- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Assessing Experience-dependent Tuning of Song Preference in Fruit Flies (Drosophila melanogaster)
Published: Vol 8, Iss 14, Jul 20, 2018 DOI: 10.21769/BioProtoc.2932 Views: 7494
Reviewed by: Zinan ZhouXiaoliang ZhaoAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Caste Transition and Reversion in Harpegnathos saltator Ant Colonies
Comzit Opachaloemphan [...] Hua Yan
Aug 20, 2023 1576 Views

A Method for Studying Social Signal Learning of the Waggle Dance in Honey Bees
Shihao Dong [...] Ken Tan
Aug 20, 2023 1382 Views
Habituation of Sugar-Induced Proboscis Extension Reflex and Yeast-Induced Habituation Override in Drosophila melanogaster
Swati Trisal [...] Mani Ramaswami
Dec 5, 2023 1450 Views
Abstract
In songbirds and higher mammals, early auditory experience during childhood is critical to detect and discriminate sound patterns in adulthood. However, the neural and molecular nature of this acquired ability remains elusive. Here, we describe a new behavioral paradigm with Drosophila melanogaster to investigate how the auditory experience shapes sound perception. This behavioral paradigm consists of two parts: training session and test session. In the training session, we keep the flies singly in a training capsule and expose them to training sound for 6 days after eclosion. After the training session, flies are subjected to the test session, in which the mating behaviors of flies are monitored upon sound playback. As the training and test sounds, we use two types of artificial sound, which correspond to the pattern of conspecific and heterospecific courtship songs of fruit flies. By applying this method, we can measure how the acoustic experience with the conspecific song as a young adult sharpens the song preference and mate selection as a breeding adult in the fruit fly.
Keywords: Courtship songBackground
For successful mating, animals actively evaluate candidate partners for mating by detecting and preferring species-specific sensory signals. In higher animals, both innate instinct and social experience acquired after birth contribute to the mating preference. Indeed, in species such as birds, fishes, sheep and goats, innate preference is dominant, while later social interactions with parents or siblings fine-tune the phenotypic discrimination and mating preference (Owens et al., 1999; Ten Cate and Vos, 1999; Kozak et al., 2011).
The fruit fly, Drosophila melanogaster, has been an invaluable model to study the neural mechanism underlying mating behaviors at the level of genes, cells, and circuits (Kazama, 2015). A male fly courts a female with a stereotyped ritual before he succeeds in copulation (Yamamoto and Koganezawa, 2013). Both male and female flies have the innate ability to evaluate species-specific sensory signals emitted during this courtship ritual, such as visual cues and pheromones, as well as courtship songs, and choose a conspecific mating partner (Zhou et al., 2014; Clowney et al., 2015; Kallman et al., 2015). Although it is traditionally believed that the courtship behavior of flies is innate (Hall, 1994; Baker et al., 2001; Auer and Benton, 2016), the programmed courtship circuitry is susceptible to variables in development such as sleep deprivation (Kayser et al., 2014), social isolation (Kim and Ehrman, 1998; Pan and Baker, 2014), and auditory experience (Li et al., 2018). The courtship behavior of flies is also affected by previous courtship outcomes, which has been used as a courtship conditioning assay to evaluate learning and memory in male flies (Keleman et al., 2012; Koemans et al., 2017). Moreover, researchers developed a sensitive system to record and analyze the courtship songs (Arthur et al., 2013). By combining this system with computational modeling, Coen et al. (2014) found that males courtship song can be modulated by dynamic sensory experience acutely. However, to what extent social experience and developmental plasticity contribute to the perception of the courtship song is not well understood.
Here we report a behavioral paradigm to assess how an experience of hearing the conspecific song as a young adult sharpens the song preference and mate selection as a breeding adult in the fruit fly. This paradigm consists of training session and test session. At the training session, we expose single males or females to an artificial courtship song. At the test session, we use the male-female courtship behavior and male-male chaining behavior to measure the response in females and males, respectively. By combining this brand-new behavioral paradigm with sophisticated genetic tools established in flies, such as the GAL4/UAS binary expression system to manipulate individual genes and neurons, this novel approach gives the potential to clarify the neural mechanism on how experience-dependent tuning of the mating preference is built upon both innate and experience-dependent auditory systems.
