- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Quantitative Determination of Ca2+-binding to Ca2+-sensor Proteins by Isothermal Titration Calorimetry
Published: Vol 10, Iss 7, Apr 5, 2020 DOI: 10.21769/BioProtoc.3580 Views: 6339
Reviewed by: Geoffrey C. Y. LauAndrew L. EagleAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Determination of the Intracellular Calcium Concentration in Peritoneal Macrophages Using Microfluorimetry
Silvia González-Ramos [...] Lisardo Boscá
Dec 5, 2013 18128 Views
Cation (Ca2+ and Mn2+) Partitioning Assays with Intact Arabidopsis Chloroplasts
Anna Harms [...] Anja Schneider
Jan 5, 2017 9047 Views
Abstract
Diverse and complex molecular recognitions are central elements of signal transduction cascades. The strength and nature of these interaction modes can be determined by different experimental approaches. Among those, Isothermal titration calorimetry (ITC) offers certain advantages by providing binding constants and thermodynamic parameters from titration series without a need to label or immobilize one or more interaction partners. Furthermore, second messenger homeostasis involving Ca2+-ions requires in particular knowledge about stoichiometries and affinities of Ca2+-binding to Ca2+-sensor proteins or Ca2+-dependent regulators, which can be obtained by employing ITC. We used ITC to measure these parameters for a set of neuronal Ca2+-sensor proteins operating in photoreceptor cells. Here, we present a step wise protocol to (a) measure Ca2+ interaction with the Ca2+-sensor guanylate cyclase-activating protein 1, (b) to design an ITC experiment and prepare samples, (c) to remove Ca2+ nearly completely from Ca2+ binding proteins without using a chelating agent like EGTA.
Background
Isothermal titration calorimetry (ITC) is a label free method to investigate bio-molecular interaction processes by measuring heat release or heat consumption during a binding process. These processes could occur between different forms of molecules including macromolecules and/or small ligands. The principle design of the measuring device consists of two cells (a reference and sample cell) in an adiabatic jacket. One binding partner is present in the sample cell, while the other interacting molecule is injected into the sample cell. The reference cell contains a buffer solution of water. Temperature differences between sample cell and reference cell are sensed by detectors and an equal temperature between both cells is maintained by a combination of a heater system and a feedback circuit. Interaction of two molecule species either releases or consumes heat, which is detected based on the heater/feedback system. ITC can be applied for example to study a wide range of interaction processes including protein-protein (Pierce et al., 1999), protein-carbohydrate, protein-DNA, enzyme-substrate and cation-protein (Wiseman et al., 1989; García-Hernández et al., 2003; Kozlov and Lohmann, 2012; Mazzei et al., 2014). Alternative methods like surface plasmon resonance (SPR), back scattering interferometry or fluorescence-based techniques utilize different principles and each technique has its own advantages and disadvantages. SPR requires immobilization of one binding partner and fluorescence would in most cases need site-specific labelling. ITC is a method of choice to measure direct ion binding to proteins for deriving binding constants. Furthermore, ligand binding or protein-protein interactions can be studied without the need to label or immobilize one binding partner and has also shown promises in drug discovery science (Ward and Holdgate, 2001; Weber and Salemme, 2003; Lim et al., 2009; Sulmann et al., 2015). The values extracted from ITC measurements are the binding constants (Ka), binding stoichiometry (n), free energy (ΔG), enthalpy (ΔH) and entropy (ΔS). In another words, it provides thermodynamic parameters in addition to binding constants. Since Ca2+ ions play important roles in intracellular signaling, it’s important to understand the intracellular detection of Ca2+ by Ca2+-sensor proteins and to investigate the interaction processes in quantitative terms. Among Ca2+-sensors, one subgroup of neuronal Ca2+-sensor (NCS) proteins is important for the primary processes of vision in photoreceptor cells, where they are involved in Ca2+-feedback mechanisms to control deactivation processes and sensitivity of photoreceptor cells to light. Guanylate cyclase-activating proteins (GCAPs) are NCS proteins that regulate the activity of membrane bound guanylate cyclases in a Ca2+-dependent manner, thereby controlling the cytoplasmic concentration of cyclic GMP, the second messenger of photoexcitation in photorecepotor cells. Several point mutations in GCAP1 lead to severely impaired vision or blindness and amino acid substitutions in different positions appear to cause some common features (Marino et al., 2015; Vocke et al., 2017; Marino et al., 2018; Peshenko et al., 2019). For example, mutant GCAP1 proteins undergo different conformational changes due to impaired Ca2+-binding, and it often results in constitutive activity of the GCAP1/guanylate cyclase complex. Eventually it causes an imbalance of the Ca2+/cyclic GMP homeostasis. ITC has been applied for measuring the binding of Ca2+ and Mg2+ to GCAPs (Lim et al., 2009; Sulmann et al., 2015; Peshenko et al., 2019), but also to recoverin (Dell'Orco et al., 2010; Abbas et al., 2019), calmodulin (Beccia et al., 2015) visinin-like protein 3 (Li et al., 2016) neuronal calcium sensor 1 (NCS-1) (Tsvetkov et al., 2018) and other Ca2+ sensor proteins. An alternative method specifically developed for measuring Ca2+-protein bindings is the Chelator assay, which utilizes the presence of a detectable and titrable Ca2+-chelator (e.g., BAPTA). It measures the binding between the protein of interest and free Ca2+ (Linse, 2002). Both techniques are to some degree complementary and have been used in combination in a recent publication (Elbers et al., 2018).
In this protocol, we provide a step-by-step description for using ITC in Ca2+- and Mg2+-binding studies. Since Ca2+-sensor proteins can contain sometimes binding sites of very high affinity, it is a challenging task to remove bound Ca2+-ions nearly completely from the protein. Therefore, we also provide a protocol to decalcify Ca2+-sensor proteins without using a chelating substance like EGTA. This protein decalcification protocol can be used for preparing Ca2+-binding proteins used in other techniques (surface plasmon resonance, backscattering interferometry, and Chelator assay). In general, the protocol defines the essentials to design an ITC experiment and analysis of ITC data.
