- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
TUNEL Assay to Assess Extent of DNA Fragmentation and Programmed Cell Death in Root Cells under Various Stress Conditions
Published: Vol 7, Iss 16, Aug 20, 2017 DOI: 10.21769/BioProtoc.2502 Views: 15070
Reviewed by: Marisa RosaYingnan HouAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Efficient Transient Gene Knock-down in Tobacco Plants Using Carbon Nanocarriers
Gozde S. Demirer and Markita P. Landry
Jan 5, 2021 6401 Views

Faster Bacterial Gene Cloning Using the Brick into the Gateway (BiG) Protocol
Flaviani G. Pierdoná [...] Fabio T. S. Nogueira
Dec 20, 2022 2391 Views
Abstract
DNA damage is one of the common consequences of exposure to various stress conditions. Different methods have been developed to accurately assess DNA damage and fragmentation in cells and tissues exposed to different stress agents. However, owing to the presence of firm cellulosic cell wall and phenolics, plant cells and tissues are not easily amenable to be subjected to these assays. Here, we describe an optimized TUNEL (terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling) assay-based protocol to determine the extent of DNA fragmentation and programmed cell death in plant root cells subjected to various stress conditions. The method described here has the advantages of simplicity, reliability and reproducibility.
Keywords: DNA fragmentationBackground
Exposure to various stresses generally leads to at least some degree of DNA damage resulting in various lesions such as thymine dimerization, alkylation of bases, single stranded nicks, and double-stranded breaks (Bray and West, 2005; Manova and Gruszka, 2015). Of all types of DNA damage, DNA fragmentation is of particular concern during stress conditions, which may either be a direct effect of the stress (as observed, for example, upon treatment with genotoxic agents) or an indirect effect (predominantly, via excessive generation of reactive oxygen species) or may even be a cumulative consequence of both (Bray and West, 2005; Kapoor et al., 2015). This DNA damage must be accurately repaired by the cell’s repair machinery, failing which there may be deleterious consequences including cell death. For maintaining the normal state, cells utilize the DNA damage response which relies on three non-exclusive events viz. detection/recognition of the damage, its access by the repair machinery and finally its repair (Smerdon, 1991).
One of the major molecular mechanisms of stress adaptation at the cellular level involves the resistance to DNA damage and/or efficient repair of the damaged DNA caused due to stress. Therefore, to assess the stress adaptability of a genotype, accurate assessment of DNA damage is often needed. Two widely-used assays to detect DNA fragmentation in plants are Single Cell Gel Electrophoresis–also known as Comet assay (Santos et al., 2015), and TUNEL [Terminal deoxynucleotidyl Transferase (TdT)-mediated dUTP Nick-End Labeling] assay. In comet assay, the tissue of interest is sliced and the resulting cell suspension containing nuclei is embedded in an agarose matrix followed by its alkaline electrophoresis and staining with DAPI/ethidium bromide. After electrophoresis, micrographs show the appearance of broken DNA like a tail similar to that of a comet while the undamaged and condensed DNA appears like a spherical mass forming the head of the comet (Wang et al., 2013). Comet assay, though quite useful, has a few limitations. For instance, it requires isolated nuclei, and hence gives no information on the distribution of DNA damage in a given tissue as well as regarding programmed cell death (PCD). The other widely-used assay–TUNEL assay, can be used to detect in situ DNA strand breaks. TUNEL assay is based on incorporation of labeled dUTP in the DNA (mediated by the enzyme terminal deoxynucleotidyl transferase) which occurs only at the regions with free 3’ termini (i.e., breaks or extreme ends of the chromosome) (Gavrieli et al., 1992). Besides, as breaks in inter-nucleosomal DNA often lead to programmed cell death, TUNEL assay provides significant information about PCD. TUNEL assay, in its basic form, also offers the advantages of simplicity and can give an idea about the distribution of DNA fragmentation (TUNEL-positive cells) in the tissue being studied.
