- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
MPM-2 Mediated Immunoprecipitation of Proteins Undergoing Proline-directed Phosphorylation
Published: Vol 6, Iss 23, Dec 5, 2016 DOI: 10.21769/BioProtoc.2046 Views: 8841
Reviewed by: Oneil G. BhalalaEmma PuighermanalPia Giovannelli

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Ultrafast Isolation of Synaptic Terminals From Rat Brain for Cryo-Electron Tomography Analysis
Rong Sun and Qiangjun Zhou
Sep 5, 2025 3587 Views
Protocol for the Preparation of a Recombinant Treacle Fragment for Liquid–Liquid Phase Separation (LLPS) Assays
Nadezhda V. Petrova [...] Artem K. Velichko
Sep 20, 2025 1869 Views

Optimized Secretome Sample Preparation From High Volume Cell Culture Media for LC–MS/MS Proteomic Analysis
Basil Baby Mattamana [...] Peter Allen Faull
Dec 20, 2025 1344 Views
Abstract
Immunoprecipitation (IP) represents a widely utilized biochemical method to isolate a specific protein from a complex mixture taking advantage of an antibody that specifically recognizes that particular target molecule. This procedure is extremely versatile and can be applied to concentrate a specific protein, to identify interacting partners in complex with it or to detect post-translational modifications. The mitotic protein monoclonal 2 (MPM-2) is an antibody originally raised against extracts of synchronized mitotic HeLa cells to identify proteins selectively present in mitotic, and not in interphase-cells (Davis et al., 1983). MPM-2 recognizes phosphorylated serine or threonine residues followed by proline (pS/T-P), consensus epitopes generated by the concerted action of proline-directed protein kinases and phosphatases (Lu et al., 2002). These reversible phosphorylation events have emerged to control various cellular processes by promoting conformational changes on the target that are not simply due to the phosphorylation event per se. These motifs, once phosphorylated, are able to recruit Pin1 (Peptidyl-prolyl Isomerase NIMA interacting protein 1) (Lu et al., 1996; Lu and Zhou, 2007), a chaperone which drives the cis/trans isomerization reaction on the peptide bond, switching the substrate between functionally diverse conformations (Lu, 2004; Wulf et al., 2005). This protocol describes a general MPM-2 based immunoprecipitation strategy using the scaffolding molecule postsynaptic density protein-95 (PSD-95) (Chen et al., 2005), a neuronal Pin1 target (Antonelli et al., 2016), as an example to illustrate the detailed procedure.
Keywords: MPM-2 antibodyBackground
Identification of antigens recognized by MPM-2 antibody represents a useful starting point for the discovery of target molecules undergoing post-phosphorylation prolyl-isomerization regulatory mechanism. The prolyl isomerase Pin1, in fact, shares with MPM-2 antibody the same recognition motif as well as the phospho-dependency of the binding. The main advantage of using the MPM-2 antibody in immunoprecipitation experiments is the achievement of a selective enrichment of the phosphorylated targets over the unphosphorylated counterparts, which are frequently present in much greater quantities in the cell. Precipitated antigens by virtue of such post-translational modification can be then easily identified by standard western blotting using highly specific primary antibodies.
