- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Real-time Three-dimensional Tracking of Endocytic Vesicles
Published: Vol 10, Iss 20, Oct 20, 2020 DOI: 10.21769/BioProtoc.3794 Views: 4091
Reviewed by: Guillaume BompardYong TengAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Monitoring of Sperm-Independent Calcium Oscillations in Immature Oocytes of Mice
Sae Horiike [...] Hidehiko Ogawa
Feb 5, 2026 242 Views
Time-Lapse Into Immunofluorescence Imaging Using a Gridded Dish
Nick Lang [...] Andrew D. Stephens
Feb 20, 2026 235 Views

How to Train Custom Cell Segmentation Models Using Cell-APP
Anish J. Virdi and Ajit P. Joglekar
Feb 20, 2026 338 Views
Abstract
Endocytic trafficking and recycling are fundamental cellular processes that control essential functions such as signaling protein complexes transport and membrane identity. The small GTPase Rabs are indispensable component of the endosomal recycling machinery. The Rabs bind to effectors to mediate their functions, such as protein sorting and degradation, membrane tethering or lipid modification, and organelle motility. Due to the complex and dynamic nature of endosomal compartments and tracking route, detailed multiparametric analyses of three-dimensional data by quantitative methods are challenging. Here, we describe a detailed time-lapse imaging protocol designed for the quantitative tracking of single endosomal vesicles, using GFP-Rab4-positive recycling endosomes. This method permits automated tracking of single endocytic vesicles in three-dimensional live cell imaging, allowing the study of multiple parameters such as abundance, speed, directionality, and subcellular localization, as well as protein colocalization. This protocol can be broadly used in any kind of cellular models, under various contexts, including growth factors stimulation, gene knockdowns, drug treatments, and is suitable for high throughput screens.
Keywords: Single-vesicle 3D trackingBackground
Accumulating evidence emphasizes the importance of coordinated endosomal recycling in such diverse processes as cell migration, adhesion, morphogenesis, proliferation, cytokinesis, as well as learning and memory (Grant and Donaldson, 2009; Parachoniak and Park, 2012; Wandinger-Ness and Zerial, 2014; Zaoui et al., 2019a and 2019b). There are over 70 Rab GTPases in mammals that have distinct localizations and functions in membrane trafficking. Further compounding complexity, while most Rab GTPases are ubiquitous, some display tissue specificity expression (van der Sluijs et al., 1992; McCaffrey et al., 2001; Grant and Donaldson, 2009; Wandinger-Ness and Zerial, 2014). Among them, the small GTPase Rab4, localized in the early endosome, regulates the trafficking along the endocytic route, from early and recycling endosomes, toward the plasma membrane. Rab4 has been involved in the recycling of receptors such as Tfn and receptor tyrosine kinase (RTK), integrins, ubiquitin ligases, as well as other machinery regulators associated with vesicle formation, budding, transport, and fusion (McCaffrey et al., 2001; Grant and Donaldson, 2009; Stenmark, 2009; Parachoniak et al., 2011; Wandinger-Ness and Zerial, 2014; Zaoui et al., 2019a). We recently reported strict requirement of the MT plus-end tracking proteins CLIP-170 for Rab4-mediated Met RTK recycling at the plasma membrane in response to its ligand, hepatocyte growth factor (HGF). The increased localization of Met/Rab4 vesicles towards the MT plus-ends in response to HGF results in enhanced cell protrusion dynamic and cell migration (Zaoui et al., 2019a).