Plant tissues are not easily amenable to some of the steps of TUNEL assay. The major reasons for this are: difficulty in permeabilization due to the presence of cellulosic cell wall and potential inhibition of TdT-catalyzed reaction by phenolics present in the plant cells. Due to these reasons, TUNEL assay is not a frequently utilized procedure for assessment of DNA fragmentation and PCD in plants. A few recent studies, nonetheless, have shown the application of TUNEL assay in rice (Kwon et al., 2013) and Arabidopsis (Phan et al., 2011; Yang et al., 2014). However, most of these studies have used microtomy/ultramicrotomy and ‘paraffin section’ preparation–a procedure which is not very easy, and requires somewhat expensive instrumentation and technical expertise. Given the range of information which TUNEL assay can provide, especially when determining the stress adaptability of plant genotypes, and its advantages in comparison to other methods, there is a need to develop a standardized, easy-to-follow and relatively inexpensive protocol for TUNEL assay using plant tissues.
Here, we describe an optimized TUNEL assay-based protocol to assess the extent of DNA fragmentation and programmed cell death in plant root cells under various stress conditions. The protocol presented here describes, in detail, a more generalized version of the methodology used for TUNEL assay in our recent study (Tripathi et al., 2016). While we often use this method to study DNA damage and PCD in root tissue from rice and Arabidopsis, it can also be utilized to study these phenomena in root tissue from other herbaceous plants with some minor modifications as detailed in the ‘Procedure’ section. The method presented here is quite easy-to-follow, reliable and reproducible.
Materials and Reagents
- 1-200 μl pipet tips (DNase-free) (Corning, USA)
- 0.2-2 μl pipet tips (DNase-free) (Corning, USA)
- 100-1,000 μl pipet tips (DNase-free) (Corning, USA)
- Razor blade (any standard make)
- 1.5 ml and 2 ml microcentrifuge tubes (DNase-free) (Corning, USA)
- 15 ml and 50 ml centrifuge tubes (DNase-free) (Corning, USA)
- Aluminium foil (any standard make)
- Glass slides (any standard make)
- Cover slips (any standard make)
- 1-4 weeks old rice (Oryza sativa cv. IR64) seedlings (see Note at the beginning of the ‘Procedure’ section)
- Ethanol (Sigma-Aldrich, catalog number: 24102 )
Note: This product has been discontinued. - ProLong® Gold Antifade mountant with DAPI (Thermo Fisher Scientific, InvitrogenTM, catalog number: P36931 )
- DeadEndTM Fluorometric TUNEL System (Promega, catalog number: G3250 )
Note: *In case the DeadEndTM Fluorometric TUNEL System (Promega, catalog number: G3250 ) is being used, then the chemicals/reagents marked with an asterisk (*) need not be procured. See Note 2 below. - Sodium chloride (NaCl) (AMRESCO, catalog number: 0241 )
- Potassium chloride (KCl) (AMRESCO, catalog number: 0395 )
- Sodium phosphate dibasic (Na2HPO4) (AMRESCO, catalog number: 0404 )
- Potassium phosphate monobasic (KH2PO4) (AMRESCO, catalog number: 0781 )
- Paraformaldehyde powder (Sigma-Aldrich, catalog number: 158127 )
- Citric acid monohydrate (Sigma-Aldrich, catalog number: C1909 )
- Trisodium citrate dihydrate (Sigma-Aldrich, catalog number: S1804 )
- Triton X-100 (Sigma-Aldrich, catalog number: T8787 )
- Tris(hydroxymethyl)aminomethane (Sigma-Aldrich, catalog number: 252859 )
- Sodium cacodylate* (Sigma-Aldrich, catalog number: C4945 )
- Cobalt(II) chloride hexahydrate* (Sigma-Aldrich, catalog number: C8661 )
- Dithiothreitol (DTT) (Sigma-Aldrich, catalog number: 10708984001 )