Materials and Reagents
- Pipette tips, with volumes of 1 ml (FUKAEKASEI and WATSON, catalog number: 110-706C )
- Pasteur pipette (Borosilicate Glass Pasteur Pipette, Corning, catalog number: 7095D-5X , 6.5 mm in outer diameter, 146 mm in length)
- Stocking mesh made of nylon and polyurethane (Small pieces of stocking mesh)
- Plastic insect screen mesh (Mesh size 24: 0.84 mm)
- Rubber sheet (10 mm thick)
- Mending tape
- Empty plastic vials (Chiyoda Science, catalog number: KFB-1M ) for cold anesthesia
- Drosophila melanogaster, fly strains to be tested of both sexes
- Ice for anesthesia
- Artificial pulse song (Figure 1) (Supplemental audio file 1, Supplemental audio file 2)
A sound file comprised of the repetition of 1-sec pulse burst and a subsequent 2-sec pause, in which the inter-pulse interval (IPI) is 35 msec (“conspecific song”, Supplemental audio file 1) or 75 msec (“heterospecific song” Supplemental audio file 2). Intra-pulse frequency of both songs is 167 Hz. Use Audacity to make the artificial pulse songs. - Fly food (Standard yeast-based media) (see Recipes)
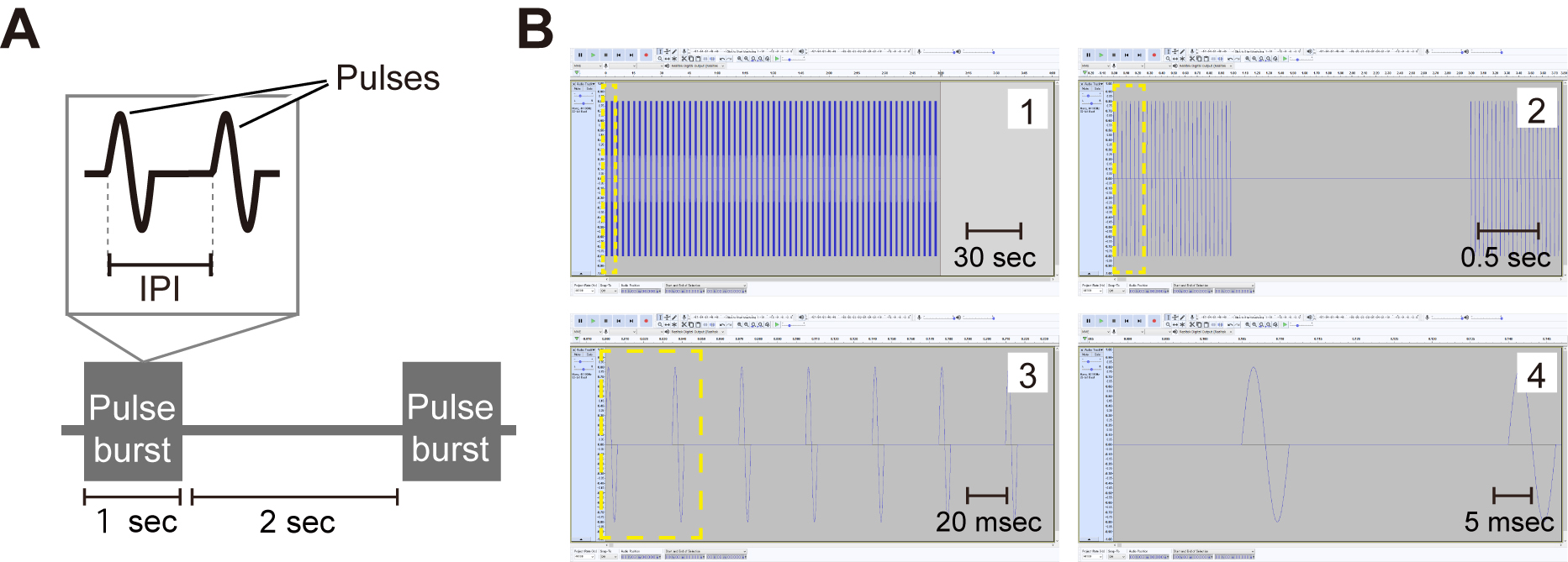
Figure 1. Artificial pulse song. A. A scheme of a sound file. Inter-pulse interval (IPI) is set to be 35 msec or 75 msec. B. Preparation of a sound file with Audacity. A series of magnified views are shown.
Equipment
- Loudspeakers (8 ohm, Ø 185 mm, Fostex, Foster Electric, catalog number: FF225WK )
Daito Voice AR-10N, 8 ohm, Ø 100 mm, Tokyo Cone Paper MFG. Co. Ltd., Saitama, Japan - Stereo zoom microscope (Leica Microsystems, model: Leica S6 E )
- Two pairs of forceps (Roboz Surgical Instrument, Dumont, model: Dumostar No. 5, catalog number: RS-4976 ) for ablating wings from flies
- Aspirator
A custom-made flexible aspirator to pick up flies gently. Assembled from a mouthpiece (Pipette tips, with volumes of 1 ml), latex tube (about 50 cm), mesh, and a glass tube (about 5 cm) to keep flies (Pellegrino et al., 2010). - Sound nest (Figure 2A)