Therefore, by mediating the compartmentalization of the endocytic pathway into early, recycling, late, and lysosomal tracking routes and by regulating the trafficking from vesicle budding to fusion, Rabs stand as essential integrators of cell signaling. Rabs control the localization, the intensity, and the duration of signaling outputs, to allow spatiotemporal regulation of signal transduction (McCaffrey et al., 2001; Stenmark, 2009; Grant and Donaldson, 2009; Parachoniak and Park, 2012; Wandinger-Ness and Zerial, 2014; Zaoui et al., 2019a). Altered endocytosis is observed in many malignancies and can lead to sustained proliferation, survival, invasiveness, and therapeutic resistance (Parachoniak and Park, 2012; Mellman and Yarden, 2013; Zaoui et al., 2019a and 2019b). Our studies aim at understanding endocytic recycling alterations on cell signaling regulating cytoskeletal rearrangement, cell migration, and invasion in oncogenic contexts.
The protocol described herein permits automated tracking of the endocytic vesicle in three-dimensional live-cell imaging, allowing the study and the quantification of multiple parameters under physiological and tumorigenic conditions. Moreover, this methodology can be easily extended to assess the mobility of other Rab family members or any other cell vesicles.
Materials and Reagents
- 35 mm glass bottom cell culture dish (Ibidi GmbH, catalog number: 81158 )
- Microcentrifuge tubes: 1.5 ml (Axygen Corning®, catalog number: MCT-150-C )
- 6-well plate (Nunc®, catalog number: 140675 )
- 100 mm dish (VWR®, catalog number: 172931 )
- SKBr3 breast carcinoma cells (ATCC® HTB-30)
- Hepatocyte growth factor, HGF (Genentech, Inc.)
- DMEM-Dulbecco's Modified Eagle Medium (Gibco®, catalog number: 11966025 )
- Rat tail collagen I (Roche®, catalog number: 11179179001 )
- Foetal Bovine Serum (FBS) (Gibco®, catalog number: 26140079 )
- Green fluorescent protein (GFP)-Rab4 plasmid [provided by Robert Lodge and cloned as described in Zaoui et al. (2019a)]
- Cell line nucleofector Kit V (Lonza®, catalog number: VCA-1003 )
- Phosphate-buffered saline (PBS) (Wisent Inc, catalog number: 311-012-LL )
- Trypsin-EDTA (Gibco®, catalog number: 25200056 )
Equipment
- Cell incubator Steri-Cycle CO2 (Thermo Fisher Scientific®, catalog number: 51030303 )
- Centrifuge (Eppendorf®, catalog number: 5424/5424R )
- Nucleofector 2b device (Lonza®, catalog number: AAB-1001 )
- Motorized Inverted microscope Axiovert 200 M (Carl Zeiss, Inc.)
- Stage top chamber with 5% (v/v) CO2 in air at 37 °C (Climabox, Carl Zeiss, Inc.).
- Objective 100x plan Apochromat NA 1.4 objective
- Hemocytometer (Sigma-Aldrich®, catalog number: Z359629-1EA )
Software
- AxioVision software (Carl Zeiss, Inc.)
- Imaris software (Bitplane Inc.)
- Microsoft Excel®
Procedure
- Collagen I coating
- Coat the 35 mm glass bottom cell culture dish with 1 ml rat-tail collagen I diluted in PBS (25 µg/ml).
- Incubate the 35 mm glass bottom cell culture dish coated with 1 ml collagen I at 37 °C, for 1 h.
- Wash the 35 mm glass bottom cell culture dish twice with 1 ml pre-warm PBS and aspirate off PBS buffer.
- Coat the 35 mm glass bottom cell culture dish with 1 ml rat-tail collagen I diluted in PBS (25 µg/ml).
- Cell transfection and seeding
- Retrieve culture plate from incubator, aspirate off media, and wash twice cells with 1 ml pre-warm PBS.
- Detach SKBr3 cells (80-90% confluency) by adding 1 ml Trypsin-EDTA and incubate the cells at 37 °C, for 5 min.
- Neutralize trypsinization reaction with supplemented culture medium (10% FBS) when most of the cells (> 90%) are detached.
- Determine cell density with hemocytometer.
- Transfer (1 x 106 cells per sample) into the microcentrifuge tubes.
- Centrifuge the cells at 200 x g for 10 min at room temperature.