Materials and Reagents
- 10 cm2 Petri dishes (Corning, catalog number: 353003 )
- Dissection plate (Sigma-Aldrich, catalog number: P7741 )
- 1.5 ml reaction tube (SARSTEDT, catalog number: 72.706.700 )
- Surgical scalpel blade number 11 (Sigma-Aldrich, catalog number: S2771 )
- 26 gauge needle (BD, PrecisionGlideTM, catalog number: 305110 )
- Amersham Protran Premium 0.2 NC (GE Healthcare, catalog number: 10600004 )
- HEK293 cells
- C57BL/6 mouse strain (or any other mouse model, both genders)
- pAcGFP1-C1 vector (Takara Bio, Clontech, catalog number: 632470 )
- FLAG-PSD-95 plasmid DNA (Craven et al., 1999)
- Dulbecco’s modified Eagle’s medium (DMEM) high glucose, GlutaMAXTM supplement (Thermo Fisher Scientific, GibcoTM, catalog number: 10566016 )
- 10% fetal bovine serum (FBS)
- Polyethylenimine, Linear (PEI) (Polysciences, catalog number: 23966-2 )
- Penicillin-streptomycin (10,000 U/ml) (Thermo Fisher Scientific, GibcoTM, catalog number: 15140148 )
- cOmpleteTM EDTA-free protease inhibitor cocktail tablets (Roche Diagnostics, catalog number: 11873580001 )
- Phosphatase inhibitor cocktail 1 (Sigma-Aldrich, catalog number: P2850 )
- IgG from mouse serum (Sigma-Aldrich, catalog number: I8765 )
- Anti-FLAG (clone M2) (Sigma-Aldrich, catalog number: F9291 )
- Phospho-Serine/Threonine-Proline MPM2 antibody (EMD Millipore, catalog number: 05-368 )
- Protein G Sepharose 4 Fast Flow (GE Healthcare, catalog number: 17-0618-01 )
- PageRulerTM prestained protein ladder (Thermo Fisher Scientific, Thermo ScientificTM, catalog number: 26616 )
- Bovine serum albumin (BSA) (Sigma-Aldrich, catalog number: A2153 )
- Goat anti-mouse HRP conjugated IgG (EMD Millipore, catalog number: 12-349 )
- ECL detection reagents (GE Healthcare, catalog number: RPN2209 )
- Sodium chloride (NaCl) (Sigma-Aldrich, catalog number: S9888 )
- Potassium chloride (KCl) (Sigma-Aldrich, catalog number: P9541 )
- Disodium hydrogen phosphate (Na2HPO4) (Sigma-Aldrich, catalog number: 7558-79-4 )
- Potassium dihydrogen phosphate (KH2PO4) (Sigma-Aldrich, catalog number: 7778-77-0 )
- Tween 20 (Sigma-Aldrich, catalog number: P9416 )
- Trizma® base (Sigma-Aldrich, catalog number: T1503 )
- NP-40 (Sigma-Aldrich, catalog number: I3021 )
- Triton X-100 (Sigma-Aldrich, catalog number: T8787 )
- Glycerol (Sigma-Aldrich, catalog number: G5516 )
- Sodium orthovanadate (Na3VO4) (Sigma-Aldrich, catalog number: S6508 )
- Sodium fluoride (NaF) (Sigma-Aldrich, catalog number: 201154 )
- TEMED (Sigma-Aldrich, catalog number: T9281 )
- Sodium dodecyl sulfate (SDS) (Sigma-Aldrich, catalog number: L3771 )
- Bromophenol blue (Sigma-Aldrich, catalog number: B0126 )
- Methanol (Sigma-Aldrich, catalog number: 322415 )
- Glycine (Sigma-Aldrich, catalog number: G8898 )
- Acrylamide/bis-acrylamide (29:1) (Sigma-Aldrich, catalog number: A2792 )
- Ammonium persulfate (Sigma-Aldrich, catalog number: A3678 )
- Phosphate buffered saline (1x PBS) (see Recipes)
- MPM-2 lysis buffer (see Recipes)
- Phosphatase inhibitors (see Recipes)
- IP wash buffer (see Recipes)
- 2x Laemmli sample buffer (see Recipes)
- Western blot running buffer (see Recipes)
- Western blot transfer buffer (see Recipes)
- Tris-buffered saline-Tween 20 (TBS-T) (see Recipes)
- BSA blocking solution (see Recipes)
Equipment
- Nikon Diaphon 300 phase contrast inverted tissue culture microscope
- Cell scraper (Sigma-Aldrich, catalog number: C5981 )
- Refrigerated centrifuge (Eppendorf, model: 5424R )
- Incubator (Panasonic Biomedical Sales Europe, model: MCO-18AC-PE )
- DNA sonicator (EpiShear, model: Cooled Sonication Platform )
- Rotator mixer (Scilogex, model: MX-RD-Pro LCD Digital Tube Rotator Mixer )
- Tissue homogenizer (Thomas Scientific, catalog number: 3409Y72 )
- Cold room, 4 °C
- Polyacrylamide gel electrophoresis system (Bio-Rad Laboratories, catalog number: 1658000EDU )
- Western blot transfer apparatus (Bio-Rad Laboratories, model : Trans-Blot® Cell )
Software
- Alliance 4.7 software (UVITECH)
- UVI band Imager software
Procedure
- FLAG-PSD-95 ectopically expressed in HEK293
- HEK293 cells are cultured in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% fetal bovine serum (FBS) and 1% 100 μg/ml penicillin and 100 μg/ml streptomycin.
