- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Bioorthogonal Labeling and Chemoselective Functionalization of Lung Extracellular Matrix
Published: Vol 11, Iss 4, Feb 20, 2021 DOI: 10.21769/BioProtoc.3922 Views: 4421
Reviewed by: Scott LaughlinWilliam C. W. Chen

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Glucosidase Inhibition to Study Calnexin-assisted Glycoprotein Folding in Cells
Hao Wang and Qingyu Wu
Jun 5, 2019 5552 Views
Permethylation and Microfractionation of Sulfated Glycans for MS Analysis
Shin-Yi Yu [...] Kay-Hooi Khoo
May 20, 2020 4672 Views

Negative Ion Mode nanoLC-ESI-MS/MS Analyses of Permethylated Sulfated Glycans
Shin-Yi Yu [...] Kay-Hooi Khoo
May 20, 2020 4400 Views
Abstract
Decellularized extracellular matrix (ECM) biomaterials derived from native tissues and organs are widely used for tissue engineering and wound repair. To boost their regenerative potential, ECM biomaterials can be functionalized via the immobilization of bioactive molecules. To enable ECM functionalization in a chemoselective manner, we have recently reported an effective approach for labeling native organ ECM with the click chemistry-reactive azide ligand via physiologic post-translational glycosylation. Here, using the rat lung as a model, we provide a detailed protocol for in vivo and ex vivo metabolic azide labeling of the native organ ECM using N-Azidoacetylgalactosamine-tetraacylated (Ac4GalNAz), together with procedures for decellularization and labeling characterization. Our approach enables specific and robust ECM labeling within three days in vivo or within one day during ex vivo organ culture. The resulting ECM labeling remains stable following decellularization. With our approach, ECM biomaterials can be functionalized with desired alkyne-modified biomolecules, such as growth factors and glycosaminoglycans, for tissue engineering and regenerative applications.
Keywords: Extracellular matrixBackground
The extracellular matrix (ECM) is a hydrated network scaffold composed of non-cellular components from a given tissue or organ, and plays key roles in supporting the activities of residential cells through its contained bioactive elements, such as fibrous proteins, growth factors and glycosaminoglycans (GAGs) (Theocharis et al., 2016). Decellularized ECM materials have been widely used for tissue engineering due to their inherent biocompatibility, highly preserved tissue architecture and biomechanical properties (Ott et al., 2008 and 2010; Petersen et al., 2010; Wagner et al., 2014). Despite the importance, one critical barrier for applying decellularized ECM to regenerative applications is that the harsh decellularization conditions usually cause dramatic loss or denaturation of key bioactive components embedded within the ECM (Reing et al., 2010; Uhl et al., 2020). To overcome this challenge, emerging efforts are underway to functionalize ECM materials via the immobilization of desired bioactive molecules, such as growth factors and GAGs, to facilitate tissue repair and regeneration.
For ECM biomaterial functionalization, amine-reactive chemistry has been widely used, due to the wide availability of amines in almost all ECM proteins. However, such high abundance of amine ligands within the ECM leads to limited control over reaction specificity and poses potential risk of compromising the desired biochemical and biomechanical properties of ECM biomaterials (Wissink et al., 2001; Chiu and Radisic, 2010; Grover et al., 2012; Davidenko et al., 2015). Chemoselective engineering of ECM materials, via the specific conjugation between two ligands that do not exist in native biological systems, offers a promising solution to this challenge. The azide-to-alkyne click conjugation is one such chemistry that has been widely used for protein engineering and has demonstrated excellent biocompatibility for in vivo and in vitro applications (Laughlin and Bertozzi, 2007; Chang et al., 2010; Sletten and Bertozzi, 2011).