A custom-made latticework of a container to hold the training capsules during the training session. We used cardboard dividers of a freezer box (AS ONE, catalog number: 1-1473-01 ). Each well of the cardboard dividers holds a training capsule. - Training capsule (Figure 2B)
A custom-made capsule made of a glass tube cut out from a Pasteur pipette, two pipette tips, a piece of stocking mesh and pieces of mending tape. Cut pipette tips (1 ml) to make their larger ends about 20 mm long. Hook two of these 20 mm pieces to a glass tube at its both ends. The size of a glass tube is about 27 mm long, with the internal diameter of 5.2 mm and the external diameter of 6.5 mm. Seal both exits of the glass tube with a piece of stocking mesh (made of nylon and polyurethane), which allowed free passage of air but not the fly. A thin layer of fly food, standard Drosophila yeast-based medium, is paved at the bottom of the glass tube. - Digital power amplifier (20 W, Lepai, model: LP-2020A+NFJ Edition )
Note: A simple and inexpensive power amplifier. Other amplifier can also be used. - Soundproof box (Sonora Technology Co. Ltd, Aichi, Japan) (Figure 2C)
Note: A custom-made steel soundproof box with a ventilating hole.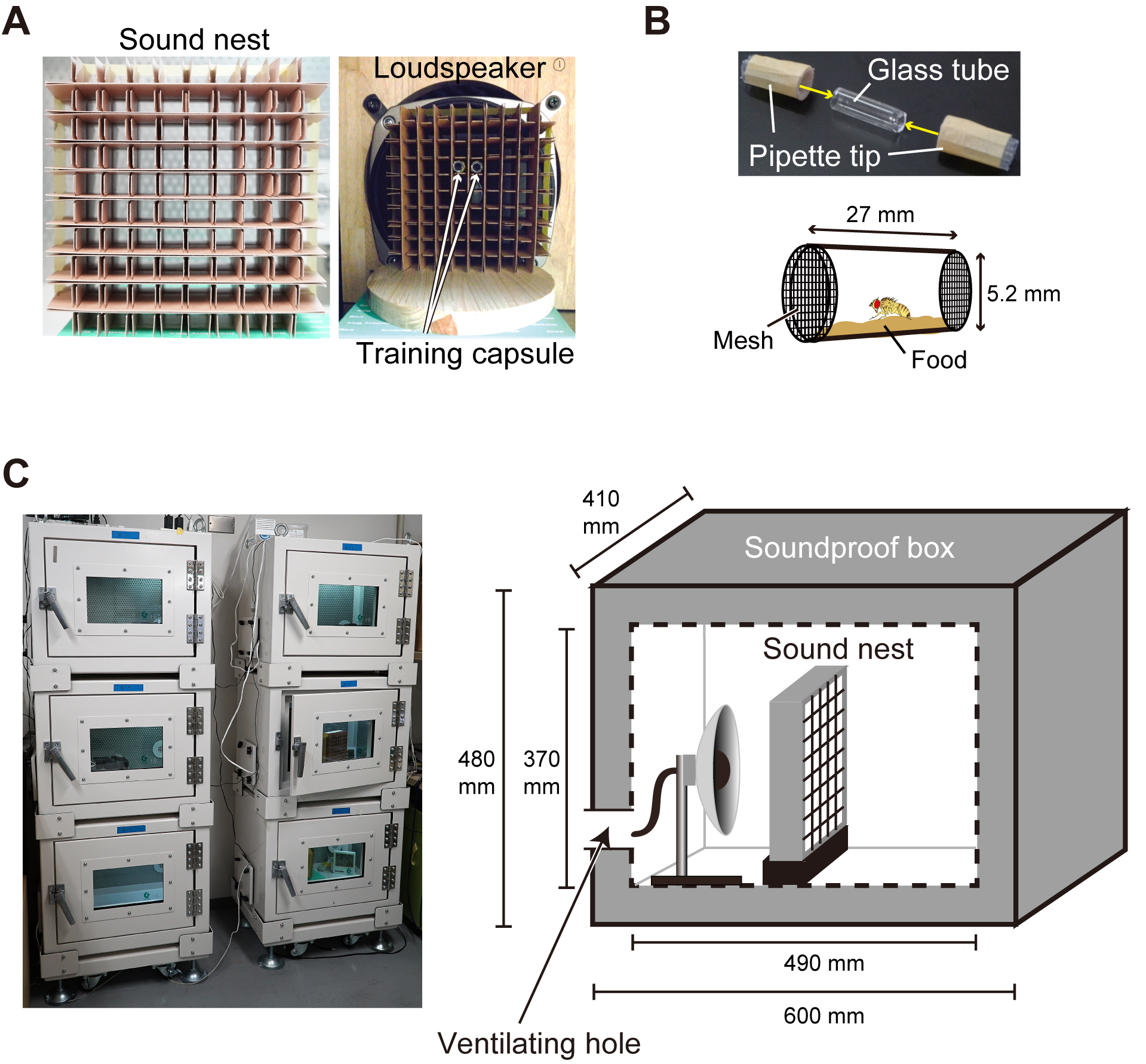
Figure 2. Training setup. A. Sound nest to hold training capsules. B. Training capsule. Two pipette tips sealed with stocking mesh cover both open ends of a glass tube (upper). A schematic of a training capsule during the training session (lower). C. Soundproof boxes for the training (left), each of which contains a loudspeaker and sound nest (right). - Tablet PC (DOSPARA, Windows 8.1, model: Diginnos DG-D08IWB )
Note: A simple and inexpensive Tablet PC. Other device to playback a sound file can also be used. - Female copulation plate (Figure 3A)