- 10 cm2 Petri dishes are transfected with 2 μg FLAG-tagged PSD-95 using PEI transfection method. In particular, 1.8 x 106 cells are plated the day before transfection. Total plasmid DNA is diluted in 500 μl serum-free DMEM (w/o) phenol red. PEI (1 μg/μl) is then added to the diluted DNA on a 7:1 ratio (μg PEI:μg DNA). The mixture is incubated for 20 min at RT and added dropwise. Cells are harvested 48 h post-transfection. Transfection efficiency is evaluated by simultaneous co-transfection of 500 ng of pGFP-C1, a vector DNA encoding the green fluorescent protein. Cells expressing GFP are directly visualized by fluorescence microscopy.
- Remove the medium and wash two times cell culture dishes with ice-cold PBS.
- Scrape adherent cells off the dish using a cell scraper, and then gently transfer the cell suspension into a fresh pre-cooled 1.5 ml reaction tube.
- Centrifuge at 13,500 x g for 10 min at 4 °C (refrigerated centrifuge). Discard the supernatant and collect the pellet.
- Mechanically resuspend the pellet in 700 μl of MPM-2 lysis buffer supplemented with protease (cOmpleteTM EDTA-free inhibitor cocktail) and phosphatase inhibitors (see Recipes).
- Incubate lysates for 30 min at 4 °C with rotation.
- Sonicate lysates with three short bursts of 10 sec each followed by intervals of 30 sec for cooling. Keep lysates at all times on ice and avoid foaming.
Note: Sonication is mainly performed to shear DNA/RNA, making the lysate progressively less viscous. - Centrifuge at 13,500 x g for 10 min at 4 °C (refrigerated centrifuge).
- Pipette out supernatant into a fresh reaction tube and discard the pellets.
- HEK293 cells are cultured in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% fetal bovine serum (FBS) and 1% 100 μg/ml penicillin and 100 μg/ml streptomycin.
- Endogenous PSD-95 from mouse brain
- Sacrifice mice according to the Italian Animal Welfare legislation (D.L. 26/2014) that implements the European Committee Council Directive (2010/63 EEC). The procedures are approved by local veterinary authorities and by the ethical committee of SISSA. After CO2 exposure with the purpose of inducing unconsciousness, postnatal day (P) 10-P15 animals (male and female) are decapitated with sharp surgical scissors. The brain is quickly removed from the skull, rinsed in ice-cold PBS, transferred to an ice-cold dissection plate and minced using a surgical scalpel blade (number 11). Broken up tissue is homogenized at 4 °C using tissue homogenizer, setting 500 rpm 10 times, in MPM-2 lysis buffer supplemented with protease and phosphatase inhibitors (as above). For each 50 mg of brain tissue is added 400 µl of lysis buffer to obtain an optimal protein concentration ranging from 10 to 20 mg/ml. For a P15 mouse brain weighing around 300-350 mg are added 3.0 ml of lysis buffer.
- Incubate tissue homogenate for 30 min-1 h at 4 °C with rotation.
- Sonicate as previously described and pass several times through a 26 gauge needle the homogenate.
- Centrifuge at 13,500 x g for 10 min at 4 °C.
- Gently remove the reaction tubes from the centrifuge, pipette out the supernatant and divide it into two 1.5 ml fresh reaction tubes pre-cooled on ice; discard the pellets.
- Sacrifice mice according to the Italian Animal Welfare legislation (D.L. 26/2014) that implements the European Committee Council Directive (2010/63 EEC). The procedures are approved by local veterinary authorities and by the ethical committee of SISSA. After CO2 exposure with the purpose of inducing unconsciousness, postnatal day (P) 10-P15 animals (male and female) are decapitated with sharp surgical scissors. The brain is quickly removed from the skull, rinsed in ice-cold PBS, transferred to an ice-cold dissection plate and minced using a surgical scalpel blade (number 11). Broken up tissue is homogenized at 4 °C using tissue homogenizer, setting 500 rpm 10 times, in MPM-2 lysis buffer supplemented with protease and phosphatase inhibitors (as above). For each 50 mg of brain tissue is added 400 µl of lysis buffer to obtain an optimal protein concentration ranging from 10 to 20 mg/ml. For a P15 mouse brain weighing around 300-350 mg are added 3.0 ml of lysis buffer.
- Phospho-Serine/Threonine-Proline immunoprecipitation
- Save 30 μl of cell lysate or homogenate as starting material (input) to be tested in SDS-PAGE analysis and 5.0 μl to determine the protein concentration.
