- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Adoptive Transfer of Basophils Enriched from Mouse Spleen
(*contributed equally to this work) Published: Vol 9, Iss 21, Nov 5, 2019 DOI: 10.21769/BioProtoc.3416 Views: 4447
Reviewed by: Meenal SinhaLaura CampisiKrisztina Vukman

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
A Rapid Protocol for Direct Isolation of Osteoclast Lineage Cells from Mouse Bone Marrow
Lei Dang [...] Jin Liu
Mar 5, 2022 4361 Views
Primary Mouse Invariant Natural Killer T (iNKT) Cell Purification and Transduction
Gloria Delfanti [...] Giulia Casorati
Jul 5, 2023 2170 Views
Isolation and Culture of Primary Pericytes from Mouse
Tamara McErlain [...] Meera Murgai
Apr 20, 2025 2985 Views
Abstract
CD49b is a member of the integrin family, expressed on basophils, natural killer (NK) cells and a subset CD4+ T cells in the spleen. This protocol describes the adoptive transfer of basophil-enriched CD49b+ cells obtained from mouse spleens by magnetic enrichment. This protocol can be used to assess the contribution of basophils or basophil-derived mediators to a certain immune response.
Keywords: Adoptive transferBackground
Basophils are evolutionarily conserved in vertebrates, despite their small numbers and short lifespan, suggesting that basophils have beneficial roles in maintaining health. However, these roles are not fully defined. The use of Cre-loxP approaches has allowed the generation of conditional mice with basophil deficiency, or deficiencies in basophil-derived mediators, which have significantly contributed to our understanding of basophil function in health and disease. For example, we recently demonstrated that mice with basophils lacking tumor necrosis factor (TNF) exhibited high morbidity and reduced ability to clear bacteria in the cecal ligation and puncture (CLP) mouse model of sepsis, highlighting a role for basophils in innate immunity against bacteria (Piliponsky et al., 2019). Moreover, we showed that basophils are recruited into the infection site, the peritoneal cavity, as early as 1 h after CLP. To rule out potential non-specific effects of the Cre-loxP approach, the ultimate proof for our findings was provided by rescuing mice with a deficiency in basophil-derived TNF. To do this, we adoptively transferred the basophil-enriched CD49b+ fraction obtained from the spleens of wild type mice into the peritoneal cavities of septic mice (Figure 1). Please note that this protocol does not compare the method used to other available methodologies, as it was specifically designed to assess the basophil-derived-TNF dependency for the enhancement of innate immune responses after CLP. More extensive studies than the ones described in this protocol are required to determine how long basophils live and how they distribute in the adoptive host. 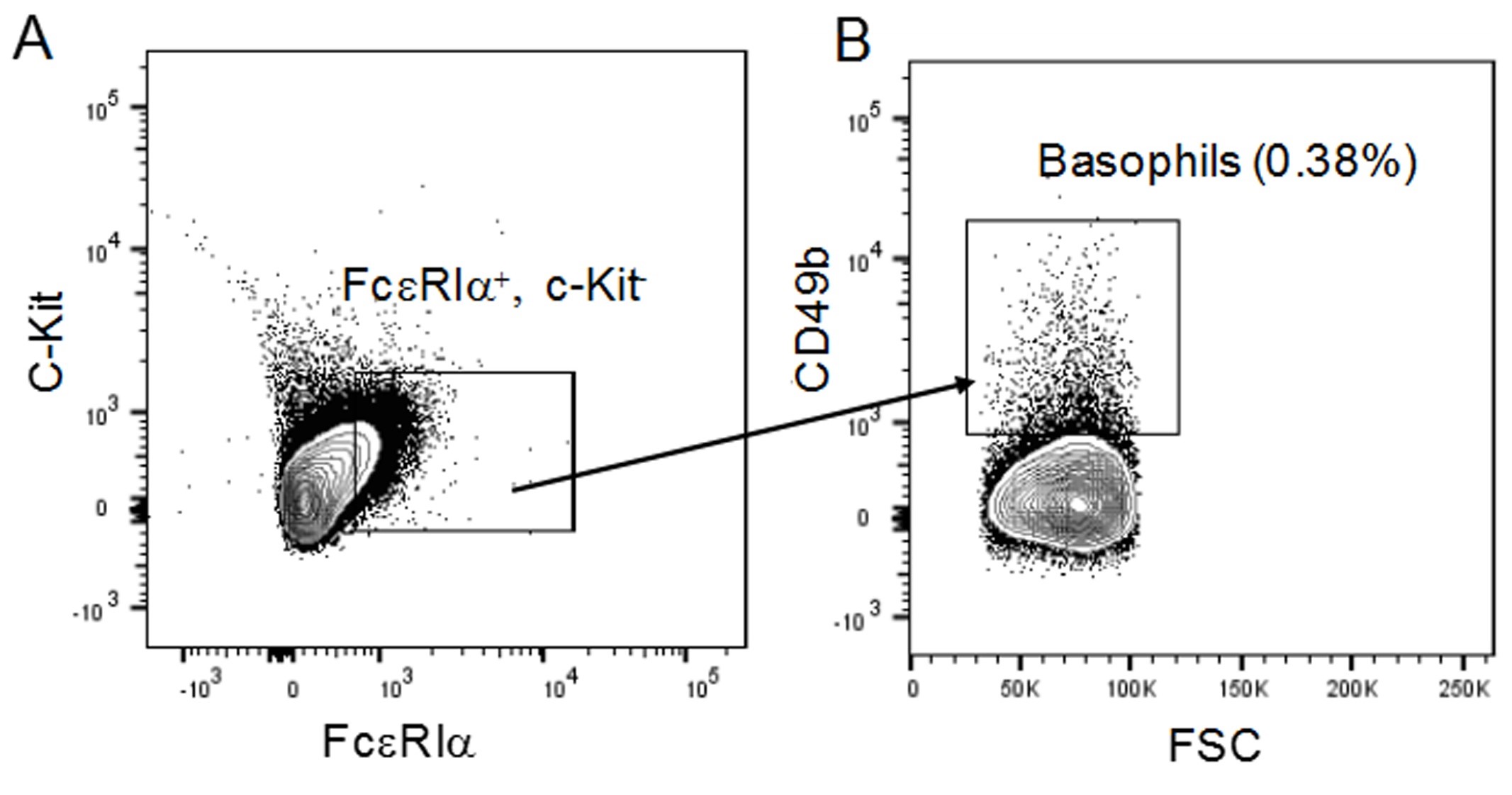