A custom-made apparatus made of Plexiglas, which consists of eight circular chambers (15 mm in diameter, 3 mm in depth) with their bottom covered with a circular sheet of insect screen mesh for sound penetration. It is made from a pair of transparent Plexiglas panels with eight holes (chamber and base) sandwiching the insect screen mesh and a circular shaped cover Plexiglas (70 mm in diameter, 1 mm in depth). The cover plate has a loading hole at the rim (3 mm in diameter) in order to load the flies in rotation. A screw fixes all the elements through the center hole (4 mm in diameter). - Rubber support
A custom-made apparatus to support the female copulation plate. Make a hole (70 mm in diameter) in the middle of a rubber sheet to hold the female copulation plate. - Male-male chaining chamber (Figure 3B)
Note: This is a custom-made apparatus made of Plexiglas. See Inagaki et al., 2010 for detailed structure and size of the apparatus. - Web camera (Logitech, Logicool®, model: HD Webcam C270 )
- Small LED light array to illuminate female copulation chambers (Figure 5B)
- LED light box (ComicMaster Tracer, Too Marker Products, Japan)
- Behavior assay room with controlled temperature (25 ± 1 °C) and humidity (50 ± 10%)
- Thermometer for monitoring the temperature at the point of the experiment
- Ultrasonic washing machine for washing the acoustic behavior chambers (AS ONE, model: ASU-6 )
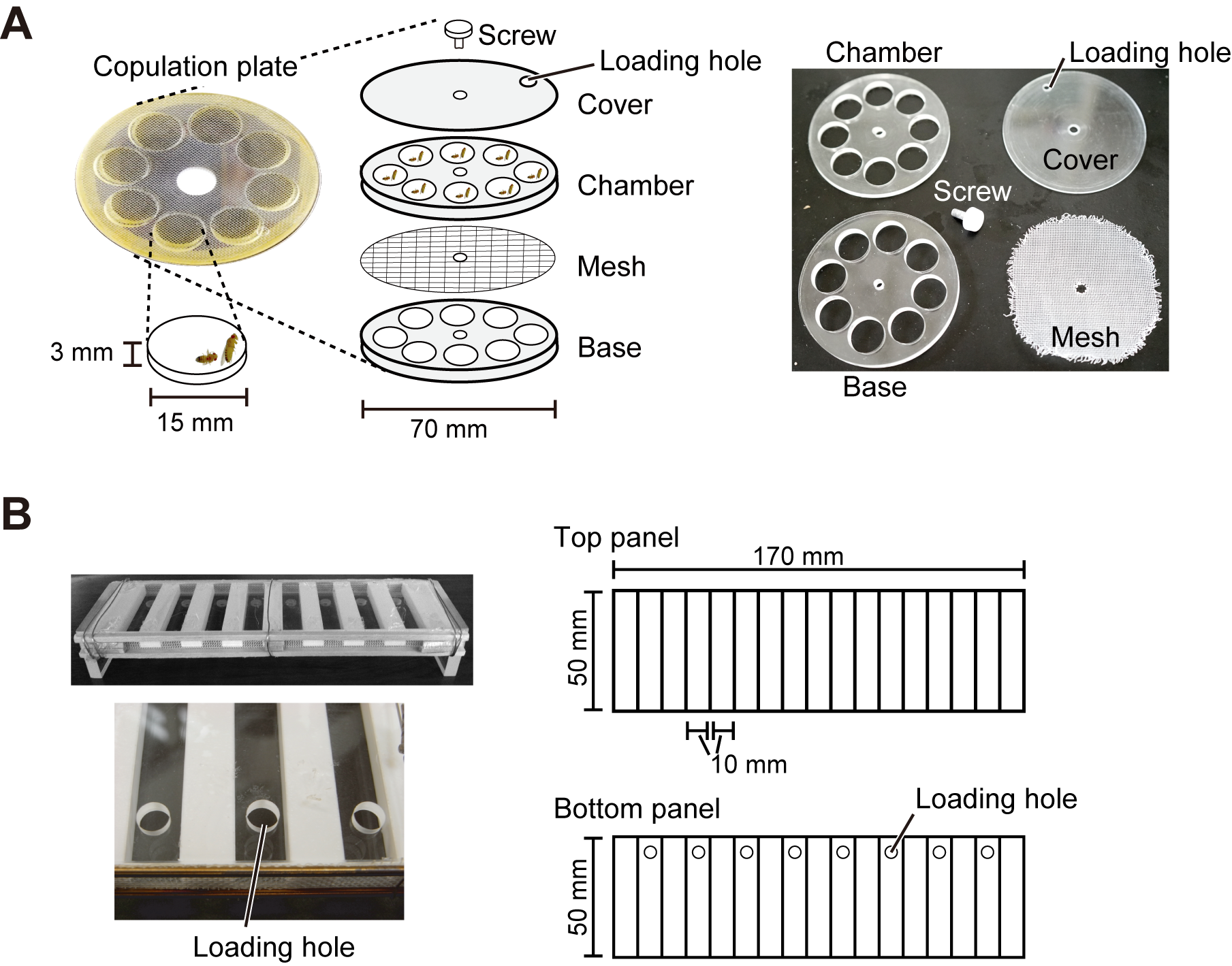
Figure 3. Apparatus for behavioral assays. A. Copulation plate for the female copulation assay. B. Apparatus for the male-male chaining assay.
Software
- Audacity (The Audacity Team) (https://www.audacityteam.org)
Free open source, cross-platform audio software - Windows Media Player (Microsoft)
Other media player can also be used. - Logitech Webcam Software
Provided with the web camera - ChaIN (http://www.bio.nagoya-u.ac.jp/~NC_home/chain_E.html)
Custom-made software (Yoon et al., 2013) - R (https://www.r-project.org)
Free open source, cross-platform for statistical computing and graphics
Procedure
- Preparing flies
- Trainee
- Culture flies at 25 ± 1 °C and 50 ± 10% relative humidity in a 12-h light/dark cycle.