Here, we describe a metabolic labeling approach to covalently incorporate the azide ligand into organ ECM using an azido monosaccharide, azidoacetylgalactosamine-tetraacylated (Ac4GalNAz). Ac4GalNAz can be incorporated as post-translational glycan modification during new ECM protein synthesis both in vivo and during ex vivo organ culture. The resulting click-reactive azide ligands within the ECM remain stable following decellularization and enable further functionalization with desired biomolecules bearing the complementary alkyne ligand using the copper-catalyzed, azide-alkyne cycloaddition (Ren et al., 2018). The protocol described here, using the rat lung as a model, is expected to be applicable to label the ECM of a wide variety of organs, such as the heart, liver, kidney and blood vessel. We also expect our protocol to be applicable to engineer ECM biomaterials derived from other animal sources, such as the mouse and porcine models (Ren et al., 2018).
Materials and Reagents
27G × 1/2 tuberculin syringe (BD, catalog number: 305620 )
Reusable Stainless Steel Dispensing Needle with Luer Lock Connection, Blunt, 18 Gauge (McMaster Carr, catalog number: 6710A44 )
Dremel rotary tool kit (Dremel, catalog number: 8220 )
Luer-Lok Syringe 1 ml (BD, catalog number: 309628 )
Luer-Lok Syringe 10 ml (BD, catalog number: 302995 )
Luer-Lok Syringe 30 ml (BD, catalog number: 302832 )
Petri Dish, PS, 145/20 mm (Greiner Bio-One, catalog number: 639102 )
2-0 PERMA-HAND Silk Suture (Ethicon, catalog number: LA55G )
Sterile Gauze Sponges (Covidien, catalog number: 2187 )
PVDF Transfer Membrane (Thermo Fisher Scientific, catalog number: 88518 ), storage: RT
Sprague Dawley rats (100-250 gram) (Charles River Laboratories, Strain Code: 400)
N-azidoacetylgalactosamine-tetraacylated (Ac4GalNAz) (Click Chemistry Tools, catalog number: 1086 ), storage: -20 °C
Dimethyl Sulfoxide (DMSO) (Fisher Scientific, catalog number: BP231-100 ), storage: room temperature (RT)
Dulbecco′s Phosphate Buffered Saline (DPBS), 1× without calcium and magnesium (Corning, catalog number: 21-031-CV ), storage: RT
Dulbecco′s Modification of Eagle′s Medium (DMEM) (Corning, catalog number: 10-013-CV ), storage: 2-8 °C
FetalClone I Serum (Cytiva, catalog number: SH30080.03 ), storage: -20 °C
Antibiotic-Antimycotic (100×) (Thermo Fisher Scientific, catalog number: 15240062 ), storage: -5 °C to -20 °C
Sodium dodecyl sulfate (SDS) (Sigma-Aldrich, catalog number: 74255 ), storage: 2-8 °C
Triton X-100 (Sigma-Aldrich, catalog number: T8787 ), storage: RT
Phosphate Buffered Saline (PBS), 20× (Boston BioProducts, catalog number: BM-220 ), storage: RT
Paraformaldehyde (Sigma-Aldrich, catalog number: P6148 ), storage: 2-8 °C
Click-iT Cell Reaction Buffer Kit (Invitrogen, catalog number: C10269 ), storage: 2-6 °C
Biotin-PEG4-Alkyne (Click Chemistry Tools, catalog number: TA105 ), storage: -20 °C
Bovine Serum Albumin (BSA) (Fisher Scientific, catalog number: BP9706100 ), storage: 2-8 °C
Rabbit Polyclonal Laminin Antibody (Novus, catalog number: NB300-144 ), storage: -20 °C