Manufacturer: Roche Diagnostics, catalog number: 10708984001 . - Bovine serum albumin (BSA) (AMRESCO, catalog number: 0332 )
- Ethylenediaminetetraacetic acid (EDTA) (Sigma-Aldrich, catalog number: 03620 )
- Fluorescein-12-dUTP* (Thermo Fisher Scientific, Thermo ScientificTM, catalog number: R0101 )
- dATP* (Promega, catalog number: U1202 )
- Terminal Deoxynucleotidyl Transferase, Recombinant (rTdT)* (Promega, catalog number: M1871 )*
- Sodium citrate
- Hydrochloric acid (HCl)
- Propidium iodide (PI) (Sigma-Aldrich, catalog number: 81845 )
- DNase-free proteinase K (Thermo Fisher Scientific, InvitrogenTM, catalog number: AM2544 )
- 1x phosphate buffered saline (PBS), pH 7.4 (see Recipes)
- Fixative buffer (see Recipes)
- 100 mM citric acid solution (see Recipes)
- 100 mM trisodium citrate dihydrate solution (see Recipes)
- 100 mM sodium citrate buffer, pH 6.0 (see Recipes)
- Permeabilization solution (see Recipes)
- Proteinase K (see Recipes)
- Equilibration buffer (see Recipes)
- Nucleotide mix (see Recipes)
- TUNEL reaction mix (see Recipes)
- 20x saline-sodium citrate (SSC) buffer (see Recipes)
- 2x saline-sodium citrate (SSC) buffer (see Recipes)
- Propidium iodide stock solution (1 μg/μl) (see Recipes)
Equipment
- 0.2-2 μl pipette (Gilson, PIPETMAN Classic®)
- 2-200 μl pipette (Gilson, PIPETMAN Classic®)
- 100-1,000 μl pipette (Gilson, PIPETMAN Classic®)
- Glass beakers of volume 20 ml, 50 ml, and 100 ml (Schott Duran, Germany)
- Reagent bottles, glass of volume 50 ml, 100 ml, 500 ml, and 1,000 ml (any standard make)
- Laboratory fume hood (any standard make)
- Magnetic stirrer (Genetix Brand, India)
- Water bath (any standard make)
- Confocal microscope (Nikon, model: Nikon A1R )
Software
- ImageJ Standard version 1.46r (http://imagej.net/mbf/)
- NIS Elements AR (Nikon, Japan) or a comparable software for confocal microscope image acquisition and analysis
Procedure
Note: The steps mentioned below have been described for carrying out in situ TUNEL assay in samples obtained from root tissue of 1-4 weeks old rice (Oryza sativa cv. IR64) seedlings. For root tissue from rice plants older than 4 weeks, or for those from other herbaceous plant species, the volume of the vessels and reagents can be adjusted accordingly.
- Germination of seeds, growth conditions and processing of samples
Seeds may be germinated in hydroponic system, or in vermiculite or on solid medium. For rice, the suggested growth conditions are 28 ± 2 °C and 16 h/8 h photoperiod (Tripathi et al., 2015). After growth for desired number of days (for example, 15 days), root tissue should be separated from the seedling by cutting using razor blades. - Fixation
- Fix root samples from the plants in which DNA fragmentation and PCD have to be assessed by incubating roots in fixative buffer for 16 h at 4 °C. Ensure that the roots are completely immersed in the fixing solution. Fixation may be performed in 50 ml tubes with their caps closed properly.
Note: Fixative buffer contains paraformaldehyde–a bronchial, eye and skin irritant. Please see Note regarding preparation and disposal of the fixative buffer in the Recipes section. - Wash the fixed roots with absolute ethanol (by adding absolute ethanol and gentle shaking for 10-15 sec) twice at room temperature. After washing, incubate the root samples in 70% ethanol for 24 h at 4 °C. Decant the solution (see Note 1).
- Incubate the samples in 1x PBS (see Recipes) for 20 min at room temperature. Remove the solution and repeat this step thrice with fresh PBS to completely remove traces of ethanol.
