- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Method for Prolonged Incubation of Brain Slices
Published: Vol 10, Iss 14, Jul 20, 2020 DOI: 10.21769/BioProtoc.3683 Views: 5225
Reviewed by: Ehsan KheradpezhouhAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Cell-attached and Whole-cell Patch-clamp Recordings of Dopamine Neurons in the Substantia Nigra Pars Compacta of Mouse Brain Slices
Stefano Cattaneo [...] Stefano Taverna
Aug 5, 2021 5815 Views
Human Skin Explant Preparation and Culture
Jessica L. Shannon [...] Jennifer Y. Zhang
Sep 20, 2022 3957 Views
Ultrafast Isolation of Synaptic Terminals From Rat Brain for Cryo-Electron Tomography Analysis
Rong Sun and Qiangjun Zhou
Sep 5, 2025 3584 Views
Abstract
Slices of neuronal tissue maintain a high degree of topographical and functional properties of neurons and glia and therefore are extensively used for measurements of neuronal activity at the molecular, cellular and network levels. However, the lifespan of slice preparations is narrow, averaging of 6-8 hours. Moreover, the average viability of brain slices varies according to animal age and region of interest, leading to the high variability and low reproducibility of recorded data.
Previous techniques to increase the viability of brain slices focused on reducing cytotoxicity by chemical means, including alterations of the artificial cerebrospinal fluid (aCSF) composition to alleviate the direct damage of the slicing procedure or adding protective antioxidants to reduce cellular deterioration. In this protocol, we use a combination of hypothermia with firm control of the aCSF conditions in the recovery chamber (pH, temperature, and bacteria levels) to extend the slice viability significantly.
Given the breadth of its usage, improving slice viability and longevity can considerably increase data reproducibility and reduce the cost, time, and number of animals used in neurophysiological studies.
Background
Neuronal tissue slices provide unique advantages in basic neuroscience research as it allows direct investigation of neuronal network activity and the influence of pharmacological compounds on the central nervous system. Hence, it is imperative to prepare and maintain slices viability at the highest quality for experimental purposes. The viability of neuronal slices relies on several intrinsic and extrinsic factors (Buskila et al., 2014), which can be divided into three periods according to the slicing procedure: before (pre-slicing), during (slicing) and after (post-slicing) slicing procedure. The main factors that decrease slice viability in the pre-slicing period include ischemia and hypoxia occurring due to elimination of continuous blood supply (Buskila et al., 2020). Both hypoxia and ischemia, starting seconds after decapitation, lead to excitotoxic processes such as mitochondrial dysfunction and Ca2+ influx, which contribute to neuronal hyperexcitability and decrease in slice viability. The slicing procedure itself leads to physical damage of the semi-permeable membrane of neurons, resulting in neurotransmitter spill-over and inflow of compounds (i.e., Ca2+, Na2+, etc.) into the cell, which enhances neuronal cytotoxicity (Reid et al., 1988). The main factors affecting the slice viability during the post-slicing episode includes the temperature of the incubation solution and the stability of its pH, as these factors affect processes which impact the cells deterioration rate, including redox metabolism and the reduction in the oxygen transfer into the deep layers of the tissue (Reid et al., 1988). For long-term incubation of the slices (> 4 h), the level of bacteria is also an essential factor, as it contributes to slice deterioration via releasing of endotoxins, glial activation and neurodegeneration (Qin et al., 2007; Breen and Buskila, 2014).
To reduce the impact of the factors affecting the viability of the slices, we capitalize on previous protocols that found effective in increasing slice viability by reducing excitotoxicity during the pre-slicing and slicing steps and additionally use an active recovery (post-slicing) incubation chamber, the BraincubatorTM, which controls several parameters, such as temperature, pH and bacteria levels. In this system, slices are kept in a temperature-controlled chamber, while the aCSF flows through a secondary tank exposed to an ultraviolet C (UVC) light source that eradicates bacteria. The design and use of the BraincubatorTM have been described previously (Breen and Buskila, 2014; Buskila et al., 2014; Cameron et al., 2016 and 2017). Hypothermia has been proven highly efficient in reducing the metabolic rate and post-slicing cellular damage, hence, we have incubated brain slices at 15-16 °C. The combination of hypothermia with the UVC filtration system can extend slice viability for > 24 h (Buskila et al., 2014).
