- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Ex vivo Analysis of Lipolysis in Human Subcutaneous Adipose Tissue Explants
Published: Vol 8, Iss 3, Feb 5, 2018 DOI: 10.21769/BioProtoc.2711 Views: 8026
Reviewed by: Salma MerchantAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Metabolomic and Lipidomic Analysis of Bone Marrow Derived Macrophages
Gretchen L. Seim [...] Jing Fan
Jul 20, 2020 9039 Views

OrganoPlate Micro-fluidic Microvessel Culture and Analysis
Abidemi Junaid and Thomas Hankemeier
Jul 5, 2021 4560 Views
Mass Spectrometry-based Lipidomics, Lipid Bioenergetics, and Web Tool for Lipid Profiling and Quantification in Human Cells
Liang Cui [...] Kuan Rong Chan
Aug 20, 2023 3039 Views
Abstract
Most studies of human adipose tissue (AT) metabolism and functionality have been performed in vitro on isolated mature adipocyte or in situ using the microdialysis technique (Lafontan, 2012). However, these approaches have several limitations. The use of mature isolated adipocytes is limiting as adipocytes are not in their physiological environment and the collagenase digestion process could affect both adipocyte survival and functionality. While metabolic studies using microdialysis have brought the advantage of studying the lipolytic response of the adipose tissue in situ, it provides only qualitative measures but does not give any information on the contribution of different adipose tissue cell components. Moreover, the number of microdialysis probes that can be used concomitantly in one subject is limited and can be influenced by local blood flow changes and by the molecular size cut-off of the microdialysis probe. Here we present a protocol to assess adipose tissue functionality ex vivo in AT explants allowing the studies of adipose tissue in its whole context, for several hours. In addition, the isolation of the different cell components to evaluate the cell-specific impact of lipolysis can be performed. We recently used the present protocol and demonstrated that fatty acid release during lipolysis impacts directly on a specific cell subset present in the adipose tissue stroma-vascular compartment. This assay can be adapted to address other research questions such as the effects of hormones or drugs treatment on the phenotype of the various cell types present in adipose tissue (Gao et al., 2016).
Keywords: Human adipocyte biologyBackground
Human white adipose tissue (WAT) plays a major role in body energy homeostasis. Adipocytes, specialized cells expressing specific lipid handling metabolic activities, constitute more than 90% of the volume of WAT (Lafontan, 2012). In addition to adipocytes, other cell types are present within human WAT e.g., vascular cells, immune cells (lymphocytes and macrophages) and progenitor cells involved in WAT remodeling and renewal. The metabolic activity of adipocytes is tightly controlled by the integration of both local and systemic pathways. Neurohumoral signals modulated in anabolic or catabolic conditions impact on the net adipocyte metabolic activity, i.e., energy storage or release. In post-prandial conditions, non-esterified fatty acids (NEFAs) originating from the hydrolysis of VLDL and chylomicron particles are taken up by the adipocytes and esterified to glycerol phosphate to form triglycerides packaged into a single lipid vacuole (Large et al., 2004). This process called lipogenesis, is mainly under the control of insulin. In conditions of energy demand such as exercise or fasting, hydrolysis of triacylglycerol through a process called lipolysis results in the release of glycerol and NEFAs into the circulation thereby providing energy to other tissues and organs. Lipolysis involves the sequential hydrolysis of triacylglycerol through the successive action of lipolytic enzymes, i.e., adipose triglyceride lipase, hormone sensitive lipase and monoacylglycerol lipase. In human adipocytes, lipolysis is mainly stimulated by catecholamines and atrial natriuretic peptide (ANP). Catecholamines mediate their pro-lipolytic effects through β-adrenoceptors (β1-AR and β2-AR), while alpha2-adrenoceptors are anti-lipolytic; ANP acts via NPR-A to activate lipolysis (Lafontan, 2012). It should be noted that in human AT the beta3-adrenergic receptor is almost not expressed and is not functional. The classical approach to evaluate adipocyte lipolytic responses was developed by Robdell (Robdell, 1964) who first described mature adipocyte isolation based on flotation after collagenase digestion. Although this technique is used worldwide, it presents several limitations. Firstly, the buoyancy of isolated mature adipocytes will prevent, with increasing time in vitro, their immersion into media and promote direct toxic effects through air contact, ultimately leading to cell damage and disintegration. Secondly, the isolation process per se alters adipocyte phenotype (Ruan et al., 2003). Thirdly, the isolated mature adipocytes are disconnected from their natural microenvironment including extracellular matrix and from other cell types (vascular cells, immune cells (lymphocytes and macrophages) and progenitor cells). Moreover, cytokines such as TNF-alpha and IL6, present in the AT microenvironment, are well described to impact adipocyte lipolysis. Finally, NEFAs originating from in situ lipolysis may have different fates: 1) release into the circulation, 2) re-esterification within the mature adipocytes, 3) potentially taken up by other cell types in the near vicinity of mature adipocytes including progenitor cells. Thus studies on isolated adipocytes do not take into account these different factors.
