- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Semi-quantitative Determination of Protein Expression Using Immunohistochemistry Staining and Analysis: An Integrated Protocol
Published: Vol 9, Iss 24, Dec 20, 2019 DOI: 10.21769/BioProtoc.3465 Views: 17717
Reviewed by: Gal HaimovichSurabhi SonamAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Quantitative Proteomics of Nitrosylated Proteins in Melanoma Using the Biotin-Switch Technique Combined With Tandem Mass Tag Labeling
Vipin K. Yadav [...] Sanjay Premi
Dec 5, 2025 1598 Views

aGPCR-HEK: A Stable High-Expression Inducible Mammalian Cell Expression System for Adhesion GPCR Structural Biology Applications
David M. Favara and Christopher G. Tate
Mar 5, 2026 118 Views
Abstract
Semi-quantitative immunohistochemistry (IHC) is a powerful method for investigating protein expression and localization within tissues that involves using software, such as the freely available Fiji (ImageJ), to conduct deconvolution and downstream analysis. Currently, there is lack of an integrated protocol that includes a detailed procedure on how to measure or compare protein expression. Publications that use semi-quantitative methods to ascertain protein expression often don’t provide enough details in their methods section, which makes it difficult for the reader to reproduce their data. The current protocol provides an example and detailed steps of conducting semi-quantitative analysis of IHC images using Fiji software.
Keywords: ImmunohistochemistryBackground
Semi-quantitative immunohistochemistry (IHC) has been widely used for investigating protein expression and localization within tissues (Matkowskyj et al., 2000; Cregger et al., 2006; Taylor and Levenson, 2006; Braun et al., 2013; Fedchenko and Reifenrath, 2014; Bauman et al., 2016; Pike et al., 2017; Lu et al., 2018; Crowe et al., 2019), and various software and methods have been used in semi-quantiative IHC to conduct deconvolution and downstream analysis. Some methods require advanced coding/mathematical experience or the use of subscription-based software that may not be feasible for every scientist (Matkowskyj et al., 2000; Shu et al., 2016; Guirado et al., 2018). Fiji (ImageJ) is a freely available software (version 1.2; WS Rasband, National Institute of Health, Bethesda, MD). Publications using ImageJ for image quantification are often directed towards the creation of specific plugins for ImageJ or lack a step-by-step protocol (Varghese et al., 2014; Chen Y, 2017; Guirado et al., 2018). However, there is no integrated protocol that includes a detailed but simple procedure of conducting such steps, including the use of ImageJ software. There are many open online forums where users post their questions about using Fiji ImageJ for image quantification purposes. This is often due to publications not providing sufficiently detailed methods sections, which makes it difficult for the reader to reproduce the data. It is also time-consuming for researchers who are not initially familiar with ImageJ software to figure out how to use this software. The current protocol provides an example for conducting semi-quantitative analysis of IHC images using ImageJ.
Equipment
Computer Specifications:
- A 64-bit operating system that has Windows 7 or greater, Mac OS X 10.11 or greater or Linux with kernel supporting GLIBC 2.14 and GLIBCXX 3.4.15 (typically kernels 2.6.39)
- NVIDIA graphics card (GeForce, Quadro, or Tesla) with CUDA capabilities 2.0 or greater, see https://developer.nvidia.com/cuda-gpus for more details
- Need up-to-date NVIDIA drivers (minimum version of 369)
- Any computer with Java-based operating system and Excel available
Software
- Free ImageJ Fiji software (Johannes Schindelin, Albert Cardona, Mark Longair, Benjamin Schmid, and others, https://imagej.net/Fiji/Downloads), version 1.2 (no specific plugin was used)
Procedure
- Staining of tissue using immunohistochemistry procedure
- Reference to the procedure used for the staining of cells for the IHC protocol can be found in a previously published manuscript (Crowe et al., 2019).
- Brief protocol for immunohistochemistry staining:
- Cut FFPE tissue blocks into 4 µm sections and mount on positively charged slides.
- Deparaffinize and rehydrate tissue.
- Retrieve antigen in buffer of choice (citrate buffer at pH 6 was used for this protocol).
- Block antigen with normal goat serum and incubate with primary antibodies (i.e., anti-OATP1B1 for this protocol).
- Incubate with horseradish peroxidase (HRP)-conjugated secondary antibody and visualized by 3,3’-diaminobenzidine (DAB) to detect the protein of interest (OATP1B1 in the current protocol).
- Stain nuclei with hematoxylin.
- Hematoxylin and eosin (H&E) staining is not necessary for this protocol but is useful when looking at the pathology of the tissue.
- Image exporting and saving
Export and save the raw immunohistochemistry (IHC) immunohistochemistry images as a .tiff or .jpg file format. Tiff format for images is preferred in order to prevent the loss of raw data. - Using Fiji for deconvolution of the IHC image.
- Download and open ImageJ Fiji software.
- Click the “File” option and click “Open”. The IHC image will open up on the computer screen.
- Click on the IHC image to make the image active.
- Click the “Image” option and select “Color” > “Color Deconvolution.”