Materials and Reagents
- Pipette tips 10 µl (Sartorius)
- Pipette tips 200 µl (Sartorius)
- Pipette tips 1,000 µl (Sartorius)
- Pipette set (SARSTEDT, Nümbrecht)
- Standard Falcon tubes 15 ml (DARSTEDT AG & CO.KG, catalog number: 9042012)
- Standard Falcon tubes 50 ml (DARSTEDT AG & CO.KG, catalog number: 9042211)
- Polypyro 0.2 µm membrane filters (Pall Corporations, catalog number: 32771646)
- Dialysis membrane (SERVA, dialysis tubing, MWCO 12,000-14,000 RC, diameter 21 mm catalog number: 44144.02)
- Ultracentrifuge tubes, 30 ml (G-Kisker GbR product, UZ-PC-38.5-2)
- Photometer cuvettes (Semi-micro Q-VETTES, 1.6 ml PS, Ratiolabs, catalog number: 2712120 )
- Disposable vinyl gloves (Semper guard)
- Plastic bottle, 100-500 ml, local supplier
- Omnifix® Solo, B.Braun 5 ml syringe
- PD10 column (GE Healthcare, catalog number: 28925232A); 2-3 used ones
- Ethanol (Sigma-Aldrich, catalog number: STBJ2652)
- Sodium dodecyl sulfate (SDS)
- HEPES (Roth, catalog number: 9105.2 )
- EGTA (Roth, catalog number: 3054.3)
- KCl (Apllicem, catalog number: 7447.40.7)
- NaCl (Sigma-Aldrich, catalog number: STB117768)
- NaOH (Fisher Scientific, catalog number: 1310.732)
- Dithiothreitol (DTT)
- Tris or trisaminomethane (Acros Organics, catalog number: A0394024)
- Chelex material (Chelex 100® sodium from Sigma-Aldrich)
- Guanidine HCl (Roth, catalog number: 6069.3)
- LB medium (Roth, catalog number: X964.4 )
- Ampicillin (Roth, catalog number: K029.2 ), store at 4 °C
- Kanamycin (Omnilife science) store at 4 °C.
- Ammonium sulphate (Roth, catalog number: 3746.1 )
- Bradford reagent (Bio-Rad, catalog number: 500-00069, store at 4 °C.
- Myristic acid (Fluka, catalog number: BCBJ9925L), store at 4 °C.
- BAPTA (1,2 Bis (2-aminophnoxy) ethane-N,N,N’,N’-tetra acetic acid tetra sodium salt) from Sigma-Aldrich
- Calcium standard solution, 10 g/L (WTW D-82362 Weilheim; https://www.fishersci.de/shop/products/es-ca-model-standard-solution/10099770)
- Urea (Roth, catalog number: X999.3 )
- Kimmwipes® lite (Kimberly-clark)
- Methanol (Sigma-Aldrich, catalog number: STBH6784)
- Decon 90 (ITC system cleaning detergent) (Decon Laboratories Limited)
- Anion exchange chromatography buffer A (see Recipes)
- Anion exchange chromatography buffer B (see Recipes)
- Size exclusion chromatography buffer (see Recipes)
- ITC buffer (see Recipes)
Equipment
- Size exclusion chromatography column (Hiload 26/60 superdex 75, GE, catalog number: 17-1070-01)
- Anion exchange chromatography column (Hiload 26/10 Q sepharose High performance, GE, catalog number: 17-1066-09)
- Glass beaker (100-200 ml, Schott Duran)
- Water bath (Memmert)
- -80 °C freezer (Thermos Electron Corporation)
- Vortex (Vortex Genie2)
- Centrifuge (Backman Coulter, Avanti J-E centrifuge, Rotor: 70Ti)
- Ultracentrifuge (Kendro Laboratory product, Rotor: AT4)
- Ultracentrifuge (SORVALL Discovery M120SE, Rotor: JA 25.50)
- Photometer (Food ALYT OMNILAB, 122197)
- UV-Vis spectrophotometer (UV-Vis SpecORD 205, 5BU1768)
- Äkta start protein purification system (GE Healthcare Life Science, catalog number: 29003181)
- Solvent filtration device (Nalgene)
- Degasser (thermoVac microcal)
- Isothermal titration calorimetry system (VP-ITC microcalorimetry, Microcal)
Software
- Origin®, Microcal, Northampton, MA https://www.malvernpanalytical.com/en
- WinASPECT spectroanalytical software https://www.analytik-jena.de/
Procedure
A general overview of the workflow is shown in Figure 1.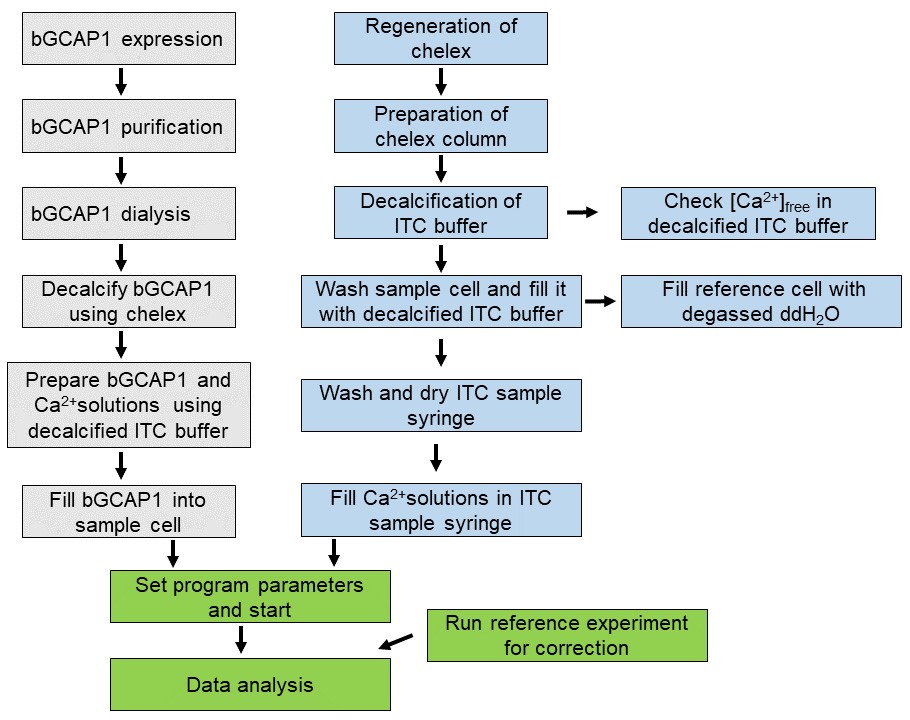
Figure 1. Workflow summary of experimental steps for determination of Ca2+-binding to bGCAP1 by ITC
- Protein expression
Note: Protein expression and purification is not in the main focus of this report, see some steps below for a general understanding.- Use E. coli cells (BL21-CodonPlus, Stratagene) containing plasmid with bovine GCAP1 (bGCAP1) sequence for overnight culture (v = 5 ml).
- Put 5 ml culture in 500 ml of LB medium to express protein in cells for up to 4 h after induction.
- Harvest the cells by centrifugation (5,000 x g, 10 min, 4 °C). Resuspend cell pellet in 50 mM Tris Buffer with pH 7.5 (20 ml for pellet resulting from a bacterial culture of 500 ml).
- The protein was purified as described previously (Hwang and Koch, 2002; Koch and Helten, 2008) using a two-step chromatography protocol (size exclusion chromatography and anion exchange chromatography).