Streptavidin, Alexa Fluor 647 conjugate (Thermo Fisher Scientific, catalog number: S21374 ), storage: -5 °C to -30 °C and protect from light
Donkey anti-Rabbit IgG (H+L) Secondary Antibody, Alexa Fluor Plus 488 (Thermo Fisher Scientific, catalog number: A32790 ), storage: 4 °C and protect from light
DAPI Fluoromount-G (SouthernBiotech, catalog number: 0100-20 ), storage: RT
Urea, ultrapure, 99% (Alfa Aesar, catalog number: J65769 ), storage: RT
HEPES (1 M) (Thermo Fisher Scientific, catalog number: 15630-080 ), storage: 2-8 °C
Protease Inhibitor Cocktail (100×) (Thermo Fisher Scientific, catalog number: 87786 ), storage: 4 °C
BCA Protein Assay Kit (Thermo Fisher Scientific, catalog number: 23225 ), storage: RT
4-15% Mini-PROTEAN TGX Precast Protein Gels (Bio-Rad Laboratories, catalog number: 4561085 ), storage: 4 °C
Tris Buffered Saline-Tween (TBST, 10×, with 0.5% Tween-20, pH 7.4) (Boston BioProducts, catalog number: IBB-181 ), storage: RT
High Sensitivity Streptavidin-HRP (Thermo Fisher Scientific, catalog number: 21130 ), storage: 4 °C
SuperSignal West Pico PLUS Chemiluminescent Substrate (Thermo Fisher Scientific, catalog number: 34580 ), storage: RT
SYPRO Ruby Protein Gel Stain (Thermo Fisher Scientific, catalog number: S12000 ), storage: RT
Ac4GalNAz Injection Solution (see Recipes)
Control Injection Solution (see Recipes)
Ex vivo Culture Medium (see Recipes)
Decell Solution (see Recipes)
2× Decell Solution (see Recipes)
1× PBS (pH 7.4) (see Recipes)
Fixation Solution (see Recipes)
Biotin-PEG4-Alkyne Click Reaction Mixture (see Recipes)
Urea Extraction Buffer (see Recipes)
1× TBST (pH 7.4) (see Recipes)
Blocking Buffer (see Recipes)
HRP-Imaging Mixture (see Recipes)
Equipment
Iris Scissors, 11.5 cm, straight (World Precision Instruments, catalog number: 503708-12 )
Pulmonary artery (PA) cannula (Figure 1)
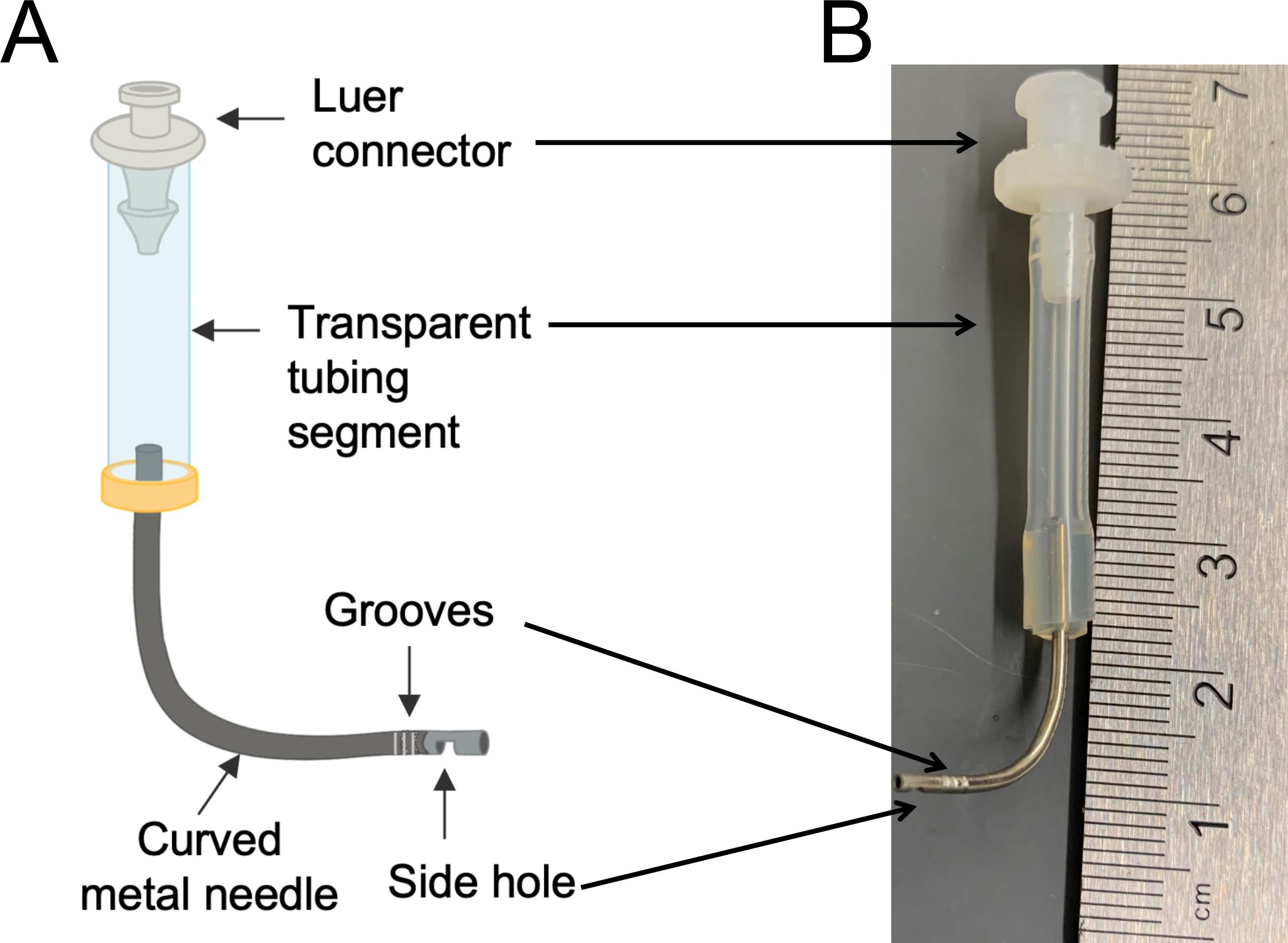
Figure 1. Custom-made PA cannula. A. Diagram. The cannula is composed of a top luer connector (1/8’, for connection with the bioreactor), a middle transparent tubing segment (1/8’, for detecting potential air bubbles), and a bottom curved metal needle (18 gauge, for connection with the PA). A small side hole at the end of the metal needle is generated using Dremel rotary tool with a sanding disc, to avoid potential perfusion blockage upon insertion into the PA. A series of grooves are created using a wire cutter around the tip of the metal needle to help stabilize the suture. B. An actual image of the cannula.Adson Forceps, straight, serrated (World Precision Instruments, catalog number: 503719-12 )
SILASTIC Thin-wall Silicone Laboratory Tubing (Dow Corning, catalog number: 508-009 )
Easy-Load Pump Head with 2-Channels for Precision Pump Tubing (Cole Parmer, catalog number: EW-77202-60 )
Masterflex L/S Digital Precision Modular Drive with Remote I/O and Benchtop Controller (Cole Parmer, catalog number: UX-07557-00 )
Autoclavable three-way valve (Cole Parmer, catalog number: EW-31200-80 )
Organ culture bioreactor (Figure 2)
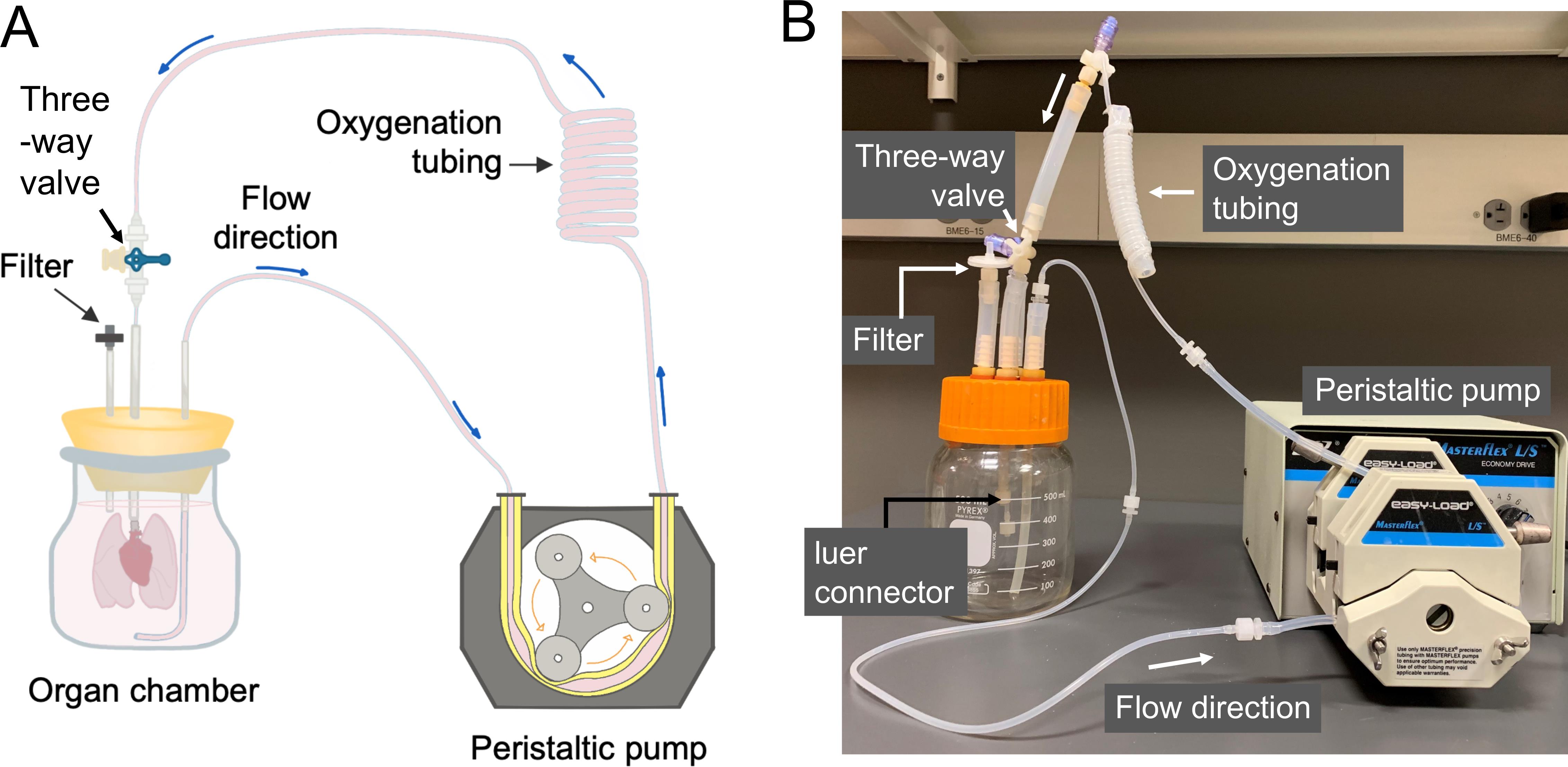
Figure 2. Organ culture bioreactor. A. Driven by a peristaltic pump, the culture medium is aspirated from the organ chamber, and perfuses through a series of thin-wall silicone tubing (for oxygenation) into the cannula leading to the PA of the lungs in culture. The filter on the organ chamber is for pressure equilibration. B. An actual image of the perfusion bioreactor.Decellularization chambers (Figure 3)
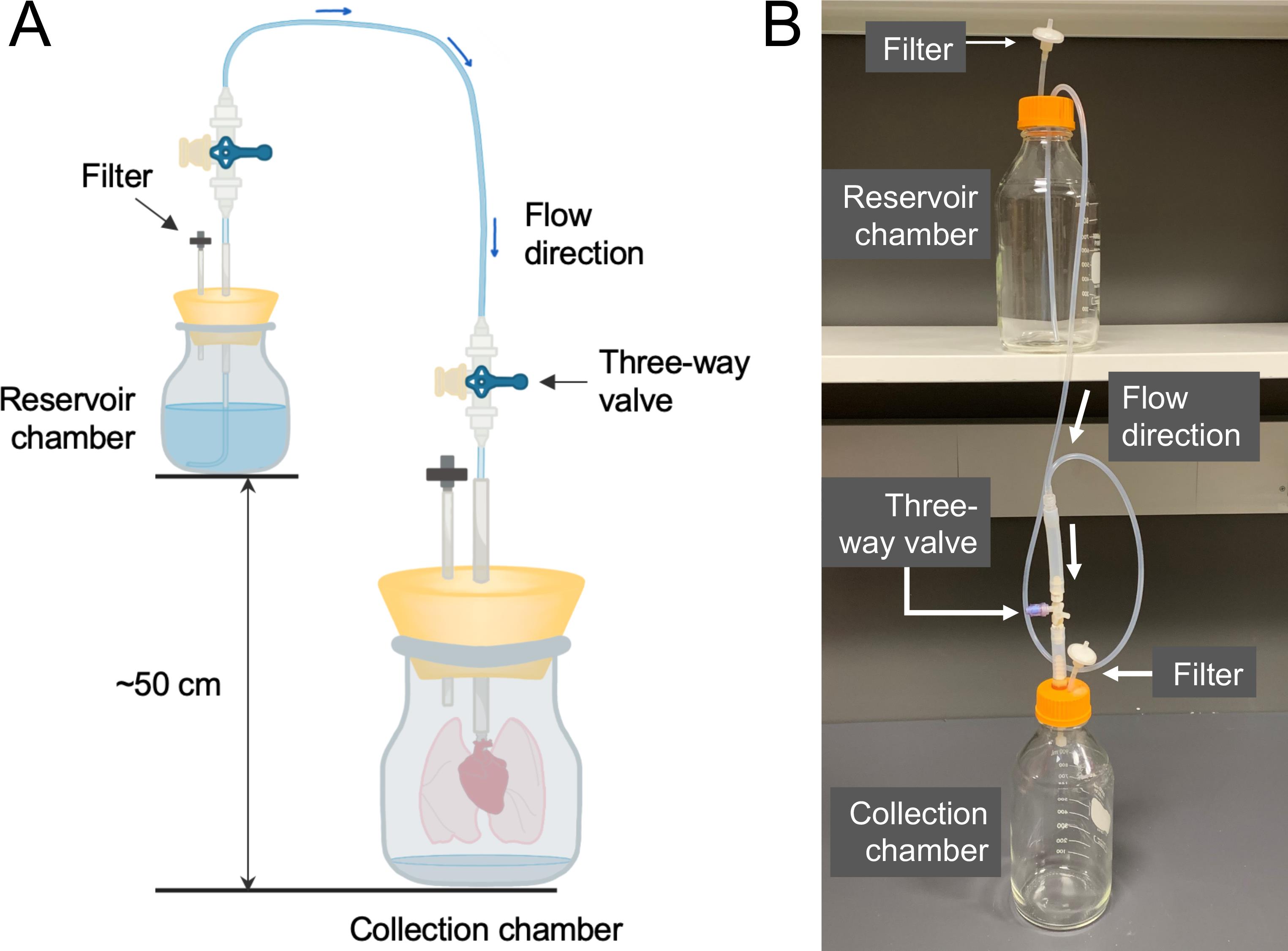