Materials and Reagents
- Masking tape (3M; RS components, catalog number: 124-0913 )
- Butterfly needles (27G; Global Medical Supply, catalog number: 91508T )
- Isoflurane (VCA I.S.O. 250 ml)
- Ketamine/Xylazine (100 mg/kg/10 mg/kg)
- Gel superglue (Loctite, 3 g)
- Carbogen (5% CO2 mixed in 95% O2; BOC Australia)
- N-methyl D-glucamine (NMDG; Sigma, catalog number: M2004-500G ), stored at RT
- KCl (Sigma, catalog number: P9333-1KG ), stored at RT
- NaH2PO4 monobasic monohydrate (Sigma, catalog number: S9638-25G ), stored at RT
- NaHCO3 (Sigma, catalog number: S5761-1KG ), stored at RT
- D-glucose (Sigma, catalog number: G5767-500G ), stored at RT
- MgSO4 (Sigma, catalog number: M7506-500G ), stored at RT
- CaCl2 (Sigma, catalog number: 223506-500G ), stored at RT
- HEPES (Sigma, catalog number: H3375-250G ), stored at RT
- Thiourea (Sigma, catalog number: T7875-100G ), stored at 4 °C
- Sodium L-ascorbate (Sigma, catalog number: 4034-100G ), stored at 4 °C
- Sodium pyruvate (Sigma, catalog number: P2256-25G ), stored at 4 °C
- NaCl (Sigma, catalog number: S5886-1KG ), stored at RT
- MgCl2 (Sigma, catalog number: M8266-100G ), stored at RT
- Perfusion aCSF (see Recipes)
- Incubation aCSF (see Recipes)
Equipment
- Anesthetic induction chamber (Darwall, catalog number: 1859 )
- 30 x 30 cm of polystyrene block (custom made)
- Large metal scissors (Fine Science Tools, catalog number: 14001-18 )
- Scalpel (Fine Science Tools, catalog number: 10004-13 )
- Scalpel blade (Fine Science Tools, catalog number: 10022-00 )
- Small scissors (Fine Science Tools, catalog number: 14088-10 )
- Forceps (Fine Science Tools, catalog number: 11271-30 )
- Spatula (Fine Science Tools, catalog number: 10090-13 )
- Hemostat (Fine Science Tools, catalog number: 13007-12 )
- Beaker (250 ml; Labdirect, catalog number: 355.018.250 )
- Vibratome (Leica Biosystems, model: VT1200S )
- Soft artist paint brush (size 2; Bunnings, catalog number: 1660207 )
- Pasteur pipette (Labdirect, catalog number: 88336 )
- BraincubatorTM (PaYo Scientific Ltd Pty; http://braincubator.com.au)
- Peristaltic pump (Masterflex, catalog number: EW-77122-20 )
- Tubing for Masterflex pump (Coleparmer, catalog number: EW-06508-15 )
- Small rodent guillotine (WPI)
Procedure
- Pre-slicing transcardial perfusion
The transcardial perfusion induces rapid hypothermia and thus reduce slice deterioration. This step is particularly important in adult animals (> 3 months), however is applicable for rodents aged 1 month onwards.- Pour 300 ml of ice-cold (4 °C) perfusion solution (see Recipe 1) into a beaker and apply carbogen flow. Carbogen is a gas mixture of 95% O2 and 5% CO2. It provides sufficient levels of oxygen consumed by neurons and required to maintain a stable pH of 7.4. Set a peristaltic pump (flow rate of 20 ml/min) to circulate the solution between the beaker and the heart (Step A5).
- Anesthetize the animal by injecting a mixture of ketamine/xylazine (100 mg/kg/10 mg/kg) intraperitoneally using 29G syringe. Once the animals are deeply anesthetized (absence of reaction to pinch of the paws or tail with forceps), place the animal in a supine position and fix the limbs onto a polystyrene block using masking tape.