- Fix root samples from the plants in which DNA fragmentation and PCD have to be assessed by incubating roots in fixative buffer for 16 h at 4 °C. Ensure that the roots are completely immersed in the fixing solution. Fixation may be performed in 50 ml tubes with their caps closed properly.
- Permeabilization
- Take out the root sample from the PBS solution and incubate the sample in 100 mM sodium citrate buffer (pH 6.0) (see Recipes) in a fresh tube for 15 min at room temperature.
- Subject root samples immersed in the sodium citrate buffer to microwave (350 W) for a duration of 30-60 sec depending on the thickness of the root tissue. For subjecting samples to microwave radiation, the root samples may be placed in 200 ml microwave-safe glass beakers. The volume of citrate buffer in these beakers should be sufficient enough to ensure complete immersion of the samples. For root samples obtained from 1-2 week-old rice seedlings, buffer volume of 100 ml is usually sufficient. Cool the samples rapidly by adding equal volume of distilled water.
- Remove the solution and incubate the samples in permeabilization solution (see Recipes) at 37 °C for 30 min in water bath with intermittent shaking.
- To digest the proteins, add DNase-free Proteinase K (see Recipes) to a final concentration of 20 µg/ml followed by incubation at 37 °C for 30 min in a water bath with intermittent shaking.
- Remove the solution and wash the root samples thrice with 1x PBS at room temperature.
- Take out the root sample from the PBS solution and incubate the sample in 100 mM sodium citrate buffer (pH 6.0) (see Recipes) in a fresh tube for 15 min at room temperature.
- TUNEL reaction
- For in situ TUNEL assay, cut root samples approximately 1 cm length from the root tip using sharp razor blades and glass plates. Collect the cut root tips in microcentrifuge tubes (2 ml), one tube for each of the samples. TUNEL reaction can now be performed in these tubes.
Note: While cutting the root samples, avoid mechanical damage, for that reason the razor blades should be sharp. - Prepare TUNEL reaction mix (see Recipes) and add 110 µl of the mix (see Note 2 below) to each of the microcentrifuge tubes. The reaction should be carried out at 37 °C for 1 h. The microcentrifuge tubes should be covered with aluminum foil so as to avoid exposure to light.
Note: The step C2 should be carried out immediately after C1. - Stop the reaction by adding 1 ml of 2x saline-sodium citrate (2x SSC) buffer (pH 7.0, see Recipes).
- Remove 2x SSC solution and stain root tips with propidium iodide by addition of 100 μl of 1 μg/ml of propidium iodide solution (final concentration, see Recipes) and incubation at 37 °C for 10 min. Remove the PI solution and wash thrice with 1x PBS.
Note: The microcentrifuge tubes should be covered with aluminum foil so as to avoid exposure to light. - Mount the root tips onto slides with an Antifade mounting reagent. For this piece of study, we have used ProLong® Gold Antifade reagent with DAPI (Thermo Fisher Scientific, USA).
- Visualize the slides under a confocal microscope using 405 nm- (for DAPI) and 488 nm- (for PI and fluorescein) -lasers, as applicable, and channel settings based on emission range of the respective dyes. Representative images have been shown in Figure 1.