Figure 3. Decellularization chambers. A. There are two chambers in the system: the reservoir chamber containing fresh solutions for decellularization; and the collection chamber containing the lungs to be decellularized. The reservoir chamber should be placed approximately 50 cm higher than the collection chamber, which generates the gravity pressure to drive the fluid flow from the reservoir chamber into the lungs through the PA cannula. The filters on both chambers are for pressure equilibration. B. An actual image of the decellularization chambers.Semi-Automated Rotary Microtome (Leica Biosystems, catalog number: RM2245 )
EVOS FL Auto 2 Imaging System (Thermo Fisher Scientific, catalog number: AMAFD2000 )
Microcentrifuge (Eppendorf, catalog number: 5415R )
BeadBug Microtube Homogenizer (Benchmark Scientific, catalog number: D1030 )
Prefilled 2.0 ml Tubes, with Acid Washed Silica (Glass) Beads, 1.0 mm (Benchmark Scientific, catalog number: D1031-10 )
Mini-PROTEAN Tetra Vertical Electrophoresis Cell for Mini Precast Gels, with Mini Trans-Blot Module (Bio-Rad Laboratories, catalog number: 1658036 )
ChemiDoc Gel and Western blot imaging system (Bio-Rad Laboratories, catalog number: 12003153 )
Reach-in IR CO2 Incubator (Caron, catalog number: 7400-25-1 )
Procedure
In vivo metabolic labeling of the native organ ECM and lung harvest
Note: All animal procedures should be performed according to protocols approved by the Institutional Animal Care and Use Committee. Consultation and/or training is advised with a veterinarian or a researcher experienced with rat intraperitoneal injection, euthanasia, dissection, and blood vessel cannulation.
Day 1:
Weigh the rat and calculate the injection volume accordingly (0.3 mg of Ac4GalNAz per gram body weight). Use the Ac4GalNAz Injection Solution prepared at 70 mg/ml, the injection volume is 4.29 μl per gram body weight. For control rat, 4.29 μl of Control Injection Solution is administered per gram body weight.
Administer the proper volume of Ac4GalNAz or Control Injection Solution by intraperitoneal injection using a 1-ml syringe with a 27-gauge needle.
House each injected rat individually with the cage marked with the type of injection solution and the date(s) of injection(s).
Day 2, Day 3: Repeat Steps A1 to A3 for two additional days.
Sacrifice the rat via CO2 inhalation. Open up the thoracic cavity using sterilized surgical tools to expose the heart and lungs without damaging them.
Note: This procedure can be performed with beating or non-beating heart.
Make a small incision (2-3 mm) at the right ventricle immediately next to the pulmonary artery (PA) as the infusion inlet and cut open the left atrial appendage as the drainage outlet (Figure 4).
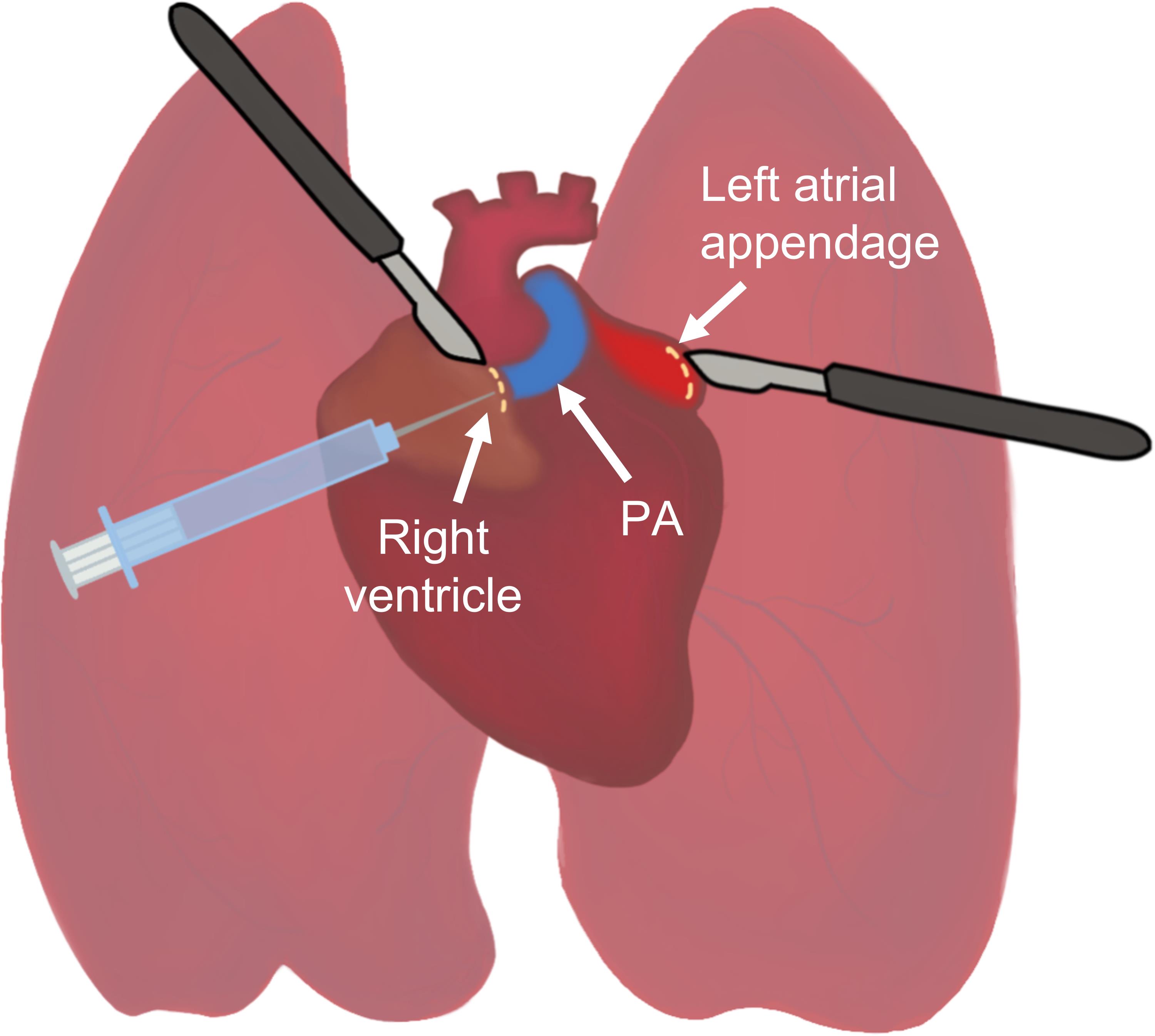
Figure 4. Surgical procedures during lung harvest. The PA (blue) is connected to the right ventricle. A small incision is made at the right ventricle immediately next to the PA to allow insertion of a blunt-end needle (for flushing) or cannula into the PA. Prior to the initial flushing, the left atrial appendage should be cut open as the drainage outlet.Prepare a syringe with 10 ml DPBS attached to an 18-gauge blunt-end needle. Insert the blunt-end needle gently into the PA via the small incision at the right ventricle, and use forceps to gently secure the needle in the PA. Infuse 10 ml DPBS over approximately 10 s to flush the remaining blood out of the pulmonary vasculature. Successful flushing is indicated by initial blood-colored fluid coming out of left atrial appendage, which gradually loses the blood color over the infusion course, and by the entire lungs turning white.