- Collect virgin females and virgin males within 8 h after eclosion under ice anesthesia.
- Clip the male wings with forceps (Figure 4). Keep the female wings intact.
- Subject these flies immediately to the training session after recovering from the anesthesia.

Figure 4. Operation for clipping fly wings. Flies are anesthetized on an ice-cold aluminum plate. Wings of male flies are clipped at their bases (dotted line).
- Culture flies at 25 ± 1 °C and 50 ± 10% relative humidity in a 12-h light/dark cycle.
- Partner male
- Use wild-type male flies for the female copulation assay as partner males. Clip the wings of these males right after eclosion.
- Keep them singly in a plastic vial with fly food at 25 °C in a 12-h light/dark cycle until experiments. Transfer each fly to a new plastic vial with fly food every 2 to 3 days, but not on the day of experiment. Use 7-day old males for the female copulation assay.
- Use wild-type male flies for the female copulation assay as partner males. Clip the wings of these males right after eclosion.
- Trainee
- Training session
- Keep the room temperature at 25 ± 1 °C and relative humidity 50 ± 10%.
- Introduce each fly gently to a training capsule on the day of eclosion.
- Set training capsules in the sound nest.
- Place the sound nest in front of a loudspeaker ( FF225WK ). One of the mesh-ends of each training capsule faces the loudspeaker, so that sound is delivered to each chamber with minimal disturbance. The distance between the loudspeaker and the near end of the training capsules is 24 mm.
- Start an artificial pulse song (conspecific song or heterospecific song) playback for experienced flies. For Naïve flies, no sound is played. The mean baseline-to-peak amplitude of sound particle velocity is 8.6 mm/sec when measured at the near end of the training capsules, and 6.6 mm/sec at the far end of the training capsule. The sound particle velocity is identical for all training sounds.
- The artificial pulse song, 3-min long, is stored in a WAV format audio file (monaural, 44,100 Hz, 32-bit float). Play the audio file repeatedly with Windows media player for 6 days. Renew food in each training capsule every 36 h.
- After the 6-day training, collect male flies into a vial containing fly food in a group of seven without anesthesia until male-male chaining assay. Keep the female flies in the training capsules singly without sound playback until the female copulation assay.
- Keep the room temperature at 25 ± 1 °C and relative humidity 50 ± 10%.
- Test session
Notes:- Keep the room temperature at 25 ± 1°C and relative humidity 50 ± 10%.
- Perform behavioral tests within 4 h after light onset on the 7th day.
- In female copulation assay, the intactness of female wings is critical for copulation success. Ruffled or crippled wings will lower the copulation success rate. Load flies (one pair into each chamber) rapidly. To avoid early copulation and to give all the fly pairs a universal start in interaction, shake the chamber after each action of puffing.