The present technique allows the study of the lipolytic responsiveness of mature adipocytes for a longer time in their natural context 1) in a closed culture chamber avoiding direct contact with air but with adequate and modulable gas exchange, 2) without the necessity of a collagenase digestion step and 3) in a maintained viable microenvironment. This approach was recently published by our groups and clearly demonstrate that lipolytic stimulation is associated with increased fatty acid uptake by the progenitor cells leading to enhanced adipogenic capacity (Gao et al., 2016).
Materials and Reagents
- Pipettes tips (Dutscher, ClearLine®, catalog numbers: 037660CL (10 µl); 032260CL (200 µl); 027120CL (1,000 µl))
- 50 ml conical tubes (Corning, Falcon®, catalog number: 352070 )
- 20 ml syringe (Terumo, catalog number: SS-20ES1 )
- Sterile individually packaged 5 ml graduated pipettes (Corning, Falcon®, catalog number: 357543 )
- Sterile 150 x 20 mm cell culture polystyrene Petri dish (Thermo Fisher Scientific, Thermo ScienticTM, catalog number: 168381 )
- 10 ml syringe (Terumo, catalog number: SS-10ES1 )
- Sterile individually needle 21 G x 11/2” (Terumo, catalog number: NN-2138R )
- 15 ml conical tubes (Greiner Bio One International, catalog number: 188271 )
- 0.22 µm syringe filter (Dutscher, catalog number: 051732 )
- 96 wells microplate clear flat bottom (Thermo Fisher Scientific, Thermo ScienticTM, catalog number: 269620 )
- 50 ml syringe (Terumo, catalog number: SS-50L1 )
- Filtration unit for sterilization, Stericup 500 ml (Merck, catalog number: SCGPU05RE )
- Clinicell® 25 cassette (Mabio International, catalog number: 00109 )
- RNA lysis buffer (Quiazol, QIAGEN, catalog number: 79306 )
- Free glycerol reagent (Sigma-Aldrich, catalog number: F6428 )
- Wako NEFA (SOBIODA, catalog numbers: W1W434-91795 and W1W436-91995 )
- Lactate FS (Diasys Diagnostics, catalog number: 1400199109 )
- Phosphate-buffered saline (PBS) (Sigma-Aldrich, catalog number: D8537 )
- Glucose GOD FS (Diasys Diagnostics, catalog number: 1250099100 )
- Gentamycin (Sigma-Aldrich, catalog number: G1272 )
- ECBM buffer (PromoCell, catalog number: C-22210 )
- Bovine serum albumin (BSA) (Sigma-Aldrich, catalog number: A7030 )
- HEPES 1 M (PAA, catalog number: S11-001 )
- Sodium bicarbonate (Sigma-Aldrich, catalog number: S5761 )
- Krebs Ringer powder (Sigma-Aldrich, catalog number: K4002 )
- (-)Isoproterenol hydrochloride (Sigma-Aldrich, catalog number: I6504 )
- Human alpha-ANF(1-28) (R&D Systems, catalog number: 1906/1 )
- ECBM medium (see Recipes)
- BSA free fatty acid 20% (see Recipes)
- Adipose tissue explant media (ATEM) (see Recipes)
- Krebs-Ringer Bicarbonate HEPES 0.1% BSA (KRBHA) (see Recipes)
- Isoproterenol stock solution (see Recipes)
- ANP stock solution at 200 µM (see Recipes)
Equipment
- P20 pipetman (Gilson, catalog number: F123600 )
- P200 pipetman (Gilson, catalog number: F123601 )
- P1000 pipetman (Gilson, catalog number: F123602 )
- Laminar flow hood (Faster, model: BHA36 )
- Sterile steel stainless scissors, 16 cm, straight (Dutscher, catalog number: 005055 )