- Split the remaining lysate equally into two fresh reaction tubes for MPM-2 immunoprecipitation and normal mouse IgG as control sample (around 1-1.5 mg of total protein in each tube).
- Add 2 μg of MPM-2 antibody and 2 μg of control mouse IgG to the appropriate reaction tube.
Note: The appropriate ratio of MPM-2 antibody/total protein input has to be titrated in preliminary experiments, taking into account the level of expression of the protein under investigation and the number of putative pS/pT-P consensus motifs present on it. - Incubate 3 h at 4 °C with gentle rotation.
Note: The incubation time with MPM-2 antibody follows the same rationale discussed above (step C3). If the protein of interest is poorly expressed overnight incubation is recommended. - Add 100 μl (50% beads/buffer) of Protein-G Sepharose beads equilibrated into MPM-2 lysis buffer to each sample.
Note: To equilibrate the beads, add MPM-2 lysis buffer to the bead, mix in the tube several times, spin down the beads, and remove the supernatant (done twice). - Pull-down the antibody-antigen complexes bound to Protein-G Sepharose beads by incubating for further 2 h at 4 °C with gentle rotation.
- Wash beads three times with lysis buffer.
Note: For wash beads, add buffer to the beads, mix the tube several times, spin down the beads, and remove the supernatant. - After the last wash, pipette out the supernatant and elute proteins by heating to 95-100 °C for 5 min with 50 µl of 2x Laemmli sample buffer. Samples can be immediately loaded onto the gel or kept frozen at -80 °C to be run later.
- Analyze samples using SDS-PAGE and Western blotting.
Load 20 μg of starting material (Input), 50 μl of the IP samples and 7 μl of prestained protein ladder onto SDS-PAGE gel (10 x 10 cm).
Note: In case of MPM-2 detection in Western blot analysis a 2 h transfer is recommended to minimize or avoid loss of post-translational modifications. - Detection of the immunoprecipitated protein using either the target specific antibody or the MPM-2 antibody.
Note: For MPM2 detection block the nitrocellulose membrane with 2.5% BSA in PBS-T (see Recipes) instead of 5% nonfat milk in TBS-T used in standard conditions. Incubate the membrane with MPM-2 antibody (1:500 dilution in BSA Blocking solution) with gentle agitation either overnight at 4 °C or 2 h at RT. After three washes of 5 min each with BSA blocking solution incubate the membrane with HRP-conjugated goat anti-rabbit secondary antibody (1:1,000 dilution in BSA blocking solution) with gentle agitation for 1 h at RT. - Western blot image acquisition is performed using the ECL detection reagents (according to the manufacturer’s protocol) and the Alliance 4.7 software.
Note: In case of aspecific binding to control IgG lysates, a pre-clearing step should be performed. To each sample add 100 μl (50% beads/buffer) of Protein G Sepharose beads and incubate 1 h with gentle rotation at 4 °C. Spin down the beads and collect the supernatants. Add the immunoprecipitating antibodies as described in step C3.
- Save 30 μl of cell lysate or homogenate as starting material (input) to be tested in SDS-PAGE analysis and 5.0 μl to determine the protein concentration.
Data analysis
Quantification of immunoprecipitated target protein was determined by densitometry analysis on the acquired images with Alliance 4.7 software (UVITECH). The volume of the bands corresponding to Input (1/20 of the total lysate) and the immunoprecipitated fraction were determined using the UVIband imager software (Amersham). The amount of immunoprecipitated is normalized to the corresponding inputs.