- Female copulation assay (Figure 5)
- Use trained females and partner males in this assay.
- Transfer a pair of female and male flies gently with an aspirator through the loading hole into one of the eight chambers in the copulation plate rapidly without anesthesia. For each recording, load up to 8 pairs of flies (Figure 5A).
- Immediately after the transfer of the flies, insert the apparatus to a rubber support and place it over a loudspeaker (Daito Voice AR-10N). The loudspeaker is placed 39 mm under the chambers. Two small LED light arrays are placed at both sides of the loudspeaker to illuminate the female copulation chambers (Figure 5B).
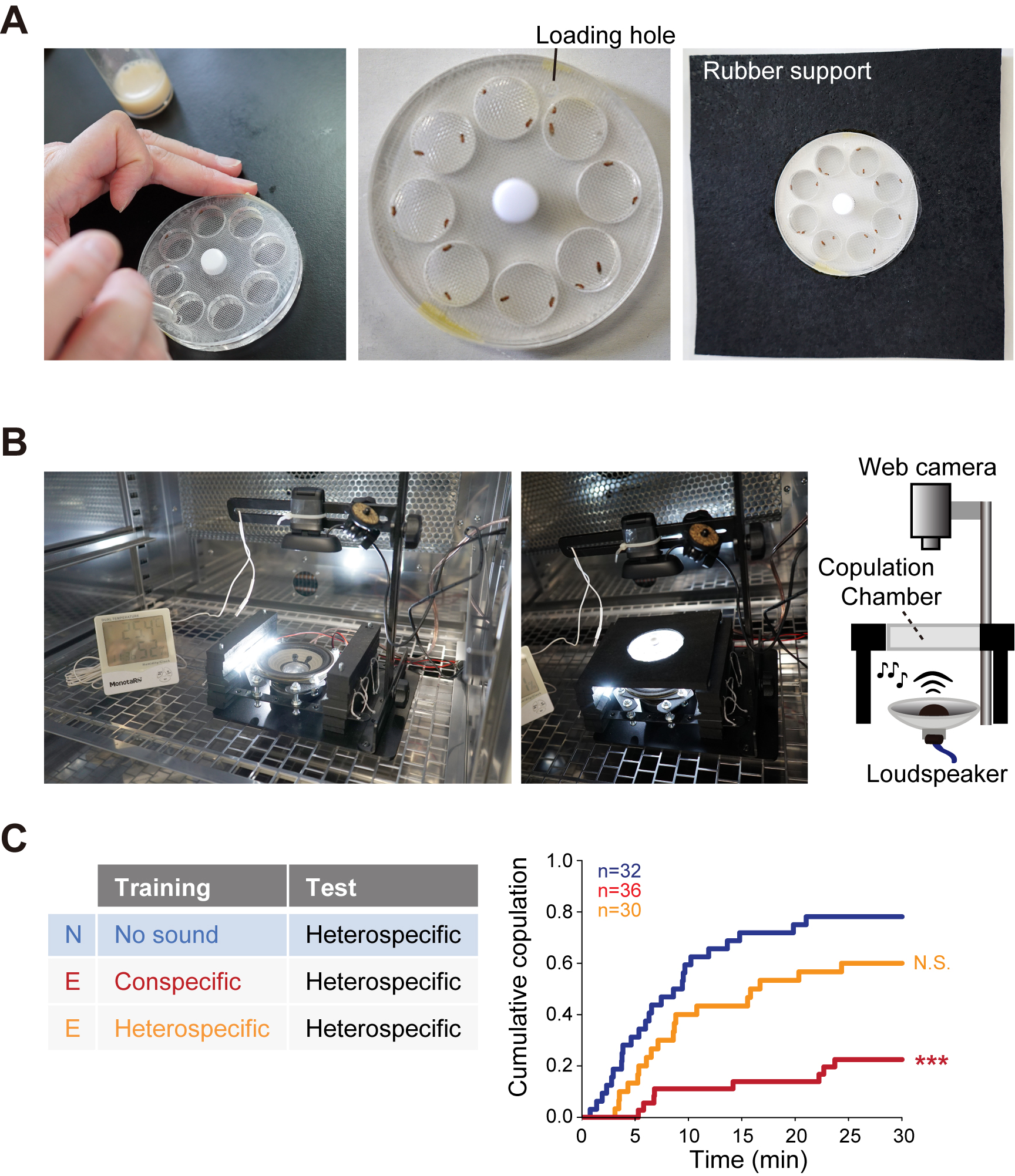
Figure 5. Female copulation assay. A. Loading flies into a chamber. A fly pair is loaded into one of eight copulation chambers by using an aspirator. Each chamber has a test female and a partner male. The copulation plate is inserted into a hole of the rubber support. B. Assay setup. Rubber support holding the copulation plate is placed over a loudspeaker with appropriate distance. C. Example graph of song response of female flies. Cumulative copulation rate in the heterospecific song test after training is plotted. Naïve group (no sound during training) and experienced groups (trained with conspecific song or heterospecific song) are shown. N, Naïve; E, experienced. N.S., not significant, P > 0.05; ***P < 0.001; Kruskal–Wallis test versus Naïve group.- Start the sound playback and video recording simultaneously. The particle velocity received at the apparatus is adjusted as 9.2 mm/sec.
- Record the behaviors of flies for 30 min with a web camera (Logitech, Logicool®) with a video capture software (Logitech Webcam Software) (Video 1).Video 1. A video clip showing female copulation assay. Copulation started in a pair out of eight pairs.
- Use trained females and partner males in this assay.
- Male-male chaining assay (Figure 6)
Use trained males in this assay. See Inagaki et al., 2010 for a protocol.
- Keep the room temperature at 25 ± 1°C and relative humidity 50 ± 10%.
Data analysis
Statistical analysis is performed with R (version 3.0.3). All data related to this study are already published in Li et al., 2018.
- Female copulation assay
Evaluate behavioral response of females to the artificial pulse songs by observing the copulation latency (time to mating from the test started) of a female paired with a partner male in the chamber. Here, copulation is defined by observing the specific features as follows: (1) females permit a male to mount them for more than 1 min, (2) females reduce their locomotor activity with the mounting partner, and (3) females part her wings during the mounting (Manning, 1967; Yamada et al., 2018).