- Using forceps, raise the skin from the xiphoid process and cut the skin through to the body cavity using blunt scissors to expose the sternum (Figures 1A and 1B).
- Remove the rib-cage and expose the heart (Figure 1C). For this step, hold the sternum and make incisions to the lateral sides to cut through the diaphragm. Then, you can cut and remove the rib-cage with scissors to have a clear view of the heart. Be careful not to damage internal organs or main blood vessels.
- Stop the carbogenation of the ice-cold perfusion aCSF solution to avoid bubbles in the tubing. This step is essential, as carbogen bubbles in the tubing system may flow into the blood circulation and block the perfusion solution from reaching all blood vessels. In case of noticing bubbles in the tubing system, try to flush them out from the tubes before continuing. Start the perfusion pump (flow rate of 20 ml/min) to fill the pipes with the solution until a few drops flow continuously through the butterfly needle that is connected to the pipe and stop the perfusion. Hold the heart with blunt forceps and gently place the needle into the left ventricle, paying attention not to push the needle through the heart (Figure 1C). Secure the needle in place with a hemostat to avoid spillover and start the perfusion pump at a flow rate of 20 ml/min.
- Immediately after operating the pump, cut the right atrium with fine scissors to allow the solution to flow through and drain out of the circulation (Figure 1D). Maintain the perfusion for 2-3 min until you notice a discoloring of the nose, liver and tail. A clear solution outflowing from the atrium will indicate that most blood has been replaced by the perfusion solution, hence successful perfusion.
- Following transcardial perfusion, decapitate the animal using a rodent guillotine or large scissors.
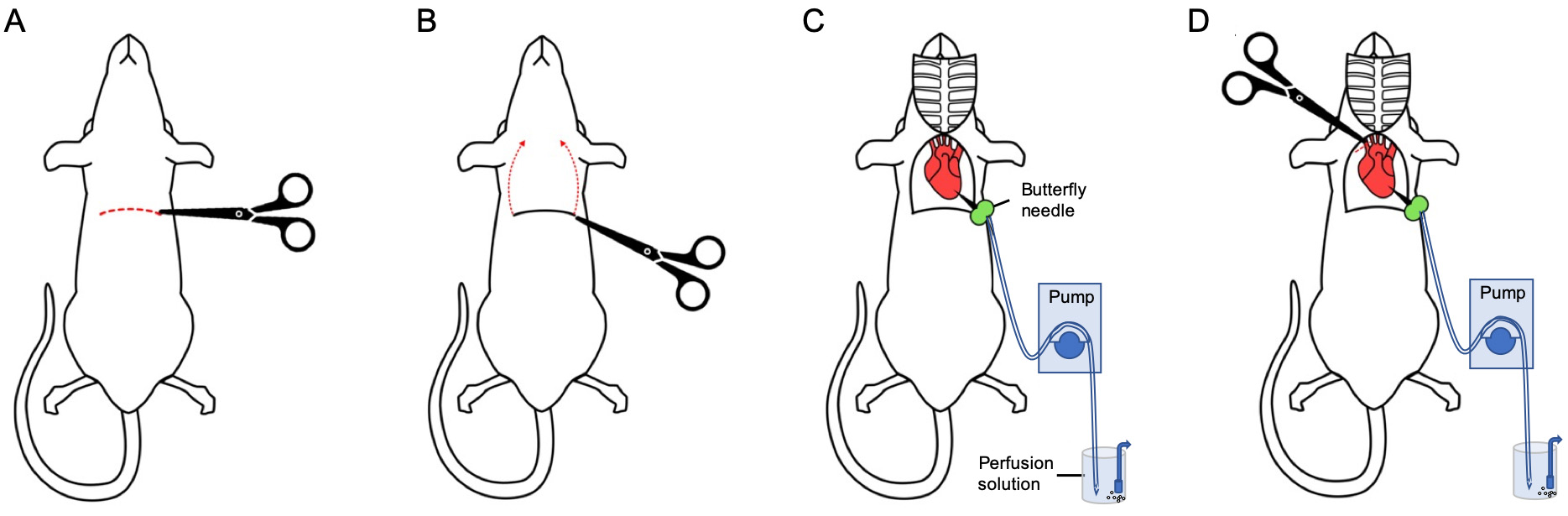
Figure 1. Major stages of transcardial perfusion. A and B. Removing the skin to expose the body cavity. C. Removal of the rib-cage to expose the heart and insertion of a butterfly needle (green) into the left ventricle. The butterfly needle is connected to a perfusion system driven by a peristaltic pump. D. Make a small incision in the right atrium to decrease blood pressure and enable the perfusion solution to be washed out from the body. - Using a scalpel, expose the bone by making a median incision from the nasal bone to the occipital surface (Figure 2A). Then clean the cervical part from remaining muscles (Figure 2B).