Representative data
Results from a representative experiment are shown in Figure 1.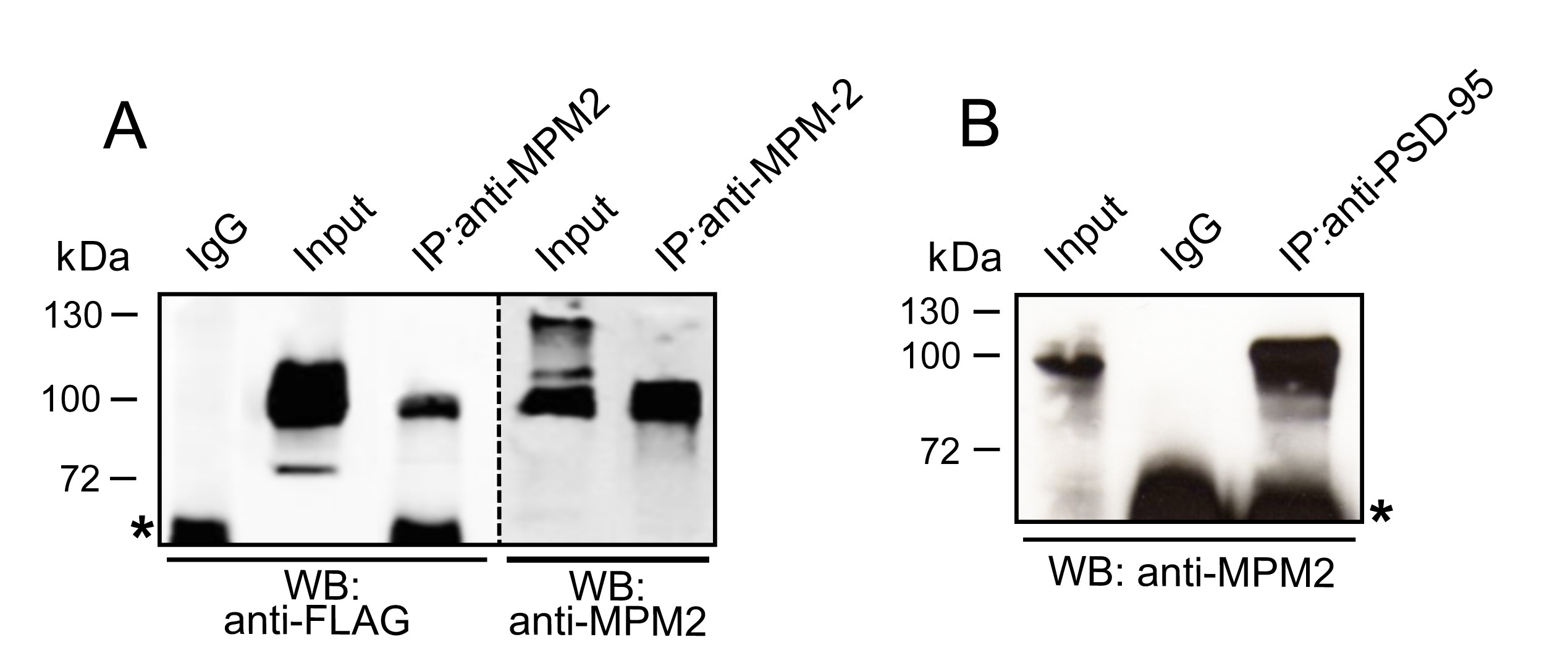
Figure 1. MPM-2 mediated immunoprecipitation of PSD-95 either ectopically expressed in HEK-293 cells or endogenously present in mouse brain. A. FLAG-PSD-95 ectopically expressed in HEK-293 cells was immunoprecipitated with anti-MPM-2 antibody. Each sample was run twice to perform immunoblots with anti-FLAG and anti MPM2 antibodies, as indicated. Mouse IgG was used as negative control. B. PSD-95 was precipitated from brain extracts and unveiled in Western blot using the anti-MPM2 antibody. Asterisks indicate background bands due to secondary antibody cross-reactivity with IgG heavy chains used in immunoprecipitation.
Note: Only a fraction of PSD-95 is phosphorylated at S-T/P consensus motifs (compare Input lanes).