- Gently resuspend cell pellet in 100 μl nucleofector solution V at room temperature. The solution V is provided with the nucleofector Kit V.
- Combine 100 μl of cell suspension with 3 μg DNA of GFP-Rab4.
- Transfer cell/DNA suspension into certified cuvette provided with nucleofector Kit V (sample must cover the bottom of the cuvette without air bubbles). Close the cuvette with the cap.
- Select the appropriate Nucleofector® Program X-005 on the Nucleofector® 2b device.
- Insert the cuvette with cell/DNA suspension into the Nucleofector® Cuvette Holder and apply the selected program by pressing the X-button.
- Take the cuvette out of the holder once the program is finished.
- Immediately add 500 μl of the culture medium (10% FBS) to the cuvette.
- Use the supplied pipettes to transfer the sample (600 µl final volume) into the collagen I pre-coated 35 mm glass bottom cell culture dish prepared in Procedure A.
- Add 2 ml supplemented culture medium with 10% FBS per 35 mm glass bottom cell culture dish and incubate at 37 °C, for 24 h.
- Retrieve culture plate from incubator, aspirate off media, and wash twice cells with 1 ml pre-warm PBS.
- Cell washing and stimulation
- Retrieve the 35 mm glass bottom cell culture dish from incubator, aspirate off media, and wash twice cells with 1 ml pre-warm PBS.
- Add 2 ml pre-warm culture medium (10% serum)
- Add HGF at 0.5 nM (or any other growth factor) for 20 min to stimulate the cells. An unstimulated condition is needed as a negative control, to evaluate the effect of Met receptor tyrosine kinase activation with HGF.
- Retrieve the 35 mm glass bottom cell culture dish from incubator, aspirate off media, and wash twice cells with 1 ml pre-warm PBS.
- Microscope setup for time-lapse imaging in 3D (Figure 1 and Video 1)
- Microscope setup
- Turn on the motorized Inverted microscope Axiovert 200 M and the connected computer.
- Select the appropriate stage holder and the 100x plan Apochromat NA 1.4 objective.
- Turn on the temperature unit and the CO2 control module, set temperature at 37 °C, humidity > 80% and CO2 at 5%. Warm up for approximately 2 h.
- Turn on the motorized Inverted microscope Axiovert 200 M and the connected computer.
- Live-cell imaging in 3D
- Open the AxioVision software and choose Multidimensional Acquisition module.
- Place the 35 mm glass bottom cell culture dish on the plate holder. Focus on the selected cells.
- Set up the cell duration of acquisition for 2 min with a capturing interval of 4 s and 10 of Z sections with 0.5 μm spacing.
- Acquire single cell images in 3D (X, Y, Z) and analyze them in real-time.
- Open the AxioVision software and choose Multidimensional Acquisition module.

Figure 1. Microscope configuration for live cell imaging. Example of a complete setup for cell tracking using Axiovert 200 M motorized inverted microscope equipped with a Perspex environmental chamber, CO2 enrichment module, and heating controller. Please refer to the McGill University Advanced Bioimaging Facility for additional details.Video 1. Visualization of GFP-Rab4 protein by time-lapse video microscopy. Movie of SKBr3 cells expressing GFP-Rab4 stimulated with HGF 0.5 nM for 20 min. - Microscope setup
Data analysis
In the following section, we will demonstrate procedures for the analyses of Rab4-positive vesicles mobility in 3D using spots tracking algorithm of Imaris® software.
- Visualization and segmentation (Figure 2)
- Transfer the measurements to Imaris® software and create a volume reconstruction of the data set select using the “Surpass mode”.
- Click on “Edit”, open “Display Adjustment” window, select as source channel the Channel 1 (green for GFP) and adjust the minimum and the maximum.
- Click on the left tab “Spot Object” button and a new object spot is now added in the object list.