- bGCAP1 dialysis
- Run polyacrylamide gel electrophoresis (PAGE) using sodium dodecyl sulfate (SDS) 12% polyacrylamide gel to confirm the protein purity (see image of gel below, Figure 2).
- Prepare (activate) the dialysis membrane with 5 mM EGTA using the following steps:
- Cut the membrane if necessary (10 cm should be sufficient) and put in a glass beaker filled with double-distilled water (ddH2O) and boil in microwave until water is boiling.
- Remove the hot water and fill with fresh water and add 5 mM EGTA and boil again.
- Remove the EGTA containing water and repeat the Step B2b twice.
- Store this activated membrane in a glass beaker with EGTA free water in a cold room at 4 °C. Storage under this condition is possible for a maximum of one month.
Note: Never store the activated dialysis membrane without water. Never touch the dialysis membrane without gloves.
- Prepare dialyzing buffer (from now until ITC buffer, see below under “Recipes”) with pH 7.5.
- Take one fraction of freshly purified bGCAP1 protein after anion exchange chromatography (maximum volume 10 ml). See fraction the gel image (Figure 2).
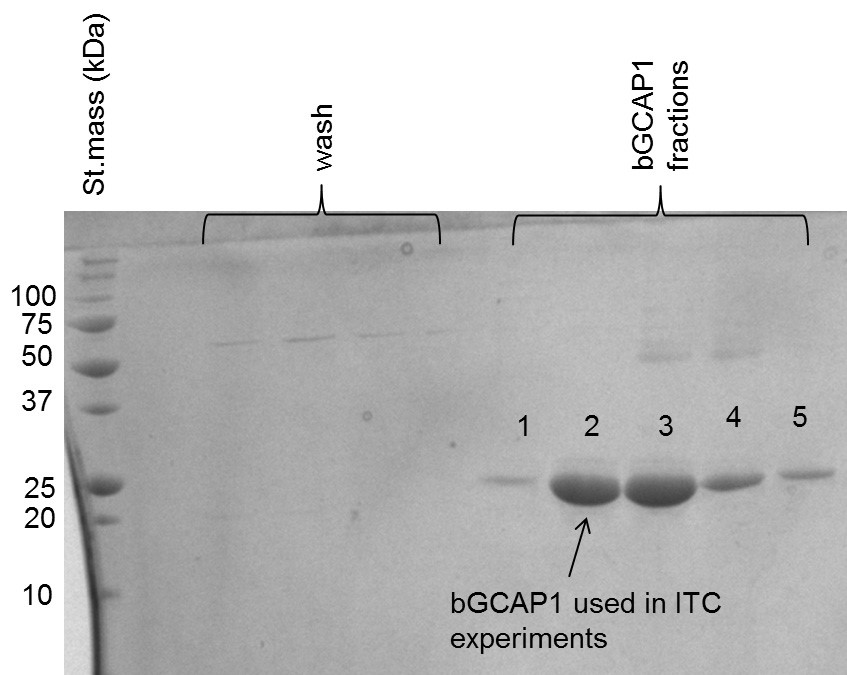
Figure 2. SDS-PAGE analysis of bGCP1 fractions. Fractions obtained by anion exchange chromatography are analyzed, “wash” indicates the non-bound fractions, 1-5 are eluted bGCAP1 fractions. Sample #2 was of highest purity (≥ 95%) and was used in ITC experiments. - Put on gloves and take the activated membrane. Put the knot at one side of the membrane (you can also use stoppers to avoid protein leakage during dialysis but doing a knot is more effective), fill the membrane with 10 ml bGCAP1 protein using a pipette. Now put another knot to the other side of the membrane and then add the stoppers to both ends (see Figure 3).
- Place the protein filled membrane into ITC buffer for overnight (minimum 3 L buffer for 10 ml of bGCAP1 solution).

Figure 3. Assembling of dialysis membrane. Positions of knots and stopper are indicated.
- Prepare chelex columns (anion exchange material for removing cations)
- Before use, the chelex material must be regenerated, regardless whether it was used before or freshly obtained from the supplier. For this purpose, a solvent filtration device is best suited. The device contains a filter holder and a chamber on top of it. The filtration device is commonly used for filtration of solutions via a pump. Further, a polypyro 0.2 µm membrane filter is needed. It is placed on a holder (part of the commercial filtration device) and keeps the chelex resin on top of it, when a vacuum pump is turned on and a washing solution is sucked through the filter. Thereby, it prevents the chelex material flowing into waste. The steps of the regeneration and washing procedure are as follows:
- Fill the chamber on top of the filter holder with chelex and wash with 500 ml of double-distilled water.
- Wash the chelex with 200 ml of 1 M HCl.
- Wash chelex with 500 ml of double-distilled water.
- Wash chelex with 200 ml of NaOH.
- Wash chelex with 500 ml of double-distilled water again. Suspend in double-distilled water and transfer the chelex material to a plastic bottle.
- Prepare a plastic bottle for chelex storage. Wash the bottle with a solution of 5 mM EGTA. Rinse with water before storing regenerated chelex.
- Adjust the pH of the chelex suspension, it should be neutral (around 7). Otherwise it might harm the protein function of bGCAP1. Adjusting the pH of the chelex material can be a very difficult and time consuming. See Notes for some tips.
- Take a used PD10 column and remove the column material (normally a gel filtration resin) and discard it. There is a small bed at the bottom of the PD10 column. Leave it inside the column. Use 6 M urea to clean the column in case the flow is slow. Fix the column to the column holder and fill it with chelex that is stored in water now (Figure 4). Let the water flows out and all chelex material stays inside the column. Your chelex bench column is ready to use.

Figure 4. Filling chelex material into a used (“old”) PD10 column. A. The PD 10 column is fixed by a conventional column holder. Chelex can be seen as white material in the pipette tip. It is filled into the PD10 column. B. Chelex is settling by gravity flow. Water or buffer will pass the bed or small platform at the bottom, which retains the chelex material. Any kind of tube can be used for collecting the flow-through.
- Before use, the chelex material must be regenerated, regardless whether it was used before or freshly obtained from the supplier. For this purpose, a solvent filtration device is best suited. The device contains a filter holder and a chamber on top of it. The filtration device is commonly used for filtration of solutions via a pump. Further, a polypyro 0.2 µm membrane filter is needed. It is placed on a holder (part of the commercial filtration device) and keeps the chelex resin on top of it, when a vacuum pump is turned on and a washing solution is sucked through the filter. Thereby, it prevents the chelex material flowing into waste. The steps of the regeneration and washing procedure are as follows:
- Remove Ca2+ from bGCAP1 (decalcify)
- Use a pipette for removing bGCAP1 out of the dialysis membrane after overnight dialysis and transfer it to a 15 ml Falcon tube. Check protein concentration with Bradford protein quantification method (Bradford, 1976). Be aware that Coomassie dye-based protein determination methods depend on the nature of the protein used for calibration.