- A new pop-up Color Deconvolution window will show up. For IHC images stained with 3,3’-diaminobenzidine (DAB) and hematoxylin (H), select the “H DAB” vector option. Leave “Show Matrices” and “Hide Legend” unchecked and click “Okay.” (Figure 1).
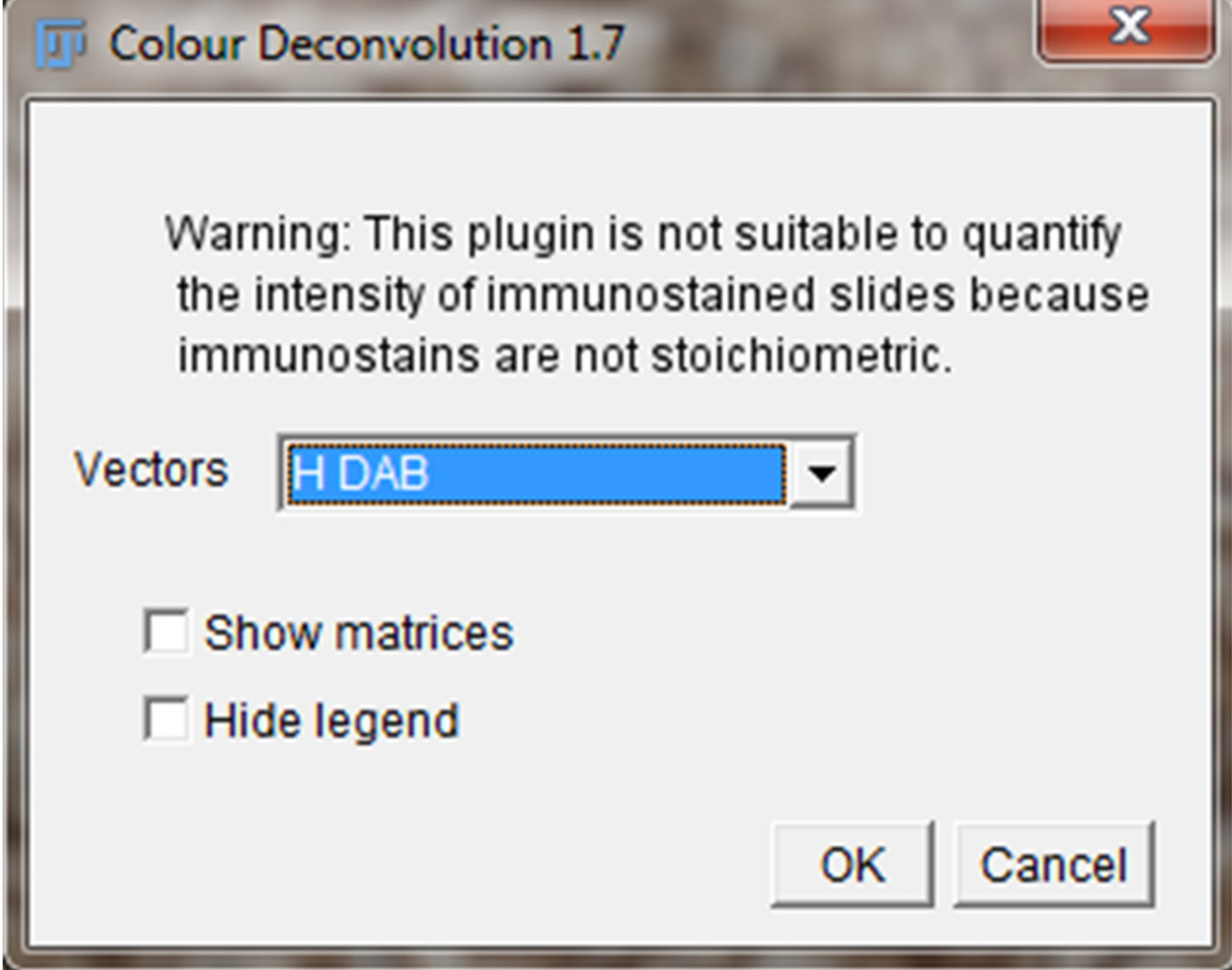
Figure 1. Color Deconvolution Window. The Color Deconvolution Window will be used to separate the staining of the IHC image. The H DAB vector separates the IHC image into DAB staining (brown staining) for the protein of interest and Hematoxylin (H) staining for the nucleus. - After selecting the H DAB option, three different images will pop up on the computer screen. Color 1 window represents only the Hematoxylin staining (blue/purple) and Color 2 window represents only the DAB staining (brown) (Figure 2).