- Once the bone is exposed, use scissors to slightly lift and cut the bone from the middle of the occipital bone to the nasal bone along the sagittal suture. For old animals with thick bone, make additional incisions on the lateral sides of the skull, to help removing the bone (Figure 2C).
- Gently remove the interparietal and parietal bones with forceps from both sides; hence, the dorsal surface of the brain becomes visible (Figure 2D). Take great care when removing the bone, not to damage the tissue with the forceps.
- Gently separate and remove the dura from the brain to avoid future damage to the tissue.
- Using a curved spatula, scoop the brain and remove it into ice-cold carbogenated perfusion aCSF. The brain must be removed carefully to avoid mechanical damage to the tissue, and rapidly to minimize hypoxia.
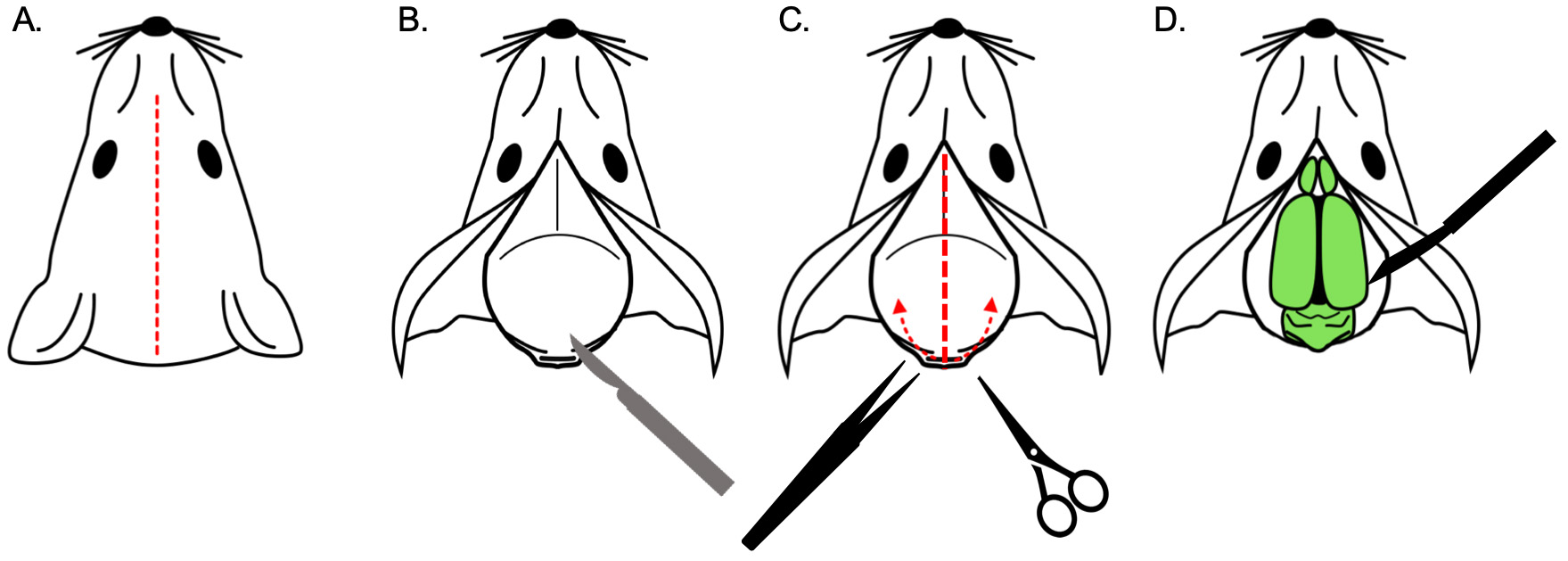
Figure 2. Major steps of brain removal. A. Make a medial incision with a scalpel to expose the bone. B. Cleaning the cervical muscles from the surface. C. Cut the parietal bones along the sagittal suture and gently remove them with forceps. D. Expose the brain and scoop it out with a curved spatula.