This copulation latency is analyzed manually from the video playback.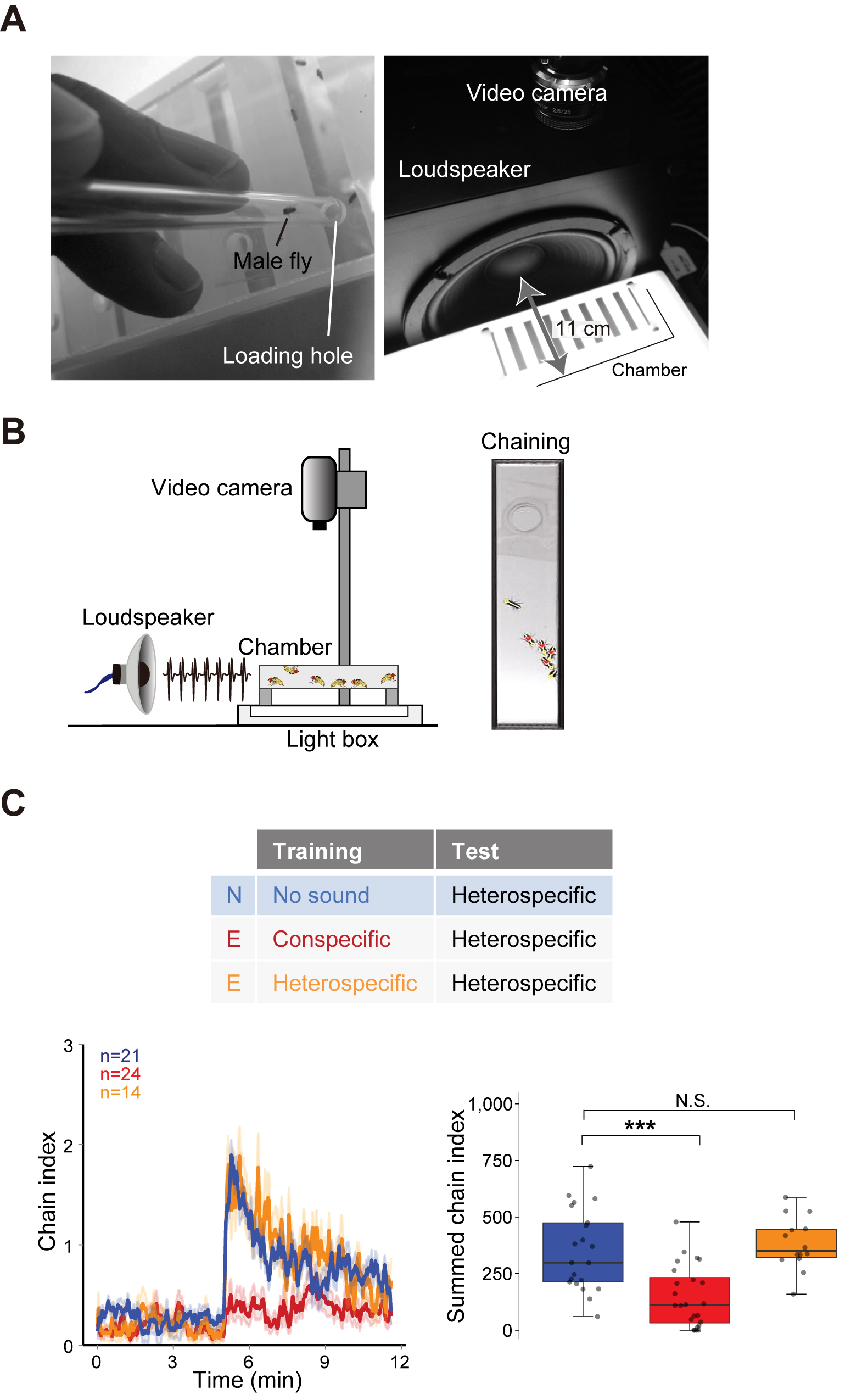
Figure 6. Male-male chaining assay. A. Six males are loaded into a chamber by using an aspirator. Put the chamber in front of a loudspeaker. B. Illustration depicts the male-male chaining assay setup (left). An actual image of male-male chaining behavior (right). C. Example graph of song response of male flies. The time-courses of the chain index in response to playback of heterospecific song are shown (Left). Sound playback starts at 5 min. The bold line and ribbon represent the average value and standard error, respectively. The box plot shows the summed chain index between 5-min and 11.5-min (right). N, Naïve group with no sound training (blue); E, experienced group with conspecific song training (red) or heterospecific song training (orange). N.S., not significant, P > 0.05; ***P < 0.001; Mann-Whitney U test versus Naïve group. - Male-male chaining assay
Measure the number of the follower flies in chains as the chain index using the custom-made software: ChaIN version 3 (Ishikawa et al., 2017).
Recipes
- Standard fly food
Use the following ingredients for 1 L of standard fly food:Ingredient Quantity Agar 8 g Cornmeal 40 g Yeast 45 g Glucose 100 g Propionic acid 4 ml 10% Methyl p-Hydroxybenzoate in 70% EtOH 3 ml
Acknowledgments
This work was supported by MEXT KAKENHI Grant-in-Aid for Scientific Research (B) (Grant 16H04655 to AK), the Grants-in-Aid for Scientific Research on Innovate Areas "Evolinguistics" (Grant 18H05069 to AK), Challenging Research (Exploratory) (Grant 17K19450 to AK), Grant-in-Aid for Scientific research (C) (15K07147 to HI), JSPS KAKENHI Grant-in-Aid for JSPS Fellows (18J15228 to XL), and Inamori Foundation Research Grant, Japan (HI). This protocol was adapted from procedures published in Li et al. (2018). Figures 2, 3, 5, and 6 were modified and reproduced with permission from Li et al. (2018).
Competing financial interests: The authors declare no competing financial interests.
References
- Arthur, B. J., Sunayama-Morita, T., Coen, P., Murthy, M. and Stern, D. L. (2013). Multi-channel acoustic recording and automated analysis of Drosophila courtship songs. BMC Biol 11: 11.
- Auer, T. O. and Benton, R. (2016). Sexual circuitry in Drosophila. Curr Opin Neurobiol 38: 18-26.
- Baker, B. S., Taylor, B. J. and Hall, J. C. (2001). Are complex behaviors specified by dedicated regulatory genes? Reasoning from Drosophila. Cell 105(1): 13-24.