Figure 1. Strategy to identify basophils among total peritoneal cells in mice that received an adoptive transfer of basophils after cecal ligation and puncture (CLP). Representative flow cytometry plots and frequencies used to identify basophils amongst peritoneal cells obtained from mice that received an adoptive transfer with the CD49b+ basophil-enriched fraction of splenocytes (4 x 105 cells) at 3 h after induction of CLP of moderate severity (50% ligation, one puncture with a 22G needle). A and B. FcεRiα+, c-Kit- (A) and CD49b+ (B) cells were gated and defined as basophils amongst peritoneal cells obtained from mice at 24 h after CLP.
Here, we describe such a protocol that can be used in other mouse models to confirm a dependency on basophils, or basophil-derived mediators, for the development of a biological response.
Materials and Reagents
- Sterile culture hood (Sterilgard)
- Surgical scissors and forceps
- Pipettes 20, 200, and 1000 μl (Eppendorf)
- Six well tissue culture plate (Olympus, catalog number: 25-105)
- Frosted glass slides (Fisher Scientific, catalog number: 12-550-143)
- 5 ml round-bottom tubes (Falcon, catalog number: 352008)
- 15 ml tubes (Falcon, catalog number: 352096)
- 50 ml conical tubes (Falcon, catalog number: 352070)
- 22 G needle (BD, catalog number: 305155)
- 1 ml syringe (BD, catalog number: 309659)
- Cell strainer, 40 mm nylon (Falcon, catalog number: 352340)
- Steritop, 0.22 mm PES, 250 ml (Millipore, catalog number: S2GPT02RE)
- CD49b (clone DX5) Microbeads (Miltenyi Biotec, catalog number: 130-052-501)
- Purified rat anti-mouse CD16/CD32 (FcBlock) (clone 2.4G2) (BD Pharmingen, catalog number: 553141)
- APC-conjugated anti-CD49b antibody (clone DX5) (Biolegend, catalog number: 108909)
- PE-conjugated anti- FcεRIα (clone MAR-1) (Biolegend, catalog number: 134307)
- APC/Cy7-conjugated anti-c-Kit (clone 2B8) (Biolegend, catalog number: 105825)
- Phosphate buffered saline (PBS) (Corning, catalog number: 21-040-CV)
- NH4Cl (Sigma-Aldrich, catalog number: A-4514)
- KHCO3 (Sigma-Aldrich, catalog number: 237205)
- EDTA (Corning, catalog number: 46-034-Cl)
- Bovine serum albumin (BSA) (Sigma-Aldrich, catalog number: A7409)
- DAPI (4′,6-Diamidino-2-Phenylindole, Dilactate) (Biolegend, catalog number: 422801)
- UltraComp eBeads (Invitrogen, catalog number: 01-2222-42)
- Dulbecco’s Modified Eagle Medium (Gibco, catalog number: 11960-044)
- Ethanol (Koptec, catalog number: V1001)
- 70% ethanol (see Recipes)
- ACK lysis buffer (see Recipes)
- MACS buffer (see Recipes)
Equipment
- Centrifuge Avanti J-15R (Beckman Coulter)
- BD LSR II (BD Biosciences)
- autoMACS Pro Separator-Starter Kit (Miltenyi Biotec, catalog number: 130-092-545)
- Biosafety cabinet
Software
- BD FACSDiva software
- FlowJo software (version 8.8.7, Tree Star)
Procedure
- Preparation of a spleen single cell suspension
- Euthanize a naive mouse by CO2 inhalation. Disinfect the skin of the mouse with 70% ethanol (Recipe 1). Extract whole spleen and place in a six-well plate with 3 ml PBS in one well. The rest of the procedures should be conducted in a sterile biosafety cabinet.