Tip: Use a protein specific calibration curve. Create this curve by using your protein of interest as standard. Prepare standard concentrations (1-10 µg/ml) by using a second independent method of protein determination for example measuring the UV absorbance. Determine the molar extinction coefficient (Pace and Schmid, 1997) and employ it by using the Lambert-Beer law; see below Step E4. - Take the chelex bench column and apply bGCAP1 (If the total column volume is 10 ml, 5 ml should be the bGCAP1 solution and 5 ml be the chelex material). Mix chelex with bGCAP1 inside column with slight hand rotations. Either leave the column in a cold room for half an hour or place it for end to end rotation in a cold room keeping bGCAP1 in direct contact with chelex.
Note: It is necessary that bGCAP1 is in direct contact with the chelex material for removing Ca2+. - Collect bGCAP1 from the column in Eppendorf tubes (2 ml) and label the tubes with protein concentrations after measurement.
- Use a pipette for removing bGCAP1 out of the dialysis membrane after overnight dialysis and transfer it to a 15 ml Falcon tube. Check protein concentration with Bradford protein quantification method (Bradford, 1976). Be aware that Coomassie dye-based protein determination methods depend on the nature of the protein used for calibration.
- Decalcify the ITC buffer
- Prepare another chelex bench column with fresh chelex and fix the column in a column holder.
- Prepare plastic bottle (Ca2+-contamination might also originate from plastic material) to collect decalcified ITC buffer as following:
- Take any 100-200 ml plastic bottle and wash it with 1 M HCl.
- Rinse the bottle with water.
- Wash bottle again with 5 mM EGTA.
- Rinse the bottle with double-distilled water.
- Position the Ca2+-free plastic bottle under the column and pass 50 ml of ITC buffer (to avoid buffer mismatch prepare a larger stock of dialysis/ITC buffer).
- Check how much Ca2+ is left in decalcified buffer by determining free Ca2+ in the buffer using BAPTA as indicator. Install WinASPECT spectroanalytical software or use any other running software for a spectrophotometer. Open the program and initialize the program by turning on the UV-lamp. Set the lamp wavelength at 254 nm. Start absorbance measurement with reference sample that contains only buffer. Afterwards, measure absorbance of buffer mixed with BAPTA for three times (separate replicates) and calculate the average.
- Buffer preparation for BAPTA measurements:
- Take 500 µl of decalcified ITC buffer for reference measurement.
- Take 495 µl buffer and mix with 5 µl of BAPTA from 1 mM stock.
- vortex the sample.
- Calculate free Ca2+ in buffer with equations listed below:
Total [BAPTA] = [BAPTAf] + [CaBAPTA]
[CaBAPTA] = [Ca2+f][BAPTAf]/KD(Ca)
KD(Ca) = 107 nM; dissociation constant of Ca2+ binding to BAPTA under conditions of the ITC buffer (room temperature 22 ± 2 °C; Tsien, 1980).
As a first step, determine the concentration of Ca2+-free BAPTA ([BAPTAf] by measuring the absorbance at 254 nm. According to Lambert-Beer's law A = ε × c × l, calculate c, concentration of [BAPTAf]; the other parameters are extinction coefficient ε = 1.6 × 104 M-1 × cm-1 and l = length of cuvette (e.g., 1 cm). With [BAPTAf] and the known concentration of total [BAPTA] (e.g., 20 µM) one can calculate [CaBAPTA].
For example, the average value of absorbance might be 0.095.
With A = ε × c × l we obtain
0.095 = 1.6 × 104 M-1 × cm-1 × 1 cm
and for concentration c = 5.9 µM
The concentration of Ca2+-bound to BAPTA is
[CaBAPTA] = total [BAPTA] - [BAPTAf]
= 10 µM - 5.9 µM
= 4.1 µM
leading to
[Ca2+f] = 0.107 µM × 4.1 µM/5.9 µM
[Ca2+f] = 0.074 µM or 74 nM
A concentration of [Ca2+f] of 74 nM might be the lower limit, which can be achieved. A concentration of [Ca2+f] below 80 nM in the absence of Ca2+-chelating reagents like EGTA might be sufficient for most titrations. One can repeat the whole buffer decalcifying process to achieve this lower limit. - Prepare the ITC system
- Wash the sample cell (the cavity for GCAP1 protein inside ITC instrument, Figure 5) with 5% Decon (system cleaning detergent). Start with 100 ml of 5% Decon followed by 400 ml of double-distilled water. Remove residual water from the sample cell by inserting the loading syringe (Figure 6) and wash the sample cell with decalcified ITC buffer. Afterwards, fill the sample cell with decalcified ITC buffer and leave it until protein sample is ready. This helps to equilibrate the sample cell and also removes free Ca2+.
- Wash the ITC sample syringe that is part of the ITC system (Figure 5) and automatically controlled by software. It contains the ligand or Ca2+ in our case. Attach a 5 ml small syringe piston (an Omnifix small 5 ml syringe or reciprocating pump, see materials) to the sample syringe port with small plastic tube (Figure 5A). Fill a special ITC tube that comes with the ITC instrument with 5% Decon and fix it in holder under the ITC sample syringe (during titrations the ITC tube carries water to wash the syringe). Use the syringe piston to catch the 5% Decon after passing through the sample syringe. Repeat the same process with at least 30 ml of water to remove any remaining detergent inside.
- Dry the ITC sample syringe. Remove the ITC tube from the syringe holder and let syringe tip stay in air. Use the small syringe piston to pull air through the sample syringe. Remove all drops of waters by repeating the process several times.
Tip: Click purge and refill button from software, this process will bring all the water at syringe tip and place a kim wipe at the tip to catch all water.
Figure 5. ITC system. A. The ITC sample syringe is connected to the Omnifix small syringe for the washing step (see text).The red arrow indicates top of the cavity that contains the sample and reference cells. For the titration, the whole sample syringe is moved to the opening of the cavity and positioned in a way that the long metal tip (blue arrow in right panel) is inserted. B. The sample tube for filling the Ca2+-solution into the sample syringe.
Figure 6. Image of the loading syringe
- Prepare protein sample for sample cell, syringe and reference cell
- Sample cell: Calculate how much bGCAP1 is required for one measurement (20 µM final concentrations; see Note #17 for calculation of optimal ratio of ligand to protein), add the rest of decalcified ITC buffer to reach final concentration. Prepare another chelex column with at least 2 ml of chelex material inside. Add the protein sample to the column and leave it on ice.
- Prepare the sample for filling the ITC sample syringe (Ca2+ in our case): use calcium standard solution, which is 10 g/L (WTW D-82362 Weilheim) and dilute with decalcified ITC buffer to prepare a Ca2+ solution with a final concentration of 0.5 mM.