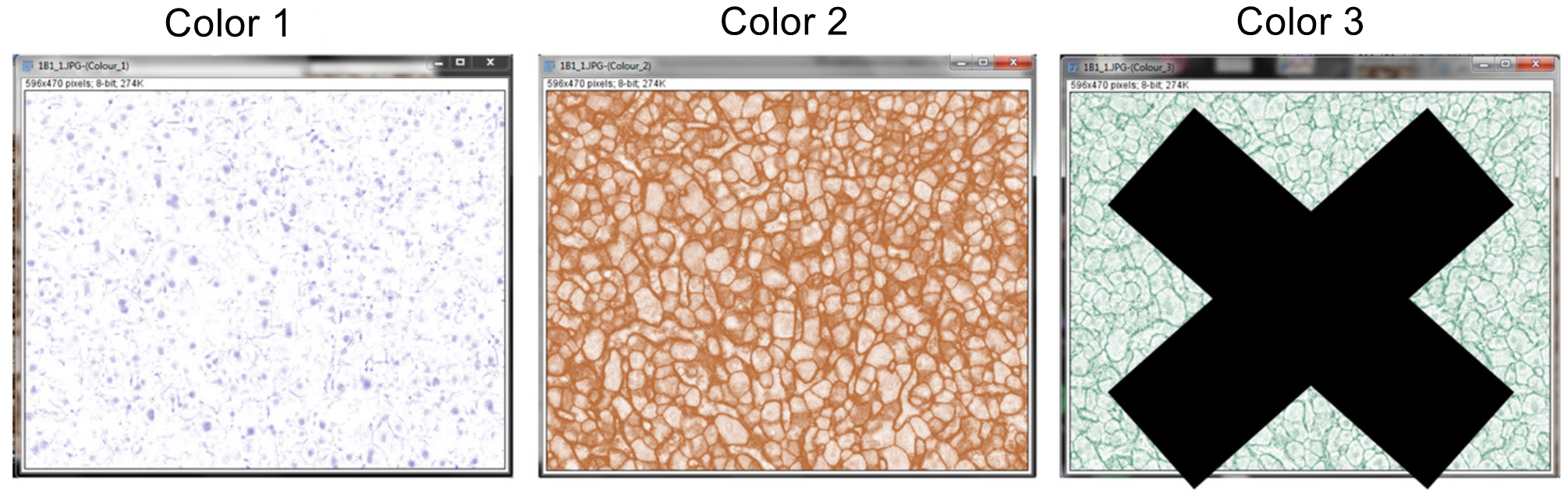
Figure 2. Deconvolution of IHC image. Separation of the IHC image into hematoxylin staining for the nuclei (Color 1) and DAB staining for OATP1B1 protein expression (Color 2) in hepatocytes within human liver tissue. Color 3 panel is for another staining, if applicable. For DAB and hematoxylin staining alone, the Color 3 panel can be eliminated. In DAB and hematoxylin staining as in current studies, a warning “X” is shown so that users understand that the amount of antibody staining (i.e., DAB staining) in this case cannot be mathematically quantified as it is not stoichiometric. - Exit out of the third Color 3 window, as this will not be needed for the image analysis.
- Thresholding the DAB-stained IHC image
- Click on the DAB Color 2 image to activate it. DAB staining represents your primary antibody of interest. In this case, liver tissue was incubated with an OATP1B1 primary antibody, and the OATP1B1 expression is detected by DAB.
- Go to “Image” and select “Adjust” and “Threshold”. After selecting the threshold, the brown image is converted into a black and white binary mask image, where all the values above the threshold are converted and represented in white. The background is black.
Note: A shortcut to threshold the image is by pressing “Ctrl” + “Shift” + “T” - A new threshold window will pop up. The top bar indicates your minimum threshold value and the bottom bar indicates your maximum threshold value (Figure 3).
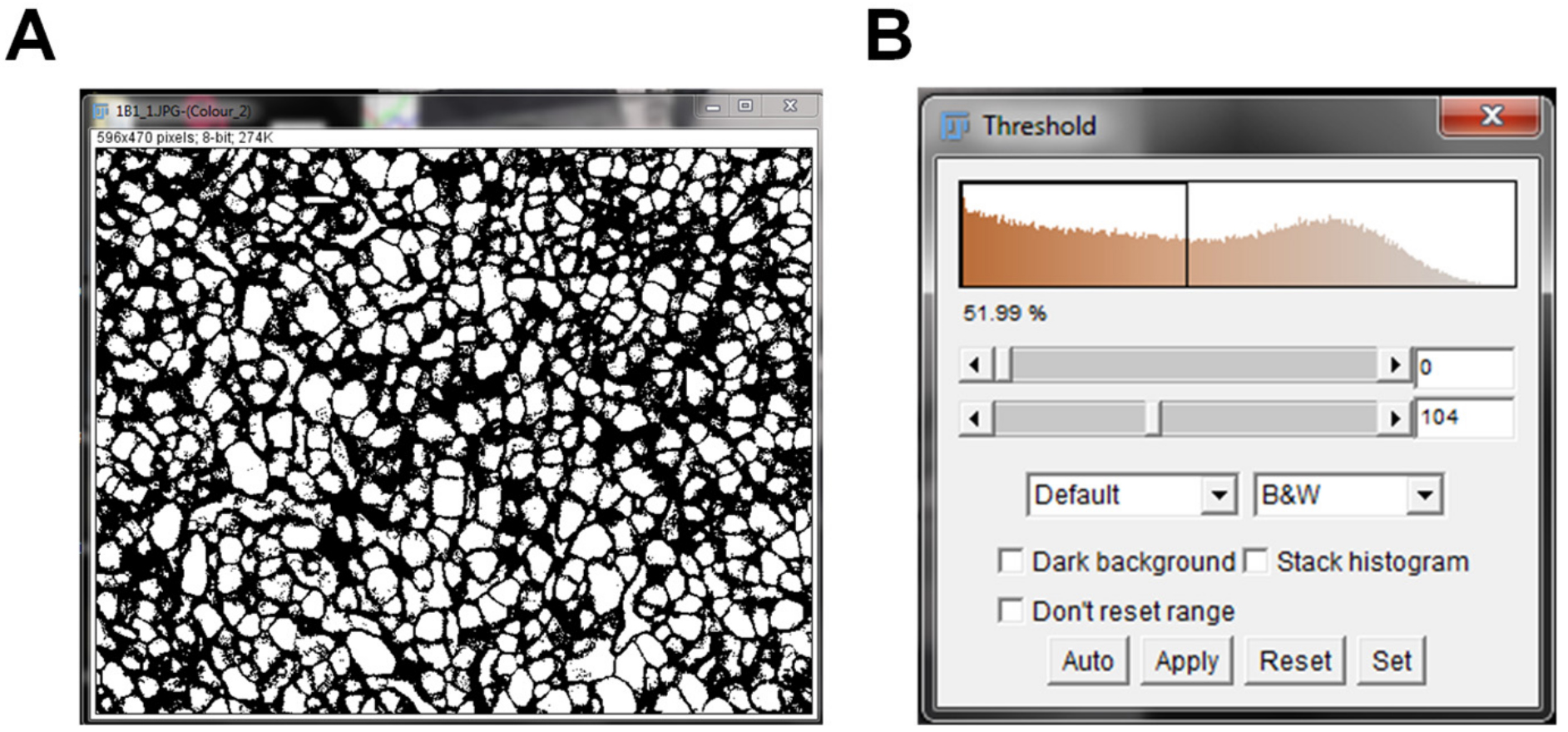
Figure 3. IHC image pre-threshold. IHC image converted to black and white pixels prior to thresholding the image (A). The threshold pop up window pre-threshold indicates the baseline threshold values (B). - Leave the minimum threshold value set at zero.