Note: The DPBS flushing step needs to be performed as soon as possible following euthanization of the animal to avoid blood clot formation in the pulmonary vasculature.
Isolate the heart-lung bloc free from surrounding tissues and place it in a Petri dish on top of a piece of gauze pre-wet with ice-cold DPBS.
To cannulate the PA, attach a custom-made cannula (Figure 1) to a 10-ml syringe pre-filled with DPBS, and insert the cannula into the PA via the small incision at the right ventricle (Figure 4) while having the DPBS slowly dripping out of the cannula by gentle syringe injection. This is to avoid accidental introduction of air bubbles into the pulmonary vasculature, which may cause air embolism and non-homogeneous perfusion. The insertion depth should be ~5 mm into the PA to ensure stable suturing. Following cannula insertion into the PA, secure the cannula at the grooves using the 2-0 silk suture. To test the cannulation, infuse DPBS through the PA cannula by gently pushing the syringe. Successful cannulation is indicated by low perfusion resistance and the expansion of the PA upon perfusion.
Ex vivo labeling of the lung ECM
Add 100 ml of pre-warmed Ex Vivo Culture Medium to the organ culture bioreactor (Figure 2), and pre-fill the entire perfusion tubing with the medium using a 30-ml syringe through the three-way valve to remove air bubbles. For metabolic ECM labeling, the Ex Vivo Culture Medium is supplemented with Ac4GalNAz (50 μM in 0.1% DMSO). For the control group, the medium is supplemented with 0.1% DMSO.
Harvest the heart-lung bloc from a rat without prior metabolic labeling, and cannulate the PA following Steps A5 to A9.
Note: Ex vivo lung culture should start within 1 h following organ harvest to ensure optimal metabolic activity of the organ to be labeled.
In a laminar hood, lift the lid of the organ culture bioreactor to allow access to the interior luer connector (Figure 2). Connect the lung cannula to the interior luer connector while having the medium slowly dripping out of the luer connector (by gentle medium injection using a syringe through the three-way valve) (Figure 2). This is to avoid introduction of air bubbles into the perfusion line. The use of one-time sterile gloves is highly recommended for this step to ensure sterility.
Note: This procedure requires the cooperation of two persons. One should gently push the medium-filled syringe while the other connect the lung cannula to the luer connector.
Attach the pump tubing of the organ culture bioreactor to a peristaltic pump and verify the perfusion direction (Figure 2). Culture the lungs in a humidified incubator supplemented with 5% CO2 for 24 h under constant-rate perfusion (5 ml/min for lungs harvested from a 100-gram rat and 10 ml/min for lungs harvested from a 200-gram rat).
Whole-lung Decellularization
Pre-fill the reservoir chamber with 1 L of Decell Solution and place it approximately 50 cm above the collection chamber (Figure 3). Pre-fill the tubing connecting between both chambers with the Decell Solution.
Attach the lungs (following in vivo or ex vivo metabolic labeling) to the luer connector inside the collection chamber while having the Decell Solution slowly dripping out of the luer connector. This is to avoid the introduction of air bubbles to the perfusion line.
Have the Decell Solution perfuse through the lungs driven by gravity pressure (determined by the height difference between the reservoir and collection chambers).
Note: During decellularization, the lungs should gradually expand in size and turn semi-transparent within the initial 5-10 min. Otherwise, it may suggest that the cannula or other parts of the perfusion line is blocked. The entire 1 L of Decell Solution should perfuse through the lungs over 1-2 h. If it takes much less time (such as less than 30 min), it may suggest damage in the lung or leakage in the tubing connection. If it takes much longer time (more than 2 h), it may suggest blockage in the perfusion line or lung vasculature by tissue debris or air bubbles, or it may be due to improper positioning of the cannula in the PA.
Pause the perfusion by turning one of the three-way valves in the perfusion line right before the fluid reservoir runs out. Discard the waste solution in the collection chamber and replace the reservoir chamber with 200 ml of H2O, and perfuse the lungs for 15 min.
Pause the perfusion, replace the reservoir chamber with 200 ml of 1% Triton X-100 in H2O, and perfuse the lungs for an additional 15 min.
Replace the reservoir chamber with 1 L of 1× PBS, and have it completely perfused through the lungs to wash away the detergents.
Repeat Step C6 for two additional times.
Histological characterization of metabolic ECM labeling
To fix the decellularized lungs, infuse 5 ml of the Fixation Solution into the trachea using a blunt-end needle connected to a 10-ml syringe, and ligate the trachea with the 2-0 silk suture. This is to open the alveolar structures for optimal histological assessment. Soak the lungs in a conical tube with an additional 15 ml of Fixation Solution and incubate at 4 °C overnight with gentle agitation.
Process the fixed lung tissues for paraffin embedding, and section the embedded tissue block at the thickness of 5 μm using a microtome.
Following standard deparaffinization and rehydration steps, wash the section with 1× PBS for 3 times, 5 min each.
To detect potential azide labeling in the decellularized lung ECM, perform biotin conjugation by adding the Biotin-PEG4-Alkyne Click Reaction Mixture to the histological section (100-200 μl per section), and incubate for 1 h at RT in a humidified staining chamber.
Wash the section with 1× PBS for 3 times, 5 min each.
Block the section with 1% BSA in 1× PBS for 20 min.
Incubate the section with primary Rabbit anti-Laminin antibody (1:500 dilution in 1% BSA in 1× PBS) at 4 °C overnight in a humidified staining chamber.
Wash the section with 1× PBS for 3 times, 5 min each.
Incubate the section with Streptavidin (Alexa Fluor 647 conjugate, 1:500 dilution in 1% BSA in 1× PBS) and Donkey anti-Rabbit-488 (1:500 dilution) for 45 min at RT.
Wash with 1× PBS for 3 times, 5 min each.
Mount the slide using Fluoromount and let it air dry.
Perform fluorescence imaging using the EVOS FL Auto 2 Imaging System (Figure 5).
Note: Standard immunofluorescence staining can be performed together with the detection of azide labeling by the click reaction. In such dual staining, it is recommended to perform the Biotin-PEG4-Alkyne click conjugation first, followed by standard antibody staining. As an example, we performed a co-staining of Laminin.
Biochemical characterization of metabolic ECM labeling
Cut a piece of the un-decellularized lung tissue (from in vivo or ex vivo metabolic labeling) and mince it using fine scissors.
Note: For biochemical detection of azide labeling in the ECM, 50-100 mg of lung tissue is usually sufficient.
Transfer the minced lung tissue into a 2.0-ml tube prefilled with glass beads, add 300 μl H2O, and homogenize on a microtube homogenizer at full speed (4,000 rpm) for 3 min. Repeat this homogenization step if large tissue pieces remain visible.
Add 300 μl of 2× Decell Solution to the tube, and agitate at RT for 30 min. This step will solubilize all cellular components in the homogenized lung tissue.
Centrifuge the tube at 16,100 × g for 20 min at RT. Discard supernatant.