- Slicing
- Incubate the whole brain for 30-60 s in the cold perfusion aCSF to allow deep hypothermia. Then, take out the brain with a spatula and prepare a flat surface by either cutting through the median line (to separate hemispheres) or removing the dorsal part of the brain, depending on the desired slice (Figure 3A). The flat surface is essential to increase the bonded tissue surface area and thus to enhance structural stability during the slicing procedure.
- Using a gel-superglue, place the flat side of the brain on the cutting stage (Figure 3B). Pay attention to the orientation of the brain in reference to the vibrating knife, as this will determine the orientation of the slices and structures that will be maintained (coronal vs. sagittal slices).
- Place the cutting stage into the chilled chamber of the vibratome (pre-filled with ice cold perfusion aCSF solution) and cut 300 μm thick brain slices in the preferred orientation that includes the region of interest. Adjust the blade vibration amplitude (0.85 to 1 mm) and speed (0.15 to 0.23 mm/s) before slicing to ensure clean cutting (Figure 3C). The entire slicing procedure should be completed in less than 10-15 min (Figure 3D).
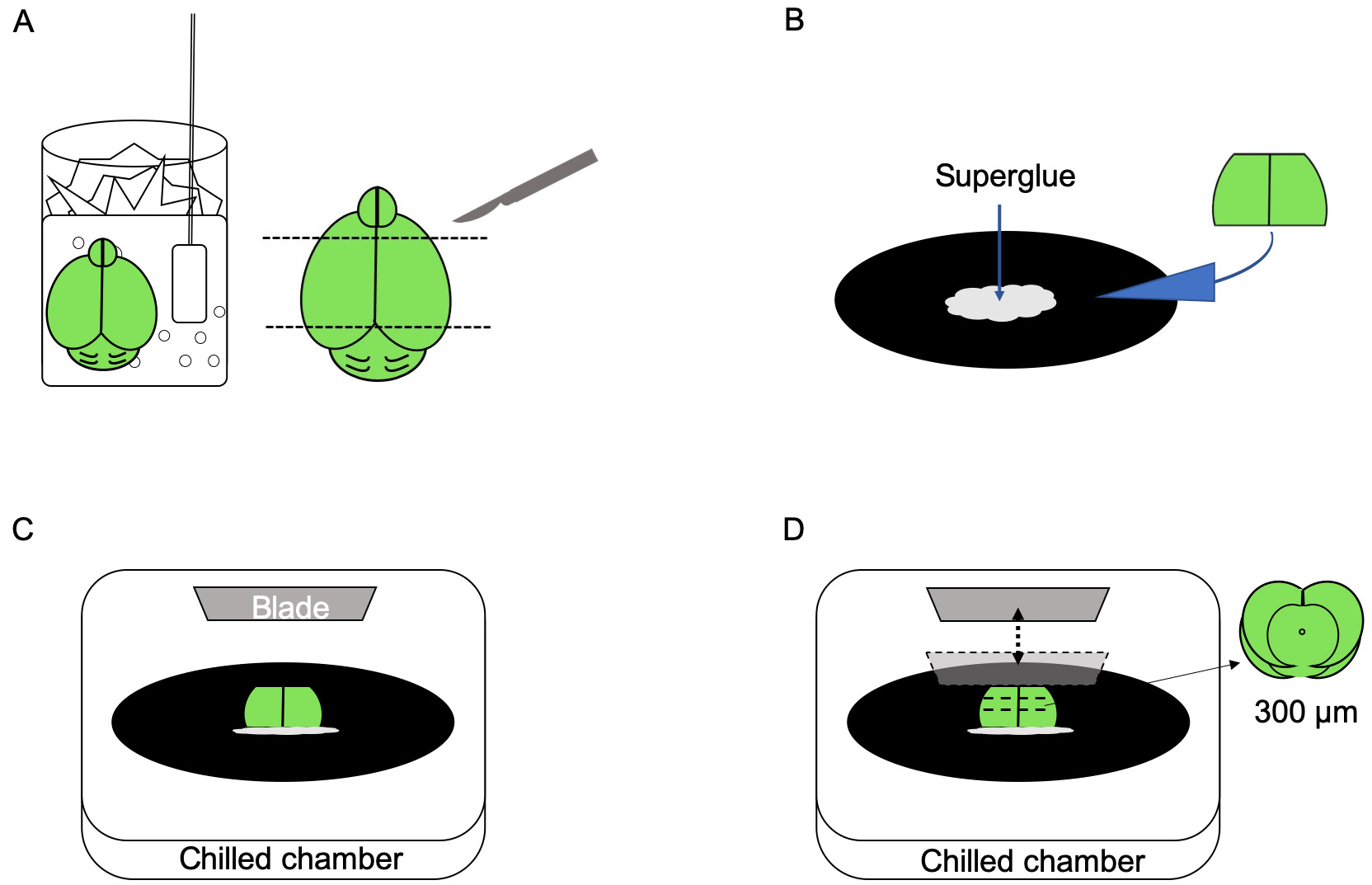
Figure 3. The slicing procedure. A. Prepare a flat surface by cutting the caudal part of the brain with a scalpel. B. Apply superglue on the slicing stage and place the brain on the flat surface in the appropriate position. C. Place the stage in the microtome cooling chamber. D. Cut 300 µm slices with the vibratome blade.