- Adjust the maximum threshold value so that the background signal is removed, without removing the true DAB signal (Figure 4A). This is an arbitrary value, since it is set by the user. The maximum threshold value should be tested for at least five images to obtain an average maximum threshold value. Once the maximum threshold value is chosen, this will be set for all future IHC images. The measured threshold will only remain relevant provided that the microscope acquisition settings (color balance, exposure time, magnification, light source, camera vendor, etc.) remain constant throughout future experiments.
- Once the maximum threshold image is set, click “Apply” on the threshold window. After clicking apply, the minimum and maximum threshold values will be 255 (Figure 4B).
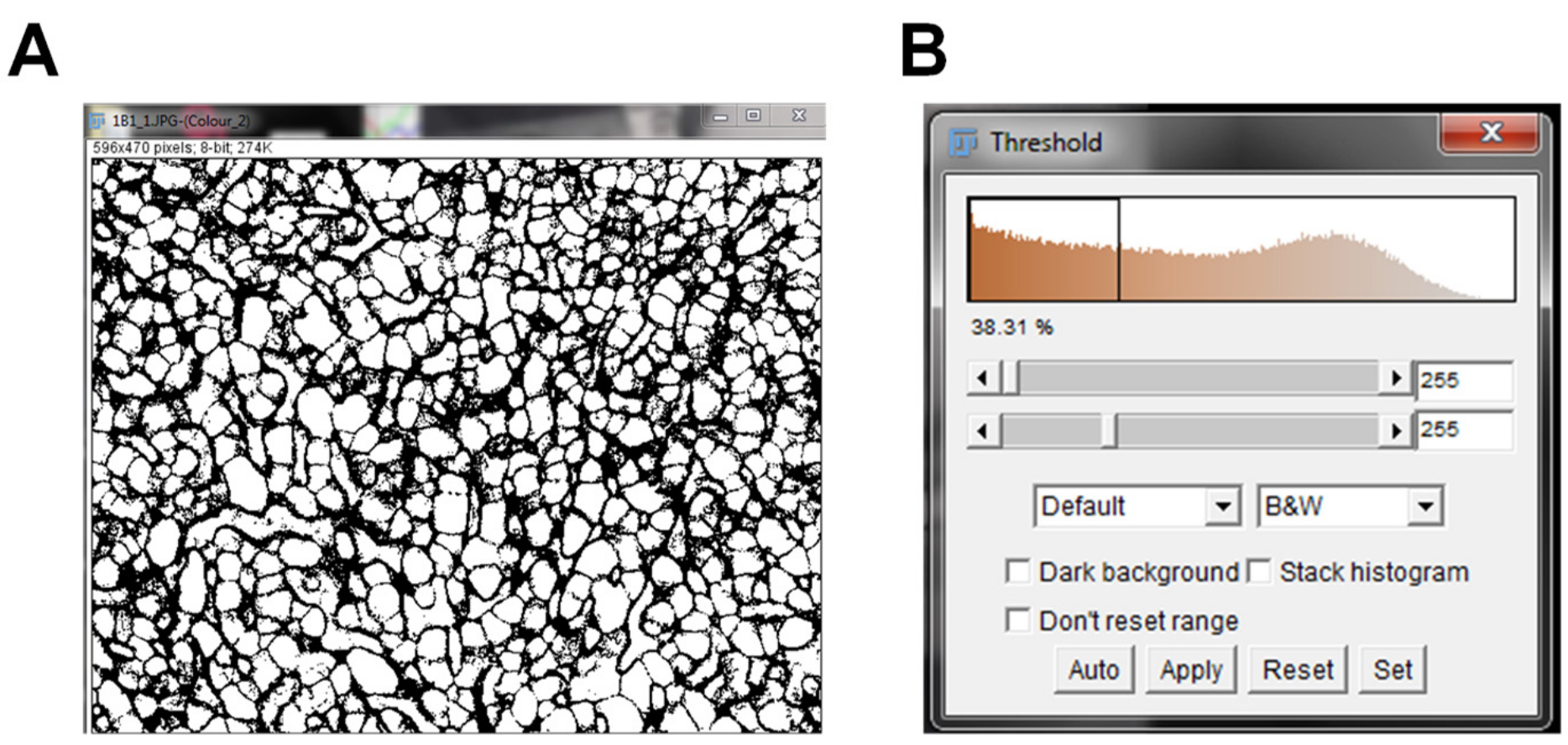
Figure 4. IHC image post-threshold. The background for the DAB staining in the IHC image was removed by adjusting the maximum threshold value (A). The minimum and maximum threshold values were set and applied (B).
- Quantifying the DAB signal area in the IHC image
- Go to “Analyze” and select “Set Measurements”.
- A “Set Measurement” pop up window will open. Select the “Area”, “Mean grey value”, and “Display Label” boxes and leave all other boxes unchecked. “Area” will give the size of the IHC image. “Mean grey value” represents the quantified signal and “Display Label” gives the information on the image name being quantified (Figure 5A).
- Select “Okay” in the Set Measurement window. These options only need to be set once for the first image and will be remembered for all other future images measured.
- Go to “Analyze” and select “Measure”.
Note: Note: A shortcut for measuring the signal area is “CTL + M”. - A “Results” window will pop up giving the name of the image (Label), size of the image (Area) and the average pixel intensity of the IHC image (Mean) (Figure 5B).
- Copy the results to an excel file for later analysis.
- Exit out of the Color 2 DAB stained image.
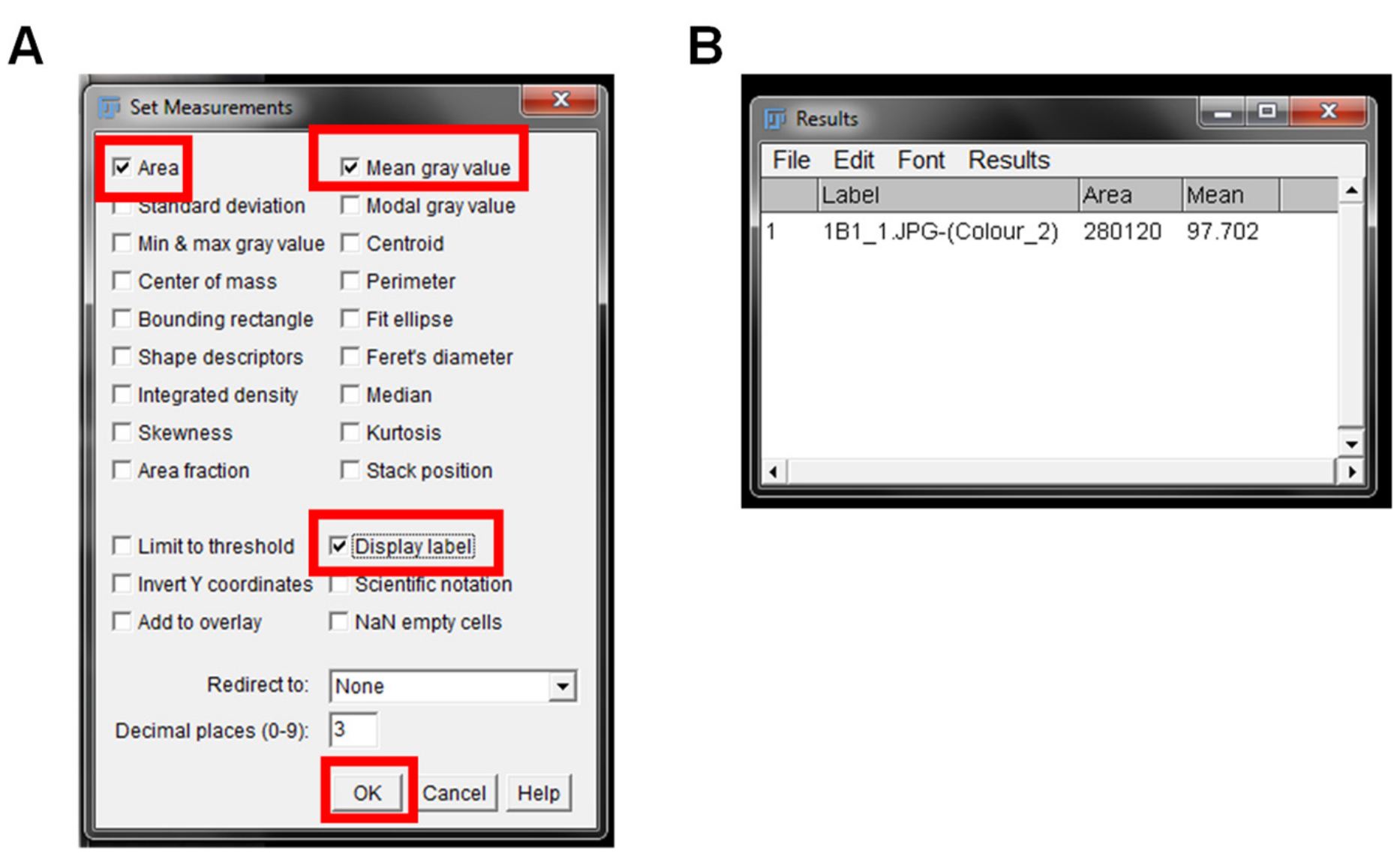