Recipes
- Phosphate buffered saline (1x PBS)
137 mM NaCl
2.7 mM KCl
4.3 mM Na2HPO4
1.47 mM KH2PO4
Adjust to a final pH of 7.4
Note: In PBS-T add 0.1% Tween 20. - MPM-2 lysis buffer
50 mM Tris-HCl (pH 7.5)
1% NP-40
0.5% Triton X-100
150 mM NaCl
10% glycerol
10 mM sodium orthovanadate
1 mM sodium fluoride
1% phosphatase inhibitor cocktail 1
1x cOmpleteTM EDTA-free protease inhibitor cocktail - Phosphatase inhibitors
1 mM sodium orthovanadate
1 mM sodium fluoride
Phosphatase inhibitor cocktail 1 - IP wash buffer
50 mM Tris-HCl (pH 7.5)
100 mM NaCl
0.1% Tween 20
0.1% SDS
10 mM sodium orthovanadate
1 mM sodium fluoride
1% phosphatase inhibitor cocktail 1
1x cOmpleteTM EDTA-free protease inhibitor cocktail - 2x Laemmli sample buffer
100 mM Tris-HCl (pH 6.8)
4% SDS
20% glycerol
1% 2-mercaptoethanol
0.001% bromophenol blue - Western blot running buffer
25 mM Tris
190 mM glycine
0.1% SDS - Western blot transfer buffer
25 mM Tris (pH 8.3)
192 mM glycine
10% methanol
0.1% SDS - Tris-buffered saline-Tween 20 (1x TBS-T)
50 mM Tris (pH 7.6)
150 mM NaCl
0.1% Tween 20 - BSA blocking solution (used also as antibody incubation buffer)
1x TBS-T
2.5% BSA
Note: All buffers are stored at RT except the BSA blocking solution which is kept at 4 °C (no longer than a week). Sample buffer is stored without 2-mercaptoethanol which is freshly added before use. Only buffer’s volumes required for the immunoprecipitation procedure are pre-cooled on ice and supplied with protease and phosphatase inhibitors just before use. All inhibitors are resuspended in sterilized MilliQ water, aliquoted and stored at -20 °C. Sodium orthovanadate is prepared as 200 mM stock solution in sterilized MilliQ water, adjusting the pH to 10.0. At this pH the solution is yellow. To ensure the presence of vanadate monomers the solution is boiled until translucent and the pH is readjusted to 10.0.
Acknowledgments
We are grateful to Dr. S. Vicini (Georgetown University School of Medicine, Washington D.C., USA), for kindly providing PSD-95 cDNA. This work was supported by grants from the Beneficentia Stiftung (BEN 2014/08) and from Telethon (GGP11043).
References
- Antonelli, R., De Filippo, R., Middei, S., Stancheva, S., Pastore, B., Ammassari-Teule, M., Barberis, A., Cherubini, E. and Zacchi, P. (2016). Pin1 modulates the synaptic content of NMDA receptors via prolyl-isomerization of PSD-95. J Neurosci 36(20): 5437-5447.
- Chen, X., Vinade, L., Leapman, R. D., Petersen, J. D., Nakagawa, T., Phillips, T. M., Sheng, M. and Reese, T. S. (2005). Mass of the postsynaptic density and enumeration of three key molecules. Proc Natl Acad Sci U S A 102(32): 11551-11556.
- Craven, S. E., El-Husseini, A. E. and Bredt, D. S. (1999). Synaptic targeting of the postsynaptic density protein PSD-95 mediated by lipid and protein motifs. Neuron 22(3): 497-509.
- Davis, F. M., Tsao, T. Y., Fowler, S. K. and Rao, P. N. (1983). Monoclonal antibodies to mitotic cells. Proc Natl Acad Sci U S A 80(10): 2926-2930.
- Lu, K. P. (2004). Pinning down cell signaling, cancer and Alzheimer's disease. Trends Biochem Sci 29(4): 200-209.
- Lu, K. P., Hanes, S. D. and Hunter, T. (1996). A human peptidyl-prolyl isomerase essential for regulation of mitosis. Nature 380(6574): 544-547.
- Lu, K. P., Liou, Y. C. and Zhou, X. Z. (2002). Pinning down proline-directed phosphorylation signaling. Trends Cell Biol 12(4): 164-172.
- Lu, K. P. and Zhou, X. Z. (2007). The prolyl isomerase PIN1: a pivotal new twist in phosphorylation signalling and disease. Nat Rev Mol Cell Biol 8(11): 904-916.
- Wulf, G., Finn, G., Suizu, F. and Lu, K. P. (2005). Phosphorylation-specific prolyl isomerization: is there an underlying theme? Nat Cell Biol 7(5): 435-441.
Article Information
Copyright
© 2016 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Readers should cite both the Bio-protocol article and the original research article where this protocol was used:
- Antonelli, R. and Zacchi, P. (2016). MPM-2 Mediated Immunoprecipitation of Proteins Undergoing Proline-directed Phosphorylation. Bio-protocol 6(23): e2046. DOI: 10.21769/BioProtoc.2046.
- Garré, J. M., Yang, G., Bukauskas, F. F. and Bennett, M. V. (2016). FGF-1 triggers Pannexin-1 hemichannel opening in spinal astrocytes of rodents and promotes inflammatory responses in acute spinal cord slices. J Neurosci 36(17): 4785-4801.
Category
Neuroscience > Cellular mechanisms > Synaptic physiology
Biochemistry > Protein > Isolation and purification
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link