Add 1 ml DPBS to the tube, mix thoroughly by inversion, centrifuge at 16,100 × g for 10 min at RT. Discard supernatant. This is to wash away any remaining cellular components.
Repeat Step E5 for two additional times.
Add 500 μl of the Biotin-PEG4-Alkyne Click Reaction Mixture to the tube, mix thoroughly, and incubate for 1 h at RT with agitation. This is to conjugate biotin to the potential azide ligand in the decellularized lung ECM.
Centrifuge the tube at 16,100 × g for 10 min at RT. Discard supernatant.
Add 1 ml DPBS to the tube and mix thoroughly. This is to wash away any remaining Biotin-PEG4-Alkyne and other click reaction reagents.
Centrifuge the tube at 16,100 × g for 10 min at RT. Discard supernatant.
Repeat E9 and E10 for two additional times.
Add 500 μl of Urea Extraction Buffer to the tube, and gently agitate for 2 h at RT.
Centrifuge the tube at 16,100 × g for 10 min at RT.
Collect the supernatant as the ECM protein sample.
Quantify the ECM protein concentration using BCA assay following manufacturer’s protocol with BSA as the protein standard.
Perform SDS-PAGE electrophoresis with equal protein loading from each sample, such as ECM protein samples extracted from lungs with and without metabolic azide labeling, and transfer proteins from the gel onto a piece of PVDF membrane blot. Block the blot for 20 min using the Blocking Buffer, incubate the blot in 10 ml of Streptavidin-HRP (1:10,000 dilution in Blocking Buffer) with agitation for 1 h at RT, and wash the blot thoroughly with TBST for 3 times, 10 min each.
Incubate the blot in the HRP-imaging Mixture for 3 min, and image using ChemiDoc Gel and Western Blot Imaging System (Figure 6A).
To validate the equal protein loading from all samples, perform SDS-PAGE electrophoresis, and stain total protein in the gel using Sypro Ruby Protein Gel Stain following manufacturer’s protocol. Image the Sypro Ruby-stained gel using ChemiDoc Gel and Western Blot Imaging System (Figure 6B).
Data analysis
Histological characterization of metabolic azide labeling: On a histological section of the decellularized lung tissue, the potential azide ligand was conjugated to biotin using the click chemistry. Streptavidin staining of biotin and Laminin staining of the ECM was imaged using two different fluorophores. Laminin staining served as a control to verify similar ECM content being present in the lung ECM with and without metabolic azide labeling. Care should be taken to ensure consistent imaging parameters for all samples under comparison. Adjust exposure time according to the sample with the strongest fluorescence signal. Compared to the control, the lung ECM derived from the rat receiving Ac4GalNAz metabolic labeling is expected to show more robust azide labeling and biotin conjugation (Figure 5).
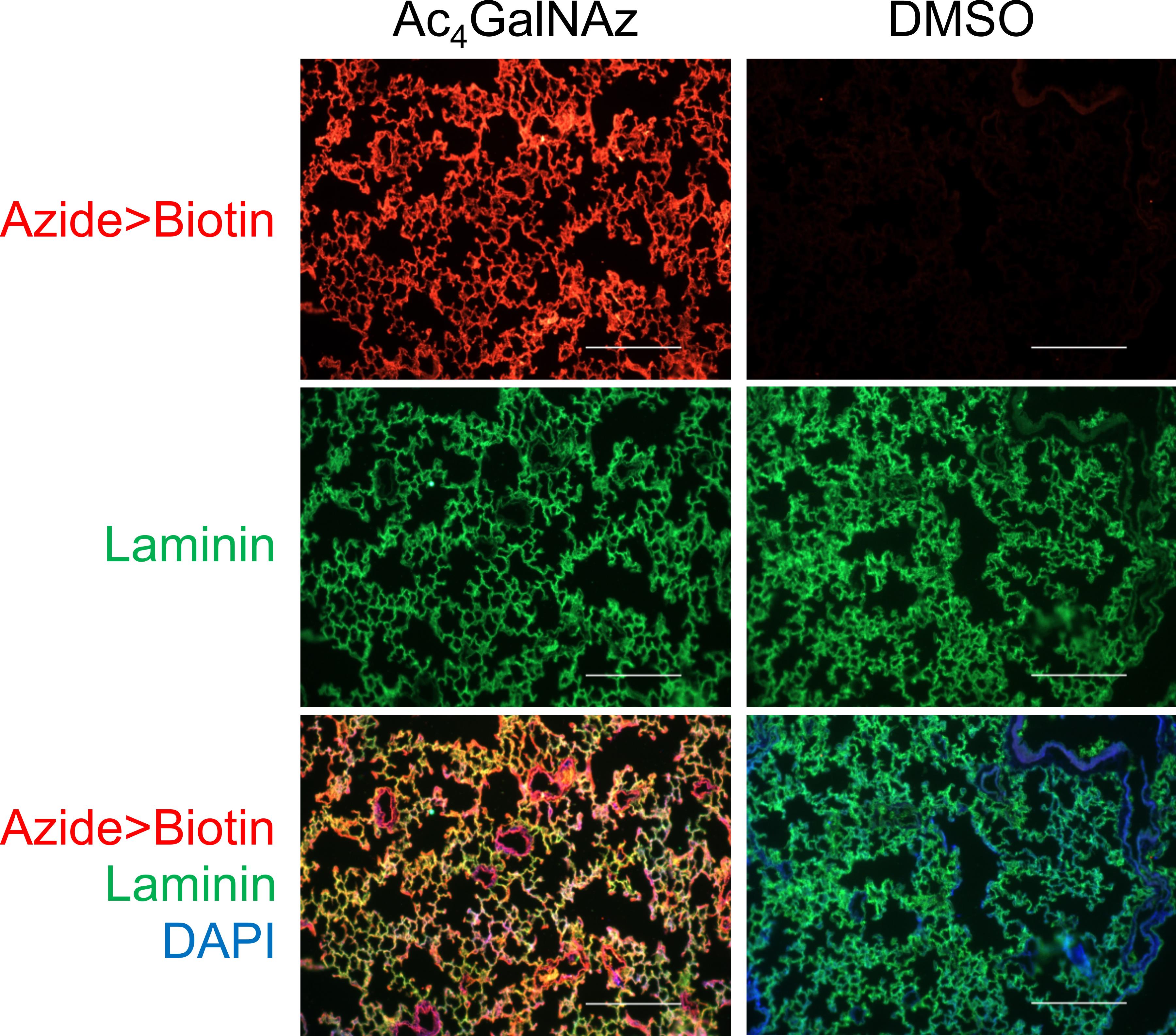
Figure 5. Histological characterization of metabolic azide labeling of the lung ECM. The representative results show two ECM samples derived from rat lungs following three-day in vivo labeling with or Ac4GalNAz or DMSO. The potential azide ligand in the ECM were conjugated to biotin and then detected using fluorophore-conjugated streptavidin (Red). The lung ECM was indicated by Laminin antibody staining (green). Scale bars: 200 μm.Biochemical characterization of metabolic azide labeling. The potential azide ligand in the extracted lung ECM protein was conjugated to biotin using the click chemistry, followed by gel electrophoresis. Using standard Western blot procedures, biotin was detected using HRP-conjugated streptavidin and chemiluminescence. Compared to the control, the ECM protein sample extracted from lungs receiving Ac4GalNAz metabolic labeling is expected to show more robust azide labeling and biotin conjugation (Figure 6).