Figure 5. Measurement of DAB staining. The Set Measurements window to choose the options for output of each IHC image is shown. The suggested options for output are highlighted with red boxes (A). The Results output window is shown in (B), and contains the name of the image (Label), area of the image (Area), and mean grey value intensity (Mean).
- Measuring the size of the nucleus
- Click on the Hematoxylin Color 1 image to activate it.
- Select the “Straight line” tool on the ImageJ Fiji panel.
- Measure the distance of a nucleus by drawing a line across the nucleus with the Straight line tool (Figure 6A).
- Go to “Analyze” and select “Measure”.
- A Results window will pop up with the diameter of the nucleus (“Length”) (Figure 6B).
- Measure ~10 different nuclei using Steps F1-F5 in a representative IHC image and take the average of these Lengths to determine the average size of the nuclei. This serves as the average size of the nuclei for all IHC images from hereon. Ths step only needs to be done for one image and will be used for all the rest of your images.

Figure 6. Measurement of nuclei size. The raw IHC image stained with DAB for OATP1B1 staining and hematoxylin for nuclei staining is used to measure the size of the nuclei. Using the straight-line tool, a line is drawn over the nucleus (A, white arrow pointing to yellow line). The distance of the line is measured by going to Analyze > Measure in the ImageJ Fiji toolbox panel. The Length (highlighted in red box) represents the diameter of the nucleus (B).
- Thresholding the hematoxylin-stained IHC image
- Click on the Hematoxylin Color 1 image to activate it.
- Go to “Image” and select “Adjust” and “Threshold”. After selecting threshold, the blue/purple image is now converted to a black and white image.
- Set the minimum threshold value to zero.