- Clowney, E. J., Iguchi, S., Bussell, J. J., Scheer, E. and Ruta, V. (2015). Multimodal chemosensory circuits controlling male courtship in Drosophila. Neuron 87(5): 1036-1049.
- Coen, P., Clemens, J., Weinstein, A. J., Pacheco, D. A., Deng, Y. and Murthy, M. (2014). Dynamic sensory cues shape song structure in Drosophila. Nature 507(7491): 233-237.
- Hall, J. C. (1994). The mating of a fly. Science 264(5166): 1702-1714.
- Inagaki, H. K., Kamikouchi, A. and Ito, K. (2010). Protocol for quantifying sound-sensing ability of Drosophila melanogaster. Nat Protoc 5(1): 26-30.
- Ishikawa, Y., Okamoto, N., Nakamura, M., Kim, H. and Kamikouchi, A. (2017). Anatomic and physiologic heterogeneity of subgroup-A auditory sensory neurons in fruit flies. Front Neural Circuits 11: 46.
- Kallman, B. R., Kim, H. and Scott, K. (2015). Excitation and inhibition onto central courtship neurons biases Drosophila mate choice. Elife 4: e11188.
- Kayser, M. S., Yue, Z. and Sehgal, A. (2014). A critical period of sleep for development of courtship circuitry and behavior in Drosophila. Science 344(6181): 269-274.
- Kazama, H. (2015). Systems neuroscience in Drosophila: Conceptual and technical advantages. Neuroscience 296: 3-14.
- Keleman, K., Vrontou, E., Krüttner, S., Yu, J. Y., Kurtovic-Kozaric, A. and Dickson, B. J. (2012). Dopamine neurons modulate pheromone responses in Drosophila courtship learning. Nature 489(7414): 145-149.
- Kim, Y. K. and Ehrman, L. (1998). Developmental isolation and subsequent adult behavior of Drosophila paulistorum. IV. Courtship. Behav Genet 28(1): 57-65.
- Koemans, T. S., Oppitz, C., Donders, R. A. T., van Bokhoven, H., Schenck, A., Keleman, K. and Kramer, J. M. (2017). Drosophila courtship conditioning as a measure of learning and memory. J Vis Exp (124).
- Kozak, G. M., Head, M. L. and Boughman, J. W. (2011). Sexual imprinting on ecologically divergent traits leads to sexual isolation in sticklebacks. Proc Biol Sci 278(1718): 2604-2610.
- Li, X., Ishimoto, H. and Kamikouchi, A. (2018). Auditory experience controls the maturation of song discrimination and sexual response in Drosophila. Elife 7: e34348.
- Manning, A. (1967). The control of sexual receptivity in female Drosophila. Anim Behav 15(2): 239-250.
- Owens, I. P., Rowe, C. and Thomas, A. L. (1999). Sexual selection, speciation and imprinting: separating the sheep from the goats. Trends Ecol Evol 14(4): 131-132.
- Pan, Y. and Baker, B. S. (2014). Genetic identification and separation of innate and experience-dependent courtship behaviors in Drosophila. Cell 156(1-2): 236-248.
- Pellegrino, M., Nakagawa, T. and Vosshall, L. B. (2010). Single sensillum recordings in the insects Drosophila melanogaster and Anopheles gambiae. J Vis Exp (36): 1-5.
- Ten Cate, C. and Vos, D.R. (1999). Sexual imprinting and evolutionary processes in birds: a reassessment. Adv Study Behav 28: 1-31.
- Yamada, D., Ishimoto, H., Li, X., Kohashi, T., Ishikawa, Y. and Kamikouchi, A. (2018). GABAergic local interneurons shape female fruit fly response to mating songs. J Neurosci 38(18): 4329-4347.
- Yamamoto, D. and Koganezawa, M. (2013). Genes and circuits of courtship behaviour in Drosophila males. Nat Rev Neurosci 14(10): 681-692.
- Yoon, J., Matsuo, E., Yamada, D., Mizuno, H., Morimoto, T., Miyakawa, H., Kinoshita, S., Ishimoto, H. and Kamikouchi, A. (2013). Selectivity and plasticity in a sound-evoked male-male interaction in Drosophila. PLoS One 8(9): e74289.
- Zhou, C., Pan, Y., Robinett, C. C., Meissner, G. W. and Baker, B. S. (2014). Central brain neurons expressing doublesex regulate female receptivity in Drosophila. Neuron 83(1): 149-163.
Article Information
Copyright
Li et al. This article is distributed under the terms of the Creative Commons Attribution License (CC BY 4.0).
How to cite
Readers should cite both the Bio-protocol article and the original research article where this protocol was used:
- Li, X., Ishimoto, H. and Kamikouchi, A. (2018). Assessing Experience-dependent Tuning of Song Preference in Fruit Flies (Drosophila melanogaster). Bio-protocol 8(14): e2932. DOI: 10.21769/BioProtoc.2932.
- Li, X., Ishimoto, H. and Kamikouchi, A. (2018). Auditory experience controls the maturation of song discrimination and sexual response in Drosophila. Elife 7: e34348.
Category
Neuroscience > Behavioral neuroscience > Learning and memory
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link