- Sterile stainless steel dressing forceps 11.5 cm, straight (Dutscher, catalog number: 711202 )
- Stainless steel round-point needle 14 G x 6” (Dutscher, catalog number: 075515 )
- Refrigerated tabletop centrifuge for 15 and 50 ml conical tube (Eppendorf, model: 5810 R )
- 37 °C, 5% CO2 water jacketed incubator (Thermo Fisher Scientific, Thermo ScienticTM, model: HeraCellTM 150i )
- Pipettor PipetGirl (Integra Biosciences, catalog number: 155 021 )
- Benchtop dry bath (Thermo Electron LED, catalog number: D-63505 )
- Autoclave
- -20 °C freezer
- 100 ml beaker (Corning, PYREX®, catalog: 1000-100 )
- 1 L beaker (Corning, PYREX®, catalog: 1000-1L )
- Microplate spectrophotometer (Labsystem, model: iEMS Reader MF )
- pH-meter (Mettler-Toledo, model: EL20 )
Software
- Excel 2013
- GraphPad Prism6 software
Procedure
Notes:
- Pre-warm KRBHA buffer and ATEM (see Recipes) to 37 °C prior to the experiment
- This protocol was performed in sterile conditions under a laminar flow hood.
- Adipose tissue explant preparation (Figure1)
- Cut adipose tissue into pieces of around 1-2 cm3 and put it in a 50 ml conical tubes (Figure 1A). Do not exceed 25 ml of AT explant for each 50 ml conical tube (this amount is sufficient to perform 4-5 x Clinicell AT explant)
- Cut with scissors for 2 min (Figure 1B).
- Add 20 ml of KRBHA, close the tube, and mix gently by inverting the tube 3-4 times.
- Centrifuge the tube at 100 x g for 2 min (Figure 1C).
- Remove the infranatant by using a 14 G stainless cannula and a 20 ml syringe (Figures H-I).
- Repeat Steps A2-A6 until AT explant is able to be pipetted with a 5 ml plastic pipet.
- When AT explants are sufficiently cut, replace the KRBHA by 20 ml of ATEM (see Recipes).
- Close the tube, mix gently by inverting the tube and leave it for 15 min at 37 °C in the benchtop dry bath.
- Remove the infranatant by using the 14 G stainless cannula and a 20 ml syringe (Figures G-I).

Figure 1. Preparation of adipose tissue explants. A. AT pieces before mincing; B. First 2 min mincing; C. AT explants after a first mincing, washing and centrifugation; D-F. Each photograph shows the aspect of the adipose tissue explant after 2 min mincing with scissors and after ATEM addition and 100 x g centrifugation. G-I. AT explants after last ATEM wash, 15 min rest and infranatant removal using a 14 G stainless cannula and a 20 ml syringe.
- Cut adipose tissue into pieces of around 1-2 cm3 and put it in a 50 ml conical tubes (Figure 1A). Do not exceed 25 ml of AT explant for each 50 ml conical tube (this amount is sufficient to perform 4-5 x Clinicell AT explant)
- Clinicell filling (Figure 2)
- Under the hood, take a 150 mm sterile Petri dish and use the lower part.
- Prepare 10 ml of pre-warmed ATEM in a 50 ml conical tube for each Clinicell. For lipolysis with isoproterenol and ANP, the final concentrations used are typically 1 and 0.5 µM respectively. Taking into account that the volume of explant represents 1/3 of the total cassette volume, the real concentration of the lipolytic drug in the ATEM should be then 1.33 and 0.667 µM for isoproterenol and ANP respectively.