- Adjust the maximum threshold value so that the background signal is removed, without removing the true hematoxylin/nucleus signal. This is an arbitrary value, since the user sets it. The maximum threshold value should be tested for at least five images to get an average maximum threshold value. Once the maximum threshold value is chosen, this will be set for all future IHC images.
- Once the minimum and maximum threshold values for the image are set, click “Apply” on the threshold window. After clicking apply, the minimum and maximum threshold values will be 255.
Of note, automatic thresholds are preferred, since manual thresholds must be adequately tested and kept constant for all other images in the experiment.
- Quantifying the hematoxylin/nucleus signal in the IHC image
- Select the Color 1 Hematoxylin image to activate it again.
- Go to “Process” and select “Binary” > “Watershed”. This action will split the nuclei that are joined together into multiple nuclei.
- Go to “Analyze” and select “Analyze Particles”.
- An “Analyze Particles” window will pop up giving multiple options (Figure 7A).
- Size (pixel^2): Set the size of the nuclei to the average nuclei size measured in Step D6 to infinity (i.e., 6-Infinity).
- Circularity: Leave it set at 0.00-1.00.
- Show: Leave it set to “Nothing”.
- Select “Summarize” and “Exclude on Edges” in the window. Summarize will give a summary of the particle’s measurements. Exclude on the edges means that nuclei on the outer edge of the image will not be included in the measurement.
- Select “Okay” on the Analyze Particles window and a Summary window with the output will pop up (Figure 7B). The output data includes:
- Count indicates the number of nuclei in the IHC image.
- Total area indicates the total area of the image.
- Average area indicates the average size of the nuclei in the IHC image.
- Copy the results in the Summary window to the same excel file for later analysis.
- Exit out of the Color 1 Hematoxylin/Nuclei stained image.