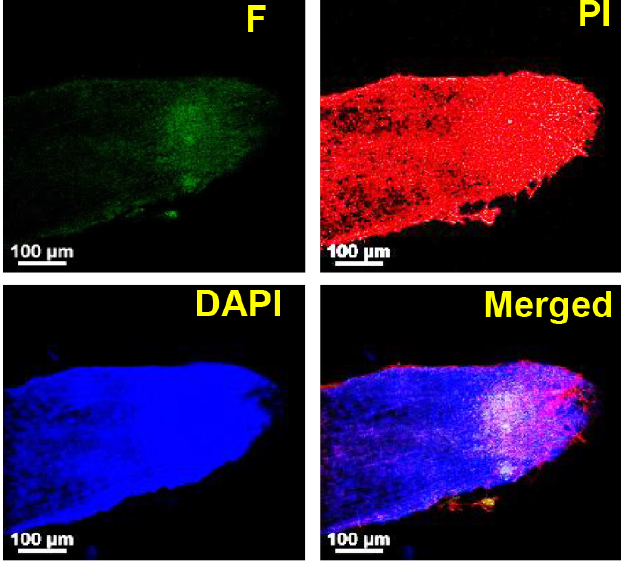
Figure 1. Micrographs showing salt stress-treated rice root tissue subjected to TUNEL assay to assess in situ DNA fragmentation. Root tissue from 15-day old rice seedlings was subjected to salinity stress (200 mM NaCl) for 60 h followed by TUNEL assay as per the protocol described above. F: Fluorescein (green), PI: Propidium Iodide (red), DAPI: 4’,6-diamidino-2-phenylindole (blue). Samples were mounted on slides in a mountant (ProLong® Gold Antifade with DAPI, Thermo Fisher Scientific) and the slides were examined under Nikon A1R (Nikon, Japan) confocal microscope using a 20x objective. NIS Elements AR software (Nikon, Japan) was used to acquire and process the images. DAPI and PI were used to stain DNA (both damaged and undamaged). Fluorescein fluorescence (green) is due to incorporation of fluorescein-12-dUTP during the TUNEL reaction and would correspond to the number of free DNA ends. Scale bars = 100 µm.
- For in situ TUNEL assay, cut root samples approximately 1 cm length from the root tip using sharp razor blades and glass plates. Collect the cut root tips in microcentrifuge tubes (2 ml), one tube for each of the samples. TUNEL reaction can now be performed in these tubes.
Data analysis
The incorporation of labelled dUTP during the TUNEL reaction would theoretically be directly proportional to the frequency of free DNA termini (which are generated majorly due to breaks in DNA) and therefore, would indicate the extent of DNA fragmentation which in turn can provide a rough estimate of PCD. The extent of DNA fragmentation can be quantified through measurement of fluorescence intensity in the micrographs (as shown in Figure 1) in a well-defined region of interest (ROI) using various micrograph analysis software like ImageJ (http://imagej.net/mbf/) or NIS Elements (Nikon, Japan). Analysis of fluorescence intensity through ImageJ has been described in the documentation section of ImageJ and can be found at the URL: https://imagej.nih.gov/ij/docs/. One of the paths for choosing ROI and analyzing the image intensity is Analyze>Tools>ROI Manager, through which ROI Manager can be accessed. A selection tool may be chosen and the ROI can be selected by drawing. Next, to analyze the intensity, in the ROI manager window click ‘Add [t]’ then ‘Measure’ which will give the values of maximum, minimum and mean intensity along with area of the ROI. Detailed instructions regarding image processing and analysis can be found in ImageJ documentation section and in the Tutorials and Examples section (https://imagej.nih.gov/ij/docs/examples/index.html). Alternatively, when longitudinal sections of roots are used for TUNEL assay, the frequency of TUNEL-positive cells can be determined via counting the number of cells showing fluorescein-fluorescence and the total number of cells in the defined ROI. Irrespective of the method chosen, the capture settings of microscope, and the area of the ROI should be kept identical for the samples which are to be compared. When examining the stress adaptability of different plants, a relative assessment of the extent of DNA fragmentation by analyzing the fluorescence intensity often serves the purpose.
Notes
- Incubation of whole root samples in buffers or ethanol is to be carried out in beakers of appropriate volume. During the incubation period, the beakers/tubes should be properly covered with aluminum foil. Decanting/removal of solution after washing or incubation should be carefully carried out so as not to disturb the root samples and the next step should be carried out immediately to avoid drying of the samples.
- To carry out the TUNEL reaction, for our recent study (Tripathi et al., 2016), we used equilibration buffer, nucleotide mix (containing fluorescein-12-dUTP) and recombinant terminal deoxynucleotidyl transferase (rTdT) supplied with the DeadEndTM Fluorometric TUNEL System (from Promega, USA). However, buffers and reaction mixes prepared using reagents (molecular biology-grade), along with separately procured terminal deoxynucleotidyl transferase (TdT) enzyme and fluorescently labelled dUTP, can also be used to carry out the TUNEL reaction. Buffer preparation and composition of the reaction mixes have been provided below.