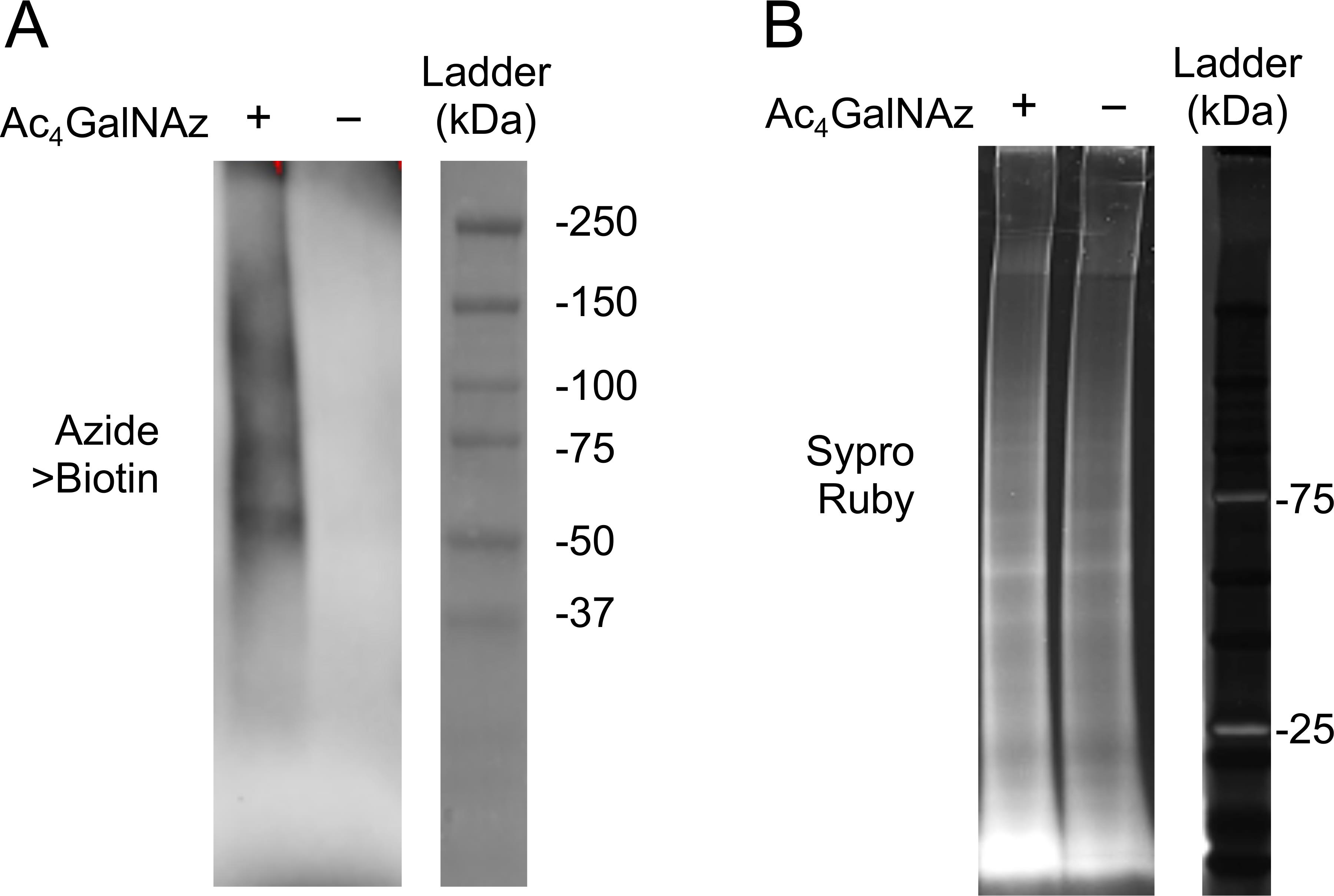
Figure 6. Biochemical characterization of azide labeling in the lung ECM. The representative results show two ECM samples derived from rat lungs following one-day ex vivo labeling with Ac4GalNAz or DMSO. A. Detection of the azide ligand by Western blot analysis of the conjugated biotin. B. Protein loading was verified by in-gel staining of total protein with Sypro Ruby.
Recipes
Ac4GalNAz Injection Solution
70 mg/ml Ac4GalNAz in DPBS with 70% DMSO
Control Injection Solution
70% DMSO in DPBS
Ex vivo Culture Medium
DMEM supplemented with 10% FetalClone I Serum
1% antibiotic/antimycotic
Decell Solution
0.1% SDS in autoclaved deionized water
2× Decell Solution
0.2% SDS in autoclaved deionized water
1× PBS (pH 7.4)
Dilute 20× PBS (pH 7.4) in deionized water, sterilize by autoclave
Fixation Solution
4% paraformaldehyde (PFA) in 1× PBS (pH 7.4)
Biotin-PEG4-Alkyne Click Reaction Mixture
Note: Components from the Click-iT Cell Reaction Buffer Kit (Table 1).
Table 1. Recipe for Biotin-PEG4-Alkyne Click Reaction Mixture
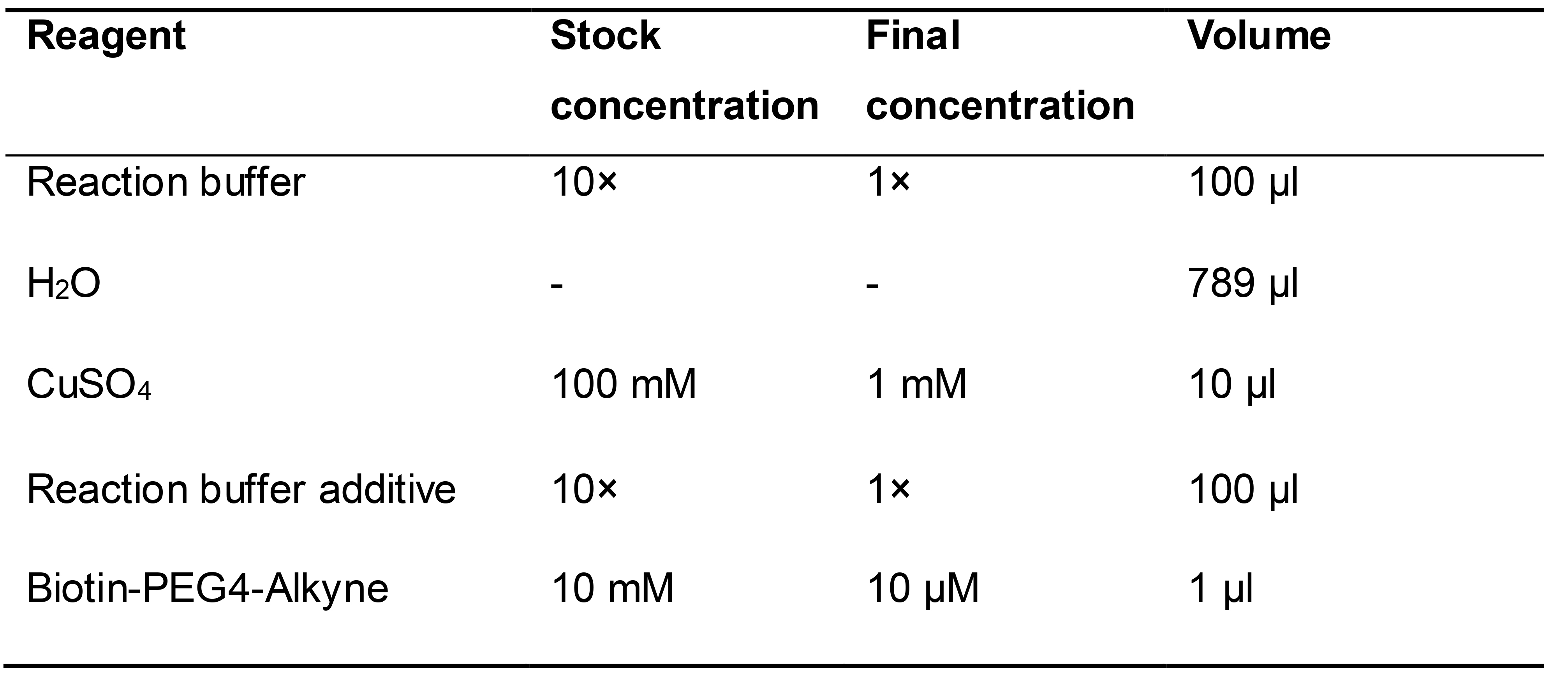
Urea Extraction Buffer
6 M Urea
10 mM HEPES
1× protease inhibitor
Adjust pH to 8.0
1× TBST (pH 7.4)
Dilute 10× TBST (pH 7.4) in deionized water
Blocking Buffer
5% BSA in TBST
HRP-Imaging Mixture
SuperSignal West Pico PLUS Luminol/Enhancer solution and SuperSignal West Pico PLUS Peroxide solution, 1:1 (freshly prepared)
Acknowledgments
This works is supported by the Department of Biomedical Engineering at Carnegie Mellon University. Z.L. and Y.X. are supported by scholarships from the China Scholarship Council. This protocol is originally adapted from Ren et al. (2018). We thank Pitt Biospecimen Core (University of Pittsburgh) for histological processing. We also thank the Vivarium at Carnegie Mellon University for animal husbandry. Finally, we thank Piyumi Wijesekara and Wai Hoe Ng for their assistance in gathering information regarding materials and reagents.