- Fill a 10 ml syringe connected with a 21 G needle.
- Open the Clinicell by one side (be cautious to keep the cap sterile) and open a half turn the cap of the other side in order to fill the Clinicell with AT explants (Figure 2B).
- Take the AT explant under the hood and aspirate the ATEM with the14 G stainless cannula and a 20 ml syringe.
- Pipette 3.5 ml of AT explant with a 5 ml pipette connected to a pipettor (Figure 2B).
- Completely fill the Clinicell with the ATEM using the 10 ml syringe connected to the 21 G needle by one side and by tilting it lightly (Figures 2E-2F). When the level of media is nearly to the top of the other side (Figure 2G), close this side and completely fill the cassette and close the second side (Figure 2H).
- Gently mix AT explant and media by tilting the Clinicell by rotating movement and lay it horizontally in the 150 mm Petri dish (Figure 2I).
- Put all the system in the incubator (37 °C, 5% CO2) and leave it for the time you need. Typically 4 h incubation time is sufficient to study short term lipolysis. The incubation time could be extended to 48 h for studying long term effect on adipose tissue stroma cells.

Figure 2. Clinicell cassette filling. A. Disposition of the Clinicell before filling; B-D. Filling of the Clinicell with AT explants with a 5 ml pipet; E-H. Complete filling of the Clinicell with ATEM with a 10 ml syringe connected to a 21 G needle; I. Disposition of the Clinicell in the Petri dish before incubation in the incubator.
- Under the hood, take a 150 mm sterile Petri dish and use the lower part.
- Explant and media recovery (Figure 3)
- Prepare a 15 ml conical tube, a 10 ml syringe and the stainless cannula.
- Take the Clinicell out of the incubator and transfer under the laminar flow hood. After tilting vigorously (Figure 3B) open one side, put it vertically above the 15 ml conical tube, then open the second side and let the explant and the media pour out (Figures 3C-3E).
- Centrifuge the tube at 100 x g for 2 min.
- Recover the conditioned media by using the 10 ml syringe connected to the stainless steel cannula (Figure 3F). When done, vertically return the syringe and cannula, aspirate 1 ml of air, discard the cannula, connect a 0.22 µm filter to the syringe and filtrate the conditioned media Figure 3G).
- Make aliquots of the conditioned media and freeze it at -20 °C or -80 °C for long-term storage. Keep an aliquot of 300 to 500 µl at -20 °C to perform metabolite determination subsequently.
- The AT explants could either be dissociated using collagenase digestion to isolate the stroma-vascular fraction and the mature adipocytes or aliquoted by 0.5 ml and frozen and stored at -80 °C in 0.5 ml RNA lysis buffer.

Figure 3. Recovery of AT explant and conditioned medium. A. Aspect of the explant 24 h after incubation; B-E. Recovery of AT explant and medium; F. Recovery of the conditioned medium after 100 x g centrifugation; G. Representative photography of the explants and the medium after filtration through a 0.22 µm filter.
- Prepare a 15 ml conical tube, a 10 ml syringe and the stainless cannula.
- Lipolysis and metabolites determinations (Figure 4)
Glycerol and free fatty acid determination could be performed using the commercial kits described in the materials and reagents in 96-well microplates and a microplate spectrophotometer. Typically 20 µl of media is sufficient for glycerol and NEFA determination and 5 to 10 µl for glucose and lactate. The standard curves are performed in ATEM medium except for the glucose one which is done in PBS (the glucose concentration in the ATEM medium, which is around 5.5 mM, should be measured in order to be able to calculate the glucose consumption [difference between glucose concentration in ATEM and in AT explants conditioned medium]). Examples of data obtained are presented in Figure 4.