Recipes
- 1x phosphate buffered saline (PBS), pH 7.4 (1,000 ml)
8 g NaCl
0.2 g KCl
1.44 g Na2HPO4
0.24 g KH2PO4
Dissolve in 800 ml deionized water and bring the volume to 1,000 ml with deionized water. Store at room temperature - Fixative buffer (4% paraformaldehyde [prepared in 1x PBS, pH 7.4])
20 g paraformaldehyde is to be weighed and 1x PBS is added so as to bring the volume to 500 ml. For proper dissolution, heat the closed bottle at 65 °C in a water bath for 1.5-2 h. The solution may be stored at 4 °C
Note: It may be noted that paraformaldehyde is quite hazardous and is a bronchial, eye and skin irritant and therefore, it should be weighed in a fume hood. The fixative buffer after use should be discarded by emptying in a hazardous waste container. - 100 mM citric acid solution
2.10 g citric acid monohydrate in 100 ml deionized water. The solution may be stored at 4 °C - 100 mM trisodium citrate dihydrate solution
2.94 g trisodium citrate dihydrate in 100 ml deionized water. The solution may be stored at 4 °C - 100 mM sodium citrate buffer (100 ml), pH 6.0
Mix 11.5 ml of 100 mM citric acid solution and 88.5 ml of 100 mM trisodium citrate dihydrate solution. Ensure the pH of the resulting solution is 6.0. The solution may be stored at 4 °C - Permeabilization solution
0.1% Triton X-100 in 100 mM sodium citrate buffer, pH 6.0. The solution should be prepared fresh - Proteinase K
20 mg/ml prepared in 10 mM Tris-Cl, pH 7.5. The enzyme solution should be stored at -20 °C - Equilibration buffer*
200 mM sodium cacodylate (pH 6.5)
25 mM Tris-Cl (pH 6.5)
2.5 mM cobalt chloride
200 µM dithiothreitol
250 µg/ml bovine serum albumin
The solution should be prepared fresh - Nucleotide mix*
10 mM Tris-HCl (pH 7.5)
1 mM EDTA
60 μM fluorescein-12-dUTP
100 μM dATP
The mix should be stored at -20 °C
*Note: In case the DeadEndTM Fluorometric TUNEL System (Promega, USA) is being used, then the reagents marked with an asterisk (*) need not be prepared as similar reagents are supplied with the kit. See Note 2 above. - TUNEL reaction mix (per reaction)
100 µl equilibration buffer
10 µl nucleotide mix
60 U (2 µl) of rTdT (Promega, USA)
The mix should be freshly prepared - 20x saline-sodium citrate (SSC) buffer (100 ml), pH 7.0
3 M (17.54 g) sodium chloride (NaCl)
0.3 M (8.82 g) sodium citrate
Dissolved in 80 ml deionized water; pH is adjusted to 7.0 with hydrochloric acid (HCl) and final volume is to be brought to 100 ml with deionized water. The solution may be stored at room temperature - 2x saline-sodium citrate (SSC) buffer (100 ml)
10 ml 20x SSC, pH 7.0
90 ml deionized water - Propidium iodide stock solution (1 mg/ml)
10 mg of propidium iodide dissolved in 10 ml 1x PBS
Note: This solution is to be stored at 4 °C in dark. Besides, propidium iodide is potentially carcinogenic. Avoid contact with skin and wear proper protective clothing while handling the solution. Propidium iodide solution after use should be discarded by emptying in a hazardous waste container.
Acknowledgments
Research in our laboratory is supported by funds from the Department of Biotechnology (DBT), Ministry of Science and Technology, Government of India, and ICGEB. AKT has been a recipient of senior research fellowship from DBT, Government of India, which is gratefully acknowledged. The protocol presented here describes, in detail, a more generalized version of the methodology used for TUNEL assay to assess DNA fragmentation and programmed cell death in root tissue under various stress conditions in our recent study (Tripathi et al., 2016) published in the journal Plant Physiology.