- Post-slicing
Transfer slices to a custom-built incubation system (BraincubatorTM), containing probes for measuring pH levels (7.2-7.4), carbogen flow and temperature, as previously described (Breen and Buskila, 2014; Buskila et al., 2014; Cameron et al., 2017). Before putting slices into the main chamber of the BraincubatorTM, you should prepare and preset the conditions (desired temperature, carbogen flow, pH) of the incubation aCSF in the main chamber.- Pour 200 ml of post slicing incubation aCSF into the main chamber of the BraincubatorTM and turn it on. Set the temperature to 32 °C and the carbogen flow to the slow-mid range to allow proper carbogenation (pH should be 7.2-7.4). Turn on the peristaltic pump to circulate the aCSF (flow rate of 12 ml/min) through a secondary UVC chamber periodically exposed to 1.1W UVC light (Figure 4A). This step will eradicate bacteria floating in the aCSF.
- Transfer slices using a cutoff plastic Pasteur pipet (internal diameter of 1cm) into the appropriate slice holder in the main chamber of the BraincubatorTM (Figure 4B).
- For the initial incubation period, slices can also be transferred into a chamber containing perfusion aCSF for 12 min at 32-34 °C, as previously described by (Ting et al., 2014), and then placed into the BraincubatorTM, which includes incubation aCSF (Figure 4C).
- Following 10 min at a temperature of 32 °C, decrease the temperature to 15 °C and let the slices to rest for an additional 45 min before use (Figure 4C).