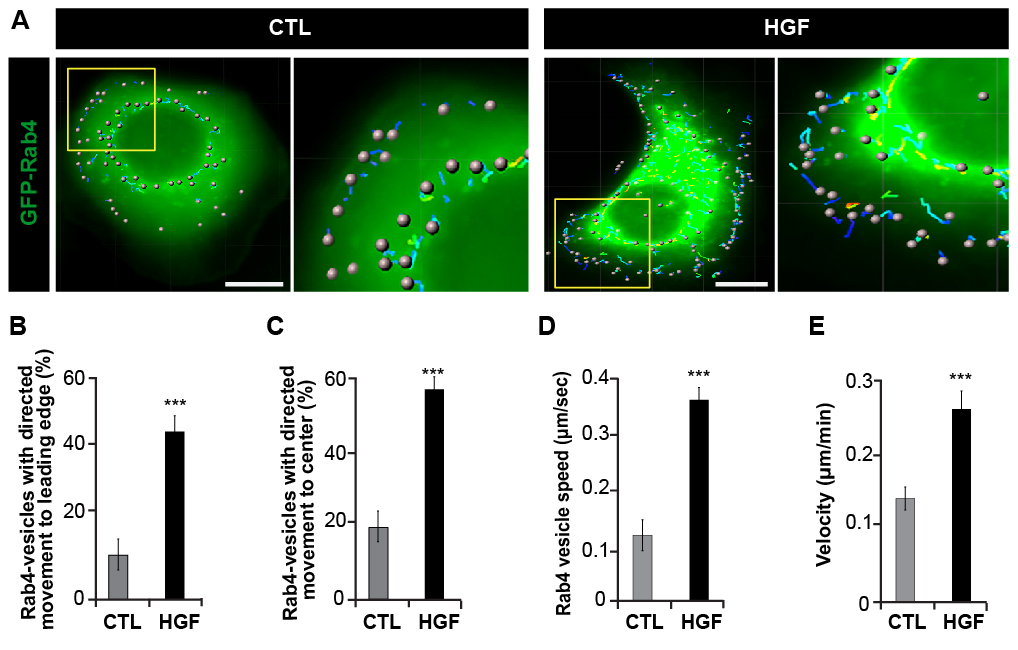
Figure 2. Three-dimensional analysis of Rab4-GFP movement by live cell imaging. A. Representative image of spot assignment of the Rab4-GFP vesicle using the Spots tracking algorithm of Imaris® software. Stack image of SKBr3 cells transfected with GFP-Rab4 and treated or not (CTL) with HGF 0.5 nM for 20 min. Insets show enlargement from the cell cortex. B-E. Individual Rab4-positive vesicles were tracked over time (2 min) after 20 min stimulation with HGF 0.5 nM. The percentage of directed movement of vesicles to leading edge (B), to center (C) and, the speed of Rab4-positive vesicles (D) were analyzed. E) Cell velocity was determined by tracking cells every 5 min for 420 min. Ninety cells were tracked for CTL and HGF treated cells. Scale bars = 10 μm. Graphed data represent the average values ± SEM from at least three independent experiments. Statistical significance was assessed using a two-tailed Student’s t test. *** P < 0.001. Representative movie of SKBr3 cells expressing GFP-Rab4 stimulated with HGF 0.5 nM for 20 min (Video 1).
- Tracking algorithm
- Select “Track Spots (over time)” in create properties. Click on “Next”.
- Select as Source Channel the Channel 1 (green for GFP); set the “Estimated Diameter” to 0.5 µm. Add “Background Subtraction” option. Click on “Next”.
- Select “Track Spots (over time)” in create properties. Click on “Next”.
- (Optional) Selecting the manual option
- Click on “Manual” option and select a subset of the detected spots by modifying the lower and higher threshold of the green fluorescence. Put the Lower Threshold value at 10 and to verify spots detection click on play button. Click on “Next”.
- Add or delete spots at this step (if needed). Click on “Next”.
- Select “Brownian Motion” algorithm. Set Maximum Distance at 10 µm and Maximum Gap Size at 3 µm. Click on “Next”.