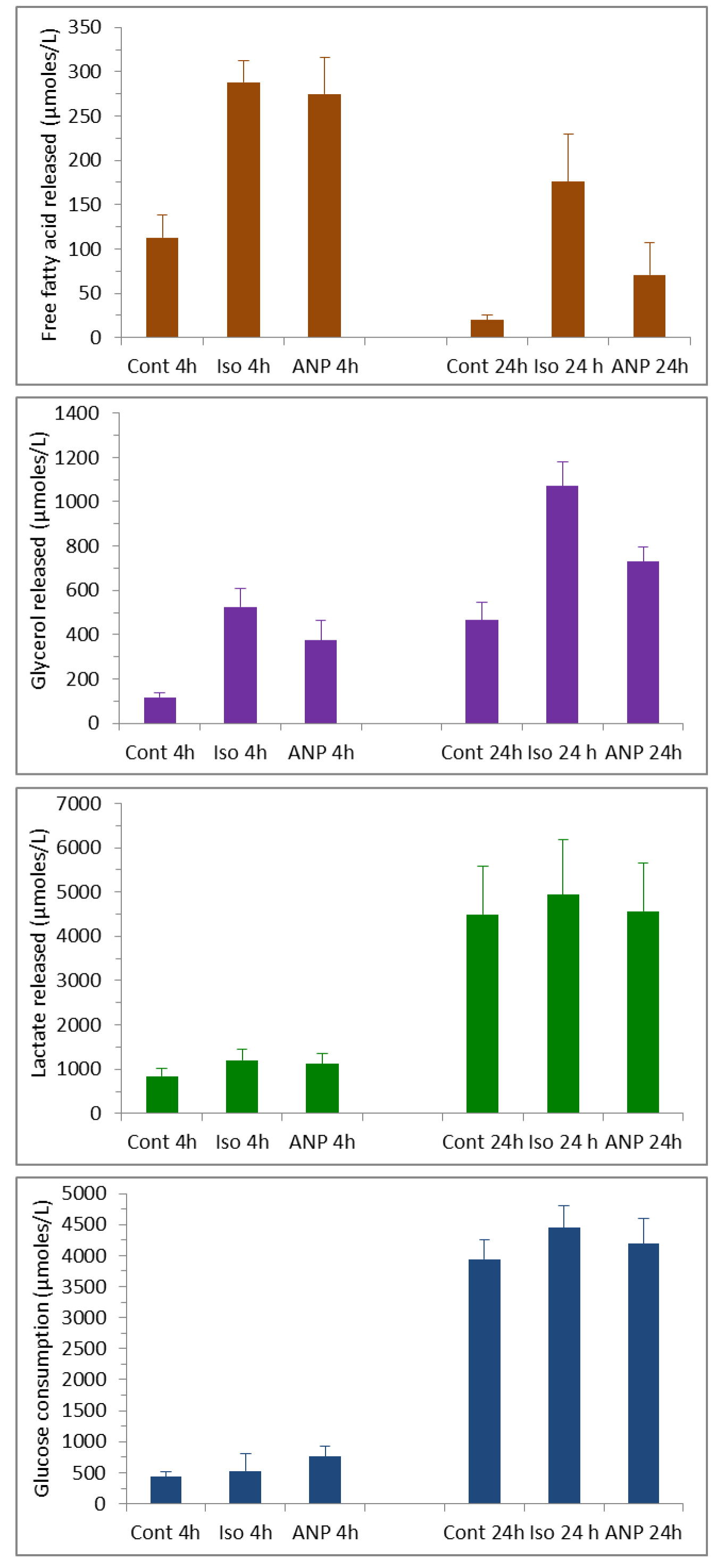
Figure 4. Typical metabolites determination in conditioned media of human AT explants. Data represent the mean ± SEM of 4 to 8 experiments. Lipolysis was induced by either the β-agonist isoprenaline at 1 µM or ANP at 500 nM for the time indicated. Cont represents the basal condition without drugs.
Data analysis
Data calculation and analysis were performed using Excel 2013 and GraphPad Prism6 software.