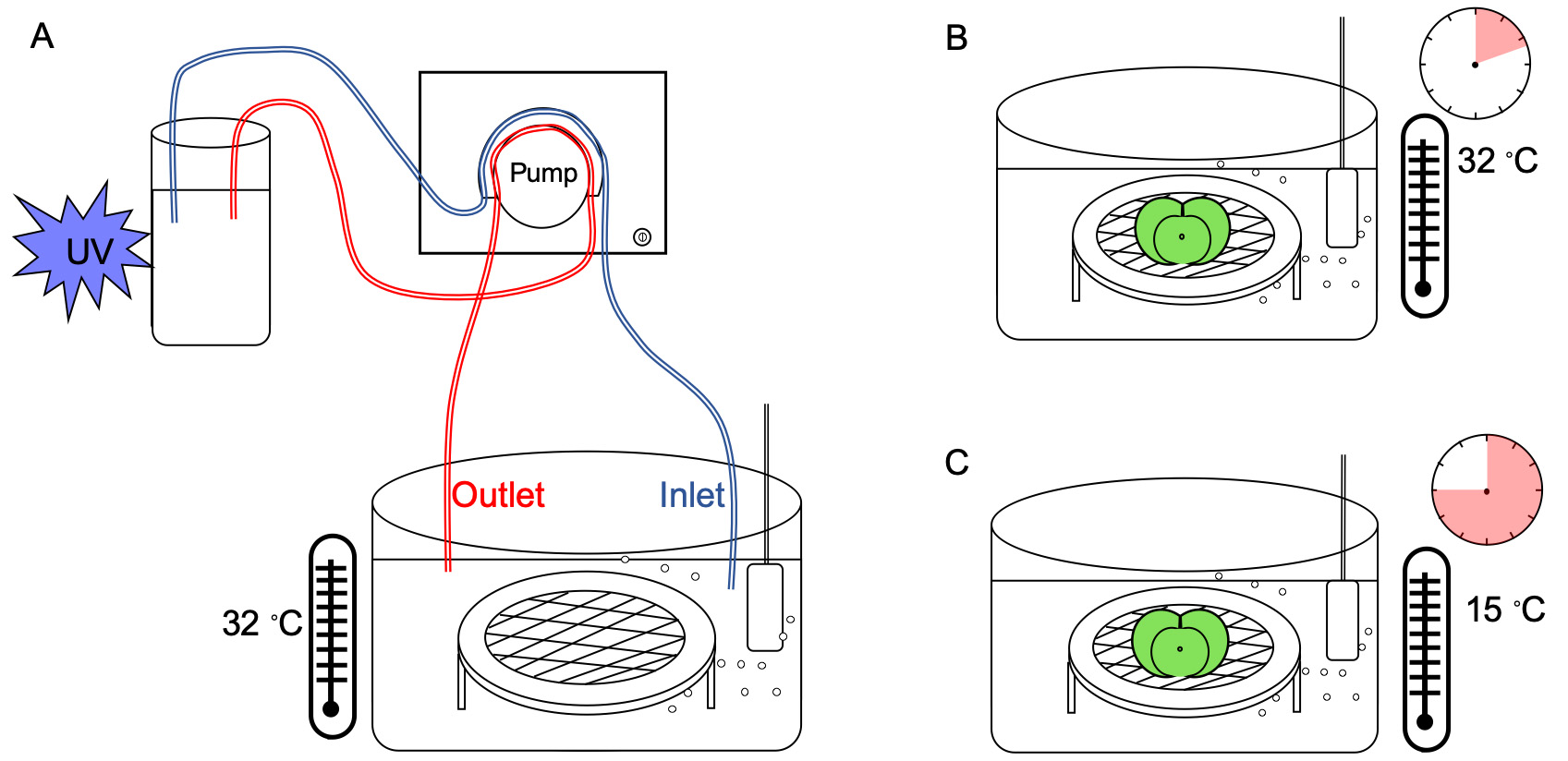
Figure 4. Post-slicing incubation. A. Prepare the Braincubator. Pour 200 ml incubation aCSF to the main chamber and start the circulation between main and secondary chamber. Set the temperature to 32 °C. B. Place slices on the mesh in the main chamber and incubate for 12 min. C. Decrease the temperature to 15 °C and incubate for a minimum of 45 min before use.
Notes
- It is imperative to ensure that the UVC chamber is "light-tight", as UV light can be hazardous to human tissue and can damage the slices.
- Control the UVC light timing via a programmable timer using a random feature that turns ON at times varying between 15 and 26 min every 15-30 min to avoid excessive heating of the aCSF.