- Collect the bGCAP1 sample from the chelex column in a tube (Tip: every time wash the tubes with decalcified buffer before collecting a bGACP1 sample from the column to avoid any contamination of free calcium that might come from the tube walls). Put small magnet stir bars in the tubes and degas both samples (Ca2+ and bGCAP1 solution) for at least 10 min. Set the degasser temperature at 25 °C as the experiment is also performed at 25 °C. Degasser is also part of ITC system and provided by ITC suppliers.
- Reference cell (second cavity inside ITC instrument to hold water or buffer as reference): Fill double-distilled water in a fresh tube and degas it for 10 min. Transfer the water to the reference cell with a loading syringe (the loading syringe is only used to load a protein solution into the sample cell and water or buffer into the reference cell). The syringe also comes with ITC instrument (be very careful to avoid adding air bubbles in cells).
- Load samples
- Fix the tube containing the Ca2+-solution under the ITC sample syringe and load sample very carefully (Figure 5B). Sometimes a few air bubbles are left inside the sample holder. To avoid trapping of air bubbles, click close port button in the software and pull the piston very carefully at the same time. (This saves sample and avoids adding air bubbles). Click purge and refill button in the software twice to release the entire sample back into the tube and let it slowly load again. This trick removes all small air bubbles.
- Wash the loading syringe with water and remove the entire decalcified ITC buffer from the sample cell, which was used to wash the sample cell. Remove the buffer from the loading syringe and make sure there is no buffer left in the sample cell. Do not touch the loading syringe with anything at this moment and fill it with bGCAP1 sample after degassing. Insert the loading syringe needle into sample cell and release the sample slowly. (Tip: loading sample with the loading syringe can easily create small air bubbles that can affect the measurement. To avoid loading air bubbles proceed as follows: when the sample is visible in the sample reservoir, load back a small volume and release with a short pressure pulse so that all bubbles appear on the top, then remove them). Remove all excess sample from the sample reservoir.
- Start the titration experiment
- Hold the ITC sample syringe in one hand and wash its tip with decalcified ITC buffer (the syringe tip was inside the Ca2+ solution and we need to prevent any contamination of bGCAP1 sample with Ca2+ before starting the experiment). Use a minimum of 1 ml decalcified ITC buffer to wash the syringe tip. Afterwards, remove extra buffer drops around the syringe tip with kimwipes and place the syringe to the sample cell.
Note: Do it very slowly and be very careful not to hit the tip with anything that can bend the tip easily. - After loading to the sample cell, take a small volume (a few µl) of bGCAP1 to check the final protein concentration in case it might have been changed due to previous steps (GCAP1 sample might be diluted due to passing over the chelex column). It is important to mention the correct protein concentration to the software before starting the experiment.
- Set program parameters as following:
Total number of injects = 50
Cell temp °C = 25
reference power (µcal/s) = ideal is between 3-10
cell concentration (mM) = 0.02
syringe concentrations (mM) = 0.5
injection volume (µl) = 5
Initial delay (s) = 210
Injection space time (s) = 60
Feedback mode/gain = High
Stirring speed (rmp) = 307
See the following link for more details about ITC instrument.
https://ctrstbio.org.uic.edu/manuals/vpitc_manual.pdf
- Hold the ITC sample syringe in one hand and wash its tip with decalcified ITC buffer (the syringe tip was inside the Ca2+ solution and we need to prevent any contamination of bGCAP1 sample with Ca2+ before starting the experiment). Use a minimum of 1 ml decalcified ITC buffer to wash the syringe tip. Afterwards, remove extra buffer drops around the syringe tip with kimwipes and place the syringe to the sample cell.
- Measurement of reference experiments
Reference experiments are necessary for data analysis to correct the any heat change by dilution. These can originate from diluting the ligand into buffer or from injecting a buffer solution into the macromolecule (bGCAP1) solution, whereby dilution of the ligand into the buffer solution is more significant, because ligand concentrations are typically 20 times larger than that of macromolecules.- Load Ca2+ solution in ITC sample syringe with the same concentration as above (0.5 mM) and load (fill) decalcified ITC buffer in the sample cell. Record the heat change of dilution from ligand injection into buffer.
- Load protein solution in sample cell with same concentration of bGCAP1 (20 µM) and load decalcified buffer to the syringe. Record the heat change of dilutions from buffer into macromolecule titration as outlined above.
Data analysis
- Binding isotherms are normalized as kcal/mol of ligand and plotted against the molar ratio of macromolecule (Pierce et al., 1999). For data analysis, use Origin software provided by Malvern MicroCal and upload the experimental and reference files from the corresponding drive of the computer. The experimental isotherm contains heat changes of dilution and also contributions of heat changes from stirring. To correct for heat of dilution it is important to substract the reference isotherm from the experimental isotherm. For this purpose, it is important that all concentrations are the same, when both experimental files are recorded. For example, when bGCAP1 concentration is 20 µM and Ca2+ is 0.5 mM, in the reference experiment Ca2+ need to be 0.5 mM and the sample cell must contain the corresponding buffer solutions (indicating a cell concentration of 0 at the time of experiment). Now change the cell concentration to 20 µM as for bGCAP1. Subtract the reference file from experiment that will correct the heat changes originating from dilutions. Afterwards, select a fitting model to fit the ITC binding isotherm based on the number of ligands that bind to the molecule present in the measuring cell. Based on experimental and theoretical evidence one GCAP molecule can bind up to three Ca2+ ions at EF-hand Ca2+-binding sites EF2, EF3 and EF4 (Hwang et al., 2004; Peshenko et al., 2006; Dell’Orco et al., 2010). Selecting the sequential binding model to fit the data will provide fitting parameter for more than two binding sites. One can select as many number of ligands in the sequential binding model as seems appropriate. The Origin software provides two other fitting models (one site model and two site model) that do not account for putative cooperativity between the single binding steps of a ligand.
- Processing the raw data by applying the selected fitting model will yield an integrated binding isotherm as shown in the lower panel of Figure 7. Two parameters of the binding process can be extracted, the change in enthalpy ΔH and the equilibrium constant KA (or 1/KA = KD, dissociation constant). These parameters allow a full thermodynamic characterization according to
ΔG = -RT InKA and ΔG = ΔH - TΔS
providing values for free energy (ΔG) and entropy (ΔS). R is the gas constant and T is temperature.