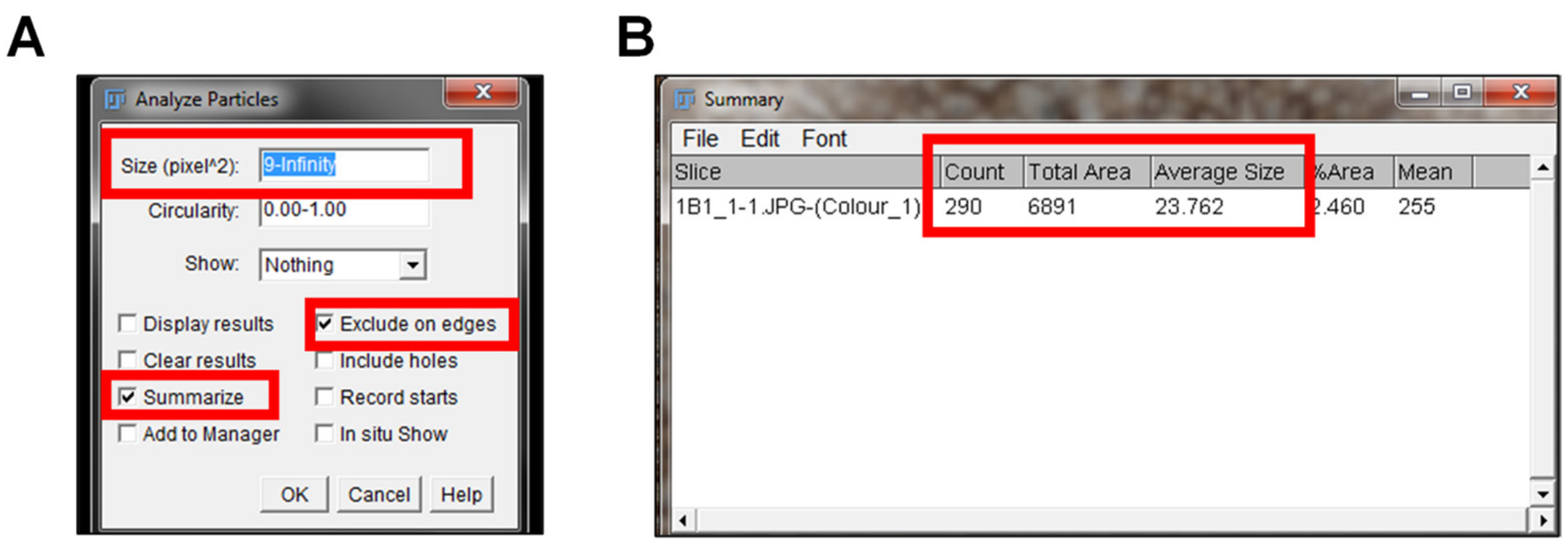
Figure 7. Quantification of nuclei in IHC image. To measure the number of nuclei in each IHC image, the Analyze Particle window pops up after selecting Analyze > Analyze Particles (A). The size of the particle measured is set to the average diameter of nuclei to infinity. The options to summarize and exclude on the edges are selected prior to clicking okay. The summarize option leads to the summarized output of the count, total area, and the average size of the nuclei particles in the IHC image (B). Exclude on the eges indicates that no nuclei particles will be included in the analysis.
- Semi-quantitative analysis of the IHC image
- Open the excel file containing the DAB and hematoxylin results outputted from the Fiji software.
- For each image, divide the Mean grey intensity value from Step E5 by the number of nuclei measured in Step H5. This value represents the area of DAB stain normalized to the number of nuclei.
- Take the average normalized area of DAB of for all IHC images for each sample/treatment, etc. to obtain an average and standard deviation values.
Data analysis
Expression of OATP1B1 and OATP1B3 were compared in genotyped human liver tissue stained with OATP1B1 or OATP1B3 DAB staining and hematoxylin. Using 79 genotyped human liver IHC samples, there was no significant difference between the genotypes for OATP1B1 (c.521 TC) polymorphism using a Student’s t-test (Crowe et al., 2019).
Application of this protocol to other studies should be done with caution, as it may not work for others if their microscope or staining protocols differ slightly, even if they were to imagestain the same protein with the same antibody. In addition, this protocol is not intended to replace the ImageJ manual, which is a great resource for users, and can be found at https://www.researchgate.net/publication/260261544_Analyzing_fluorescence_microscopy_images_with_ImageJ or https://petebankhead.gitbooks.io/imagej-intro/content/chapters/rois/rois.html.
Acknowledgments
This research was supported by NIH R01 GM094268 [W. Y]. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. Alexandra Crowe is an American Foundation of Pharmaceutical Education pre-doctoral Fellow. We thank Drs. Wei Zheng, Kar-Ming Fung, Feng Yin, and Erin Rubin for providing the FFPE tissues for this research.
Competing interests
No competing financial interests for this study.
Ethics
Use of human tissues was approved by the Institutional Review Board at the University of Oklahoma Health Sciences Center. A total of 79 formalin-fixed, paraffin-embedded (FFPE) archived human liver (42 from surgical resection and 37 from liver biopsy) and normal kidney tissue blocks were obtained from OUHSC Stephenson Cancer Center Biospecimen Acquisition Core and the Bank from the Department of Pathology at the University of Oklahoma Health Sciences Center.