Recipes
- ECBM medium
Add 2.5 ml of gentamycin solution for one 500 ml bottle of ECBM
- BSA free fatty acid 20% (50 ml)
- Prewarm at 37 °C 40 ml of ECBM medium in a 100 ml beaker
- Weight 10 g of BSA in a 50 ml conical tube
- Add BSA on the top of ECBM medium and let it dissolve at 37 °C
- Adjust pH to 7.4 and complete to 50 ml with ECBM
- Sterilize with a 0.22 µm filter connected to a 50 ml syringe
- Store at 4 °C for several weeks
- Prewarm at 37 °C 40 ml of ECBM medium in a 100 ml beaker
- Adipose tissue explant medium (ATEM) (100 ml)
- Add 1 ml 1 M HEPES and 2.5 ml of 20% BSA free fatty acid to 97.5 ml ECBM
- Adjust to pH = 7.4
- Sterilize by 0.22 µm filtration on a 500 ml Stericup connected to vacuum
- Store at 4 °C
- Add 1 ml 1 M HEPES and 2.5 ml of 20% BSA free fatty acid to 97.5 ml ECBM
- Krebs-Ringer Bicarbonate HEPES 0.1% BSA (KRBHA) (1 L)
- Weight 1,260 g of sodium bicarbonate in a 15 ml conical tube
- Weight 2.38 g of HEPES in a 15 ml conical tube
- In a 1 L beaker put the HEPES, the sodium bicarbonate and one vial of Krebs Ringer powder
- Add 950 ml of ddH2O
- Add 5 ml of 20% BSA , FFA free (Recipe 2)
- Adjust pH to 7.4
- Complete the volume to 1 L with ddH2O
- Sterilize by 0.22 µm filtration on a 500 ml Stericup connected to vacuum
- Store at 4 °C
- Weight 1,260 g of sodium bicarbonate in a 15 ml conical tube
- Isoproterenol stock solution (10 mM)
- Weight 5 mg of isoproterenol and dilute in 2,018 µl of double distilled water
- Sterilize by 0.22 µm filtration
- Make 50 µl aliquots and store at -20 °C
- Weight 5 mg of isoproterenol and dilute in 2,018 µl of double distilled water
- ANP stock solution at 200 µM
- Dilute 0.5 mg of human ANP in 812 µl of sterile water
- Make 10 µl aliquots and store at -20 °C
- Dilute 0.5 mg of human ANP in 812 µl of sterile water
Acknowledgments
This work was financially supported by the INSERM and Clarins Dermocosmetique. We acknowledge Grolleau-Raoux from aesthetic surgery department, Rangueil Hospital for the collection of AT samples. This approach was recently published by our groups (Gao et al., 2016). The authors have no conflict or competing interests to disclose.
References
- Gao, H., Volat, F., Sandhow, L., Galitzky, J., Nguyen, T., Esteve, D., Åström, G., Mejhert, N., Ledoux, S., Thalamas, C., Arner, P., Guillemot, J. C., Qian, H., Rydén, M. and Bouloumié, A. (2016). CD36 is a marker of human adipocyte progenitors with pronounced adipogenic and triglyceride accumulation potential. Stem Cells 35(7): 1799-1814.
- Lafontan, M. (2012). Historical perspectives in fat cell biology: the fat cell as a model for the investigation of hormonal and metabolic pathways. Am J Physiol Cell Physiol 302(2): C327-359.
- Large, V., Peroni, O., Letexier, D., Ray, H. and Beylot, M. (2004). Metabolism of lipids in human white adipocyte. Diabetes Metab 30(4): 294-309.
- Robdel, M. (1964). Metabolism of isolated fat cells. I. Effects of hormones on glucose metabolism and lipolysis. J Biol Chem 239: 375-380.
- Ruan, H., Zarnowski, M. J., Cushman, S. W. and Lodish, H. F. (2003). Standard isolation of primary adipose cells from mouse epididymal fat pads induces inflammatory mediators and down-regulates adipocyte genes. J Biol Chem 278(48): 47585-93.
Article Information
Copyright
© 2018 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Decaunes, P., Bouloumié, A., Ryden, M. and Galitzky, J. (2018). Ex vivo Analysis of Lipolysis in Human Subcutaneous Adipose Tissue Explants. Bio-protocol 8(3): e2711. DOI: 10.21769/BioProtoc.2711.
Category
Cell Biology > Cell metabolism > Lipid
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link