- Smash spleen thoroughly between frosted glass slides by placing the spleen on the rough side of the frosted part of the slide (wetted with PBS) and carefully grind it between the two slides until spleen is dissociated.
- Pipette up the PBS buffer containing splenocytes with a 5 ml pipette into a 15 ml tube.
- Filter through a 40 μm cell strainer over a 50 ml conical tube.
- Spin at 335 x g at room temperature (RT) for 5 min.
- Discard the supernatant and resuspend the pellet in 2 ml of ACK lysis buffer at RT for 5 min.
- Add 8 ml of PBS to cell suspension.
- Spin at 335 x g at RT for 5 min.
- Discard the supernatant and resuspend in 10 ml of PBS.
- Count 1:10 dilution of cells.
- Place splenocytes on ice until ready for CD49b+ cell enrichment.
- Magnetic labeling
- Spin at 335 x g at 4 °C for 5 min.
- Discard the supernatant and resuspend in 90 μl of “MACS buffer” per 107 total cells (approximately 108 total cells are expected from one spleen from an adult mouse).
- Add 10 μl of CD49b (DX5) microbeads per 107 total cells.
- Mix well by pipetting up and down several times and incubate for 30 min on ice.
- Add 1 ml of MACS buffer per 107 total cells.
- Spin at 335 x g at 4 °C for 5 min.
- Discard the supernatant and resuspend up to 108 cells in 500 μl of “MACS buffer”.
- Place cells on ice until the autoMACS Separator is ready for use. Magnetic separation can be performed with MACS columns for positive selection (MS) or with other separators (e.g., MiniMACS, OctoMACS, VarioMACS, SuperMACS) if the autoMACS Separator is not available.
- Magnetic separation with the autoMACS Separator
- Prepare and prime autoMACS Separator. This procedure can be done while cells are being magnetically labeled (Step B4).
- Place tube containing the magnetically labeled cells in the autoMACS Separator choosing the positive selection program (“Possel”).
- Collect the purified CD49b+ cell fraction in outlet port “pos1”.
- Spin the enriched CD49b+ cell fraction at 335 x g at 4 °C for 5 min.
- Discard the supernatant and resuspend in 1 ml DMEM.
- Count cells (approximately 106 cells are expected from one spleen).
- Place cells on ice until assessment of basophil purity by staining and flow cytometry analysis. Approximately 20-30% of the CD49b+ cells enriched from one mouse spleen are expected to be basophils of > 90% viability.
- Staining for flow cytometry
- Dilute cells to 1 x 106 cells/ml for staining. Two samples are needed: one sample will be unstained and the second sample will be incubated with the antibody and DAPI mix (generating the stained sample).
- Spin both samples at 335 x g at 4 °C for 5 min.
- Discard the supernatant and block both samples with 10 l of diluted FcBlock (1:200 in PBS).
- Incubate both samples on ice for 15 min.
- Stain one sample with 20 μl of antibody and DAPI mix: PE FcεRIα (1:200), APC/Cy7 c-Kit (1:100), APC CD49b and DAPI (1:100). Add the equivalent volume of PBS to the unstained sample tube. Place samples on ice in a dark place to stain for 20-30 min.