The authors have noticed some errors in the original protocol that were now amended and clarified in the current protocol. The authors apologize for any inconvenience caused.
References
- Bauman, T. M., Ricke, E. A., Drew, S. A., Huang, W. and Ricke, W. A. (2016). Quantitation of protein expression and co-localization using multiplexed immuno-histochemical staining and Multispectral Imaging. J Vis Exp (110). doi: 10.3791/53837.
- Braun, M., Kirsten, R., Rupp, N. J., Moch, H., Fend, F., Wernert, N., Kristiansen, G. and Perner, S. (2013). Quantification of protein expression in cells and cellular subcompartments on immunohistochemical sections using a computer supported image analysis system. Histol Histopathol 28(5): 605-610.
- Chen Y., Qi Y., and Xu C. B.(2017) A convenient method for quantifying collagen fibers in atherosclerotic lesions by ImageJ software. Int J Clin Exp Med 10(10): 14904-14910.
- Cregger, M., Berger, A. J. and Rimm, D. L. (2006). Immunohistochemistry and quantitative analysis of protein expression. Arch Pathol Lab Med 130(7): 1026-1030.
- Crowe, A., Zheng, W., Miller, J., Pahwa, S., Alam, K., Fung, K. M., Rubin, E., Yin, F., Ding, K. and Yue, W. (2019). Characterization of plasma membrane localization and phosphorylation status of organic anion transporting polypeptide (OATP) 1B1 c.521 T>C Nonsynonymous single-nucleotide polymorphism. Pharm Res 36(7): 101.
- Fedchenko, N. and Reifenrath, J. (2014). Different approaches for interpretation and reporting of immunohistochemistry analysis results in the bone tissue - a review. Diagn Pathol 9: 221.
- Guirado, R., Carceller, H., Castillo-Gomez, E., Castren, E. and Nacher, J. (2018). Automated analysis of images for molecular quantification in immunohistochemistry. Heliyon 4(6): e00669.
- Jensen, K., Krusenstjerna-Hafstrom, R., Lohse, J., Petersen, K. H. and Derand, H. (2017). A novel quantitative immunohistochemistry method for precise protein measurements directly in formalin-fixed, paraffin-embedded specimens: analytical performance measuring HER2. Mod Pathol 30(2): 180-193.
- Kokolakis, G., Panagis, L., Stathopoulos, E., Giannikaki, E., Tosca, A. and Kruger-Krasagakis, S. (2008). From the protein to the graph: how to quantify immunohistochemistry staining of the skin using digital imaging. J Immunol Methods 331(1-2): 140-146.
- Li, H., Spagnol, G., Naslavsky, N., Caplan, S. and Sorgen, P. L. (2014). TC-PTP directly interacts with connexin43 to regulate gap junction intercellular communication. J Cell Sci 127(Pt 15): 3269-3279.
- Lu, Z., Liu, Y., Xu, J., Yin, H., Yuan, H., Gu, J., Chen, Y. H., Shi, L., Chen, D. and Xie, B. (2018). Immunohistochemical quantification of expression of a tight junction protein, claudin-7, in human lung cancer samples using digital image analysis method. Comput Methods Programs Biomed 155: 179-187.
- Matkowskyj, K. A., Schonfeld, D. and Benya, R. V. (2000). Quantitative immunohistochemistry by measuring cumulative signal strength using commercially available software photoshop and matlab. J Histochem Cytochem 48(2): 303-312.
- Pike, J. A., Styles, I. B., Rappoport, J. Z. and Heath, J. K. (2017). Quantifying receptor trafficking and colocalization with confocal microscopy. Methods 115: 42-54.
- Shu, J., Dolman, G. E., Duan, J., Qiu, G. and Ilyas, M. (2016). Statistical colour models: an automated digital image analysis method for quantification of histological biomarkers. Biomed Eng Online 15: 46.
- Sysel, A. M., Valli, V. E., Nagle, R. B. and Bauer, J. A. (2013). Immunohistochemical quantification of the vitamin B12 transport protein (TCII), cell surface receptor (TCII-R) and Ki-67 in human tumor xenografts. Anticancer Res 33(10): 4203-4212.
- Taylor, C. R. and Levenson, R. M. (2006). Quantification of immunohistochemistry--issues concerning methods, utility and semiquantitative assessment II. Histopathology 49(4): 411-424.
- Varghese, F., Bukhari, A. B., Malhotra, R. and De, A. (2014). IHC Profiler: an open source plugin for the quantitative evaluation and automated scoring of immunohistochemistry images of human tissue samples. PLoS One 9(5): e96801.
Article Information
Copyright
© 2019 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Crowe, A. R. and Yue, W. (2019). Semi-quantitative Determination of Protein Expression Using Immunohistochemistry Staining and Analysis: An Integrated Protocol. Bio-protocol 9(24): e3465. DOI: 10.21769/BioProtoc.3465.
Category
Biochemistry > Protein > Quantification
Molecular Biology > Protein > Expression
Cell Biology > Cell imaging > Fixed-tissue imaging
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link