Note: The “Brownian Motion” algorithm is appropriated for the tracking of random movements without discernable pattern and frequent directional changes. Here, this algorithm has been used to track the Rab4-GFP vesicles because in absence of HGF the Rab4-GFP vesicles move slowly, in a random manner, without any directional persistence. However, the Imaris® software provides “Autoregressive Option”, this algorithm tolerates high rate of directional change during the displacement and can be used to track objects with continuous motion. - Sort and filter the result tracks by selecting “Track Speed Average” in “Filter Type menu”. Click on Finish.
- Export the data to Microsoft Excel® software for further analysis. The tracking measurements are stored in the “Detailed Tab” found under “Tab Statistics”.
- Summarize vesicle speed of at least 500 vesicles from 30 cells as desired (i.e., mean ± SEM) and perform statistical analysis as desired (such as unpaired t-test).
- Plot summarized information using preferred graph (such as bar graph).
- Click on “Manual” option and select a subset of the detected spots by modifying the lower and higher threshold of the green fluorescence. Put the Lower Threshold value at 10 and to verify spots detection click on play button. Click on “Next”.
Acknowledgments
This work was performed as a part of the publication of Zaoui et al. (2019a), Traffic and was supported by a Susan G. Komen for the Cure Postdoctoral fellowship (KG111467) to K. Zaoui.
Competing interests
The authors declare no competing financial interests.
References
- Grant, B. D. and Donaldson, J. G. (2009). Pathways and mechanisms of endocytic recycling. Nat Rev Mol Cell Biol 10(9): 597-608.
- McCaffrey, M. W., Bielli, A., Cantalupo, G., Mora, S., Roberti, V., Santillo, M., Drummond, F. and Bucci, C. (2001). Rab4 affects both recycling and degradative endosomal trafficking. FEBS Lett 495(1-2): 21-30.
- Mellman, I. and Yarden, Y. (2013). Endocytosis and cancer. Cold Spring Harb Perspect Biol 5(12): a016949.
- Parachoniak, C. A., Luo, Y., Abella, J. V., Keen, J. H. and Park, M. (2011). GGA3 functions as a switch to promote Met receptor recycling, essential for sustained ERK and cell migration. Dev Cell 20(6): 751-763.
- Parachoniak, C. A. and Park, M. (2012). Dynamics of receptor trafficking in tumorigenicity. Trends Cell Biol 22(5): 231-240.
- Stenmark, H. (2009). Rab GTPases as coordinators of vesicle traffic. Nat Rev Mol Cell Biol 10(8): 513-525.
- van der Sluijs, P., Hull, M., Webster, P., Male, P., Goud, B. and Mellman, I. (1992). The small GTP-binding protein rab4 controls an early sorting event on the endocytic pathway. Cell 70(5): 729-740.
- Wandinger-Ness, A. and Zerial, M. (2014). Rab proteins and the compartmentalization of the endosomal system. Cold Spring Harb Perspect Biol 6(11): a022616.
- Zaoui, K., Duhamel, S., Parachoniak, C. A. and Park, M. (2019a). CLIP-170 spatially modulates receptor tyrosine kinase recycling to coordinate cell migration. Traffic 20(3): 187-201.
- Zaoui, K., Rajadurai, C. V., Duhamel, S. and Park, M. (2019b). Arf6 regulates RhoB subcellular localization to control cancer cell invasion. J Cell Biol 218(11): 3812-3826.
Article Information
Copyright
© 2020 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Duhamel, S. and Zaoui, K. (2020). Real-time Three-dimensional Tracking of Endocytic Vesicles. Bio-protocol 10(20): e3794. DOI: 10.21769/BioProtoc.3794.
Category
Cancer Biology > Cancer biochemistry > Protein
Cell Biology > Cell imaging > Live-cell imaging
Cell Biology > Cell imaging > Confocal microscopy
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link