- Add 500 μl of PBS to the samples, vortex gently and spin at 335 x g at 4 °C for 5 min.
- Discard the supernatants and resuspend in 300 μl PBS.
- Filter cells into a new FACS tube trough the cell strainer to remove clumps and place samples on ice until flow cytometry analysis.
- Flow cytometry
- On the BD FACS DIVA software, select for a “New Experiment” to set up.
- Select for Area, Height, Width for FSC and SSC; select Log and Area for the colors PE, APC, APC/Cy7 and Indo I-Violet.
- Create compensation controls and adjust the gating for unstained cells (adjust FSC and SSC voltages as necessary).
- Adjust each color of single stains to center each fluorescence histogram on 102 + 1 tick mark.
- For single stains for compensation, use the UltraComp eBeads following manufacturer’s instructions. Use cells stained with DAPI to generate a single color for compensation.
- Check that P2 (i.e., fluorescence positive) gates look appropriate for each single-stained tube and make any necessary adjustments to P2 gates before calculating compensation for an experiment.
- Calculate compensation controls and record events. Set up to collect 50,000 events.
- Adoptive transfer of spleen basophils into the peritoneal cavity
- Spin the enriched basophils at 335 x g at 4 °C for 5 min.
- Discard the supernatants and resuspend 3 x 105 spleen basophils defined as FcεRIα+, CD49b+ and c-Kit- cells in 200 μl DMEM.
- Inject the recipient mice with 200 μl intraperitoneally using a 22 G needle and a 1 ml syringe.
Data analysis
Staining followed by flow cytometry (as described in Procedures D and E above) was used to confirm that recipient mice that received basophils from either donor wild-type mice or mice with basophils lacking TNF show similar numbers of basophils in the peritoneal cavity at 24 h after CLP (Fig. 8C in Piliponsky et al., 2019).
Recipes
- 70% ethanol (for 100 ml)
70 ml 99.9% ethanol
30 ml demineralized water
Store at RT for up to a year - ACK lysis buffer
8.0235 g NH4Cl
0.10012 g KHCO3
200 μl 0.5 M EDTA, pH 8.0
Bring to pH 7.3 and filter using a Steritop filter
Store at RT for up to a year - MACS buffer
Phosphate buffered saline (PBS), pH 7.2
0.5% bovine serum albumin (BSA)
2 mM EDTA
Store at 4 °C and use within 3 days of preparation
Acknowledgments
This protocol was adapted from Wada et al., (2010) and Piliponsky et al., (2019). Research in the Piliponsky lab was supported by the National Institute of Health (NIH) (R01 HL113351, R01 HL141094) and the American Heart Association (AHA) (12GRNT9680021). Research in the Galli lab was supported by the NIH (R01 CA72074, R37 AI23990, R01 AI070813 and R01 AR067145) and by the Department of Pathology, Stanford University.
Competing interests
The authors declare no competing financial interests.
Ethics
All the procedures were conducted in accordance with the current National Institutes of Health guidelines and with the approval of the Seattle Children’s Research Institute Institutional Animal Care and Use Committee (ID: IACUC00020, Validate period: 8/22/2019-8/21/2019).
References
- Piliponsky, A. M., Shubin, N. J., Lahiri, A. K., Truong, P., Clauson, M., Niino, K., Tsuha, A. L., Nedospasov, S. A., Karasuyama, H., Reber, L. L., Tsai, M. and Mukai, K. (2019). Basophil-derived tumor necrosis factor can enhance survival in a sepsis model in mice. Nat Immunol 20(2): 129-140.
- Wada, T., Ishiwata, K., Koseki, H., Ishikura, T., Ugajin, T., Ohnuma, N., Obata, K., Ishikawa, R., Yoshikawa, S., Mukai, K., Kawano, Y., Minegishi, Y., Yokozeki, H., Watanabe, N. and Karasuyama, H. (2010). Selective ablation of basophils in mice reveals their nonredundant role in acquired immunity against ticks. J Clin Invest 120(8): 2867-2875.
Article Information
Copyright
© 2019 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Piliponsky, A. M. and Galli, S. J. (2019). Adoptive Transfer of Basophils Enriched from Mouse Spleen. Bio-protocol 9(21): e3416. DOI: 10.21769/BioProtoc.3416.
Category
Immunology > Animal model > Mouse
Immunology > Host defense > Murine
Cell Biology > Cell isolation and culture > Cell isolation > Dynabead
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link









