by the electrode are essentially local field potentials (LFP) with single neuron action potentials, which can be filtered out using 0-300Hz and 500-3000Hz bandpass filters. In particular, monitor the level of anesthesia using the LFP response of the cortical region, and include some of the delta-theta waves (0-4Hz brain waves) usually observed during the stable unconscious stage. Regularly monitor the animal response to tail and paw pinching to ensure the animal does not return to consciousness. This procedure should be performed during the recording, using the same setup as multi-unit recording.
Acknowledgments
This work was supported by an NIH (RO1NS099596) award to LK, and partially supported by a Georgia Partners in Regenerative Medicine seed grant from the Regenerative Engineering and Medicine (REM) research center to LK, and an Alliance for Regenerative Rehabilitation Research and Training (AR3T) technology development grant to LK and C-FL. Original work (Latchoumane et al., 2021), DOI: 10.1126/sciadv.abe0207.
Competing interests
There are no conflicts of interest or competing interests.
Ethics
All procedures on animals were approved by the Institutional Animal Care and Use Committee (IACUC). Protocols were performed in accordance with the Guide for the Care and Use of Laboratory Animals published by the National Institution of Health (NIH), protocol numerb A2020-06-002 (period 2020-2023).
References
- Dewan, M. C., Rattani, A., Gupta, S., Baticulon, R. E., Hung, Y. C., Punchak, M., Agrawal, A., Adeleye, A. O., Shrime, M. G. and Rubiano, A. M. (2018). Estimating the global incidence of traumatic brain injury. J Neurosurg 1-18.
- Fernández-García, L., Marí-Buyé, N., Barios, J. A., Madurga, R., Elices, M., Pérez-Rigueiro, J., Ramos, M., Guinea, G. V. and González-Nieto, D. (2016). Safety and tolerability of silk fibroin hydrogels implanted into the mouse brain. Acta Biomater 45262-275.
- Hao, P., Duan, H., Hao, F., Chen, L., Sun, M., Fan, K. S., Sun, Y. E., Williams, D., Yang, Z. and Li, X. (2017). Neural repair by NT3-chitosan via enhancement of endogenous neurogenesis after adult focal aspiration brain injury. Biomaterials 14088-102.
- Latchoumane, C. V., Betancur, M. I., Simchick, G. A., Sun, M. K., Forghani, R., Lenear, C. E., Ahmed, A., Mohankumar, R., Balaji, N. and Mason, H. D. (2021). Engineered glycomaterial implants orchestrate large-scale functional repair of brain tissue chronically after severe traumatic brain injury. Sci Adv 7(10): eabe0207.
- Tan, H. X., Borgo, M. P. D., Aguilar, M. I., Forsythe, J. S., Taylor, J. M. and Crack, P. J. (2020). The use of bioactive matrices in regenerative therapies for traumatic brain injury. Acta Biomater 1021-12.
- Yang, Z., Zhang, A., Duan, H., Zhang, S., Hao, P., Ye, K., Sun, Y. E. and Li, X. (2015). NT3-chitosan elicits robust endogenous neurogenesis to enable functional recovery after spinal cord injury. Proc Natl Acad Sci U S A 112(43): 13354-13359.
Article Information
Copyright
© 2021 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Readers should cite both the Bio-protocol article and the original research article where this protocol was used:
- Latchoumane, C. V., Forghani, R. and Karumbaiah, L. (2021). Cortical Laminar Recording of Multi-unit Response to Distal Forelimb Electrical Stimulation in Rats. Bio-protocol 11(22): e4153. DOI: 10.21769/BioProtoc.4153.
- Latchoumane, C. V., Betancur, M. I., Simchick, G. A., Sun, M. K., Forghani, R., Lenear, C. E., Ahmed, A., Mohankumar, R., Balaji, N. and Mason, H. D. (2021). Engineered glycomaterial implants orchestrate large-scale functional repair of brain tissue chronically after severe traumatic brain injury. Sci Adv 7(10): eabe0207.
Category
Biophysics > Electrophysiology
Neuroscience > Nervous system disorders > Animal model
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link