For bGCAP1 three titrations series are performed on a VP-ITC from MicroCal (Northhampton, MA) to determine the Ca2+ binding affinities. Measurements are done without Mg2+. Heat responses are both exothermic and endothermic for bGCAP1.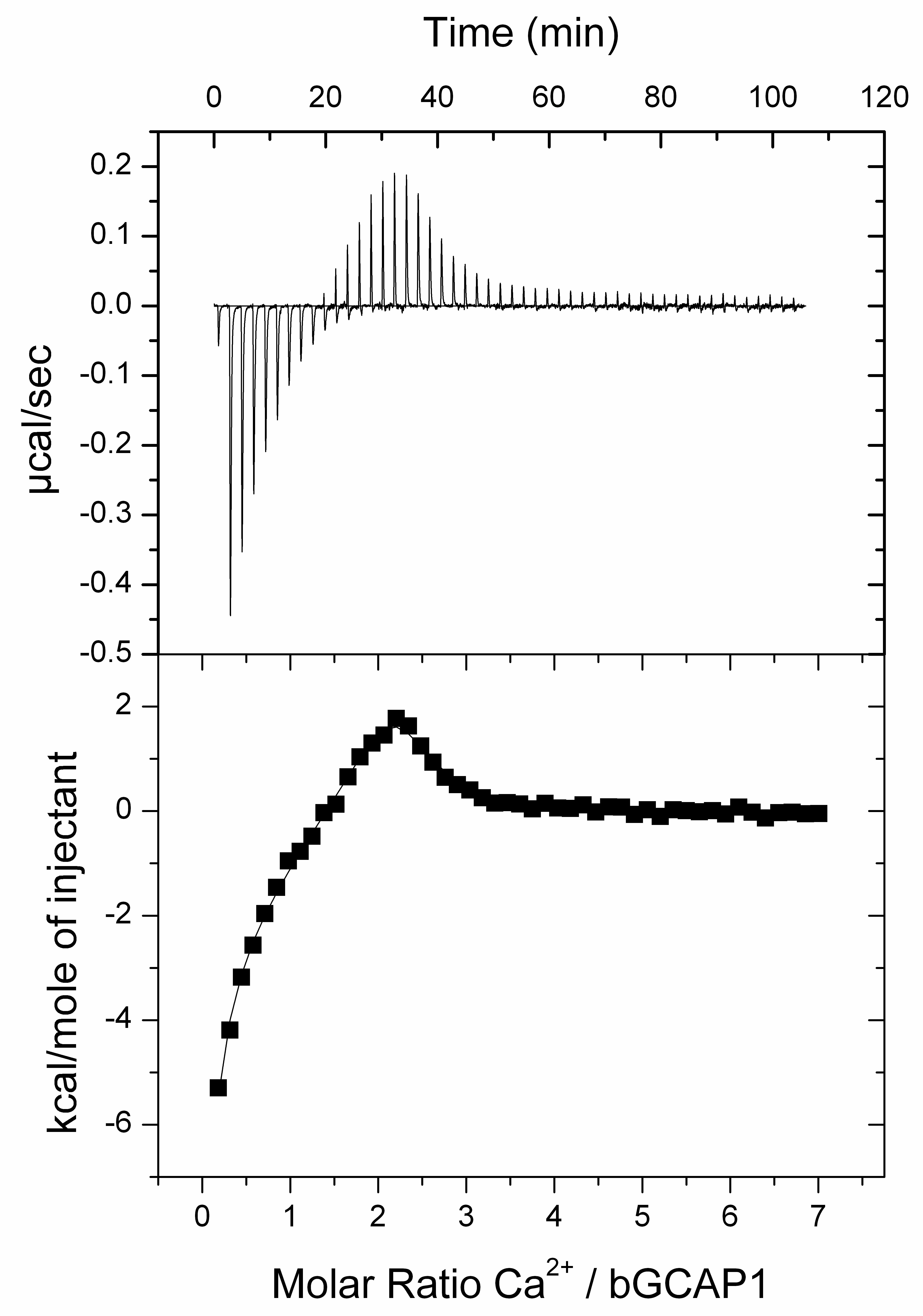
Figure 7. Ca2+-binding to bGCAP1. ITC titration measured with 20 µM bGCAP1. Upper panel shows heat release upon Ca2+-injections as described. Lower panel shows the integrated binding isotherm fitted with the three site sequential binding model plotted as a function of increasing bGCAP1 concentration (see also Note #19). Data analysis after curve fitting yielded the equilibrium constants KA, the dissociations constants KD derived from KA and enthalpy changes (ΔH), all shown in Table 1.
Table 1. Thermodynamic parameter of Ca2+-binding to bovine GCAP1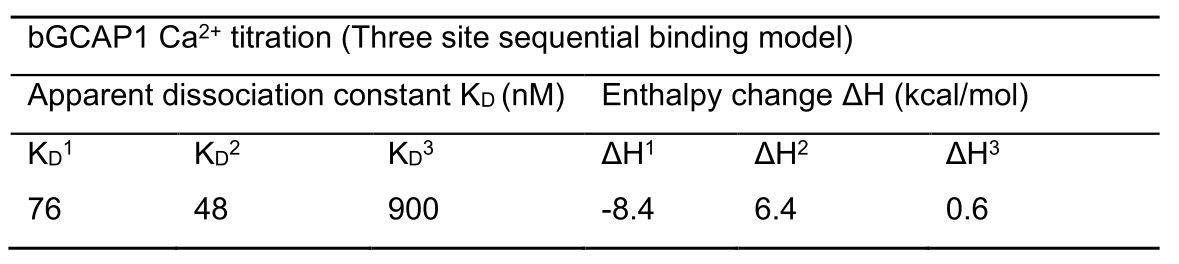
Comments
Heat release that is detected by Ca2+-binding to bGCAP1 was both exothermic (negative peak) and endothermic (positive peak). It was similar to an ITC study by Lim et al. (2015), but differed from recent ITC data observed with human GCAP1 (Figure 7A in Peshenko et al., 2019), where strong exothermic responses were observed and occasionally much smaller endothermic responses. However, Peshenko et al. (2019) fit their Ca2+-binding data to a sequential binding model resulting in two apparent dissociation constants. They interpreted their findings with a high affinity binding site in human GCAP1 that is partially or fully occupied in the presence of residual free Ca2+ in their experiments (90-200 nM). Species differences might account for different Ca2+-affinities, which are also observed with previously published data for zebrafish GCAPs in comparison to mammalian GCAP forms (Figure 5 in Sulmann et al., 2015).
Mg2+-binding to Ca2+-binding proteins can be investigated by the same protocol (see for example Sulmann et al., 2015 and Peshenko et al., 2019).
Notes
- Buffer mismatch can cause a significant experimental error, which can be minimized or avoided by overnight protein dialysis and extensive and careful washing of the system.
- Choice of buffer can be very critical for ITC experiments. Some buffers (e.g., Tris) show large enthalpy changes of ionization. Thus, measured enthalpy values are a combination of enthalpy coming from complex formation of ligand and macromolecule plus enthalpy of buffer ionization. Use phosphate buffers or HEPES buffer for initial experiments. The ionization enthalpy of sodium phosphate buffer is significantly less than of Tris buffer (1 kcal/mol vs. 11 kcal/mol) (Pierce et al., 1999).