References
- Bray, C. M. and West, C. E. (2005). DNA repair mechanisms in plants: crucial sensors and effectors for the maintenance of genome integrity. New Phytol 168(3): 511-528.
- Gavrieli, Y., Sherman, Y. and Ben-Sasson, S. A. (1992). Identification of programmed cell death in situ via specific labeling of nuclear DNA fragmentation. J Cell Biol 119(3): 493-501.
- Kapoor, D., Sharma, R., Handa, N., Kaur, H., Rattan, A., Yadav, P., Gautam, V., Kaur, R., and Bhardwaj, R. (2015). Redox homeostasis in plants under abiotic stress: role of electron carriers, energy metabolism mediators and proteinaceous thiols. Front Environ Sci 3: 13.
- Kwon, Y. I., Abe, K., Endo, M., Osakabe, K., Ohtsuki, N., Nishizawa-Yokoi, A., Tagiri, A., Saika, H. and Toki, S. (2013). DNA replication arrest leads to enhanced homologous recombination and cell death in meristems of rice OsRecQl4 mutants. BMC Plant Biol 13: 62.
- Manova, V. and Gruszka, D. (2015). DNA damage and repair in plants - from models to crops. Front Plant Sci 6: 885.
- Phan, H. A., Iacuone, S., Li, S. F. and Parish, R. W. (2011). The MYB80 transcription factor is required for pollen development and the regulation of tapetal programmed cell death in Arabidopsis thaliana. Plant Cell 23(6): 2209-2224.
- Santos, C., Pourrut, B., and Ferreira de Oliveira, J. (2015). The use of comet assay in plant toxicology: recent advances. Front Genet 6: 216.
- Smerdon, M. J. (1991). DNA repair and the role of chromatin structure. Curr Opin Cell Biol 3(3): 422-428.
- Tripathi, A. K., Pareek, A. and Singla-Pareek, S. L. (2016). A NAP-family histone chaperone functions in abiotic stress response and adaptation. Plant Physiol 171(4): 2854-2868.
- Tripathi, A. K., Singh K., Pareek, A. and Singla-Pareek, S. L. (2015). Histone chaperones in Arabidopsis and rice: genome-wide identification, phylogeny, architecture and transcriptional regulation. BMC Plant Biol 15: 42.
- Wang, Y., Xu, C., Du, L. Q., Cao, J., Liu, J. X., Su, X., Zhao, H., Fan, F. Y., Wang, B., Katsube, T., Fan, S. J. and Liu, Q. (2013). Evaluation of the comet assay for assessing the dose-response relationship of DNA damage induced by ionizing radiation. Int J Mol Sci 14(11): 22449-22461.
- Yang, Z. T., Wang, M. J., Sun, L., Lu, S. J., Bi, D. L., Sun, L., Song, Z. T., Zhang, S. S., Zhou, S. F. and Liu, J. X. (2014). The membrane-associated transcription factor NAC089 controls ER-stress-induced programmed cell death in plants. PLoS Genet 10(3): e1004243.
Article Information
Copyright
© 2017 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Readers should cite both the Bio-protocol article and the original research article where this protocol was used:
- Tripathi, A. K., Pareek, A. and Singla-Pareek, S. L. (2017). TUNEL Assay to Assess Extent of DNA Fragmentation and Programmed Cell Death in Root Cells under Various Stress Conditions. Bio-protocol 7(16): e2502. DOI: 10.21769/BioProtoc.2502.
- Tripathi, A. K., Pareek, A. and Singla-Pareek, S. L. (2016). A NAP-family histone chaperone functions in abiotic stress response and adaptation. Plant Physiol 171(4): 2854-2868.
Category
Plant Science > Plant molecular biology > DNA > DNA modification
Molecular Biology > DNA > DNA labeling
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link