Competing interests
X.R. has a patent application (US20170362266A1) related to this work.
Ethics
All animal procedures were performed according to the protocol approved by the Institutional Animal Care and Use Committee at Carnegie Mellon University (Approval ID#TR202000006, valid 2018-2022).
References
- Chang, P. V., Prescher, J. A., Sletten, E. M., Baskin, J. M., Miller, I. A., Agard, N. J., Lo, A. and Bertozzi, C. R. (2010). Copper-free click chemistry in living animals. Proc Natl Acad Sci U S A 107(5): 1821-1826.
- Chiu, L. L. and Radisic, M. (2010). Scaffolds with covalently immobilized VEGF and Angiopoietin-1 for vascularization of engineered tissues. Biomaterials 31(2): 226-241.
- Davidenko, N., Schuster, C. F., Bax, D. V., Raynal, N., Farndale, R. W., Best, S. M. and Cameron, R. E. (2015). Control of crosslinking for tailoring collagen-based scaffolds stability and mechanics. Acta Biomater 25: 131-142.
- Grover, C. N., Gwynne, J. H., Pugh, N., Hamaia, S., Farndale, R. W., Best, S. M. and Cameron, R. E. (2012). Crosslinking and composition influence the surface properties, mechanical stiffness and cell reactivity of collagen-based films. Acta Biomater 8(8): 3080-3090.
- Laughlin, S. T. and Bertozzi, C. R. (2007). Metabolic labeling of glycans with azido sugars and subsequent glycan-profiling and visualization via Staudinger ligation. Nat Protoc 2(11): 2930-2944.
- Ott, H. C., Clippinger, B., Conrad, C., Schuetz, C., Pomerantseva, I., Ikonomou, L., Kotton, D. and Vacanti, J. P. (2010). Regeneration and orthotopic transplantation of a bioartificial lung. Nat Med 16(8): 927-933.
- Ott, H. C., Matthiesen, T. S., Goh, S. K., Black, L. D., Kren, S. M., Netoff, T. I. and Taylor, D. A. (2008). Perfusion-decellularized matrix: using nature's platform to engineer a bioartificial heart. Nat Med 14(2): 213-221.
- Petersen, T. H., Calle, E. A., Zhao, L., Lee, E. J., Gui, L., Raredon, M. B., Gavrilov, K., Yi, T., Zhuang, Z. W., Breuer, C., Herzog, E. and Niklason, L. E. (2010). Tissue-engineered lungs for in vivo implantation. Science 329(5991): 538-541.
- Reing, J. E., Brown, B. N., Daly, K. A., Freund, J. M., Gilbert, T. W., Hsiong, S. X., Huber, A., Kullas, K. E., Tottey, S., Wolf, M. T. and Badylak, S. F. (2010). The effects of processing methods upon mechanical and biologic properties of porcine dermal extracellular matrix scaffolds. Biomaterials 31(33): 8626-8633.
- Ren, X., Evangelista-Leite, D., Wu, T., Rajab, T. K., Moser, P. T., Kitano, K., Economopoulos, K. P., Gorman, D. E., Bloom, J. P., Tan, J. J., Gilpin, S. E., Zhou, H., Mathisen, D. J. and Ott, H. C. (2018). Metabolic glycan labeling and chemoselective functionalization of native biomaterials. Biomaterials 182: 127-134.
- Sletten, E. M. and Bertozzi, C. R. (2011). From mechanism to mouse: a tale of two bioorthogonal reactions. Acc Chem Res 44(9): 666-676.
- Theocharis, A. D., Skandalis, S. S., Gialeli, C. and Karamanos, N. K. (2016). Extracellular matrix structure. Adv Drug Deliv Rev 97: 4-27.
- Uhl, F. E., Zhang, F., Pouliot, R. A., Uriarte, J. J., Rolandsson Enes, S., Han, X., Ouyang, Y., Xia, K., Westergren-Thorsson, G., Malmstrom, A., Hallgren, O., Linhardt, R. J. and Weiss, D. J. (2020). Functional role of glycosaminoglycans in decellularized lung extracellular matrix. Acta Biomater 102: 231-246.
- Wagner, D. E., Bonenfant, N. R., Parsons, C. S., Sokocevic, D., Brooks, E. M., Borg, Z. D., Lathrop, M. J., Wallis, J. D., Daly, A. B., Lam, Y. W., Deng, B., DeSarno, M. J., Ashikaga, T., Loi, R. and Weiss, D. J. (2014). Comparative decellularization and recellularization of normal versus emphysematous human lungs. Biomaterials 35(10): 3281-3297.
- Wissink, M. J., Beernink, R., Pieper, J. S., Poot, A. A., Engbers, G. H., Beugeling, T., van Aken, W. G. and Feijen, J. (2001). Immobilization of heparin to EDC/NHS-crosslinked collagen. Characterization and in vitro evaluation. Biomaterials 22(2): 151-163.
Article Information
Copyright
© 2021 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Ling, Z., Xing, Y., Reinoso Jacome, E., Fok, S. W. and Ren, X. (2021). Bioorthogonal Labeling and Chemoselective Functionalization of Lung Extracellular Matrix. Bio-protocol 11(4): e3922. DOI: 10.21769/BioProtoc.3922.
Category
Cell Biology > Tissue analysis
Biochemistry > Carbohydrate > Glycoprotein
Biochemistry
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link