- Boil dialysis membrane twice in water to make sure EGTA is removed after membrane activation.
- Chelex of white cloudy color indicates when it is ready to use during the regeneration process.
- To adjust the pH of the chelex suspension it is better to start with high molar concentration of HCl or NaOH. When pH is around 6 or 8, use solutions of lower molar concentrations like 1 M HCl or NaOH.
- If the bed (or platform) of the used and recycled PD10 column is clogged and flow of buffer is really slow, apply 6 M urea inside column and leave it for few minutes. Afterwards wash column extensively with water until flow has increased.
- During decalcifying of ITC buffer, pass the same 50 ml twice through the same column to remove the maximum amount of Ca2+.
- Always set ITC thermostat temperature at 25 °C shortly after turning on the system before starting with other steps of the procedure to save time. Sometimes it takes very long to reach the required temperature.
- During set-up of the ITC system bring three plastic beakers, beaker A with double-distilled water, beaker B with double-distilled water and beaker C for waste. Reason to keep two beakers for water is that every time when washing the system, first wash the syringe in beaker A, then take water from beaker B to wash the system. Proceeding like this avoids contamination of the system with dirty particles.
- Never switch off the system when it is running, ITC syringe will immediately releases all the syringe solution into the sample cell. If something is going wrong, an initial delay can be used to stop the reaction and to start again.
- Be very careful for air bubbles when loading sample, which is why degassing samples twice is effective. Air bubbles can cause poor baseline and can also effect the feedback circuit between reference and sample cells. Do not pipet up and down the protein samples too often as this can create air bubbles.
- When removing Ca2+ from proteins, passing protein twice to chelex column can increase the efficiency.
- Reference cell can be filled with ITC buffer or just water without macromolecule. Water is an ideal choice, because cleaning of the reference cell is a major issue and often neglected.Double-distilled water brings in much less contamination.
- Load fresh buffer or water in the reference cell on a weekly basis when instrument is in constant use. It is not necessary to change buffer or water in the reference cell after every experiment.
- Always calibrate your pipettes before sample preparations and always use the same pipette set. The ITC experiment is very sensitive concerning any concentration mismatch.
- After finishing experiment wash the system as explained above and fill 300 µl of 100% methanol (HPLC grade) into the sample cell. Close the fill port of the syringe before switching off the system.
- To obtain a sigmoidal curve from ITC measurements always check the combination of protein and ligand concentrations. Syringe ligand should be 20 times more concentrated than protein sample in the sample cell. The following equation can be used for designing an experiment. Unitless value c can be in a range from 1 to 1,000. Always increase your protein concentration or decrease the ligand concentration, if c value is around 1. High c values facilitate the determination of an association constant (Ka) (Wiseman et al., 1989).c = Ka [M] n
[M] is the concentration of the macromolecule
Ka determines the binding constant for the protein of interest and the ligang, if already known or can be guessed;
n determines the number of ligands binding to the macromolecule. - It is important to measure both the ligand and macromolecule concentration with high degree of accuracy when determing association constants (Ka).
- Fitting of data to sequential binding sites using the Origin software is based on Fisher and Singh (1995) and ITC Data Analysis in Origin–Tutorial Guide, Version 7.0 (2004) (Reference 10). Briefly:
Binding constants K1, K2, K3 ….Kn are defined as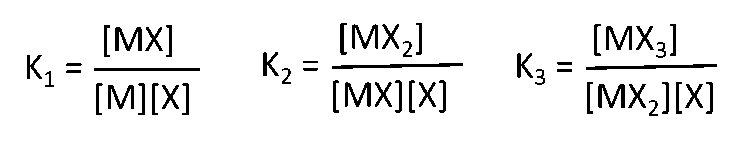
[M] is the free concentration of macromolecule in the active cell volume V0.
[X] is the free concentration of ligand.
The fraction of total macromolecule having n bound ligands, Fn, is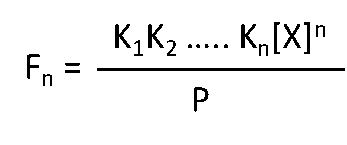
Wherein P is defined as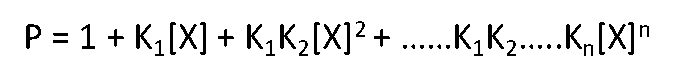
The heat absorbed or evolved after the nth injection is given by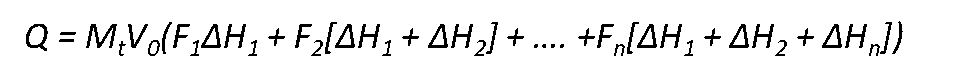
Mt is the bulk concentration of macromolecule.
Recipes
- Anion exchange chromatography buffer A
20 mM Tris/HCl
2 mM EGTA
1 mM DTT
pH 7.5 - Anion exchange chromatography buffer B
20 mM Tris/HCl
2 mM EGTA
1 M NaCl
1 mM DTT
pH 7.5 - Size exclusion chromatography buffer
20 mM Tris/HCl
2 mM EGTA
150 mM NaCl
1 mM DTT
pH 7.5 - ITC buffer
20 mM HEPES
60 mM KCl
4 mM NaCl
pH 7.4 (adjusted with 1 M or 0.1 M KOH)
Acknowledgments
We thank the German Academic Exchange Program (DAAD) and the Deutsche Forschungsgemeinschaft (DFG, grant number GRK1885) for financial support. This protocol was modified from previous work (Sulmann et al., 2015; Peshenko et al., 2019).
Competing interests
The authors declare not competing interest.
Ethics
Experiments were performed with recombinant proteins according to safety regulations in category S1.
References
- Abbas, S., Marino, V., Dell'Orco, D. and Koch, K. W. (2019). Molecular Recognition of Rhodopsin Kinase GRK1 and Recoverin Is Tuned by Switching Intra- and Intermolecular Electrostatic Interactions. Biochemistry 58(43): 4374-4385.
- Bradford, M. M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72: 248-254.
- Beccia, M. R., Sauge-Merle, S., Lemaire, D., Bremond, N., Pardoux, R., Blangy, S., Guilbaud, P. and Berthomieu, C. (2015). Thermodynamics of Calcium binding to the Calmodulin N-terminal domain to evaluate site-specific affinity constants and cooperativity. J Biol Inorg Chem 20(5): 905-919.
- Dell'Orco, D., Muller, M. and Koch, K. W. (2010). Quantitative detection of conformational transitions in a calcium sensor protein by surface plasmon resonance. Chem Commun (Camb) 46(39): 7316-7318.
- Elbers, D., Scholten, A. and Koch, K. W. (2018). Zebrafish recoverin isoforms display differences in calcium switch mechanisms. Front Mol Neurosci 11: 355.
- Fisher, H. F. and Singh, N. (1995). Calorimetric methods for interpreting protein-ligand interactions. Methods Enzymol 259: 194-221.