- Remove the carbogen bubbles from the mesh slice holder in the main chamber, as these can lead to lipid peroxidation and slice deterioration.
Recipes
- Perfusion aCSF
NMDG 92 mM HCl 20 ml KCl 2.5 mM NaH2PO4(monohydrate) 1.2 mM NaHCO3 25 mM CaCl2 0.5 mM D-glucose 25 mM HEPES 20 mM Thiourea 2 mM Na-ascorbate 5 mM Na-pyruvate 3 mM MgSO4 10 mM pH 7.2 Storage temperature (4 °C) Osmolarity 300-310 mOsm - Incubation aCSF
KCl 2.5 mM NaH2PO4(monohydrate) 1.25 mM NaHCO3 25 mM CaCl2 2 mM NaCl 125 mM MgCl2 1 mM D-glucose 15 mM Thiourea 2 mM Na-ascorbate 5 mM Na-pyruvate 3 mM pH 7.4 Storage temperature (4 °C) Osmolarity 300-310 mOsm
Acknowledgments
This work was supported by a seed funding grant (WSU), and the and Ainsworth Medical Research Innovation Fund to Y.B. O.K. is supported by WSU postgraduate scholarship (UWSPRA).
This protocol was derived from previous publications (Cameron et al., 2017; Buskila et al., 2020).
Ethics
All procedures performed in this study involving animals were in accordance with the ethical standards of Western Sydney University (Animal Research Authority, catalog number: A11199) at which the studies were conducted. This article does not contain any studies with human participants performed by any of the authors.
References
- Breen, P. P. and Buskila, Y. (2014). Braincubator: An incubation system to extend brain slice lifespan for use in neurophysiology. In: 36th Annual International Conference of the IEEE Engineering in Medicine and Biology Society. EMBC 2014, pp. 4864-4867.
- Buskila, Y., Bellot-Saez, A., Kékesi, O., Cameron, M. and Morley, J. (2020) Extending the life span of acute neuronal tissue for imaging and electrophysiological studies. In: Wright, N. (Ed.). Basic Neurobiology Techniques. Neuromethods, vol 152. Humana, New York, NY.
- Buskila, Y., Breen, P. P., Tapson, J., van Schaik, A., Barton, M. and Morley, J. W. (2014). Extending the viability of acute brain slices. Sci Rep 4: 5309.
- Cameron, M., Kekesi, O., Morley, J. W., Tapson, J., Breen, P. P., van Schaik, A. and Buskila, Y. (2016). Calcium imaging of AM Dyes following prolonged incubation in acute neuronal tissue. PLoS One 11(5): e0155468.
- Cameron, M. A., Kekesi, O., Morley, J. W., Bellot-Saez, A., Kueh, S., Breen, P., van Schaik, A., Tapson, J. and Buskila, Y. (2017). Prolonged incubation of acute neuronal tissue for electrophysiology and calcium-imaging. J Vis Exp 15(120).
- Qin, L., Wu, X., Block, M. L., Liu, Y., Breese, G. R., Hong, J. S., Knapp, D. J. and Crews, F. T. (2007). Systemic LPS causes chronic neuroinflammation and progressive neurodegeneration. Glia 55(5): 453-462.
- Reid, K. H., Edmonds, H. L., Jr., Schurr, A., Tseng, M. T. and West, C. A. (1988). Pitfalls in the use of brain slices. Prog Neurobiol 31(1): 1-18.
- Ting, J. T., Daigle, T. L., Chen, Q. and Feng, G. (2014). Acute brain slice methods for adult and aging animals: application of targeted patch clamp analysis and optogenetics. Methods Mol Biol 1183: 221-242.
Article Information
Copyright
© 2020 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Kekesi, O. and Buskila, Y. (2020). Method for Prolonged Incubation of Brain Slices. Bio-protocol 10(14): e3683. DOI: 10.21769/BioProtoc.3683.
Category
Neuroscience > Basic technology > Acute slice preparation
Neuroscience > Basic technology > Cryopreservation
Cell Biology > Tissue analysis > Tissue culture
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link