- García‐Hernández, E., Zubillaga, R. A., Chavelas-Adame, E. A., Vázquez-Contreras, E., Rojo-Dominguez, A. and Costas, M. (2003). Structural energetics of protein-carbohydrate interactions: Insights derived from the study of lysozyme binding to its natural saccharide inhibitors. Protein Sci 12(1): 135-142.
- Hwang, J. Y. and Koch, K. W. (2002). The myristoylation of the neuronal Ca2+ -sensors guanylate cyclase-activating protein 1 and 2. Biochim Biophys Acta 1600(1-2): 111-117.
- Hwang, J. Y., Schlesinger, R. and Koch, K. W. (2004). Irregular dimerization of guanylate cyclase-activating protein 1 mutants causes loss of target activation. Eur J Biochem 271(18): 3785-3793.
- ITC Data Analysis in Origin–Tutorial Guide, Version 7.0 (2004).
- Koch, K. W. and Helten, A. (2008). Chap. 6: Guanylate cyclase-based signaling in photoreceptors and retina. In: Signal Transduction in the retina. Fliesler, S. J. and Kisselev, O. G. (Eds.). Taylor and Francis CRC Press, North Tonawanda, NY. 121-143.
- Kozlov, A. G. and Lohman, T. M. (2012). SSB binding to ssDNA using isothermal titration calorimetry. Methods Mol Biol 922: 37-54.
- Li, C., Lim, S., Braunewell, K. H. and Ames, J. B. (2016). Structure and calcium binding properties of a neuronal calcium-myristoyl switch protein, visinin-like protein 3. PLoS One 11(11): e0165921.
- Lim, S., Peshenko, I., Dizhoor, A. and Ames, J. B. (2009). Effects of Ca2+, Mg2+, and myristoylation on guanylyl cyclase activating protein 1 structure and stability. Biochemistry 48(5): 850-862.
- Linse, S. (2002). Calcium binding to proteins studied via competition with chromophoric chelators. Methods Mol Biol 173: 15-24.
- Marino, V., Scholten, A., Koch, K. W. and Dell'Orco, D. (2015). Two retinal dystrophy-associated missense mutations in GUCA1A with distinct molecular properties result in a similar aberrant regulation of the retinal guanylate cyclase. Hum Mol Genet 24(23): 6653-6666.
- Marino, V., Dal Cortivo, G., Oppici, E., Maltese, P. E., D'Esposito, F., Manara, E., Ziccardi, L., Falsini, B., Magli, A., Bertelli, M. and Dell'Orco, D. (2018). A novel p.(Glu111Val) missense mutation in GUCA1A associated with cone-rod dystrophy leads to impaired calcium sensing and perturbed second messenger homeostasis in photoreceptors. Hum Mol Genet 27(24): 4204-4217.
- Mazzei, L., Ciurli, S. and Zambelli, B. (2014). Hot biological catalysis: isothermal titration calorimetry to characterize enzymatic reactions. J Vis Exp(86). Doi: 10.3791/51487.
- Pace, N. C. and Schmid, F.X. (1997). How to determine the molar absorbance coefficient of a protein. In: Protein Structure – A Practival Approach (2nd Edition). Creighton,T. E. (Ed.). Oxford University Press, pp. 253-259.
- Peshenko, I. V. and Dizhoor, A. M. (2006). Ca2+ and Mg2+ binding properties of GCAP-1. Evidence that Mg2+-bound form is the physiological activator of photoreceptor guanylyl cyclase. J Biol Chem 281(33): 23830-23841.
- Peshenko, I. V., Cideciyan, A. V., Sumaroka, A., Olshevskaya, E. V., Scholten, A., Abbas, S., Koch, K. W., Jacobson, S. G. and Dizhoor, A. M. (2019). A G86R mutation in the calcium-sensor protein GCAP1 alters regulation of retinal guanylyl cyclase and causes dominant cone-rod degeneration. J Biol Chem 294(10): 3476-3488.
- Pierce, M. M., Raman, C. S. and Nall, B. T. (1999). Isothermal titration calorimetry of protein-protein interactions. Methods 19(2): 213-221.
- Sulmann, S., Vocke, F., Scholten, A. and Koch, K. W. (2015). Retina specific GCAPs in zebrafish acquire functional selectivity in Ca2+-sensing by myristoylation and Mg2+-binding. Sci Rep 5: 11228.
- Tsien, R. Y. (1980). New calcium indicators and buffers with high selectivity against magnesium and protons: design, synthesis, and properties of prototype structures. Biochemistry 19(11): 2396-2404.
- Tsvetkov, P. O., Roman, A. Y., Baksheeva, V. E., Nazipova, A. A., Shevelyova, M. P., Vladimirov, V. I., Buyanova, M. F., Zinchenko, D. V., Zamyatnin, A. A., Jr., Devred, F., Golovin, A. V., Permyakov, S. E. and Zernii, E. Y. (2018). Functional status of neuronal calcium sensor-1 is modulated by zinc binding. Front Mol Neurosci 11: 459.
- Vocke, F., Weisschuh, N., Marino, V., Malfatti, S., Jacobson, S. G., Reiff, C. M., Dell'Orco, D. and Koch, K. W. (2017). Dysfunction of cGMP signalling in photoreceptors by a macular dystrophy-related mutation in the calcium sensor GCAP1. Hum Mol Genet 26(1): 133-144.
- Ward, W. H. and Holdgate, G. A. (2001). Isothermal titration calorimetry in drug discovery. Prog Med Chem 38: 309-376.
- Wiseman, T., Williston, S., Brandts, J. F. and Lin, L. N. (1989). Rapid measurement of binding constants and heats of binding using a new titration calorimeter. Anal Biochem 179(1): 131-137.
- Weber, P. C. and Salemme, F. R. (2003). Applications of calorimetric methods to drug discovery and the study of protein interactions. Curr Opin Struct Biol 13(1): 115-121.
Article Information
Copyright
© 2020 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Readers should cite both the Bio-protocol article and the original research article where this protocol was used:
- Abbas, S. and Koch, K. W. (2020). Quantitative Determination of Ca2+-binding to Ca2+-sensor Proteins by Isothermal Titration Calorimetry. Bio-protocol 10(7): e3580. DOI: 10.21769/BioProtoc.3580.
- Peshenko, I. V., Cideciyan, A. V., Sumaroka, A., Olshevskaya, E. V., Scholten, A., Abbas, S., Koch, K. W., Jacobson, S. G. and Dizhoor, A. M. (2019). A G86R mutation in the calcium-sensor protein GCAP1 alters regulation of retinal guanylyl cyclase and causes dominant cone-rod degeneration. J Biol Chem 294(10): 3476-3488.
Category
Biochemistry > Other compound > Ion > Calcium
Biochemistry > Protein > Quantification
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link